Abstract
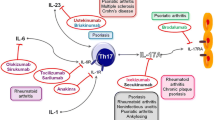
The definition of the CD3+ CD4+ Th17 cell subset and the identification of the IL-23-Th17 axis have introduced new paradigms to explain the origin of autoimmune events in animal models, subverting the established Th1-Th2 paradigm. IL-17A has been pivotal for the discovery of the Th17 lineage, which probably evolved as an arm of the adaptive immune system for host protection against extracellular bacteria and fungi. IL-17A, is the founding member of the IL-17 family composed of six members. Th17 cells and IL-17A have been implicated in a variety of inflammatory and autoimmune diseases in rodents. In these models, Th17 cells are pivotal in the pathogenesis of the disease and IL-17A appears to be the main mediator, but the situation might be different in humans. In some human pathological conditions, in addition to Th17 cells, other IL-17A-producing cells have been described, including CD8+ T cells, astrocytes, macrophages and Langerhans cells. The therapeutic effect of some new biologics can now, at least in part, be explained by their interference with mediators involved in the generation of Th17 cells, but more specific treatments would be valuable to dissect these intricate networks. An antibody neutralizing IL-17A is being evaluated under different autoimmune conditions. This approach might not only benefit patients, but, by neutralizing IL-17A selectively, might also help to define the role of this cytokine in autoimmune disorders and contribute to a new wave of selective and targeted therapies.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25: 821–852
Komiyama Y, Nakae S, Matsuki T, Nambu A, Ishigame H, Kakuta S, Sudo K, Iwakura Y (2006) IL-17 plays an important role in the development of experimental autoimmune encephalomyelitis. J Immunol 177: 566–573
Korn T, Bettelli E, Gao W, Awasthi A, Jager A, Strom TB, Oukka M, Kuchroo VK (2007) IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature 448: 484–487
Wilson NJ, Boniface K, Chan JR, McKenzie BS, Blumenschein WM, Mattson JD, Basham B, Smith K, Chen T, Morel F et al (2007) Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nat Immunol 8: 950–957
Fujino S, Andoh A, Bamba S, Ogawa A, Hata K, Araki Y, Bamba T, Fujiyama Y (2003) Increased expression of interleukin 17 in inflammatory bowel disease. Gut 52: 65–70
Tzartos JS, Friese MA, Craner M, Palace J, Newcombe J, Esiri MM, Fugger L (2007) Interleukin-17 production in CNS-infiltrating T-cells and glial cells is associated with active disease in multiple sclerosis. Am J Pathol 172: 146–155
Coury F, Annels N, Rivollier A, Olsson S, Santoro A, Speziani C, Azocar O, Flacher M, Djebali S, Tebib J et al (2008) Langerhans cell histiocytosis reveals a new IL-17Adependent pathway of dendritic cell fusion. Nat Med 14: 81–87
Spriggs, MK (1997) Interleukin-17 and its receptor. J Clin Immunol 17: 366–369
Yao, Z, Fanslow WC, Seldin MF, Rousseau AM, Painter SL, Comeau MR, Cohen JI, Spriggs MK (1995) Herpesvirus saimiri encodes a new cytokine, IL-17, which binds to a novel cytokine receptor. Immunity 3: 811–821
Moseley TA, Haudenschild DR, Rose L, Reddi AH (2003) Interleukin-17 family and IL-17 receptors. Cytokine Growth Factor Rev 14: 155–174
Kolls JK, Linden A (2004) Interleukin-17 family members and inflammation. Immunity 21: 467–476
Kawaguchi M, Adachi M, Oda N, Kokubu F, Huang SK (2004) IL-17 cytokine family. J Allergy Clin Immunol 114: 1265–1273
Starnes T, Robertson MJ, Sledge G, Kelich S, Nakshatri H, Broxmeyer HE, Hromas R (2001) Cutting edge: IL-17F, a novel cytokine selectively expressed in activated T cells and monocytes, regulates angiogenesis and endothelial cell cytokine production. J Immunol 167: 4137–4140
McAllister F, Henry A, Kreindler JL, Dubin PJ, Ulrich L, Steele C, Finder JD, Pilewski JM, Carreno BM, Goldman SJ et al (2005) Role of IL-17A, IL-17F, and the IL-17 receptor in regulating growth-related oncogene-a and granulocyte colony-stimulating factor in bronchial epithelium: Implications for airway inflammation in cystic fibrosis. J Immunol 175: 404–412
Lubberts E (2003) The role of IL-17 and family members in the pathogenesis of arthritis. Curr Opin Investig Drugs 4: 572–577
Yang XO, Chang SH, Park H, Nurieva R, Shah B, Acero L, Wang YH, Schluns KS, Broaddus RR, Zhu Z et al (2008) Regulation of inflammatory responses by IL-17F. J Exp Med 205: 1063–1075
Akimzhanov AM, Yang XO, Dong C (2007) Chromatin remodeling of interleukin-17 (IL-17)-IL-17F cytokine gene locus during inflammatory helper T cell differentiation. J Biol Chem 282: 5969–5972
Liang SC, Long AJ, Bennett F, Whitters MJ, Karim R, Collins M, Goldman SJ, Dunussi-Joannopoulos K, Williams CMM, Wright JF et al (2007) An IL-17F/A heterodimer protein is produced by mouse Th17 cells and induces airway neutrophil recruitment. J Immunol 179: 7791–7799
Wright JF, Guo Y, Quazi A, Luxenberg DP, Bennett F, Ross JF, Qiu Y, Whitters MJ, Tomkinson KN, Dunussi-Joannopoulos K et al (2007) Identification of an interleukin 17F/17A heterodimer in activated human CD4+ T cells. J Biol Chem 282: 13447–13455
Chang SH, Dong C (2007) A novel heterodimeric cytokine consisting of IL-17 and IL-17F regulates inflammatory responses. Cell Res 17: 435–440
Kuestner RE, Taft DW, Haran A, Brandt CS, Brender T, Lum K, Harder B, Okada S, Ostrander CD, Kreindler JL et al (2007) Identification of the IL-17 receptor related molecule IL-17RC as the receptor for IL-17F. J Immunol 179: 5462–5473
Lee J, Ho WH, Maruoka M, Corpuz RT, Baldwin DT, Foster JS, Goddard AD, Yansura DG, Vandlen RL, Wood WI et al (2001). IL-17E, a novel proinflammatory ligand for the IL-17 receptor homolog IL-17Rh1. J Biol Chem 276: 1660–1664
Zrioual S, Toh ML, Tournadre A, Zhou Y, Cazalis MA, Pachot A, Miossec V, Miossec P (2008) IL-17RA and IL-17RC receptors are essential for IL-17A-induced ELR+ CXC chemokine. expression in synoviocytes and are overexpressed in rheumatoid blood. J Immunol 180: 655–663
Toy D, Kugler D, Wolfson M, Vanden Bos T, Gurgel J, Derry J, Tocker J, Peschon J (2006) Cutting edge: Interleukin 17 signals through a heteromeric receptor complex. J Immunol 177: 36–39
Acosta-Rodriguez EV, Rivino L, Geginat J, Jarrossay D, Gattorno M, Lanzavecchia A, Sallusto F, Napolitani G (2007) Surface phenotype and antigenic specificity of human interleukin 17-producing T helper memory cells. Nat Immunol 8: 639–646
Kotake S, Udagawa N, Takahashi N, Matsuzaki K, Itoh K, Ishiyama S, Saito S, Inoue K, Kamatani N, Gillespie MT et al (1999) IL-17 in synovial fluids from patients with rheumatoid arthritis is a potent stimulator of osteoclastogenesis. J Clin Invest 103: 1345–1352
Infante-Duarte C, Horton HF, Byrne MC, Kamradt T (2000) Microbial lipopeptides induce the production of IL-17 in Th cells. J Immunol 165: 6107–6115
Nistala K, Moncrieffe H, Newton KR, Varsani H, Hunter P, Wedderburn LR (2008) Interleukin-17-producing T cells are enriched in the joints of children with arthritis, but have a reciprocal relationship to regulatory T cell numbers. Arthritis Rheum 58: 875–887
Page G, Sattler A, Kersten S, Thiel A, Radbruch A, Miossec P (2004) Plasma cell-like morphology of Th1-cytokine-producing cells associated with the loss of CD3 expression. Am J Pathol 164: 409–417
Liu Y (2002) Uncover the mystery of plasmacytoid dendritic cell precursors or type 1 interferon producing cells by serendipity. Hum Immunol 63: 1067–1071
Fricke I, Mitchell D, Mittelsta J, Lehan N, Heine H, Goldmann T, Bohle A, Brandau S (2006) Mycobacteria induce IFN-? production in human dendritic cells via triggering of TLR2. J Immunol 176: 5173–5182
Teunissen MB, Koomen CW, de Waal Malefyt R, Wierenga EA, Bos JD (1998) Interleukin-17 and interferon-gamma synergize in the enhancement of proinflammatory cytokine production by human keratinocytes. J Invest Dermatol 111: 645–649
Li J, Li D, Tan Z (2004) The expression of interleukin-17, interferon-gamma, and macrophage inflammatory protein-3 alpha mRNA in patients with psoriasis vulgaris. J Huazhong Univ Sci Technol Med Sci 24: 294–296
Arican O, Aral M, Sasmaz S, Ciragil P (2005) Serum levels of TNF-alpha, IFN-gamma, IL-6, IL-8, IL-12, IL-17, and IL-18 in patients with active psoriasis and correlation with disease severity. Mediators Inflamm 2005: 273–279
Bos JD, Hagenaars C, Das PK Krieg SR, Voorn WJ, Kapsenberg ML (1989) Predominance of’ memory’ T cells (CD4+, CDw29+) over’ naive’ T cells (CD4+, CD45R+) in both normal and diseased human skin. Arch Dermatol Res 281: 24–30
Bos JD, Zonneveld I, Das PK Krieg SR, van der Loos CM, Kapsenberg ML (1987) The skin immune system (SIS): distribution and immunophenotype of lymphocyte subpopulations in normal human skin. J Invest Dermatol 88: 569–573
Bovenschen HJ, Seyger MMB, Van De Kerkhof PCM. (2005) Plaque psoriasis vs. atopic dermatitis and lichen planus: A comparison for lesional T-cell subsets, epidermal proliferation and differentiation. Br J Dermatol 153: 72–78
Lowes MA, Kikuchi T, Fuentes-Duculan J, Cardinale I, Zaba LC, Haider AS, Bowman EP, Krueger JG (2008) Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J Invest Dermatol 128: 1207–1211
Nielsen OH, Kirman I, Rudiger N, Hendel J, Vainer B (2003) Upregulation of interleukin-12 and-17 in active inflammatory bowel disease. Scand J Gastroenterol 38: 180–185
Gottfried E, Kunz-Schughart LA, Weber A, Rehli M, Peuker A, Muller A, Kastenberger M, Brockhoff G, Andreesen R, Kreutz M (2008) Expression of CD68 in non-myeloid cell types. Scand J Immunol 67: 453–463
Friese MA, Fugger L (2005) Autoreactive CD8(+) T cells in multiple sclerosis: A new target for therapy? Brain 128: 1747–1763
Meeuwsen S, Persoon-Deen C, Bsibsi M, Ravid R, van Noort JM (2003) Cytokine, chemokine and growth factor gene profiling of cultured human astrocytes after exposure to proinflammatory stimuli. Glia 43: 243–253
Lock C, Hermans G, Pedotti R, Brendolan A, Schadt E, Garren H, Langer-Gould A, Strober S, Cannella B, Allard J et al (2002) Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat Med 8: 500–508
Ishizu T, Osoegawa M, Mei FJ, Kikuchi H, Tanaka M, Takakura Y, Minohara M, Murai H, Mihara F, Taniwaki T et al (2005) Intrathecal activation of the IL-17/IL-8 axis in opticospinal multiple sclerosis. Brain 128: 988–1002
Laman JD, Leenen PJM, Annels NE, Hogendoorn PCW, Egeler RM (2003) Langerhanscell histiocytosis’ insight into DC biology’. Trends Immunol 24: 190–196
Hizawa N, Kawaguchi M, Huang SK, Nishimura M (2006) Role of interleukin-17F in chronic inflammatory and allergic lung disease. Clin Exp Allergy 36: 1109–1114
Andoh A, Zhang Z, Inatomi O, Fujino S, Deguchi Y, Araki Y, Tsujikawa T, Kitoh K, Kim-Mitsuyama S, Takayanagi A et al (2005) Interleukin-22, a member of the IL-10 subfamily, induces inflammatory responses in colonic subepithelial myofibroblasts. Gastroenterol 129: 969–984
Zaba LC, Cardinale I, Gilleaudeau P, Sullivan-Whalen M, Fariñas MS, Fuentes-Duculan J, Novitskaya I, Khatcherian A, Bluth MJ, Lowes MA et al (2007) Amelioration of epidermal hyperplasia by TNF inhibition is associated with reduced Th17 responses. J Exp Med 204: 3183–3194
Massarotti EM (2008) Clinical and patient-reported outcomes in clinical trials of Abatacept in the treatment of rheumatoid arthritis. Clin Therapeutics 30: 429–442
Chen Z, Tato CM, Muul L, Laurence A, O’Shea JJ (2007) Distinct regulation of interleukin-17 in human T helper lymphocytes. Arthritis Rheum 56: 2936–2946
Ohsugi Y, Kishimoto T (2008) The recombinant humanized anti-IL-6 receptor antibody tocilizumab, an innovative drug for the treatment of rheumatoid arthritis. Expert Opin Biol Ther 8: 669–681
Leonardi CL, Kimball AB, Papp KA, Yeilding N, Guzzo C, Wang Y, Li S, Dooley LT, Gordon KB (2008) Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1). Lancet 371: 1665–1674
Papp KA, Langley RG, Lebwohl M, Krueger GG, Szapary P, Yeilding N, Guzzo C, Hsu MC, Wang Y, Li S et al (2008) Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 52-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 2). Lancet 371: 1675–1684
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Birkhäuser Verlag Basel/Switzerland
About this chapter
Cite this chapter
Di Padova, F. (2009). IL-17A and Th17 cells as therapeutic targets for autoimmune diseases. In: Quesniaux, V., Ryffel, B., Di Padova, F. (eds) Th 17 Cells: Role in Inflammation and Autoimmune Disease. Progress in Inflammation Research. Birkhäuser Basel. https://doi.org/10.1007/978-3-7643-8681-8_19
Download citation
DOI: https://doi.org/10.1007/978-3-7643-8681-8_19
Publisher Name: Birkhäuser Basel
Print ISBN: 978-3-7643-8680-1
Online ISBN: 978-3-7643-8681-8
eBook Packages: MedicineMedicine (R0)