Abstract
Deep-sea chemosynthesis-based ecosystems are inhabited by diverse groups of metazoans. Although remote from the euphotic layer, and thus from photosynthetic primary producers, these ecosystems harbour high animal biomasses, orders of magnitude above biomasses usually reported in the deep-sea (Sibuet and Olu 1998; Van Dover 2000). The key to this high productivity is chemoautotrophy, a type of metabolism by which many prokaryotes fix inorganic carbon into organic molecules using the chemical energy resulting from the oxidation of reduced compounds present in their environment (Madigan et al. 2002). Indeed, hydrothermal vents and cold seeps are characterized by the occurrence of fluid emissions originating from the subsurface, which bring reduced compounds into mixing with bottom oxygenated seawater (reviewed in Von Damm 1995; Sibuet and Olu 1998; Van Dover 2000; Tunnicliffe et al. 2003). To summarize, hydrothermal vents occur mostly on oceanic ridges, where bottom seawater circulates into the newly formed crust. The high geothermic gradient linked with the presence of a magma chamber few kilometres below the ridge causes water to heat and to be enriched in reduced compounds (metals, sulphide…). Heated fluids, displaying lower densities, then reach back the seafloor along cracks and are emitted. Fluid interaction with cold seawater provokes the precipitation of dissolved metals and minerals in the form of complexes with sulphide, yielding typical (and often spectacular) chimneys. At cold seeps, which are mostly located along continental margins, fluids originate from the subsurface. The thermogenic or biogenic reduction of buried organic matter produces methane and other hydrocarbons which seep to the seafloor, and processes such as the anaerobic oxidation of methane coupled with sulphate-reduction can account for local enrichments in sulphide.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
6.1 Introduction
Deep-sea chemosynthesis-based ecosystems are inhabited by diverse groups of metazoans. Although remote from the euphotic layer, and thus from photosynthetic primary producers, these ecosystems harbour high animal biomasses, orders of magnitude above biomasses usually reported in the deep-sea (Sibuet and Olu 1998; Van Dover 2000). The key to this high productivity is chemoautotrophy, a type of metabolism by which many prokaryotes fix inorganic carbon into organic molecules using the chemical energy resulting from the oxidation of reduced compounds present in their environment (Madigan et al. 2002). Indeed, hydrothermal vents and cold seeps are characterized by the occurrence of fluid emissions originating from the subsurface, which bring reduced compounds into mixing with bottom oxygenated seawater (reviewed in Von Damm 1995; Sibuet and Olu 1998; Van Dover 2000; Tunnicliffe et al. 2003). To summarize, hydrothermal vents occur mostly on oceanic ridges, where bottom seawater circulates into the newly formed crust. The high geothermic gradient linked with the presence of a magma chamber few kilometres below the ridge causes water to heat and to be enriched in reduced compounds (metals, sulphide…). Heated fluids, displaying lower densities, then reach back the seafloor along cracks and are emitted. Fluid interaction with cold seawater provokes the precipitation of dissolved metals and minerals in the form of complexes with sulphide, yielding typical (and often spectacular) chimneys. At cold seeps, which are mostly located along continental margins, fluids originate from the subsurface. The thermogenic or biogenic reduction of buried organic matter produces methane and other hydrocarbons which seep to the seafloor, and processes such as the anaerobic oxidation of methane coupled with sulphate-reduction can account for local enrichments in sulphide. Co-occurrence of effective electron donors in the form of reduced compounds such as sulphide or methane, and of good acceptors such as oxygen or nitrate, provides large amounts of energy to prokaryotes able to use them, and triggers significant primary production (Jannasch and Mottl 1985). Although free-living chemoautotrophs are abundant, the most remarkable feature of hydrothermal vents and cold seeps is the occurrence of symbiotic associations between chemoautotrophic bacteria and dominant invertebrates. Although no definition of ‘symbiosis’ is universally accepted, the term is used herein to describe a close association between a metazoan host and bacteria allowing the host to gain novel metabolic capabilities (Douglas 1994). Many metazoan taxa, such as siboglinid tubeworms, molluscs and arthropods indeed live in symbiosis with chemoautotrophic bacteria, mostly sulphur-oxidizers (this volume, Dubilier et al. 2008).
Among symbiotic metazoans found at deep-sea hydrothermal vents and cold seeps, members of a clade within the family Mytilidae are the group which has evolved interactions with the most diversified bacteria. Although related to coastal mytilids, they represent a highly specialized group with a restricted habitat range. Deep-sea mytilids occur worldwide at hydrothermal vents and cold seeps at depths from 200 to 3,600 m (Plate 11, Table 6.1). They are also reported from large sunken organic debris such as wood falls and carcasses of large vertebrates, types of habitats which present analogies with vents and seeps, in particular due to the presence of reduced compounds (Distel et al. 2000; Baco and Smith 2003; Pailleret et al. 2007). To date, no such mytilid was reported from the background deep-sea fauna. The most remarkable biological feature distinguishing deep-sea mussels from their coastal relatives is the presence of chemosynthetic bacterial symbionts associated with their gill epithelial cells. Understanding this symbiotic association has stimulated the interest of many investigators since its discovery in the early 1980s. Many species harbour sulphur-oxidizing symbionts comparable to those described from other bivalves families (Lucinidae, Thyasiridae, Solemyidae and Vesicomyidae), but some species harbour methane-oxidizing symbionts, a very rare feature among bivalves. Multiple symbioses involving two to six distinct bacterial types are another originality of these deep-sea mussels, and appear as a good way to deal with highly variable environments they are experiencing (Fisher et al. 1993; Distel et al. 1995).
Although no symbiont has yet been isolated in pure culture, the diversity, role, physiology, ecology and evolution of mussel symbioses have been documented. The development of tools applied to deep-sea biology, such as molecular techniques or experimental devices, and the input from other disciplines such as deep-sea imagery or in situ chemistry has allowed a better understanding of the ecology of chemosynthesis-based ecosystems. The present contribution gives a general overview of the diversity of mussels associated with deep-sea chemosynthesis-based ecosystems and their bacterial symbionts, with an emphasis on recent advances in the understanding of interactions between hosts, bacteria, and their environment. Finally, some new questions and directions for future research are presented.
6.2 Mussel Distribution and Habitats
Mussels have been collected over the years from many deep-sea chemosynthesis-based environments spread over the world including hydrothermal vents, cold seeps and organic falls such as whale bone or wood deposits (Plate 11). Mussels are mostly epibenthic and live attached by byssal threads to hard substrates, sometimes forming tight aggregates (Fig. 6.1, Plate 12). Notable exceptions are Bathymodiolus boomerang from the Barbados accretionary prism and Gigantidas horikoshi, which live two-thirds buried into the sediment (Hashimoto et al. 1995; von Cosel and Olu 1998; Desbruyères et al. 2006). The mussels’ reliance on chemosynthetic symbionts located in their gills implies that reduced compounds are needed in the surrounding fluid, as mussels do not have access to the anoxic sediment. This does not normally occur in the deep-sea despite the anoxy of the sediment because fluxes are not sufficient to emit reduced compounds into the bottom seawater. On the contrary, fluids emitted at cold seeps and hydrothermal vents contain huge amounts of reduced compounds such as sulphide and methane (Van Dover 2000; Le Bris et al. 2006b; Sarrazin et al. 2006). Similar compounds, for example sulphide and methane, are also produced during the microbially-mediated decay of large debris of organic matter such as whale or wood falls (Leschine 1995; Baco and Smith 2003; Goffredi et al. 2008; Treude et al. 2009).
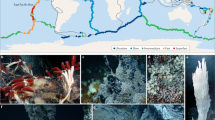
Morphology and habitats of bathymodiolin mussels. (a) Idas sp. Med attached to a carbonate crust recovered from cold seeps in the eastern Mediterranean (MEDECO cruise, 2007, photo by O. Gros). Note the two whitish siphons in the posterior part. Shell length 1 cm. (b) Bathymodiolus azoricus specimens attached to a fluid-emitting edifice at the Rainbow hydrothermal vent site (Mid Atlantic Ridge, 2,277 m depth). Notice the iron deposits on chimneys and shells, and the presence of Rimicaris exoculata shrimps on the right of the image. Image © Ifremer, MOMARDREAM cruise, 2008 (chief scientist: J. Dyment). (c) Dense bed of the mussel Bathymodiolus sp. at a cold seep site situated in a depression on top of a diapir structure in the lower Congo Basin (2,700 m depth). The area displayed gas bubble sources. Other metazoans include tubeworms (Lamellibrachia sp., in the back), shrimps, and limpets (small white dots visible on mussel shells). Image © Marum, University of Bremen, GUINECO M76/3a cruise, 2008 (chief scientist: A. Boetius). (d) Idas sp. Med, opened valves. Note the white foot, brownish visceral mass below and gills in the left and right valves. A colour version of this figure can be found in Appendix (Plate 12)
Around vents, mussels usually occur on chimneys or in the vicinity of cracks where diffuse flow occurs (Fig. 6.1b) (Johnson et al. 1994; Fisher 1995; Cuvelier et al. 2009; Le Bris and Duperron in press). At seeps mussels usually live attached to authigenic carbonates, sometimes covering large areas (Fig. 6.1c), or other hard substrates such as tubes of vestimentiferans (Olu-LeRoy et al. 2007b). Living at the oxic-anoxic interface can be risky. In a documented example, specimens of B. childressi form a ring, a few meters wide, surrounding a 190 m2 methane-rich anoxic hypersaline brine pool in the northern Gulf of Mexico (GoM). However, some mussels located in the inner rim were dead after being entirely submerged by an elevation of the brine level (MacDonald et al. 1990). Similarly, small specimens of B. azoricus reach densities above 10,000 individuals m−2 on the walls of active venting chimneys at Lucky Strike despite the risk of exposure to deadly hot fluids (von Cosel et al. 1999). Specimens of B. brevior were shown to survive in acidic waters with pH values as low as 5.36 at the Eifuku volcano (Mariana Arc), although with shell thickness and daily growth rates only half those measured in mussels occurring in neutral waters (Tunnicliffe et al. 2009). Aside of such occasional extremes, mussels have to tolerate only a relatively moderate temperature range. B. brevior has an upper temperature limit of 35°C and tolerates 2 h exposure at 19°C (Henry et al. 2008). Heat shock experiments for 2 h at 25°C and 30°C were shown to result in global depression in gene expression in B. azoricus, suggesting that mussels are not adapted to ‘hot’ temperatures (Boutet et al. 2009). At vents, mussels are indeed usually out-competed at the hottest habitats by specialists such as the gastropod Ifremeria nautilei at Lau Basin, the annelid polychaete Alvinella pompejana at sites of the East Pacific Rise (EPR), or the shrimp Rimicaris exoculata at sites of the Mid Atlantic Ridge (MAR). They rather occupy more peripheral areas with more diffuse venting and temperatures slightly above those of ambient seawater, up to 15°C (Sarradin et al. 1999). Although in situ measurement of physico-chemical parameters is difficult, and some compounds such as methane can not be monitored directly, several studies have described local conditions around mussels. An important point is that physico-chemical characteristics are highly variable in both space and time, particularly at vents, resulting in varying sharp gradients to which the organisms must adapt (Johnson et al. 1994; Le Bris et al. 2006b). Mussels thrive in environments with moderate concentrations of reduced compounds (compared to end-member fluids), in the range of micromolars for sulphide and methane. For example, sulphide concentrations between 0.5 and 62 μM were reported in the vicinity of mussel beds at the Lucky Strike vent site (<0.5 to 26 μM at Menez Gwen), with larger mussels found at sulphide-rich locations (Sarradin et al. 1999), and up to 325 μM sulphide was recorded at Rose Garden (Galapagos Rift) (Johnson et al. 1986, 1994; Fisher et al. 1988). Methane concentrations up to 33.7 μM were reported from mussel beds at the Régab cold seep (Duperron et al. 2005; Olu-LeRoy et al. 2007b). The presence of additional chemical compounds can influence the habitat. For example, end member fluids at the Rainbow vent site display low sulphide concentrations, and the abundance of dissolved iron (which forms deposits on shells, see Fig. 6.1b) further limits the bioavailability of sulphide for mussels (Le Bris et al. 2006a). At vents, toxic heavy metals can also be abundant, even in diffuse flow, and mussels use a variety of systems to protect themselves (Geret et al. 1998; Rousse et al. 1998). Mussels ‘engineer’ their vent habitat by redirecting fluid flow and dispersing it throughout the mussel bed (Johnson et al. 1994). They also display a limited mobility, and can move a bit to optimize access to their substrates (Childress 1988; Johnson et al. 1994). Compared to vent and seep habitats, precise characteristics of organic falls have rarely been documented, although some data indicate enrichment in sulphide in the sediment close to whale bones, and the occurrence of methane (Smith et al. 1998; Fujiwara et al. 2007; Treude et al. 2009). Recent results confirm the direct emission of sulphide from wood deployed in a shallow mangrove habitat (Laurent et al. 2009). In summary, observations and results indicate that mussels require habitats where oxygen is available as well as electron donors and energy sources for their bacterial symbionts.
6.3 The Diversity of Mussels
6.3.1 General Description
Around 37 mussel species reported from chemosynthesis-based ecosystems have been assigned a name (22 large mussels within the genera Bathymodiolus and Gigantidas, and around 16 small species within the genera Idas and relatives including Adipicola, Benthomodiolus, or Tamu) (Dell 1987; von Cosel 2002; von Cosel and Marshall 2003; von Cosel and Janssen 2008; Pelorce and Poutiers 2009). Many more possible ‘species’ have been identified based on molecular studies (Smith et al. 2004; McKiness and Cavanaugh 2005; McKiness et al. 2005; Jones et al. 2006; Olu-LeRoy et al. 2007a; Samadi et al. 2007; Lorion et al. 2009). They display several common features. Shell is modioliform and usually brownish (Fig. 6.1). Adult shell length varies from ∼10 to 360 mm, larger species (>40 mm) being restricted to hydrothermal vents and cold seeps. The mantle delimiting the pallial cavity displays different degrees of fusion. In some species (such as Idas sp. from the eastern Mediterranean, see Fig. 6.1a), siphons comparable to those of infaunal bivalve species have been described on living specimens despite their epibenthic lifestyle (von Cosel 2002; Okutani et al. 2003; Fujiwara et al. 2007). Compared to their shallow, non-symbiotic relatives, mussels display reduced labial palps and guts, the latter varying among species from curved or S-shaped to straight (Page et al. 1991; von Cosel 2002). Gills are typically enlarged, occupying for example 77% of shell length in B. boomerang, and very often dark coloured, a common trend in symbiotic bivalves (Fisher 1990; von Cosel and Olu 1998). The gills are filibranch. They display inner and outer demibranchs composed of ascending and descending lamellae, with few-to-no inter-lamellar junctions, and ciliary junctions between consecutive filaments (von Cosel et al. 1999). Gill epithelium is constituted of one layer of cells, of which some harbour symbionts (see below and Fig. 6.2) while others are almost symbiont-free and harbour cilia (intercalary cells) (Fiala-Médioni 1984). Gills retains the ability to filter feed at rates comparable to shallow mussels, as shown in B. azoricus and “B.” childressi (the use of quotation marks is explained in Section 6.3.2), and the digestive tract is functional (Page et al. 1991; Pile and Young 1999). To colonize scattered habitats efficiently, mussels have developed specific strategies. Bathymodiolinae are dioecious or successive hermaphrodites, but simultaneous hermaphrodites rarely occurred in the species investigated (Le Pennec and Beninger 2000). For example Idas washingtonia displayed evidence of protandric hermaphroditism (Tyler et al. 2009). Males represented 74% of individuals investigated and included the smallest mussels. Female and hermaphrodites represented 10% and 14% of specimens, respectively. Although not well-documented, the development of mussels appears to involve small and numerous gametes and eggs (Tyler et al. 2007, 2009). The large size of the larval shell indicative of a long larval life, planktotrophic larvae probably have large dispersal abilities (Lutz et al. 1980, 1984; Comtet et al. 2000; Arellano and Young 2009, reviewed in Le Pennec and Beninger 2000 and Tyler and Young 2003). Mussel lifespan is hard to estimate compared to that of many coastal bivalves, because no comparable typical cycles (seasonal, lunar, circadian, tidal) are proven to be linked with shell increments. Rough estimates of an age around 18 years for a 14 cm-long specimen of B. brevior were suggested, and B. thermophilus specimens may get older than 25 years (Schöne and Giere 2005). This is not an unusual lifespan among mytilids, as Modiolus modiolus, a non symbiotic mussel, was reported to live 50 years (von Cosel and Olu 1998).
(a) Frontal view of a single mussel gill. Arrow indicates how transverse sections through filaments (used for electron and fluorescence microscopy) are obtained. (b–c) Detail of two mussel gill epithelial cells as seen in transverse sections. Notice the basal lamina underlying the gill epithelium and separating cells from the hemolymph, and the nucleus and phagolysosome-like bodies (pll) in the basal part of cells. Arrows indicate the direction of water flow. Sulphur-oxidizing bacteria are displayed in dark grey. (b) Extracellular bacteria located between microvilli, or engulfed in a large vacuole, as typically observed in some small bathymodiolines from organic falls; (c) Intracellular bacteria inside of host cells within vacuoles, as observed in most large species from hydrothermal vents and cold seeps
6.3.2 Taxonomy and Classification
The taxonomy of mussels from chemosynthesis-based ecosystems is under discussion. Not many distinctive features allow comparisons among species, and some could be attributed to allometric growth (von Cosel and Olu 1998). Most large species found at hydrothermal vents and cold seeps are classified within the genus Bathymodiolus of which the type species is B. thermophilus from the EPR (Kenk and Wilson 1985). However, detailed morphological investigations indicate that Bathymodiolus can be subdivided in three groups: the “B.” childressi, theB. thermophilus, and the B. aduloides group (von Cosel 2002; von Cosel and Janssen 2008). A second genus of large mussels is Gigantidas, morphologically distinct from Bathymodiolus and described from vents around New Zealand (von Cosel and Marshall 2003). Although known since the end of the nineteenth century from trawls (example in Jeffreys 1876), smaller species (shell lengths <3 cm, Fig. 6.1a) have been less investigated and morphological descriptions are sometimes ancient and rarely backed by molecular data. Members of the genera Idas and Adipicola (classified within the sub-family Modiolinae), or the more recently erected genus Tamu, have been reported from cold seeps as well as organic falls from various locations. Except for their smaller size, their morphology resembles that of large Bathymodiolus. Soft parts have however not been studied in great detail.
Molecular phylogenies based on host nuclear and mitochondrial genes support the monophyly of symbiont-associated mussels within the family Mytilidae. The subfamily name ‘Bathymodiolinae’ has been proposed. The monophyly of Bathymodiolus is not supported. Analyses of COI, ND4 and 28S rRNA-encoding genes distinguish three Bathymodiolus clades, not contradictory to the groups suggested by von Cosel (Miyazaki et al. 2004; Jones et al. 2006; Samadi et al. 2007; Lorion et al. 2009). The three groups emerge from a multifurcation which includes smaller mussels classified as Idas, Tamu or Adipicola. To distinguish between ‘true’ Bathymodiolus from the B. thermophilus group and others, Gustafson suggested the use of quotation marks for representatives of other groups until the taxonomy is fully clarified (Gustafson et al. 1998). Molecular studies also point to the existence of cryptic species (Moraga et al. 1994; Won et al. 2003a; Smith et al. 2004; Olu-LeRoy et al. 2007a). A re-assessment of the nomenclature is thus needed which should combine morphological as well as molecular data, as used for example by Maas and co-workers to distinguish between mid-Atlantic ridge (MAR) vent species (Maas et al. 1999).
6.4 The Diversity of Bacterial Symbionts
Bacteria have been shown to occur in all Bathymodiolinae species investigated to date. As for other chemosynthetic symbioses, no symbiont has been isolated in pure culture despite attempts (Nelson et al. 1995). Bacteria are associated with gill epithelial cells located in the lateral zone of gill filaments (Fig. 6.2). This allows bacteria to gain access to compounds present in circulating fluid, including energy and carbon sources. Comparable gill symbioses, though involving only sulphur-oxidizers (thiotrophs), have also been reported from the bivalve families Thyasiridae, Lucinidae, Solemyidae and Vesicomyidae (Fisher 1990; Stewart et al. 2005; Stewart and Cavanaugh 2006; Taylor and Glover 2006).
6.4.1 Sulphur-Oxidizing Bacteria
Of the 27 mussel ‘species’ for which molecular information about symbiosis is available, 24 harbour bacteria related to chemoautotrophic sulphur-oxidizing (or thiotrophic) Gammaproteobacteria, based on comparative analysis of their16S rRNA-encoding gene (Table 6.1). Initially discovered in the EPR vent species B. thermophilus, sulphur oxidizing symbionts are small (0.5 μm diameter on electron micrographs) Gram negative bacteria (Rau and Hedges 1979; Felbeck et al. 1981; Cavanaugh 1983; Fiala-Médioni 1984). They fix carbon autotrophically using the Calvin Benson Bassham (CBB) cycle as demonstrated by the presence of the functional form I of Ribulose 1,5 bisphosphate carboxylase/oxygenase (RubisCO) and its encoding gene in several species (Fisher et al. 1988; Robinson et al. 1998a; Pimenov et al. 2002; Spiridonova et al. 2006). Measurements of enzyme activities and gene sequencing also demonstrated the presence of adenosine phosphosulfate (APS) reductase and ATP sulfurylase, two enzymes involved in sulphur metabolism (Nelson and Fisher 1995). Thiosulphate and sulphide stimulate the fixation of inorganic carbon based on incubation experiments of animal tissue or living specimens of B. thermophilus and B. azoricus (Belkin et al. 1986; Nelson et al. 1995; Riou et al. 2008). The use of alternate energy sources, such as hydrogen, has been suspected but has not been proven to date. Contrary to what is observed for example in lucinid clams, symbionts do not deposit sulphur granules (Dando et al. 1986).
In most species, thiotrophic symbionts occur within vacuoles containing one to several bacteria each that are located inside of host gill epithelial cells named ‘bacteriocytes’ (Fig. 6.2) (Fiala-Médioni 1984; Dubilier et al. 1998). Recently however, thiotrophs were shown to occur extracellularly between microvilli of epithelial cells in several small species such as MOTU 16, A. longissima, Idas sp. C and Idas sp. D from organic falls of the western Pacific (Fig. 6.2) (Gros and Gaill 2007; Gros et al. 2007; Duperron et al. 2008a; Lorion et al. 2009). Idas simpsoni, I. washingtoniana and Adipicola sp. JDF (for Juan de Fuca ridge) also display extracellular bacteria as evident from microscopic investigation (Southward 2008). In many species, symbiont localization was not investigated, or ambiguous such as in Bathymodiolus sp. from the Juan de Fuca ridge (McKiness et al. 2005; Duperron et al. 2008b; Won et al. 2008). Either intra- or extracellular, symbiont localization appears to be consistent and conserved within a given host species (Lorion et al. 2009).
All sulphur-oxidizing symbionts of mussels cluster within a clade of Gammaproteobacteria that also includes sequences from a few free-living uncultivated bacteria, and a clade of maternally-inherited endosymbionts associated with vesicomyid clams (Distel et al. 1988; Peek et al. 1998; Duperron et al. 2008a). Grouping free-living bacteria as well as extracellular, intracellular, and maternally-inherited (with reduced genome) symbionts, this clade is a good target group to study the processes leading from free-living to strictly endosymbiotic bacteria. One 16S rRNA symbiont phylotype (a phylotype is a version of the sequence) is usually associated specifically with a single mussel species, although exceptions occur. The two MAR vent species B. azoricus and B. puteoserpentis for example share a single 16S rRNA symbiont phylotype (Duperron et al. 2006), and the co-occurrence of two distinct thiotroph-related 16S rRNA phylotypes representing two distinct symbiont strains was reported in B. heckerae and in Idas sp. Med (Duperron et al. 2007, 2008b).
6.4.2 Methane-Oxidizing Bacteria
To date, at least ten species from cold seeps and hydrothermal vents are reported to harbour bacteria related to Type I methane-oxidizing (or methanotrophic) Gammaproteobacteria, of which three have only methanotrophs, five also have sulphur-oxidizers, and two – Bathymodiolus heckerae and Idas sp. Med – have four and six symbiont types in total, respectively (Table 6.1). Methanotrophic symbiosis is rare among metazoans, and was suggested only for some sponges (Vacelet et al. 1996), a few siboglinid annelids (Schmaljohann et al. 1990), the gastropod Ifremeria nautilei (Borowski et al. 2002), the thyasirid bivalve Conchocele bisecta (Kamenev et al. 2001), and epibionts of the shrimp Rimicaris exoculata (Zbinden et al. 2008). Methanotrophic symbionts were initially discovered in mussels from the Gulf of Mexico cold seeps. B. childressi grows using methane as a carbon and energy source (Childress et al. 1986; Cary et al. 1988). Bathymodiolus heckerae harbours large bacteria with a diameter of 1.5 μm and stacked internal membranes typical of free-living Type I methanotrophs. This species also displays unexpectedly negative δ13C tissue values, very close to those reported for local methane (Cavanaugh et al. 1987). Methanol dehydrogenase activity, the second enzyme of the aerobic oxidation of methane, was shown (Fisher et al. 1987; Robinson et al. 1998b; Pimenov et al. 2002). The presence of methane mono-oxygenase, the diagnostic enzyme for aerobic methane oxidation, was confirmed based on gene sequence analysis as well as in situ hybdridization of its mRNA. However, its activity is difficult to measure because of its unstability (Pernthaler and Amann 2004; Spiridonova et al. 2006; Duperron et al. 2007). Besides methane utilization, RubisCO appears to be present in methane-oxidizing symbionts of a Bathymodiolus from Okinawa Through, indicating the possible presence of a functional CBB cycle (Elsaied et al. 2006). When present, methanotrophs are always intracellular, located within vacuoles inside bacteriocytes.
Symbiotic methanotrophs form a clade within the Gammaproteobacteria which is the sister-group of free-living Type I methanotrophs (Distel and Cavanaugh 1994; Duperron et al. 2005). To date, very few free-living bacteria cluster within this clade questioning the existence of active free-living forms of symbionts. Similar to thiotrophs, one 16S rRNA symbiont phylotype is usually associated with a single mussel species. Exceptions again are B. azoricus and B. puteoserpentis that share a single methanotroph-related phylotype (Duperron et al. 2006).
6.4.3 Additional Symbionts and Multiple Symbiosis
Besides the presence of methanotrophic symbionts, the main originality of bathymodiolines is the occurrence of multiple symbioses. ‘Dual symbiosis’ involving sulphur- and methane-oxidizers co-occurring within a single bacteriocyte was first suspected in B. heckerae (Cavanaugh et al. 1987), hypothesized in B. brooksi from the Gulf of Mexico based on microscopy (Fisher et al. 1993), and demonstrated formally using 16S rRNA gene sequencing and fluorescence in situ hybridization (FISH) in the vent species B. puteoserpentis (Distel et al. 1995). Dual symbiosis has subsequently been documented in B. azoricus, B. aff. azoricus from Lost City, B. boomerang from the Barbados accretionary prism, B. cf. boomerang from Régab (Plate 13a–b) and re-investigated in B. brooksi (Olu et al. 1996; Fiala-Médioni et al. 2002; Duperron et al. 2005, 2007; DeChaine et al. 2006).
Recently, thanks to the sequencing of larger bacterial 16S rRNA clone libraries from gill tissues, new types of bacterial symbionts have been identified in the gills of two cold seep species, B. heckerae and Idas sp. Med, and confirmed using FISH (Duperron et al. 2007, 2008b). Both mussel species harbour one methanotroph- and two distinct thiotroph-related 16S rRNA symbiont phylotypes, labelled M, S1 and S2 in Table 6.1, respectively (Plate 13d). Both mussels also harbour phylotypes related to free-living methylotrophs of the genus Methylophaga (Plate 13c). Although the metabolism of these symbionts is unknown, it can be hypothesized that they are actually methylotrophs, i.e. bacteria utilizing mono carbon compounds such as methanol, but not methane. Use of methane is restricted to the subgroup of methanotrophs (Anthony 1982). Finally, Idas sp. has two additional symbionts compared with B. heckerae. One is a Gammaproteobacterium unrelated to any cultivated bacterium, making it impossible to propose any metabolic scheme. The other is a member of the Bacteroidetes group, related to members of the genus Tenacibaculum and cellulose-degrading bacteria such as Polaribacter dokdonensis. Its role is unknown, and could be linked either to the degradation of organic compounds or pathogenicity. Intra- or extracellular localization of these recently discovered symbionts has not yet been ascertained.
Bacteria suspected to be deleterious have also been identified in mussel gill tissues. “Candidatus Endonucleobacter bathymodioli”, a Gammaproteobacterium related to sequences from a bacterium causing mass mortalities in Pacific razor clams, has recently been identified within nuclei of gill intercalary cells in several species of Bathymodiolus from various geographical origins (Zielinski et al. 2009). This filament-shaped bacterium apparently establishes itself within host cells nuclei, multiplies and destroys the content of the nucleus. It then causes the nucleus to break open, releasing offspring bacteria to the environment, similar to the way that is used by some viruses. “Ca. E. bathymodioli” is apparently closely related to several bacterial species based on sequences recovered from marine animal samples, including two which also have been hypothesized to originate from endonuclear bacteria (Kerk et al. 1992). This led Zielinski et al. (2009) to propose that endonuclear bacteria could be widespread among marine invertebrates.
Clearly, recent discoveries of new bacterial partners and of the occurrence of up to six symbionts within a single mytilid host indicate that the diversity of bacteria associated with mussel tissues has been underestimated. Cheaper high-throughput sequencing technologies should allow in the future to gain a more exhaustive and systematic overview of bacteria associated with bathymodioline mussels.
6.5 The Symbiotic System: How Does It Work?
Mussels and their sometimes diverse associated bacteria form complex ‘symbiotic systems’ in which each partner interacts with others and with the environment. Although the nature of these interactions is rarely well-understood, the following chapter aims at summarizing current knowledge concerning aspects of these interactions.
6.5.1 Interactions Between Mussels and Their Bacterial Symbionts
How bacteria are acquired by host tissue has not been fully resolved. Although early developmental stages of dual symbiotic species display sulphur- and methane-oxidizing bacteria (Salerno et al. 2005), these were not observed from gonad tissue or eggs, suggesting an early environmental acquisition. The extracellular localization of thiotrophs in several small mussel species, and the observations of open vacuoles engulfing sulphide-oxidizing bacteria in the most apical part of bacteriocytes in many species with intracellular symbionts support this hypothesis (Dubilier et al. 1998; Duperron et al. 2008a; Southward 2008; Lorion et al. 2009). Detailed genetic investigations have shown the co-occurrence of several 16S rRNA or ITS (Internal Transcribed Spacer, a more variable marker gene allowing to distinguish between closely related bacterial strains) phylotypes of sulphide-oxidizers in the gills of mussels from the Mid-Atlantic Ridge, Gulf of Mexico, eastern Mediterranean and western Pacific (Won et al. 2003b; Duperron et al. 2007, 2008b; Lorion et al. 2009). Because some of these strains were usually found in mussel species occurring nearby, these observations are interpreted as further support for environmental acquisition. The transmission of methanotrophs and other types of symbionts has not been studied in detail. To date only one 16S rRNA methanotroph-related phylotype was reported from a given mussel species. Genomic investigations will help documenting whether bacteria can live outside of their hosts, and how hosts and bacteria recognize each other and establish the association.
Contrary to tubeworms such as Riftia pachyptila, or clams such as Calyptogena spp., no specific H2S transporter has been identified in mussels. In tubeworms, bacteria are located inside a special organ, the trophosome, remote from the gill where H2S is taken up. In Calyptogena (vesicomyid) clams the foot mines for sulphide in the sediment or emitted waters whereas bacteria sit in the gills (Childress et al. 1993; Scott and Fisher 1995; Flores et al. 2005). Both are highly-integrated symbioses in which reduced sulphur needs to be transported to the symbionts. In mussels, symbionts occurring in (or on) gill epithelial cells are very close to the circulating fluid that contains both their electron donors, including sulphide, and acceptors (Fig. 6.2). Transporters are therefore not needed.
The nutritional role of symbionts has been well-documented. Stable isotope analyses are a classical tool used to trace carbon sources in ecology (Lajhta and Michener 1994). Tissue carbon isotope signatures can be compared with signatures of carbon sources to estimate their quantitative importance in the nutrition of animals. In hydrothermal vents and cold seeps, the role of sulphur- or methane-oxidizing symbionts in mussel nutrition can be, to a certain extent, evaluated using this approach (Fisher 1995). Along with direct incubations of gill material or whole living mussels in the presence of radio-labelled inorganic carbon and methane, these methods have demonstrated the transfer of bacterially-acquired carbon to the host tissue from both sulphur- and methane-oxidizing symbionts (Table 6.1) (Childress et al. 1986; Fisher et al. 1987; Fisher and Childress 1992; Kochevar et al. 1992; Jahnke et al. 1995; Nelson et al. 1995; Riou et al. 2008). Estimates for B. heckerae mussels from Blake Ridge show 60% of their organic carbon from methane- and 40% from sulphur-oxidizers based on stable isotopes investigation (Van Dover et al. 2003). How carbon transfer is achieved is however still debated. Early work has shown the digestion of bacteria within phagolysosome-like bodies located in the basal part of bacteriocytes (Fig. 6.2) (Fisher et al. 1987; Fiala-Médioni et al. 1994). No such phagolysosome-like bodies were convincingly observed in extracellular sulphur-oxidizers, questioning their role as carbon providers (Duperron et al. 2008a). Other ways of transfer are known from other symbioses, for example the release of organic compounds by photosynthetic symbionts in corals. Another possibility was suggested following the observation of bacteria-filled bacteriocytes, detached from the gill, in the food groove. Mussels could possibly perform a type of autophagy. Direct evidence is lacking, but these alternate pathways can not be ruled out and could play a role. Besides their role in carbon nutrition, thiotrophs were also suggested to detoxify sulphide, which is toxic for host cytochrome oxidase, protecting mussels against occasional bursts of sulphide-rich fluids (Powell and Somero 1986; Dubilier et al. 1998; Duperron et al. 2005). Methanotrophs were suggested to contribute nitrogen uptake (Lee and Childress 1994; Lee et al. 1999; Dattagupta et al. 2004).
6.5.2 Interactions Among Bacteria
Dividing stages of sulphur- and methane-oxidizers have been observed in many mussel species, confirming that symbionts benefit from their cellular environment (examples in Fiala-Médioni et al. 1986; Duperron et al. 2005). In the limited volume available for growth within bacteriocytes, competition is likely. In multiple symbioses, direct competition between symbionts with distinct metabolisms is possibly prevented by their use of distinct carbon and energy sources. Relative densities appear to depend on environmental parameters that favour either metabolism (see below). Competition between two symbionts with the same presumed metabolism was suggested based on the observation of mutual exclusion between the two thiotrophs S1 and S2 from B. heckerae. They very rarely co-occur within a single bacteriocyte, but instead tend to occupy distinct areas of the gill filaments (Plate 13d) (Duperron et al. 2007). A similar co-occurrence of two thiotrophs has been reported in Idas sp. Preliminary observations indicate that one phylotype occurs mostly in the periphery of filaments while the other is spread over the whole lateral zone, but mutual exclusion has not been observed (Duperron et al. 2008b; Halary et al. personal observation). In both cases, it is not clear if the presence of two thiotrophs leads to true functional redundancy, or whether they display slight differences in their metabolisms.
Besides competition, syntrophic relationships have been suggested between methanotrophs and methylotroph-related bacteria in B. heckerae (Duperron et al. 2007). Methylotrophs were suggested to use methanol, a C1 intermediate compound in the methane oxidation pathway used by methanotrophs. This hypothesis was based on the close proximity between the two symbiont types observed in hybridized sections (Plate 13c). Syntrophy among symbionts was previously hypothesized between the sulphur-oxidizing and sulphate-reducing bacteria associated with gutless oligochetes (Dubilier et al. 2005; Woyke et al. 2006).
The nature of inter-bacteria interactions is very hard to study. Furthermore, the host likely controls what occurs within bacteriocytes, maybe not only through digestion. In vesicomyid clams for example, recent investigation of the genome content of the thiotrophic symbiont Vesicomyosocius okutanii indicated the absence of ftsZ, an important gene involved in bacterial division. Authors suggested a close control of bacterial division directly by the host (Kuwahara et al. 2007).
6.5.3 Interaction Between the Symbiotic System and Its Environment
Generally, even mussels associated with symbionts can filter-feed. Filter feeding can significantly contribute to mussel nutrition, as suggested by stable isotope-based evidence from “B.” childressi (Page et al. 1991; Pile and Young 1999). Recently, modelling approaches have indicated a shift from reliance on photosynthesis-derived carbon towards total reliance on bacterial-derived carbon during the growth of B. azoricus (Martins et al. 2008). However, transplant experiments in which B. thermophilus specimens were transferred from their vent habitat to bare rock or sediment devoid of fluid emissions have shown that mussels were quickly losing their symbionts and dying (Raulfs et al. 2004). This fits with the observation that specimens from a waning vent site were in bad condition, displaying low condition indexes and thin bacteriocytes with almost no symbionts. Altogether, this makes clear that organic matter available in the deep-sea is not sufficient to sustain symbiotic mussels without the help of their symbionts (Raulfs et al. 2004).
Deep-sea mussels can not survive in the absence of reduced compounds. However, detailed studies of the interaction between mussel symbioses and environmental parameters are still in their infancy due to our lack of precise knowledge about the physico-chemical environment of mussels and to the difficulties of experimentation on deep-sea animals. Deep-sea environments, in particular vent habitats, are highly variable in space and time even at the scale of centimeters. These steep gradients are caused by multiple distinct sources of fluids, biological activity, convective processes and fluid circulation, currents, and so on (see section 6.2). Determination of local physico-chemical parameters in the deep-sea requires the use of adapted in situ sensors, if they exist, and necessitates time-series measurements that are expensive to perform. Despite these limits, transplant experiments in which specimens of “B.” childressi were transferred from a low-methane to a high-methane site (and vice versa) have clearly demonstrated increased mussel growth caused by increased abundance of methane, confirming the direct link between environmental parameters and mussels health (Nix et al. 1995; Bergquist et al. 2004). Experimentation is another difficulty. Certain mussel species from shallower sites, such as B. azoricus and “B”. childressi, can be kept alive in aquaria at ambient pressure (Kadar et al. 2005; Riou et al. 2008). For deeper-dwelling species, pressurized aquaria in which specific compounds can be injected and monitored, such as the IPOCAMP™ device, are becoming available (Shillito et al. 2001). However, without detailed knowledge of habitat characteristics, it is not possible to properly simulate a realistic deep-sea vent or seep environment in an experimental setting. Even though observed trends can usually be extrapolated to the ‘real’ environment, exact physiological rates must therefore be taken with caution (Cary et al. 1988; Kochevar et al. 1992).
Mussels from different sites can display markedly different carbon stable isotope signatures that indicate different relative contributions of sulphur- and methane-oxidizers, even within a single species. (Trask and Van Dover 1999). This was shown for mussels from the Mid-Atlantic ridge associated with both sulphur- and methane-oxidizing symbionts, where isotope signatures were correlated with sulphide concentration in the end-member fluid (Colaco et al. 2002). To test whether these differences in the nutritional role of symbionts arise from differences in bacterial abundances, accurate quantification methods are required. However quantifying bacterial densities is not straightforward. Using bacteria-specific indicators such as lipids or quinones, densities of 1.7 × 1010 to 2 × 1011 bacteria per gram of gill tissue were estimated (Powell and Somero 1986; Yamamoto et al. 2002). Methanotrophic symbionts of “B.” childressi were estimated at 5.6 × 109 bacteria per gram tissue (Kochevar et al. 1992). Quantifying variations of symbiont densities is also difficult. Several methods have been employed, including direct counts on TEM pictures (benefiting from the distinct morphologies of sulphur- and methane-oxidizers), quantification of 16S rRNA, and quantification of volumes occupied by distinct symbionts in bacteriocyte sections (Fiala-Médioni 1984; Trask and Van Dover 1999; Fiala-Médioni et al. 2002; Duperron et al. 2007; Halary et al. 2008; Riou et al. 2008). Using such methods on mussels collected from several MAR hydrothermal vent sites, the authors demonstrated that symbiont populations were flexible. Relative abundances of methane- and sulphur-oxidizers were correlated with the composition of the end-member vent fluids, with the former becoming predominant at methane-rich sites and vice versa. Symbiont densities in B. azoricus specimens that were experimentally exposed to either sulphide or methane responded within hours to changes in their environment, with an increased abundance of sulphur- or methane-oxidizers, respectively (Kadar et al. 2005; Halary et al. 2008; Riou et al. 2008). Dynamic multiple symbioses thus appear to allow mussels to quickly optimize their access to available resources in variable environments, and cope with more diverse types of habitats, as suggested shortly after their discovery (Distel et al. 1995). Recently, we suggested that the specific bacterial densities observed in specimens of a single mussel species found on wood or whale falls are a consequence of sulphide availability (Lorion et al. 2009). Tools are becoming available to study the interaction between mussels, symbiont populations, and the environment in detail. However, more interdisciplinary studies coupling biological, chemical and modelling approaches are required (Martins et al. 2008).
6.6 The Symbiotic System: How Did It Evolve?
6.6.1 The Evolution of Mussels
Mussels occurring at chemosynthesis-based ecosystems and organic falls group in a single clade within the Mytilidae. The question of how the group evolved, what its common ancestor looked like, what the environment it lived in was or whether symbiotic bacteria were present, is hard to answer. Bathymodiolines are known from fossil seeps since the late Eocene, but the occurrence of symbionts is not possible to establish in the fossil record (Kiel and Goedert 2006). Molecular studies suggest an age between 23.7 and 74.3 MY for the origin of the group (Little and Vrijenhoek 2003; Jones et al. 2006). Early work suggested the evolution of vent mussel species from seep ancestors (Craddock et al. 1995). The later addition of small species from organic falls in phylogenetic reconstructions lead Distel et al. to propose the hypothesis that vent and seep mussels derived from ancestors associated with wood and whale fall ecosystems (Distel et al. 2000). However, results from recent bathymodioline phylogenies draw a more complex picture. Some species associated with organic falls such as Benthomodiolus lignicola or Idas sp. Sal 3 appear to be basal to seep and vent species (Samadi et al. 2007) while at least one vent species, “Bathymodiolus” NZ3, possibly a relict from an early vent colonization event, is also quite basal (Jones et al. 2006). Apart from these deep-branching species, the bathymodioline tree today is a large multifurcation which includes at least three vent/seep clades (named the B. thermophilus, “B.” childressi and “B.” aduloides clades) as well as numerous small species from whale and wood falls (Iwasaki et al. 2006; Jones et al. 2006; Lorion et al. 2009). Authors identified a recurrent trend from shallow (<1,000 m) to deep habitats in the evolution of the three groups of Bathymodiolus-like mussels. This would advocate multiple events of seep and vent colonization from a pool of small species associated with organic falls (Fig. 6.3, Plate 12). Habitat shifts during the evolution of mussels are plausible. Indeed, although most species today are specialists of a single type of environment, several ubiquist species were recently identified using reliable genetic approaches. B. platifrons and B. japonicus occur both at seeps and vents around Japan, and Adipicola crypta and Idas sp. C were both associated with wood- and whale-falls in the western Pacific (Miyazaki et al. 2004; Lorion et al. 2009).
The ‘stepping stone’ hypothesis. Ancestors of bathymodiolines were shallow species (on the right), which might have acquired the ability to associate with bacteria (1), most likely sulphur oxidizers. Mussels able to derive nutrition from both filter-feeding and bacterial chemoautotrophs colonized organic habitats such as wood and whale falls occurring at various depths (2). From such habitats, mussels then colonized hydrothermal vents at ridges and cold seeps on margins (3), giving rise to large Bathymodiolus-like species. This event probably occurred several times during the evolution of the group. Shifts between vent and seep probably occur, as evidenced by the existence of species colonizing both habitats (dotted arrow). A color version of this figure can be found in Appendix (Plate 12)
6.6.2 The Evolution of Mussel Symbiosis
Bacterial symbionts do not co-speciate with their mussel hosts (Won et al. 2008). However, very closely related mussel species, for example B. azoricus and B. puteoserpentis from the MAR, B. boomerang and B. aff. boomerang from the Gulf of Mexico (GoM) and Gulf of Guinea (GoG), or Idas sp. C and Idas sp. D from the Vanuatu and Philippine waters, harbour very closely related bacteria (Duperron et al. 2006, 2007; Lorion et al. 2009). Overall, symbiont phylogenies are not well resolved and sometimes ambiguous due to the low variation levels observed in 16S rRNA gene sequences (Duperron et al. 2005, 2008b; DeChaine et al. 2006; Won et al. 2008). Clearly, the understanding of symbiont biogeography and evolution would greatly benefit from the use of multi-marker approaches besides 16S rRNA. Reconstructions indicate that the occurrence of thiotrophic symbionts in the latest common ancestor of bathymodiolines is likely (Won et al. 2008; Duperron et al. 2009). Symbiont localization in the ancestor is however not clear. Thiotrophs are consistently intra- or extracellular depending on the species and mostly intracellular in seep and vent mussels and often extracellular in small Idas-like species from organic falls. However, symbiont localization is not documented or ambiguous for the deepest-branching species such as Bathymodiolus from the Juan de Fuca Ridge, Benthomodiolus lignicola or “Bathymodiolus” NZ3, thus not allowing a prediction (McKiness et al. 2005; Lorion et al. 2009). Whatever the localization, the acquisition of a flexible sulphur-oxidizing symbiosis in the common ancestor of bathymodiolines must have constituted an advantage for the mussels. Such an adaptation opened new potential ecological niches, and could explain the subsequent specialization of the group towards reduced ecosystems. It probably triggered a rapid diversification of the group which translated into the large multifurcation observed in host phylogenetic trees. Based on our current knowledge, methanotrophic symbiosis seems to appear several times in host trees, and could be more opportunistic from the animal’s point of view. It apparently occurred only at methane-rich sites. However, host – symbiont specificity appears to be high as evidenced by the consistent occurrence of two distinct methanotrophs in the gills of B. brooksi and B. childressi, two mussels which co-occur within the same mussel beds at Alaminos Canyon (Fujiwara et al. 2000; Duperron et al. 2007). Other symbioses involving methylotrophs, Bacteroidetes or other Gammaproteobacteria have been discovered recently and it is too early to propose any hypothesis. Nevertheless, it is intriguing that dual and multiple symbioses are to date reported only from the Atlantic area, including MAR, the GoM, GoG and Mediterranean (Table 6.1). Noteworthy, at least three mussel species from this area, two Bathymodiolus and one Idas, display amphi-Atlantic distributions (Olu-LeRoy et al. 2007a; Duperron et al. 2008b; Génio et al. 2008). No mussel with multiple symbionts is reported to date from the Pacific, despite 16 mussel ‘species’ investigated in the area. Clearly, although hypotheses can be proposed based on current knowledge, much information is needed to clarify our understanding of symbiosis evolution in mytilids.
6.7 Conclusion
Mytilids associated with deep-sea chemosynthesis-based ecosystems provide fascinating examples of symbiosis. How the diversity of symbionts is accommodated within a single bacteriocyte in some species is not understood, but the worldwide occurrence of mytilids at seeps, vents and organic falls and the diversity of their associated bacteria illustrate the success of this group. The ability to associate with metabolically diverse bacteria and to adapt symbiont densities to environmental parameters is probably a key to this success, allowing mytilids to cope with diverse and variable habitats.
Many questions remain. Our inability to cultivate symbionts, and difficulties of both in situ and ex situ experimentation on living mussels are major issues hampering research on these ‘symbiotic systems’. The existence of free-living forms of symbionts remains to be proven, and how associations establish is not known yet. The metabolic potential and diversity of symbionts are probably underestimated. Genomic approaches should provide indications, yield testable hypotheses, and allow comparisons between mytilid symbioses and other well-documented associations for which genomic information is available, be they chemosynthetic (Riftia, Calyptogena…) or not (insects).
Finally, geographical trends observed and evolutionary hypotheses proposed to date result from the study of a limited number of species from regions where oceanographic research has focused over the last decades (GoM, GoG, MAR…), leaving many blank spots on the map (Indian Ocean, southern Atlantic, Antarctic…). There is no doubt that new species discovered in newly explored areas will in the future considerably change our views, and yield a more complete picture of the biogeography and evolution of symbiosis in deep-sea mytilids.
References
Anthony C (1982) The biochemistry of methylotrophs. Academic, London
Arellano SM, Young CM (2009) Spawning development and the duration of larval life in a deep-sea cold-seep mussel. Biol Bull 216:149–162
Baco AR, Smith CR (2003) High species richness in deep-sea-chemoautotrophic whale skeleton communities. Mar Ecol Prog Ser 260:109–114
Barry JP, Buck KR, Kochevar RK, Nelson DC, Fujiwara Y Goffredi, SK HJ (2002) Methane-based symbiosis in a mussel Bathymodiolus platifrons from cold seeps in Sagami Bay, Japan. Invertebr Biol 121:47–54
Belkin S, Nelson DC, Jannash HW (1986) Symbiotic assimilation of CO2 in two hydrothermal vent animals the mussel Bathymodiolus thermophilus and the tube worm Riftia pachyptila. Biol Bull 170:110–121
Bergquist DC, Fleckenstein C, Szalai EB, Knisel J, Fisher CR (2004) Environment drives physiological variability in the cold seep mussel Bathymodiolus childressi. Limnol Oceanogr 49:706–715
Borowski C, Giere O, Krieger J, Amann R, Dubilier N (2002) New aspects of the symbiosis in the provannid snail Ifremeria nautilei from the North Fiji Nack Arc Basin. Cah Biol Mar 43:321–324
Boutet I, Tanguy A, Le Guen D, Piccino P, Hourdez S, Legendre P, Jollivet D (2009) Global depression in gene expression as a response to rapid thermal changes in vent mussels. Proc R Soc Lond B 276:3071–3079
Cary SC, Fisher CR, Felbeck H (1988) Mussel growth supported by methane as sole carbon and energy source. Science 240:78–80
Cavanaugh CM (1983) Symbiotic chemoautotrophic bacteria in marine invertebrates from sulphide-rich habitats. Nature 302:58–61
Cavanaugh CM, Levering PR, Maki JS, Mitchell R, Lidstrom ME (1987) Symbiosis of methylotrophic bacteria and deep-sea mussels. Nature 325:346–347
Childress JJ (1988) Biology and chemistry of a deep-sea hydrothermal vent on the Galapagos Rift; the Rose Garden in 1985 an introduction. Deep Sea Res 35:1677–1680
Childress JJ, Fisher CR, Brooks JM, Kennicutt MC II, Bidigare R, Anderson AE (1986) A methanotrophic marine molluscan (Bivalvia Mytilidae) symbiosis: mussels fueled by gas. Science 233:1306–1308
Childress JJ, Fisher CR, Favuzzi JA, Arp AJ, Oros DR (1993) The role of a zinc-based serum-borne sulphide-binding component in the uptake and transport of dissolved sulphide by the chemoautotrophic symbiont containing clam Calyptogena elongata. J Exp Biol 179:131–158
Colaco A, Dehairs F, Desbruyères D, Le Bris N, Sarradin PM (2002) d13C signature of hydrothermal mussels is related with the end-member fluid concentration of H2S and CH4 at the Mid-Atlantic Ridge hydrothermal vent fields. Cah Biol Mar 43:259–262
Comtet T, Jollivet D, Khripounoff A, Segonzac M (2000) Molecular and morphological identification of settlement-stage vent mussel larvae Bathymodiolus azoricus (Bivalvia Mytilidae) preserved in situ at active vent fields on the Mid-Atlantic Ridge. Limnol Oceanogr 45:1655–1661
Craddock C, Hoeh WR, Gustafson RG, Lutz RA, Hashimoto J, Vrijenhoek RJ (1995) Evolutionary relationships among deep-sea mytilids (Bivalvia: Mytilidae) from hydrothermal vents and cold water methane/sulfide seeps. Mar Biol 121:477–485
Cuvelier D, Sarrazin J, Colaço A, Copley J, Desbruyères D, Glover AG, Tyler P, Santos RS (2009) Distribution and spatial variation of hydrothermal faunal assemblages at Lucky Strike (Mid-Atlantic Ridge) revealed by high-resolution video image analysis. Deep Sea Res I 56:2026–2040
Dando PR, Southward AJ, Southward EC (1986) Chemoautotrophic symbionts in the gills of the bivalve mollusc Lucinoma borealis and the sediment chemistry of its habitat. Proc R Soc Lond B 227:227–247
Dattagupta S, Bergquist DC, Szalai EB, Macko SA, Fisher CR (2004) Tissue carbon nitrogen and sulfur stable isotope turnover in transplanted Bathymodiolus childressii mussels: relation to growth and physiological condition. Limnol Oceanogr 49:1144–1151
DeChaine EG, Bates AE, Shank T, Cavanaugh CM (2006) Off-axis symbiosis found: characterization and biogeography of bacterial symbionts of Bathymodiolus mussels from Lost City hydrothermal fields. Environ Microbiol 8:1902–1912
Dell RK (1987) Mollusca of the family Mytilidae (Bivalvia) associated with organic remains from deep water off New Zealand with revisions of the genera Adipicola Dautzenberg 1927 and Idasola Iredale 1915. Natl Mus N Z Rec 3:17–36
Desbruyères D, Segonzac M, Bright M (2006) Handbook of deep-sea hydrothermal vent fauna, Denisia 18. Linz Museum, Austria, p 544
Distel DL, Cavanaugh CM (1994) Independent phylogenetic origins of methanotrophic and chemoautotrophic bacterial endosymbioses in marine bivalves. J Bacteriol 176:1932–1938
Distel D, Lane D, Olsen G, Giovannoni S, Pace B, Pace NR, Stahl DA, Felbeck H (1988) Sulfur-oxidizing bacterial endosymbionts - Analysis of phylogeny and specificity by 16S ribosomal RNA sequences. J Bacteriol 170:2506–2510
Distel DL, Lee HKW, Cavanaugh CM (1995) Intracellular coexistence of methano- and thioautotrophic bacteria in a hydrothermal vent mussel. Proc Natl Acad Sci USA 92:9598–9602
Distel D, Baco A, Chuang E, Morrill W, Cavanaugh C, Smith C (2000) Do mussels take wooden steps to deep-sea vents? Nature 403:725–726
Douglas AE (1994) Symbiotic interactions. Oxford University Press, Oxford
Dubilier N, Windoffer R, Giere O (1998) Ultrastructure and stable carbon isotope composition of the hydrothermal vent mussel Bathymodiolus brevior and B. sp affinis brevior from the North Fiji Basin western Pacific. Mar Ecol Prog Ser 165:187–193
Dubilier N, Blazejak A, Ruehland C (2005) Symbioses between bacteria and gutless marine oligochaetes. In: Overmann J (ed) Molecular basis of symbiosis. Springer, Berlin, pp 251–275
Dubilier N, Bergin C, Lott C (2008) Symbiotic diversity in marine animals: the art of harnessing chemosynthesis. Nat Rev Microbiol 6:725–740
Duperron S, Nadalig T, Caprais JC, Sibuet M, Fiala-Médioni A, Amann R, Dubilier N (2005) Dual symbiosis in a Bathymodiolus mussel from a methane seep on the Gabon continental margin (South East Atlantic): 16S rRNA phylogeny and distribution of the symbionts in the gills. Appl Environ Microbiol 71:1694–1700
Duperron S, Bergin C, Zielinski F, McKiness ZP, DeChaine EG, Cavanaugh CM, Dubilier N (2006) A dual symbiosis shared by two mussel species Bathymodiolus azoricus and B. puteoserpentis (Bivalvia: Mytilidae) from hydrothermal vents along the northern Mid-Atlantic Ridge. Environ Microbiol 8:1441–1447
Duperron S, Sibuet M, MacGregor BJ, Kuypers MM, Fisher CR, Dubilier N (2007) Diversity, relative abundance and metabolic potential of bacterial endosymbionts in three Bathymodiolus mussels (Bivalvia: Mytilidae) from cold seeps in the Gulf of Mexico. Environ Microbiol 9:1423–1438
Duperron S, Laurent MCZ, Gaill F, Gros O (2008a) Sulphur-oxidizing extracellular bacteria in the gills of Mytilidae associated with wood falls. FEMS Microbiol Ecol 63:338–349
Duperron S, Halary S, Lorion J, Sibuet M, Gaill F (2008b) Unexpected co occurrence of 6 bacterial symbionts in the gill of the cold seep mussel Idas sp (Bivalvia:Mytilidae). Environ Microbiol 10:433–445
Duperron S, Lorion J, Samadi S, Gros O, Gaill F (2009) Symbioses between deep-sea mussels (Mytilidae: Bathymodiolinae) and chemosynthetic bacteria: diversity, function and evolution. C R Biol 332:298–310
Elsaied HE, Kaneko R, Naganuma T (2006) Molecular characterization of a deep-sea methanotrophic mussel symbiont that carries a RuBisCO gene. Mar Biotechnol 8:511–520
Felbeck H, Childress JJ, Somero GN (1981) Calvin-Benson cycle and sulphide oxidation enzymes in animals from sulphide-rich habitats. Nature 293:291
Fiala-Médioni A (1984) Mise en évidence par microscopie électronique à transmission de l’abondance de bactéries symbiotiques dans la branchie de Mollusques bivalves de sources hydrothermales profondes. C R Acad Sci 298:487–492
Fiala-Médioni A, Métivier C, Herry A, Le Pennec M (1986) Ultrastructure of the gill filament of an hydrothermal vent mytilid Bathymodiolus sp. Mar Biol 92:65–72
Fiala-Médioni A, Michalski JC, Jollès J, Alonso C, Montreuil J (1994) Lysosomic and lysozyme activities in the gill of bivalves from deep hydrothermal vents. C R Acad Sci 317:239–244
Fiala-Médioni A, McKiness ZP, Dando P, Boulègue J, Mariotti A, Alayse-Danet AM, Robinson JJ, Cavanaugh CM (2002) Ultrastructural biochemical and immunological characterisation of two populations of he Mytilid mussel Bathymodiolus azoricus from the Mid Atlantic Ridge: evidence for a dual symbiosis. Mar Biol 141:1035–1043
Fisher CR (1990) Chemoautotrophic and methanotrophic symbioses in marine invertebrates. Rev Aquat Sci 2:399–613
Fisher CR (1995) Towards an appreciation of hydrothermal-vent animals: their environment physiological ecology and tissue stable isotope values. In: Humphris SE, Zierenberg RA, Mullineaux LS, Thomson RE (eds) Seafloor hydrothermal systems: physical, chemical, biological, and geochemical interactions. American Geophysical Union, Washington, DC, pp 297–316
Fisher CR, Childress JJ (1992) Organic carbon transfer from methanotrophic symbionts to the host hydrocarbon-seep mussel. Symbiosis 12:221–235
Fisher CR, Childress JJ, Oremland RS, Bidigare RR (1987) The importance of methane and thiosulphate in the metabolism of the bacterial symbionts of two deep-sea mussels. Mar Biol 96:59–71
Fisher CR, Childress JJ, Arp AJ, Brooks JM, Distel DL, Favuzzi JA, Felbeck H, Hessler R, Johnson KS, Kennicutt MC, Macko SA, Newton A, Powell MA, Somero GN, Soto T (1988) Microhabitat variation in the hydrothermal vent mussel Bathymodiolus thermophilus at the Rose Garden vent on the Galapagos Rift. Deep Sea Res 35:1769–1791
Fisher CR, Brooks JM, Vodenichar JS, Zande JM, Childress JJ, Burke RA Jr (1993) The co-occurrence of methanotrophic and chemoautotrophic sulfur oxidizing bacterial symbionts in a deep-sea mussel. Mar Ecol 14:277–289
Flores JF, Fisher CR, Carney SL, Green BN, Freytag JK, Schaeffer SW, Royer WEJ (2005) Sulfide binding is mediated by zinc ions discovered in the crystal structure of a hydrothermal vent tubeworm hemoglobin. Proc Natl Acad Sci USA 102:2713–2718
Fujiwara Y, Takai K, Uematsu K, Tsuchida S, Hunt JC, Hashimoto J (2000) Phylogenetic characterization of endosymbionts in three hydrothermal vent mussels: influence on host distribution. Mar Ecol Prog Ser 208:147–155
Fujiwara Y, Kawato M, Yamamoto T, Yamanaka T, Sato Okoshi W, Noda S et al (2007) Three-year investigations into sperm whale-fall ecosystems in Japan. Mar Ecol 28:219–232
Génio L, Johnson SB, Vrijenhoek RC, Cunha MR, Tyler PA, Kiel S, Little CTS (2008) New record of “Bathymodiolus” mauritanicus Cosel 2002 from the Gulf of Cadiz (NE Atlantic) mud volcanoes. J Shellfish Res 27:53–61
Geret F, Rousse N, Riso R, Sarradin PM, Cosson R (1998) Metal compartmentalization and metallothionein isoforms in mussels from the Mid-Atlantic Ridge; preliminary approach to the fluid-organism relationship. Cah Biol Mar 39:291–293
Goffredi SK, Wilpiszeski R, Lee RW, Orphan VJ (2008) Temporal evolution of methane cycling and phylogenetic diversity of archaea in sediments form a deep-sea whale fall in Monterey Canyon California. ISME J 2:204–220
Gros O, Gaill F (2007) Extracellular bacterial association in gills of “wood mussels”. Cah Biol Mar 48:103–109
Gros O, Guibert J, Gaill F (2007) Gill-symbiosis in Mytilidae associated with wood-fall environments. Zoomorphology 126:163–172
Gustafson R, Turner R, Lutz R, Vrijenhoek R (1998) A new genus and five new species of mussels (Bivavlia Mytilidae) from deep-sea sulfide/hydrocarbon seeps in the Gulf of Mexico. Malacologia 40:63–112
Halary S, Riou V, Gaill F, Boudier T, Duperron S (2008) 3D FISH for the quantification of methane- and sulphur-oxidising endosymbionts in bacteriocytes of the hydrothermal vent mussel Bathymodiolus azoricus. ISME J 2:284–292
Hashimoto J, Ohta S, Fujikura K, Miura T (1995) Microdistribution pattern and biogeography of the hydrothermal vent communities of the Minami-Ensei Knoll in the mid-Okinawa trough Western Pacific. Deep Sea Res I 42:577–598
Henry MS, Childress JJ, Figueroa D (2008) Metabolic rates and thermal tolerances of chemoautotrophic symbioses from Lau Basin hydrothermal vents and their implication for species distribution. Deep Sea Res I 55:679–695
Iwasaki H, Kyuno A, Shintaki M, Fujita Y, Fujiwara Y, Fujikura K, Hashimoto J, Martins LD, Gebruk A, Miyazaki JI (2006) Evolutionary relationships of deep-sea mussels inferred by mitochondrial DNA sequences. Mar Biol 149:1111–1122
Jahnke LL, Summons RE, Dowling LM, Zahiralis KD (1995) Identification of methanotrophic lipid biomarkers in cold seep mussel gills: chemical and isotopic analysis. Appl Environ Microbiol 61:576–582
Jannasch HW, Mottl MJ (1985) Geomicrobiology of deep-sea hydrothermal vents. Science 229:717–725
Jeffreys JG (1876) New and peculiar Mollusca of the Pecten Mytilus and Arca families procured in the ‘Valorous’ expedition. Ann Mag Nat Hist 4:424–436
Johnson KS, Beehler CL, Sakamoto-Arnold CM, Childress JJ (1986) In situ measurements of chemical distributions in a deep-sea hydrothermal vent field. Science 231:1139–1141
Johnson KS, Childress JJ, Beehler CL, Sakamoto CM (1994) Biogeochemistry of hydrothermal vent mussel communities – the deep-sea analog to the intertidal zone. Deep Sea Res I 41:993–1011
Jones WJ, Won YJ, Maas PAY, Smith PJ, Lutz RA, Vrijenhoek RC (2006) Evolution of habitat use by deep-sea mussels. Mar Biol 148:841–851
Kadar E, Bettencourt R, Costa V, Serrão Santos R, Lobo-da-Cunha A, Dando PR (2005) Experimentally induced endosymbiont loss and re-acquirement in the hydrothermal vent bivalve Bathymodiolus azoricus. J Exp Mar Biol Ecol 318:99–110
Kamenev GM, Nadtochy VA, Kuznetsov AP (2001) Conchocoele bisecta (Conrad, 1849) (Bivalvia: Thyasiridae) from cold-water methane-rich areas of the sea of Okhotsk. Veliger 44:84–94
Kenk VC, Wilson BR (1985) A new mussel (Bivlavia: Mytilidae) from hydrothermal vents in the Galapagos Rift zone. Malacologia 26:253–271
Kerk D, Gee A, Dewhirst FE, Drum AS, Elston RA (1992) Phylogenetic placement of “Nuclear Inclusion X (NIX)” into the gamma subclass of proteobacteria on the basis of 16S ribosomal RNA sequence comparisons. Syst Appl Microbiol 15:191–196
Kiel S, Goedert JL (2006) A wood-fall association from late Eocene deep-water sediments of Washington State, USA. Palaios 21:548–556
Kochevar RE, Childress JJ, Fisher CR, Minnich E (1992) The methane mussel: roles of symbiont and host in the metabolic utilization of methane. Mar Biol 112:389–401
Kuwahara H, Yoshida T, Takaki Y, Shimamura S, Nishi S, Harada M, Matsuyama K, Takishita K, Kawato M, Uematsu K, Fujiwara Y, Sato T, Kato C, Kitagawa M, Kato I, Maruyama T (2007) Reduced genome of the thioautotrophic intracellular symbiont in a deep-sea clam Calyptogena okutanii. Curr Biol 17:881–886
Lajhta K, Michener RH (1994) Stable isotopes in ecology and environmental sciences. Oxford University Press, Oxford
Laurent MCZ, Gros O, Brulport JP, Gaill F, Le Bris N (2009) Sunken wood habitat for thiotrophic symbiosis in mangrove swamps. Mar Environ Res 67:83–88
Le Bris N, Duperron S (2010) Chemosynthetic communities and biogeochemical energy pathways along the MAR: the case of Bathymodiolus azoricus. In: Rona PA, Devey CW, Dyment J, Murton BJ (eds): Diversity of hydrothermal systems on slow spreading ocean ridges. Geophysical Monograph Series, American Geophysical Union 188:350
Le Bris N, Rodier P, Sarradin PM, Le Gall C (2006a) Is temperature a good proxy for sulfide in hydrothermal vent habitats? Cah Biol Mar 47:465–470
Le Bris N, Govenar B, Le Gall C, Fisher CR (2006b) Variability of physico-chemical conditions in 9°50N EPR diffuse flow vent habitats. Mar Chem 98:167–182
Le Pennec M, Beninger PG (2000) Reproductive characteristics and strategies of reducing-system bivalves. Comp Biochem Physiol A 126:1–16
Lee RW, Childress JJ (1994) Assimilation of inorganic nitrogen by marine invertebrates and their chemoautotrophic symbionts. Appl Environ Microbiol 60:1852–1858
Lee RW, Robinson JJ, Cavanaugh CM (1999) Pathways of inorganic nitrogen assimilation in chemoautotrophic bacteria-marine invertebrate symbioses: expression of host and symbiont glutamine synthetase. J Exp Biol 202:289–300
Leschine SB (1995) Cellulose degradation in anaerobic environments. Ann Rev Microbiol 49:399–426
Little CTS, Vrijenhoek RC (2003) Are hydrothermal vent animals living fossils? Trends Ecol Evol 18:582–588
Lorion J, Duperron S, Gros O, Cruaud C, Couloux A, Samadi S (2009) Several deep-sea mussels and their associated symbionts are able to live both on wood and on whale falls. Proc R Soc Lond B 276:177–185
Lutz RA, Jablonski D, Rhoads DC, Turner R (1980) Larval dispersal of a deep-sea hydrothermal vent bivalve from the Galapagos rift. Mar Biol 57:127–133
Lutz RA, Jablonski D, Turner R (1984) Larval development and dispersal at deep-sea hydrothermal vents. Science 226:1451–1454
Maas PAY, O’Mullan GD, Lutz RA, Vrijenhoek RC (1999) Genetic and morphometric characterization of mussels (Bivalvia: Mytilidae) from Mid-Atlantic hydrothermal vents. Biol Bull 196:265–272
MacDonald IR, Reilly JF II, Guinasso NL Jr, Brooks JM, Carney RS, Bryant WA, Bright TJ (1990) Chemosynthetic mussels at a brine-filled pockmark in the northern Gulf of Mexico. Science 248:1096–1099
Madigan MT, Martinko JM, Parker J (2002) Brock biology of microorganisms. Pearson Education, Upper Saddle River, NJ
Martins I, Colaço A, Dando PR, Martins I, Desbruyères D, Sarradin PM, Marques JC, Serrao Santos R (2008) Size-dependant variations on the nutritional pathway of Bathymodiolus azoricus demonstrated by a C-flux model. Ecol Model 217:59–71
McKiness ZP, Cavanaugh CM (2005) The ubiquitous mussel: Bathymodiolus aff brevior symbiosis at the Central Indian Ridge hydrothermal vents. Mar Ecol Prog Ser 295:183–190
McKiness ZP, McMullin ER, Fisher CR, Cavanaugh CM (2005) A new bathymodioline mussel symbiosis at the Juan de Fuca hydrothermal vents. Mar Biol 148:109–116
Miyazaki JI, Shintaku M, Kyuno A, Fujiwara Y, Hashimoto J, Iwasaki H (2004) Phylogenetic relationships of deep-sea mussels of the genus Bathymodiolus (Bivalvia: Mytilidae). Mar Biol 144:527–535
Moraga D, Jollivet D, Denis F (1994) Genetic differentiation across the western Pacific populations of the hydrothermal vent bivalve Bathymodiolus spp and the eastern Pacific (13°N) population of Bathymodiolus thermophilus. Deep Sea Res I 41:1551–1567
Nelson DC, Fisher CR (1995) Chemoautotrophic and methanotrophic endosymbiotic bacteria at deep-sea vents and seeps. In: Karl DM (ed) Microbiology of deep-sea hydrothermal vents. CRC Press, Boca Raton, FL, pp 125–167
Nelson DC, Hagen KD, Edwards DB (1995) The gill symbiont of the hydrothermal vent mussel Bathymodiolus thermophilus is a psychrophilic chemoautotrophic sulfur bacterium. Mar Biol 121:487–495
Nix ER, Fisher CR, Vodenichar JS, Scott KM (1995) Physiological ecology of a mussel with methanotrophic endosymbionts at three hydrocarbon seep sites in the Gulf of Mexico. Mar Biol 122:605–617
Okutani T, Fujiwara Y, Fujikura K, Miyake H, Kawato M (2003) A mass aggregation of the mussel Adipicola pacifica (Bivalvia:Mytilidae) on submerged whale bones. Venus 63:61–64
Olu K, Sibuet M, Hermeignies F, Foucher J-P, Fiala-Médioni A (1996) Spatial distribution of diverse cold seep communities living on various diapiric structures of the southern Barbados prism. Prog Oceanogr 38:347–376
Olu-LeRoy K, von Cosel R, Hourdez S, Jollivet D (2007a) Amphi-Atlantic cold-seep Bathymodiolus species complexes across the equatorial belt. Deep Sea Res 54:1890–1911
Olu-LeRoy K, Caprais JC, Fifis A, Fabri MC, Galéron J, Budzinski H, Le Menach K, Khripounoff A, Ondreas H, Sibuet M (2007b) Cold seep assemblages on a giant pockmark off West Africa: spatial patterns and environmental control. Mar Ecol 28:1–16
Page H, Fiala-Médioni A, Fisher C, Childress J (1991) Experimental evidence for filter-feeding by the hydrothermal vent mussel Bathymodiolus thermophilus. Deep Sea Res I 38:1455–1461
Pailleret M, Haga T, Petit P, Privé-Gill C, Saedlou N, Gaill F, Zbinden M (2007) Sunken woods from the Vanuatu islands: identification of wood substrates and preliminary description of associated fauna. Mar Ecol 27:1–9
Peek AS, Feldman RA, Lutz RA, Vrijenhoek RC (1998) Cospeciation of chemoautotrophic bacteria and deep sea clams. Proc Natl Acad Sci USA 95:9962–9966
Pelorce J, Poutiers JM (2009) Une nouvelle espèce de Bathymodiolinae (Mollusca Bivalvia Mytilidae) associée à des os de baleine coulés en Méditerranée. Zoosystema 31:975–985
Pernthaler A, Amann R (2004) Simultaneous fluorescence in situ hybridization of mRNA and rRNA in environmental bacteria. Appl Environ Microbiol 70:5426–5433
Pile AJ, Young CM (1999) Plankton availability and retention efficiencies of cold-seep symbiotic mussels. Limnol Oceanogr 44:1833–1839
Pimenov NV, Kalyuzhnaya MG, Khmelenina VN, Mityushina LL, Trotsenko YA (2002) Utilization of methane and carbon dioxide by symbiotrophic bacteria in gills of Mytilidae (Bathymodiolus) from the Rainbow and Logatchev hydrothermal fields on the Mid-Atlantic Ridge. Microbiology 71:587–594
Powell MA, Somero GN (1986) Adaptations to sulfide by hydrothermal vent animals: sites and mechanisms of detoxification and metabolism. Biol Bull 171:274–290
Rau G, Hedges J (1979) C-13 depletion in a hydrothermal vent mussel – Suggestion of a chemo-synthetic food source. Science 203:648–649
Raulfs EC, Macko SA, Van Dover CL (2004) Tissue and symbiont condition of mussels (Bathymodiolus thermophilus) exposed to varying levels of hydrothermal activity. J Mar Biol Assoc UK 84:229–234
Riou V, Halary S, Duperron S, Bouillon S, Elskens M, Bettencourt R, Santos RS, Dehairs F, Colaço A (2008) Influence of CH4 and H2S availability on symbiont distribution carbon assimilation and transfer in the dual symbiotic vent mussel Bathymodiolus azoricus. Biogeosciences 5:1681–1691
Robinson JJ, Stein JL, Cavanaugh CM (1998a) Cloning and sequencing of a form II Ribulose-15-bisphosphate carboxylase/oxygénase from the bacterial symbiont of the hydrothermal vent tubeworm Riftia pachyptila. J Bacteriol 180:1596–1599
Robinson JJ, Polz MF, Fiala-Médioni A, Cavanaugh CM (1998b) Physiological and immunological evidence for two distinct C1-utilizing pathways in Bathymodiolus puteoserpentis (Bivalvia:Mytilidae) a dual endosymbiotic mussel from the Mid-Atlantic Ridge. Mar Biol 132:625–633
Rousse N, Boulègue J, Cosson R, Fiala-Médioni A (1998) Bioaccumulation of metal within the hydrothermal mytilidae Bathymodiolus sp from the Mid-Atlantic Ridge. Oceanol Acta 21:597–607
Salerno JL, Macko SA, Hallam SJ, Bright M, Won Y, McKiness ZP, Van Dover CL (2005) Characterization of symbiont populations in life-history stages of mussels from chemosynthetic environments. Biol Bull 208:145–155
Samadi S, Quéméré E, Lorion J, Tillier A, von Cosel R, Lopez P, Cruad C, Couloux A, Boisselier Dubayle MC (2007) Molecular phylogeny in mytilids supports the wooden steps to deep-sea vents hypothesis. C R Acad Sci 330:446–456
Sarradin PM, Caprais JC, Riso R, Kerouel R, Aminot A (1999) Chemical environment of the hydrothermal mussel communities in the Lucky Strike and Menez Gwen vent fields Mid Atlantic Ridge. Cah Biol Mar 40:93–104
Sarrazin J, Walter C, Sarradin PM, Brind’amour A, Desbruyères D, Briand P, Fabri MC, Van Gaever S, Vanreusel A, Bachraty C, Thiébaut E (2006) Community structure and temperature dynamics within a mussel assemblage on the southern East Pacific Rise. Cah Biol Mar 47:483–490
Schmaljohann R, Faber E, Whiticar MJ, Dando PR (1990) Co-existence of methane- and sulphur-based endosymbioses between bacteria and invertebrates at a site in the Skagerrak. Mar Ecol Prog Ser 61:119–124
Schöne BR, Giere O (2005) Growth increment and stable isotope variation in shells of the deep-sea hydrothermal vent bivalve mollusk Bathymodiolus brevior from the North Fiji Basin Pacific Ocean. Deep Sea Res I 52:1896–1910
Scott KM, Fisher CR (1995) Physiological ecology of sulfide metabolism in hydrothermal vent and cold seep vesicomyid clams and vestimentiferan tubeworms. Am Zool 35:102–111
Shillito B, Jollivet D, Sarradin PM, Rodier P, Lallier FH, Desbruyères D, Gaill F (2001) Temperature resistance of Hesiolyra bergi a polychaetous annelid living on vent smoker walls. Mar Ecol Prog Ser 216:141–149
Sibuet M, Olu K (1998) Biogeography biodiversity and fluid dependence of deep sea cold seep communities at active and passive margins. Deep Sea Res II 45:517–567
Smith CR, Maybaum HL, Baco AR, Pope RH, Carpenter SD, Yager PL, Macko SA, Deming JW (1998) Sediment community structure around a whale skeleton in the deep Northeast Pacific: macrofaunal microbial and bioturbation effects. Deep Sea Res II 45:335–364
Smith PJ, McVeagh SM, Won Y, Vrijenhoek RC (2004) Genetic heterogeneity among New Zealand species of hydrothermal vent mussels (Mytilidae: Bathymodiolus). Mar Biol 144:537
Southward EC (2008) The morphology of bacterial symbioses in the gills of mussels of the genera Adipicola and Idas (Bivalvia: Mytilidae). J Shellfish Res 27:139–146
Spiridonova EM, Kuznetsov BB, Pimenov NV, Tourova TP (2006) Phylogenetic characterization of endosymbionts of the hydrothermal vents mussel Bathymodiolus azoricus by analysis of the 16S rRNA cbbL and pmoA genes. Microbiology 75:798–806
Stewart FJ, Cavanaugh CM (2006) Bacterial endosymbioses in Solemya (Mollusca:Bivalvia) - model systems for studies of symbiont-host adaptation. Antonie Leeuwenhoek 90:343–360
Stewart FJ, Newton LG, Cavanaugh CM (2005) Chemosynthetic endosymbioses: adaptations to oxic-anoxic interfaces. Trends Microbiol 13:439–448
Taylor JD, Glover E (2006) Lucinidae (Bivalvia) – the most diverse group of chemosymbioic molluscs. Zool J Linn Soc 148:421–438
Trask JL, Van Dover CL (1999) Site-specific and ontogenetic variations in nutrition of mussels (Bathymodiolus sp) from the Lucky Strike hydrothermal vent field Mid-Atlantic Ridge. Limnol Oceanogr 44:334–343
Treude T, Smith CR, Wenzhöfer F, Carney E, Bernardino AF, Hannides AK, Krüger M, Boetius A (2009) Biogeochemistry of a deep-sea whale fall: sulfate reduction sulfide efflux and methanogenesis. Mar Ecol Prog Ser 382:1–21
Tunnicliffe V, Juniper SK, Sibuet M (2003) Reducing environments of the deep-sea floor. In: Tyler PA (ed) Ecosystems of the world: the deep-sea. Elsevier, Amsterdam, The Netherlands, pp 81–110
Tunnicliffe V, Davies KTA, Butterfield DA, Embley RW, Rose JM, Chadwick WW (2009) Survival of mussels in extremely acidic waters on a submarine volcano. Nat Geosci 2:344–348
Tyler PA, Young CM (2003) Dispersal at hydrothermal vents: a summary of recent progress. Hydrobiologia 503:9–19
Tyler PA, Young CM, Dolan E, Arellano SM, Brooke SD, Baker M (2007) Gametogenic periodicity in the chemosynthetic cold-seep mussel “Bathymodiolus childressi”. Mar Biol 150:829–840
Tyler PA, Marsh L, Baco-Taylor A, Smith CR (2009) Protandric hermaphroditism in the whale-fall bivalve mollusc Idas washingtonia. Deep Sea Res II 56:1689–1699
Vacelet J, Fiala-Médioni A, Fisher CR, Boury-Esnault N (1996) Symbiosis between methane-oxidizing bacteria and a deep-sea carnivorous cladorhizid sponge. Mar Ecol Prog Ser 145:77–85
Van Dover CL (2000) The ecology of deep-sea hydrothermal vents. Princeton University Press, Princeton, NJ
Van Dover SL, Aharon P, Bernhard JM et al (2003) Blake Ridge methane seeps: characterization of a soft-sediment chemosynthetically based ecosystem. Deep Sea Res I 50:281–300
von Cosel R (2002) A new species of bathymodioline mussel (Mollusca Bivalvia Mytilidae) from Mauritania (West Africa) with comments on the genus Bathymodiolus Kenk & Wilson 1985. Zoosystema 24:259–271
von Cosel R, Janssen R (2008) Bathymodioline mussels of the Bathymodiolus (sl) childressi clade from methane seeps near Edison Seamount New Ireland Papua New Guinea. Arch Molluskenkunde 137:195–224
von Cosel R, Marshall BA (2003) Two new species of large mussels (Bivalvia: Mytilidae) from active submarine volcanoes and a cold seep off the eastern North Island of New Zealand with description of a new genus. Nautilus 117:31–46
von Cosel R, Olu K (1998) Gigantism in Mytilidae A new Bathymodiolus from cold seep areas on the Barbados accretionary Prism. C R Biol 321:655–663
von Cosel R, Comtet T, Krylova EM (1999) Bathymodiolus (Bivalvia:Mytilidae) from hydrothermal vents on the Azores Triple junction and the Logatchev hydrothermal field Mid-Atlantic Ridge. Veliger 42:218–248
Von Damm KL (1995) Controls on the chemistry and temporal variability of seafloor hydrothermal vents. In: Humphris S, Zierenberg RA, Mullineaux LS, Thomson RE (eds) Seafloor hydrothermal systems: physical chemical biological and geological interactions, Geophysical Monographs. American Geophysical Union, Washington, DC, pp 222–247
Won Y, Young CR, Lutz RA, Vrijenhoek RC (2003a) Dispersal barriers and isolation among deep-sea mussel populations (Mytilidae: Bathymodiolus) from Eastern Pacific hydrothermal vents. Mar Ecol 12:169–184
Won YJ, Hallam SJ, O’Mullan D, Pan IL, Buck KR, Vrijenhoek RC (2003b) Environmental acquisition of thiotrophic endosymbionts by deep-sea mussels of the genus Bathymodiolus. Appl Environ Microbiol 69:6785–6792
Won YJ, Jones WJ, Vrijenhoek RC (2008) Absence of cospeciation between deep-sea mytilids and their thiotrophic symbionts. J Shellfish Res 27:129–138
Woyke T, Teeling H, Ivanova N, Hunteman M, Richter M, Glockner FO, Boffelli D, Anderson IJ, Barry KW, Shapiro HJ, Szeto E, Kyrpides NC, Musmann M, Amann R, Bergin C, Ruehland C, Rubin EM, Dubilier N (2006) Symbiosis insights through metagenomic analysis of a microbial consortium. Nature 443:950–955
Yamamoto H, Fujikura K, Hiraishi A, Kato K, Maki Y (2002) Phylogenetic characterization and biomass estimation of bacterial endosymbionts associated with invertebrates dwelling in chemosynthetic communities of hydrothermal vents and cold seeps. Mar Ecol Prog Ser 245:61–67
Zbinden M, Shillito B, Le Bris N, de Villardi de Montaur C, Roussel E, Guyot F, Gaill F, Cambon-Bonavita MA (2008) New insights on the metabolic diversity among the epibiotic microbial community of the hydrothermal vent shrimp Rimicaris exoculata. J Exp Mar Biol Ecol 359:131–140
Zielinski FU, Pernthaler A, Duperron S, Raggi L, Giere O, Borowski C, Dubilier N (2009) Widespread occurrence of an intranuclear bacterial parasite in vent and seep bathymodiolin mussels. Environ Microbiol 11:1150–1167
Acknowledgments
I thank S. Kiel for his invitation to contribute this chapter, and the two reviewers for their helpful comments. S. Gaudron is gratefully acknowledged for providing helpful comments on the manuscript. Thanks to S. Halary and J. Lorion for fruitful discussions. The author’s work is funded by Université Pierre et Marie Curie, French ANR Deep Oases, and EU programs HERMES and CHEMECO.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer Science+Business Media B.V.
About this chapter
Cite this chapter
Duperron, S. (2010). The Diversity of Deep-Sea Mussels and Their Bacterial Symbioses. In: Kiel, S. (eds) The Vent and Seep Biota. Topics in Geobiology, vol 33. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-9572-5_6
Download citation
DOI: https://doi.org/10.1007/978-90-481-9572-5_6
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-481-9571-8
Online ISBN: 978-90-481-9572-5
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)