Abstract
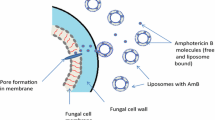
AmBisome® is a newly commercialized formulation of amphotericin B incorporated into small unilamellar liposomes composed of cohesive bilayer forming lipids (see chapter 1). An important feature of this formulation is that the amphotericin B is stably incorporated into the liposome bilayer.1 In recent years, this and other lipid formulations have been devised to improve the utility of Fungizone, a micellar formulation of amphotericin B which rapidly disassociates upon iv administration. While the standard formulation is proven clinically, it is limited by severe renal toxicity, and many life-threatening fungal infections cannot successfully be treated.2,3 For the purposes of this chapter, Fungizone is considered “free” amphotericin B (ampB) administration.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Adler-Moore J, Proffitt RT. Development, characterization, efficacy and mode of action of AmBisome,® a unilamellar liposomal formulation of amphotericin B. J Liposome Research 1993; 3(3)429–45o.
Utz JP. Amphotericin B toxicity: general side effects. Ann Int Med 1964; 61: 340–354.
McCurdy KK, Frederic M, Elkinton JR. Renal tubular acidosis due to amphotericin B. N Eng J Med 1968; 278: 124–131.
Proffitt RT, Adler-Moore J, Fujii G et al. Stability and mode of action of AmBisome (liposomal amphotericin B). J Controlled Release 1994; z8: 342–343.
McAndrews BJ, Lee MJA, Adler-Moore JP. Comparative toxicities of fungizone and AmBisome for cultured kidney cells and macrophages. General Proceeding of the ASM Meeting for American Society for Microbiology. Atlanta, Georgia 1993:11 (Abstract).
Proffitt RT, Satorius A, Chiang S et al. Pharmacology and toxicology of a liposomal formulation of amphotericin B (AmBisome) in rodents. J Antimicrobial Chemotherapy. 1991; 28 (Suppl. B): 49–61.
Fielding RM, Singer AW, Wang LH et al. Relationship of pharmacokinetics and drug distribution in tissue to increased safety of amphotericin B colloidal dispersion in dogs. Antimicrobial Agents Chemotherapy 1992; 36: 299–307.
Meunier F, Prentice HG, Ringden O. Liposomal amphotericin B (AmBisome): safety data from a phase II/III clinical trial. J Antimicrobial Chemotherapy 1991; 28 (suppl B): 83–91.
Ringden O, Andstrom E, Remberger M et al. Safety of liposomal amphotericin B (AmBisome) in 187 transplant recipients treated with cyclosporin. Bone Marrow Transplantation 1994; 14 (Supp1.5): S10 - S14.
Turner AF, Presant CA, Proffitt RT et al. In-in-labeled liposomes: dosimetry and tumor depiction. Radiology 1988; 166 (3): 761–765.
Lee JW, Amantea MA, Francis PA et al. Pharmacokinetics and safety of a unilamellar liposomal formulation of amphotericin B (AmBisome) in rabbits. Antimicrobial Agents Chemotherapy 1994; 38 (4): 713–718.
Hiles R, Bekersky I. Pharmacokinetics (PK) of liposomal amphotericin B (AmBisome) in rats. Pharmaceutical Research 1993; 10(1o):S-372 (Abstract #PPDM 8295).
Bekersky I, Hiles R. Pharmacokinetics (PK) of liposomal amphotericin B (AmBisome) in dogs. Pharmaceutical Research 1993; io(1o):S-372 (Abstract # PPDM 8296).
Walsh TJ, Bekersky I, Yeldandi V et al. Pharmacokinetics of AmBisome in persistently febrile neutropenic patients receiving empirical antifungal therapy. Proceedings of the 35th Interscience Conference on Antimicrobial Agents and Chemotherapy. Abstract A13. 1995: 3.
Croft SL, Davidson RNN, Thorton EA. Liposomal amphotericin B in the treatment of visceral leishmaniasis. J Antimicrobial Chemotherapy 1991; 28 (Suppl
Gangneux JP, Sulahian A, Garin Y et al. Therapy of visceral leishmaniasis due to Leishmania infantum: experimental assessment of efficacy of AmBisome. Antimicrobial Agents Chemotherapy 1996; 4o (5): 1214–1218.
Van Etten EWM, Van den Heuvel-de Groot C, Bakker-Woudenberg, IAJM. Efficacies of amphotericin B-desoxycholate (Fungizone), liposomal amphotericin B (AmBisome) and Fluconazole in the treatment of systemic candidosis in immunocompetent and leukopenic mice. J Antimicrobial Chemotherapy 1993; 32: 723–739.
Adler-Moore JP, Chiang S, Satorius A et al. Treatment of murine candidosis and cryptococcosis with a unilamellar liposomal amphotericin B formulation (AmBisome). J Antimicrobial Chemotherapy 1991; 28 (Suppl. B): 63–71.
Karyotakis NC, Anaissie, E J. Efficacy of escalating doses of liposomal amphotericin B (AmBisome) against hematogenous Candida lusitaniae and Candida krusei infection in neutropenic mice. Antimicrobial Agents Chemotherapy 1994; 38 (11): 2660–2662.
Francis P, Lee JW, Hoffman A et al. Efficacy of unilamellar liposomal amphotericin B in treatment of pulmonary aspergillosis in persistently granulocytopenic rabbits: the potential role of D-mannitol and galactomannan as markers of infection and therapeutic response. J Infectious Diseases 1994; 169: 356–368.
Allen SD, Sorenson KN, Nejdl MJ et al. Prophylactic efficacy of aerosolized liposomal (AmBisome) and non-liposomal (Fungizone) amphotericin B in murine pulmonary aspergillosis. J Antimicrob Chemotherapy 1994; 34: 1001–1013.
Albert MM, Stahl-Carroll TL, Luther MF et al. Comparison of liposomal amphotericin B to amphotericin B for treatment of murine cryptococcal meningitis. J Mycol Med 1995; 5: 1–6.
Graybill JR, Boccanegra R. Liposomal amphotericin B therapy of murine histoplasmosis. Antimicrobial Agents Chemotherapy 1995; 39 (8): 1885–1887.
Clemons KV, Stevens DA. Therapeutic efficacy of a liposomal formulation of amphotericin B (AmBisome) against murine blastomycosis. J Antimicrobial Chemotherapy 1993; 32: 465–472.
Clemons KV, Stevens DA. Comparison of a liposomal amphotericin B formulation (AmBisome) and desoxycholate amphotericin B (Fungizone) for the treatment of murine paracoccidioidomycosis. J Med Vet Mycol 1993; 31: 387–394.
Garcia AL. Efficacy of single high dose AmBisome for treatment of murine candidiasis and histoplasmosis. Masters thesis. Pomona CA: California State Polytechnic University, Pomona, 1996;
Prentice HG, Catovsky D, Aoun M et al. AmBisome versus amphotericin B in patients with fever unresponsive to antibiotic therapy for 96 h, or with confirmed fungal infection. 2nd International Symposium on Febrile Neutropenia. 1995; Abstract 35.
Hann IM, Stevens RF, Pinkerton CR et al. Safety and efficacy of two dose regimes of AmBisome versus amphotericin B as empiric antifungal treatment in neutropenic pediatric patients. n“ Annual Meeting of the European Group for Blood & Bone Marrow Transplantation & 11th Meeting of the Nurses Group, Davos, Switzerland, 1995; Abstract.
Ringden O, Meunier F, Tollemar J et al. Efficacy of amphotericin B encapsulated in liposomes (AmBisome) in the treatment of invasive fungal infections in immunocompromised patients. J Antimicrobial Chemotherapy 1991; 28 (suppl B): 73–82.
Tollemar J, Ringden O, Tyden G. Liposomal amphotericin B (AmBisome) treatment in solid organ and bone marrow transplant recipients. Efficacy and Safety Evaluation. Clinical Transplantation 1990; 4: 167–175.
Mills W, Chopra R, Linch DC et al. Liposomal amphotericin B in the treatment of fungal infections in neutropenic patients: a single-centre experience of 133 episodes in 116 patients. British Journal Haematology 1994; 86: 754–760.
Schurmann D, De Matos Marques B, Grunewald T et al. Safety and efficacy of liposomal amphotericin B in treating AIDS-associated disseminated cryptococcosis. J Infectious Diseases 1991; 164: 620.
Coker RJ, Viviani M, Gazzard BG et al. Treatment of cryptococcosis with liposomal amphotericin B (AmBisome) in 23 patients with AIDS. AIDS 1993; 7: 829–835.
Leenders ACAP, Reiss P, Portegies P et al. A randomized trial of liposomal amphotericin B (AmBisome) 4 mg/kg versus amphotericin B o.7 mg/kg for cryptococcal meningitis in HIV infected patients. Proceedings of the 36th Interscience Conference on Antimicrobial Agents Chemotherapy. 1996; Abstract LM35.p z87.
Viviani MA, Cofrancesco E, Boschetti C et al. Eradication of Fusarium infection in a leukopenic patient treated with liposomal amphotericin B. Mycoses 1991; 34: 255.
Fisher EW, Toma A, Fisher PH et al. Rhinocerebral mucormycosis: use of liposomal amphotericin B. J Laryngology and Otology 1991; 105: 575.
Seaman J, Boer C, Wilkinson R et al. Liposomal amphotericin B (AmBisome) in the treatment of complicated kala-azar under field conditions. Clin Inf Dis 1995; 21: 188–193.
Emminger W, Graninger W, Emminger-Schmidmeier W et al. Tolerance of high doses of amphotericin B by infusion of a liposomal formulation in children with cancer. Ann Hematol 1994; 6827–31.
Zoubek A, Emminger W, Emminger-Schmidmeier W et al. Conventional vs. liposomal amphotericin B in immunosuppressed children. Pediatric Hematology Oncology 1992; 9: 187–190.
Bindschadler DD, Bennett JE. A pharmacologic guide to the clinical use of amphotericin B. J Infectious Diseases 1969; 120(4)427–436.
Daneshmend TK, Warnock DW. Clinical pharmacokinetics of systemic antifungal drugs. Clinical Pharmacokinetics 1983; 8: 17–42.
Anaissie E, Paetznick V, Proffitt R et al. Comparison of the in vitro antifungal activity of free and liposome-encapsulated amphotericin B. Eur J Clin Microbiol Infect Dis 1991; 10: 665–668.
Garcia AL, Satorius A, Proffitt RT et al. Prophylaxis with single high dose AmBisome (AmBi) minimizes murine histoplasmosis and candidiasis. Abstract, 95`s General Meeting of the American Society for Microbiology, Washington DC 1995: i64.
Karyotakis NC, Anaissie EJ, Hachem R et al. Comparison of the efficacy of polyenes and triazoles against hematogenous Candida krusei infection in neutropenic mice. J Infectious Diseases 1993; 168: 1311–1313.
Anaissie EJ, Hachem R, Karyotakis NC et al. Comparative efficacies of amphotericin B, triazoles, and combination of both as experimental therapy for murine trichosporonosis. Antimicrobial Agents Chemotherapy 1994; 38 (11): 2541–2544.
Albert MM, Adams K, Luther MJ et al. Efficacy of AmBisome in murine coccidioidomycosis. J Med Vet Mycol 1994; 32: 467–471.
Adler-Moore JP. AmBisome targeting to fungal infections. Bone Marrow Transplantation 1994; 14 (Suppl 5): S3 - S7.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1998 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Adler-Moore, J.P., Proffitt, R.T. (1998). AmBisome®: Long Circulating Liposomal Formulation of Amphotericin B. In: Woodle, M.C., Storm, G. (eds) Long Circulating Liposomes: Old Drugs, New Therapeutics. Biotechnology Intelligence Unit. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-22115-0_13
Download citation
DOI: https://doi.org/10.1007/978-3-662-22115-0_13
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-22117-4
Online ISBN: 978-3-662-22115-0
eBook Packages: Springer Book Archive