Abstract
In most terrestrial ecosystems, allelochemical interactions are involved in vascular plants. Nevertheless, bryophytes represent also a crucial group found in many ecosystems with a specific morphology and physiology (e.g. secondary metabolism). Among bryophytes, Sphagnum genus is of particular interest because they form a dense homogeneous carpet which is slowly decomposed (the peat) in peatlands. Such ecosystems represent a terrestrial sink of carbon and so are crucial to be studied, especially under a climate change. Objectives of this chapter were (1) to synthesize current bryophyte allelochemical interactions and (2) to illustrate recent research on Sphagnum with the case of Sphagnum fallax phenolics (production and degradation) recovered in a french peatland. The top layer of living Sphagnum represents the active allelopathic part where water-soluble phenolics were mostly recovered. Their concentrations were found to change along the seasons. The transformation of phenolic compounds is performed by an enzymatic system O2 (phenoloxidases) or H2O2 (peroxidases) dependent. Sphagnum-peroxidases constituted the main oxidative system and fungal phenoloxidases were proposed to be regulated by phenolics. Moreover, Sphagnum was able to regulate its secondary metabolism under a climate forcing by decreasing its phenolic concentrations. Allelopathic potential of Sphagnum phenolics was stated with their role in the microdistribution of associated Sphagnum microorganisms. Finally, Sphagnum extracts also strongly delayed Pine and Lolium germination seeds and inhibited Lolium radicle growth and delayed Raphanus and Pinus radicles. Ecological and agronomic perspectives of Sphagnum extracts are discussed.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Since the early 1970s, research efforts on allelopathy have increased considerably. Because allelopathy meets current societal demands for environmentally sound approaches to agriculture, there is a growing interest in discovering, and understanding allelochemicals produced by plants. One way to identify allelopathic interactions is to study plants in their ecosystems. Numerous ecosystems have been studied through allelochemical interactions such as forests (Souto et al. 2000) or grasslands (Viard-Crétat et al. 2009). They involved mostly gymnosperms and angiosperms rather than nonvascular plants. Bryophytes represent one successful group of plants in terms of geographical distribution on all continents, habitat diversification, and number of species (Slack 2011). Even if bryophytes are known to produce chemicals which make mosses very unattractive for herbivores, little is known about the different roles of phenolics excreted by mosses in their surroundings. Allelopathy represents then one potential ecological interaction among all complex interactions involved in the functioning of ecosystems.
Among terrestrial ecosystems, peatlands are dominated by bryophytes of Sphagnum genus which forms a homogeneous carpet. The accumulated peat is mainly dominated by remnants of Sphagnum. Indeed, peatlands are characterized by the ability to store atmospheric carbon in the long-term accumulation of partially decomposed organic matter in form of decaying vegetation. The accumulation of organic matter in peatland is the result of production and decay of Sphagnum tissues with recalcitrant carbon such as polyphenols. The transformation of phenolic compounds is performed by an enzymatic system O2 (phenoloxidases) or H2O2 (peroxydases) dependent. The interplay polyphenols/phenoloxidases is then a key process in peatland functioning. Recent research on the organo-chemical composition of Sphagnum clarified the ability of Sphagnum to outcompete other plants for light by creating acidic nutrient poor cold and anoxic conditions (Van Breemen 1995). The role of polyphenols on peatland functioning is mostly associated with the low decomposition of peat, which is of great importance for the peat storage function (Verhoeven and Liefveld 1997). Surprisingly, the exact role of phenolics remains understudy in growing Sphagnum plants. Moreover, a few studies focused on allelopathic interactions (Michel et al. 2011).
Recently, boreal peatland is of particularly interest, because it currently represents a terrestrial sink of carbon with approximately one-third of the world’s organic carbon (390–455 Pg) (Gorham 1991; Moore 2002). The expected increase of air temperatures in boreal regions is predicted to lead to a destabilization of peatland carbon stores (Smith et al. 2004; Strack 2008). Sphagnum represents the crucial plants to study the effect of elevated temperature on peatlands. Most of the studies aim at understanding effects of temperature on the peat decomposition, but not on living Sphagnum. The study of Sphagnum phenolic metabolism under a climate forcing remains a challenge even if difficult and complex.
In this chapter, we present the main characteristics of bryophytes involved in allelochemical interactions. Then, the case of Sphagnum genus will fully illustrate why bryophytes is of interest to allelopathy based on their phenolic production, degradation, and variation.
2 Terrestrial Bryophytes
2.1 Morphology
Bryophytes represent an independent branch of the plant kingdom with more than 16,000 species, taking up a position between the algae and the pteridophytes (Slack 2011). From a taxonomic point of view they are divided into three classes namely hornworts (Anthoceratae), liverworts (Hepaticae), and mosses (Bryatae). Bryophytes lack the roots, the xylem, and the phloem of vascular plants. The dominant stage is the haploid green autotrophic gametophyte rather than the diploid sporophyte which remains parasite of the gametophyte. Bryophytes play a fundamental ecological role in many terrestrial ecosystems especially in rain forest and cold biomes where they are abundant (Tan and Pocs 2000). Many species are able to live in nutrient poor conditions and are adapted to respond physiologically to intermittent periods favorable to photosynthesis (Slack 2011).
2.2 Allelochemical Interactions
Studies in various plant communities as well as greenhouse experiments have demonstrated that germination and/or seedling emergence of vascular plants are affected by mosses (Asakawa 1990). The effect may be either positive or negative depending on the habitat and the species. The positive effects are generally explained by the moisture conditions favorable to germination and the negative ones by the low light intensity, the drier microhabitat and allelopathy (Zamfir 2000). Thus, interactions between bryophytes and vascular plants comprise a large spectrum of relations including resource competition, suppression, facilitation, and allelopathy. Among these effects allelopathy seems to be the most controversial mostly because sparse experiments were conducted and among them, differences were noticed between greenhouse and field experiments (Soudzilovskaia et al. 2011). Nevertheless, original allelochemical interactions were revealed either with native or laboratory test species (Table 3.1). In boreal ecosystems, Soudzilovskaia et al. (2011) assumed that bryophytes exclusively affect germination and very early establishment but do not influence the fitness of established seedlings. Nevertheless, nothing is much known about the importance of interspecific differences among bryophytes with respect to their effect on vascular plants or the functional traits responsible for such effects.
2.3 Bryophyte Secondary Metabolites
Recent analytical techniques allow characterizing numerous natural products from bryophytes. Phenolics or polyphenolic compounds are known to represent a main class of secondary metabolites synthesized by plants (Grooss et al. 1999). They are involved in physiological plant processes (e.g. germination) but also in plant chemical interactions (Hattenschwiler and Vitousek 2000). Compared to other plants, the chemical investigation of bryophytes is a rather young discipline. There are several reasons for this: it is difficult to collect larger amounts of material, fields cultivation is not practicable and laborious procedure to have pure species (Becker 2000). Despite these difficulties, a large number of natural products, some of them with novel and unique skeletons, have been isolated from bryophytes during the past 30 years.
Because bryophytes are nonvascular plant, their secondary metabolism is limited/different compared to vascular plants. Even if flavonoids remain one of the most studied group, only a small percentage has been studied in details because most of the effort having been devoted to species of Marchantiales, Jungermanniales and Bryales (Markham 1990). Remarkable diversity of terpenoids has also been showed to be produce by liverworts (Geiger 1990). Nevertheless, investigations directed to the biosynthesis of such compounds in nonvascular plants are still rare compared to the numerous studies on terpenoid metabolism in vascular plants. Another example is the still controversial presence of lignin in bryophytes, because degradation’s studies on bryophyte tissues did not recovered the usual lignin degradation products (Geiger 1990; Ligrone et al. 2008). In view of the limited sampling, any generalization on bryophyte secondary metabolite is then difficult to make.
3 The Case of Sphagnum
Among Bryophyte classes, Sphagnum genus takes a particular place because of its fundamental ecological role in peatlands especially for the global sequestering carbon. Even if Sphagnum is the dominant plant in peats in terms of biomass and productivity, sparse data are focused on Sphagnum chemical interaction.
3.1 Sphagnum Secondary Metabolites
The Sphagnum genus, consisting of around 300 species, is worldwide distributed and is a dominant component of peat bog vegetation (Opelt et al. 2007). Sphagnum is morphologically characterized by two distinct layers : (1) the living layer or top layer called “the capitulum” where the Sphagnum grow by around 1 mm/year and where the main physiological process occurred and (2) the decaying layer or bottom layer where the decay constitutes the accumulation of peat (Fig. 3.1). The limit between the two layers is generally considered as 0–3 or 0–6 cm for the capitulum (0 from the top of the capitulum) and 3–10 or 6–10 cm for the bottom layer. The deepest layer (>10 cm) constitutes the peat soil itself.
Representation of S. fallax top and bottom layers with their respective total water-soluble phenolic concentrations expressed in mg g−1 DM. Mean ± S.E. (n = 12). Asterisk indicates significant differences between the two Sphagnum layers (Anova test P < 0.05) (adapted from Jassey et al. 2011a)
All vascular plant organs contain varying amounts of chemicals with potential allelopathic effects. Nevertheless, here, only leaves represent the key living organ of Sphagnum because of their important biomass compared to stems. Sphagnum leaves are not fully protected from the surrounding environment because no cuticle is present. Moreover, Sphagnum leaves stored around 90 % of water in their hyaline cells. Thought that the exchange of water, from atmosphere, leachates and litter, a direct relation with Sphagnum surrounding environment occurs, and so ables easily chemical interactions.
The secondary metabolism of Sphagnum differs from vascular plants. For example, Sphagnum synthesizes specific polyphenolic compounds such as sphagnum acid (a cinnamic acid) synthesized via the Shikimate pathway (Rasmussen et al. 1995; Rudolph and Samland 1985). Another specific molecule is the flavonoid sphagnorubin, which is a red pigment which represents a natural constituent of cell wall of S. magellanicum (Tsutschek 1982).
3.2 Sphagnum Water Phenolic Production and Degradation
The first major group of secondary metabolite found in Sphagnum is the phenolics (Verhoeven and Liefveld 1997). To determine phenolic compounds produced by living sphagnum, the extraction method is crucial and depend on the goal of the study. Water to more polar solvents are commonly used and give different phenolic quantification recovery from the water extractable to the wall bound fractions. For example, Sphagnum species (S. magellanicum, S. fallax, S. cuspidatum) produced sphagnum acid, p-hydroxyacetophenone, hydroxy butenolide, p-hydroxybenzoic acid, p-coumaric acid, and trans cinnamic acid from 0.1 μmol g−1 DM up to 2.9 μmol g−1 DM (Fig. 3.2, Rasmussen et al. 1995). The phenolic profile was found to be species dependent (Opelt et al. 2007). To test allelopathic effect, the plant water extraction is commonly used.
Phenolic compounds quantified in S. magellanicum and S. fallax (adapted from Rasmussen et al. 1995)
In our work, S. fallax phenolics were extracted in cold water and quantified by using the Folin–ciocalteu reagent and gallic acid as standard (Jassey et al. 2011b). Phenolics were mainly recovered in the capitulum (1 mg g−1 DM) compared to the bottom layer (0.6 mg g−1 DM) (Fig. 3.1). These results clearly identified the active allelopathic part of Sphagnum as the top layer and the passive allelopathic part as the bottom layer. To conclude, the 0–10 cm Sphagnum layer gives a realistic allelopathic potential of Sphagnum.
The degradation of the recalcitrant polyphenolic plant residue represents a key process in the global carbon cycle of a peatland. A large diversity of extracellular enzymes, described as phenoloxidases, is involved in the degradation of polyphenolic compounds (Sinsabaugh 2010; Theuerl and Buscot 2010). Extracellular phenoloxidases are divided into phenoloxidases O2 dependent (e.g. included laccases and tyrosinases; hereafter named phenoloxidases) and phenoloxidases H2O2 dependent (e.g. lignin and manganese peroxidases; hereafter named peroxidases). Generally, peroxidases are considered to be produced by basidiomycetes or ascomycetes but a recent work demonstrated that main peroxidase activities in an oak forest litter had a plant origin (Alarcón-Gutiérrez et al. 2009). According to Sinsabaugh (2010), peatlands represent a particular focus for studies of phenoloxidases and peroxidases activities because of their large role in oxidizing recalcitrant carbon such as polyphenolics.
In a recent study, we measured variations of phenoloxidase and peroxidase activities in the Sphagnum peatland of Frasnes (Jura mountains, France). Extracellular enzymes were extracted using a specific extraction reagent (0.1 M CaCl2 with 0.05 % Tween 80 and 20 g PVPP). After centrifugation, the extracts were concentrated in a cellulose dialysis with a 10 kDa molecular mass cut-off covered with polyethylene glycol (Jassey et al. 2011c). Enzymes activities were quantified using diaminofluorene, which represents a relevant oxidative substrate to quantify both plant peroxidases and fungal phenoloxidases in an enzymatic extract (Criquet et al. 2001; Jassey et al. 2012).
A negative correlation between total phenolics and fungal phenoloxidase activities was found (respectively, r = −0.42 and r = −0.38, P < 0.01) (Jassey et al. 2011c). Phenolics at high concentration could inhibit the oxidation activity of fungal phenoloxidases. A regulation of the fungal phenoloxidases by phenolics is then suggested. Ultimately, our results also reinforce the point that phenoloxidase/polyphenol interplay is especially critical to understanding peatland functioning.
Sphagnum peroxidases constituted the main oxidative system in Sphagnum peatlands, with values 1000-fold higher than fungal phenoloxidase activities. The highest level of peroxidase activities was measured in the top layer (0–3 cm), whereas fungal phenoloxidase activities were mainly detected in the deep layer (3–10 cm) (Fig. 3.3). Similar differences have been already recorded, but in the surface litter of forest with plant peroxidases 120-fold higher than fungal phenoloxidases (Alarcón-Gutiérrez et al. 2009).
Enzyme activities of two S. fallax layers 0–3 cm (top) and 3–10 cm (bottom) (October 2010): Phenoloxidases (a) and Peroxidases (b). Enzymatic activities were expressed in enzyme units (U) defined as one μmol of substrate oxidized per min−1 (peroxidase activities) or per h−1 (laccase activities) per gram of Dry Mass. Mean ± S.E. (n = 6). Asterisks indicate significant differences of enzyme activities among Sphagnum segments (Anova tests, P < 0.05) (adapted from Jassey et al. 2012)
3.3 Seasonal Variations of Phenolics in Ambient and Warming Environment
In most ecosystems, seasonal variations of phenolics have been quantified to characterized allelochemical potential (Gallet and Lebreton 1995). Surprisingly, in peatlands, such data are scarce (Bonnett et al. 2006).
Seasonal variations of phenolics, fungal phenoloxidases involved in their degradation were quantified in the Frasne peatland (Jura mountains, France) in two ecological conditions. Briefly, the first ecological area was a transition Sphagnum-dominated poor fen characterized by a flat homogeneous carpet of S. fallax (hereafter called “Fen”) and the second one was a Sphagnum bog characterized by lawns of S. fallax and hollows of S. magellanicum associated with vascular plants (such as Eriophorum vaginatum, Vaccinium oxycoccos) (hereafter called “Bog”) (Jassey et al. 2011c). Recovered Sphagnum phenolic concentrations were found to depend on the season with a decrease in summer for the bog area and in autumn for the fen area (Fig. 3.4). Sphagnum was able to regulate its secondary metabolism by decreasing its phenolic concentration when environmental conditions changed. Indeed, in summer in the bog area the air temperature increased and the Sphagnum moisture decreased inducing a decrease of total phenolics (Jassey et al. 2011c).
Seasonal variations of water-soluble phenolic concentrations (expressed in mg g−1 DM) in two S. fallax layers (top a, c and bottom b, d) recovered in the bog (a, b) and fen (c, d) areas of the Frasne peatland. Mean ± S.E. (n = 3). Asterisk indicates significant difference among months (ANOVA tests, P < 0.05) (adapted from Jassey et al. 2011c)
Because the increased decomposition rates in peatlands with global warming might increase the release of atmospheric greenhouse gases (i.e. CO2), Sphagnums are ideal plants for climate change indication. Most of the literature focused on the effects of temperature on the decomposition of litter from boreal peatlands. Cautious conclusions suggest that not all peatlands may provide a positive feedback to global warming (i.e. differences between the fen and bog areas) (Weltzin et al. 2003). Anyway, sparse data are available on living Sphagnum and their response to a global warming.
In the Frasne peatland, an increase of air and soil temperatures was passively achieved by placing hexagonal ITEX open-top-chambers over the vegetation. In spring and summer (may to September), the OTC’s significantly increased the daily maximum air temperature by an average of 3 °C and the average air temperature by 1 °C (Jassey et al. 2011c).
In response to the climate forcing, the phenolic concentrations decreased in the fen area irrespective of the seasons (Fig. 3.5) but not in the bog area. Sphagnum carbon allocation between primary and secondary metabolism changed. Sphagnum diminished its allocation to polyphenols to favor growth or crucial synthesis to better survive (Veteli et al. 2007). Indeed, biomass allocation is an important plant trait that responds plastically to environmental perturbations. Our results also highlighted different responses of Sphagnum phenolics along the fen-bog gradient (Jassey et al. 2011c). More studies with long-term conclusion with more than 1 year are needed to draw complete conclusion on the effect on global warming on Sphagnum.
Seasonal variations of water-soluble phenolic concentrations (expressed in mg g−1 DM) in two S. fallax layers (top a and bottom b) recovered in controls and OTC in the fen areas of the Frasne peatland. Mean ± S.E. (n = 3). Asterisk indicates significant difference between control and OTC (ANOVA tests, P < 0.05) (adapted from Jassey et al. 2011c)
4 Sphagnum Allelopathic Interactions
Sphagnum-dominated peatlands were at one time erroneously believed to be devoid of microbial life because of the acid pH. In reality, a high diversity of microbial communities lives in Sphagnum mosses, among which decomposers (bacteria and fungi), primary producers (microalgae, diatoms and cyanobacteria), protozoa, and micrometazoa (ciliates, testate amoebae, rotifers, and nematodes). These microorganisms are of greatest interest in peatland functioning because they act on nutrient cycling, mainly carbon and nitrogen, via the microbial loop (Gilbert et al. 1998; Gilbert and Mitchell 2006; Thormann 2006).
Because living Sphagnum has specific morphological, physiological, and ecological properties and are associated with a specific microbial loop, we proposed to use the original term “sphagnosphere” to represent such microecosystem. Thus, we define the “sphagnosphere” as a specific microecosystem between living Sphagnum and its associated microorganisms. “Sphagnosphere” represents the first scale in peatlands allowing determining any relation with its surrounding environment.
4.1 Effect of Sphagnum Extract on Sphagnum Associated MicroOrganisms
The dynamic of Sphagnum associated microorganisms is usually explained by physical and chemical factors (e.g. water level, pH, redox potential). Unfortunately, the allelochemical interactions between Sphagnum and its associated microbial communities are rarely studied. Recently, Jassey et al. (2011a, b) suggested that phenolic compounds were involved in the structure of microbial assemblages in Sphagnum peatlands, especially on testate amoebae communities. Testate amoebae are abundant and diverse-shelled protozoa living in a wide range of habitats from soils to peatlands (Mitchell et al. 2008). They are an important group in microbial food web because they feed on a wide range of prey such as bacteria, algae, fungi, rotifers, nematodes, or other small testate amoebae. Owing to their role in microbial food web, they play a crucial role in the microbial loop and nutrient cycling (Gilbert et al. 2003; Wilkinson and Mitchell 2010). In Sphagnum peatlands, testate amoebae communities were strongly correlated to surface wetness conditions and water chemistry (Mitchell et al. 2008). Jassey et al. (2011a) demonstrated for the first time that phenolics explained a variance partitioning of 25.6% as physical factors (34.1%) and as chemical factors (16.8%) of their community composition along Sphagnum shoots. It strongly suggested the potential role of these compounds in their autecology. More precisely, a relationship between testate amoebae and polyphenols was confirmed. For example, the density of Euglypha strigosa was significantly correlated to the concentrations of Sphagnum water-soluble phenolics (Fig. 3.6). Although such result does not allow a conclusion to be strongly drawn on a possible direct positive effect (or indirect effect on the food web) of phenol released by Sphagnum on E. strigosa, it raises the issue of the possible role of phenolics on microbial communities.
Euglypha strigosa density (ind.g−1 DM) plotted against the concentrations of S. fallax water-soluble phenolics (mg g−1 DM) (ANOVA test, P = 0.02). E. Strigosa photo is reproducted, with kindly permission of E.A.D. Mitchell (adapted from Jassey et al. 2011c)
4.2 Effect of Sphagnum Extract on Germination and Early Growth of Vascular Plants
The bulk of research on allelopathy focuses on the visible effects of allelochemicals (secondary effects) particularly on germination and growth. These effects are tested with plant extract when the compounds have not been yet identified as it is the case for Sphagnum (Chiapusio et al. 1997).
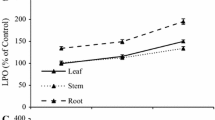
The experimental procedure involved testing the effect of S. fallax extract on the germination and early growth of three vascular species a monocot Lolium perenne, a dicot Raphanus sativa, and a local gymnosperm peatland species Pinus uncinata. Extracts of S. fallax were realized by pressing by hands Sphagnum plants. The obtained extract was (1) directly used in the germination experiments and corresponded to the 100 % extract and (2) diluted in distilled water to obtain a 75 % extract. The pH of each Sphagnum extract was measured and other controls were also realized with the same pH. As no statistical difference between the “normal control” and the “buffered pH” was found, we concluded that the pH had no effect on the germination process. A 100 % Sphagnum extract and the 75 % extract had a drastic negative effect on Lolium germination (7- and 4-times, respectively less than the control) but also on Pinus germination (two times less than the control for both treatments) (Fig. 3.7a). On the contrary, no effect on germination of radish seeds was observed. Differences between monocot and dicot could explain such germination differences but need complementary experiments. Sphagnum extract on the radicle growth was drastic for all tested species (Fig. 3.7b). Indeed, inhibition of root was noticed for radish (30 %) and for Pinus (40 %) for both treatments. Moreover, when the Lolium seeds germinate they were then not able to develop their radicle in both Sphagnum extracts. The radicle growth was totally suppressed.
Effect of S. fallax extract on A the germination (%) of Lolium perenne, Raphanus sativus, and Pinus uncinata. Mean ± S.D. (n = 5). (Mann–Whitney test, P < 0.05) and on B radicle length (mm) of L. perenne, R. sativus, and P. uncinata. Mean ± S.D. (n = 5). (Mann–Whitney test, P < 0.05) (Unpublished data)
Living Sphagnum extracts offered interesting perspectives for allelochemical interactions in the functioning of peatlands to understand vegetation dynamics. Similar results, but with other bryophytic genus, were described (Zamfir 2000). Indeed, the homogeneous mat of Sphagnum is known to be due to low nutrient availability, anoxia, low temperatures, and high acidity (Van Breemen 1995). Along our results, Sphagnum ability to compete other plant species, especially vascular plants, could also be due to allelochemical production. The term interference, combining competition, and allelopathy becomes relevant then.
Moreover, the allelochemical Sphagnum extract could also be a good candidate as biopesticides (Chiapusio et al. 2005). Observed inhibitions of germination and growth are promising comparing with other allelochemicals (e.g. 2-benzoxazolinone, Chiapusio et al. 2004; Hussain et al. 2008). Such results constitute a crucial step for further experiments.
5 Conclusions
Water-soluble phenolics produced by living Sphagnum were found to be of great interest for the functioning of peatlands. First, they were concentrated in the top layer of Sphagnum and their concentrations varied along the seasons. Sphagnum peroxidases constituted the main oxidative system and fungal-phenoloxidases were proposed to be inhibited by phenolics. In such context, the polyphenolic phenoloxidase interplay is especially crucial to understand the mechanisms by which peatlands react under a climatic change. Indeed, peatlands could become weaker sinks of carbon but depends on their proper ecological conditions (e.g. ecological gradient).
Second, our hypothesis of allelopathic interactions in peatlands made sense with the evidence of original interactions between Sphagnum phenolics and its associated microorganisms, the “sphagnosphere.” Of course, allelopathy was not considered as the only factor explaining microbial dynamic and microbial food chain but was revealed to be fully considered and not neglected. Moreover, the drastic inhibition/delay of Sphagnum extract on germination and radicle growth of vascular plants could also explain the Sphagnum dominance under surrounding plants.
We still have a lot to learn from bryophyte secondary metabolite, not only to better understand their ecological role but also to discover original natural products that could be used as natural biopesticide. However, research is still needed on the identification and the biodegradability of bryophyte allelochemicals, which are largely unknown until now.
References
Alarcón-Gutiérrez E, Floch C, Augur C, Le Petit J, Ziarelli F, Criquet S (2009) Spatial variations of chemical composition, microbial functional diversity, and enzyme activities in a Mediterranean litter (Quercus ilex L.) profile. Pedobiologia 52:387–399
Asakawa Y (1990) Terpenoids and aromatic compounds with pharmacological activity from bryophytes. In: Zinsmeister HD, Mues R (eds) Bryophytes their chemistry and chemical taxonomy. Claredon Press, Oxford, pp 367–410
Becker H (2000) Bryophyte in vitro cultures, secondary products. In: Spier RE (ed) Encyclopedia of cell technology, vol 1. Wiley, New York, pp 278–283
Bonnett SAF, Ostle N, Freeman C (2006) Seasonal variations in decomposition processes in a valley-bottom riparian peatland. Sci Total Environ 370:561–573
Chiapusio G, Sanchez AM, Reigosa MJ, Gonzalez L, Pellissier F (1997) Do germination indices adequately reflect allelochemical effects on the germination process? J Chem Ecol 23:2445–2453
Chiapusio G, Gallet C, Pellissier F (2004) Uptake and translocation of phytochemical 2-benzoxazolinone (BOA) in radish seeds and seedlings. J Exp Bot 55:1587–1592
Chiapusio G, Gallet C, Dobremez JF, Pellissier F (2005) Allelochemicals: tomorrow’s herbicides? In: Regnaud-Roger C, Philogène BJR, Vincent C (eds) Biopesticides of plantorigin. Intercept Ltd., Lavoisier Publ. Inc., Hampshire, pp 149–155
Criquet S, Joner EJ, Leyval C (2001) 2,7-Diaminofluorene is a sensitive substrate for detection and characterization of plant root peroxidase activities. Plant Sci 161:1063–1066
Equiha M, Husher MB (1993) Impact of carpets of the invasive moss Campylopus introflexus on Calluna vulgaris regeneration. J Ecol 81:359–365
Gallet C, Lebreton P (1995) Evolution of phenolic patterns in plants and associated litters and humus of a mountain forest ecosystem. Siol Biol Biochem 31:1151–1160
Geiger H (1990) Biflavonoids in bryophytes. In: Zinsmeister HD, Mues R (eds) Bryophytes their chemistry and chemical taxonomy. Claredon Press, Oxford, pp 143–161
Gilbert D, Mitchell EAD (2006) Microbial diversity in Sphagnum peatlands. In: Martini IP, Matinez Cortizas A, Chesworth W (eds) Peatlands: basin evolution and depository of records on global environmental and climatic changes. Chapman & Hall, New York, pp 287–318
Gilbert D, Amblard C, Bourdier G, Francez AJ (1998) The microbial loop at the surface of a peatland: structure, function, and impact of nutrient input. Microb Ecol 35:83–93
Gilbert D, Mitchell EAD, Amblard C, Bourdier G, Francez AJ (2003) Population dynamics and food preferences of the testate amoeba Nebela tincta major-bohemica-collaris complex (Protozoa) in a Sphagnum peatland. Acta Protozoo 42:99–104
Gorham E (1991) Northern peatlands: role in the carbon cycle and probable responses to 510 climatic warming. Ecol Appl 1:181–195
Grooss GG, hemingway RW, Yoshida T (1999). Plant polyphenols 2: chemistry, biology, pharmacology, ecology. Kluwer academic publishers, New York
Hattenschwiler S, Vitousek PM (2000) The role of polyphenols in terrestrial ecosystem nutrient cycling. Tree 15:238–243
Hussain MI, Gonzalez-Rodriguez L, Reigosa MJ (2008) Germination and growth response of four plant species to different allelochemicals and herbicides. Allelopathy J 22:101–108
Jassey VEJ, Chiapusio G, Mitchell EAD, Binet P, Toussaint ML, Gilbert D (2011a) Fine-scale horizontal and vertical micro-distribution patterns of testate amoebae along a narrow fen/bog gradient. Microb Ecol 61:374–385
Jassey VEJ, Gilbert D, Binet P, Toussaint M-L, Chiapusio G (2011b) Effect of a temperature gradient on Sphagnum fallax and its associated microbial communities: a study under controlled conditions. Can J Microb 57:226–235
Jassey VEJ, Chiapusio G, Gilbert D, Buttler A, Toussaint ML, Binet P (2011c) Experimental climate effect on seasonal variability of polyphenol/phenoloxidase interplay along a narrow fen-bog ecological gradient. Glob Change Biol 17:2945–2957
Jassey VEJ, Chiapusio G, Gilbert D, Toussaint ML, Binet P (2012) Phenoloxidase and peroxidase activities in a Sphagnum-dominated peatland in a warming climate. Soil Biol Bioch 46:49–62
Kato-Noguchi H, Seki T, Shigemori (2010) Allelopathy and allelopathic substance in the moss Rhynchostegium pallidifolium. J Plant Physiol 167:468–471
Ligrone R, Carafa A, Duckett JG, Renzaglia KS, Ruel K (2008) Immunocytochemical detection of lignin related epitopes in cell walls in bryophytes and the charalean alga Nitella. Plant Syst Evol 270:257–272
Markham KR (1990) Bryophyte flavonoids, their structures, distribution, and evolutionary significance. In: Zinsmeister HD, Mues R (eds) Bryophytes their chemistry and chemical taxonomy. Claredon Press, Oxford, pp 143–161
Michel P, Burritt DJ, William G (2011) Lee Bryophytes display allelopathic interactions with tree species in native forest ecosystems. Oikos 120:1272–1280
Mitchell EAD, Charman DJ, Warner BG (2008) Testate amoebae analysis in ecological and paleoecological studies of wetlands: past, present and future. Biodiv Conserv 17:2115–2137
Moore PD (2002) The future of cool temperate bogs. Environ Conserv 29:3–20
Nosaki H, Hayashi KJ, Nishimura N, kawaide H, Matsuo A, Takaoka D (2007) Momilactone A and B as allelochemicals from moss Hypnum plumaeforme: first occurrence in bryophytes. Biosci Biotech Biochem 71:3127–3130
Opelt K, Chobot V, Hadacek F, Schonmann S, Eberl L, Berg G (2007) Investigations of the structure and function of bacterial communities associated with sphagnum mosses. Environ Microbiol 9:2795–2809
Rasmussen S, Wolff C, Rudolph H (1995) Compartmentalization of phenolic constituents in Sphagnum. Phytochem 38:35–39
Rudolph H, Samland J (1985) Occurrence and metabolism of Sphagnum acid in the cell walls of bryophytes. Phytochem 24:745–749
Sinsabaugh RL (2010) Phenol oxidase, peroxidase and organic matter dynamics of soil. Soil Biol Biochem 42:391–404
Slack NG (2011) The ecological value of Bryophytes as indicators of climate change. In: Tuba Z, Slack NG, Stark LR (eds) Bryophytes, Ecology and climate change. Cambridge University Press, Cambridge, pp 5–12
Smith LC, MacDonald GM, Velichko AA, Beilman DW, Borisova OK, Frey KE, Kremenetski KV, Sheng Y (2004) Siberian peatlands a net carbon sink and global methane source since the early Holocene. Science 303:353–356
Soudzilovskaia NA, Graae BJ, Douma JC, Grau O, Milbau A, Shevtsova A, Wolters L, Cornelissen JHC (2011) How do bryophytes govern generative recruitment of vascular plants? New Phytol 190:1019–1031
Souto XC, Chiapusio G., Pellissier F (2000) Relationships between phenolics and soil micro-organims in spruce forest : significance for natural regeneration. J Chem Ecol 26:2025–2034
Strack M (2008) Peatlands and climate change. International Peat Society, Jyväskylä, p 235
Tan BC, Pocs T (2000) Bryogeography and conservation of bryophytes. In: Shaw AJ, Goffinet B (eds) Bryophyte biology. Cambridge University Press, Cambridge, pp 403–448
Theuerl S, Buscot F (2010) Laccases: toward disentangling their diversity and functions in relation to soil organic matter cycling. Biol Fert Soils 46:215–225
Thormann MN (2006) Diversity and function of fungi in peatlands: a carbon cycling perspective. Can J Soil Sci 86:281–293
Tsubota H, Kuroda A, Masuzaki H, Nakahara M, Deguchi H (2006) Premiminary study on allelopathic activity of bryophytes under laboratory conditions using the sandwich method. J Hattori Bot Lab 100:517–525
Tsutschek R (1982) Influence of L-α-aminooxy-β-phenylpropionic acid with cold-induced sphagnorubin synthesis in Sphagnum magellanicum BRID. Planta 155:307–309
Van Breemen N (1995) How Sphagnum bogs down other plants. Tree 10(7):270–275
Verhoeven JTA, Liefveld WM (1997) The ecological significance of organochemical compounds in Sphagnum. Acta Botanica Neerlandica 46:117–130
Veteli TO, Mattson WJ, Niemela P, Julkunen-Tiitto R, Kellomaki S, Kuokkanen K, Lavola A (2007) Do elevated temperature and CO2 generally have counteracting effects on phenolic phytochemistry of boreal trees? J Chem Ecol 33:287–296
Viard-Crétat F, Gallet C, Lefebvre M, Lavorel S (2009) A leachate a day keeps the seedlings away: mowing and the inhibitory effect of Festuca paniculata in subalpine grasslands. Ann Bot 103:1271–1278
Weltzin JF, Bridgham SD, Pastor J, Chen JQ, Harth C (2003) Potential effects of warming and drying on peatland plant community composition. Glob Change Biol 9:141–151
Wilkinson DM, Mitchell EAD (2010) Testate amoebae and nutrient cycling with particular reference to soils. Geomicrob J 27:520–533
Zamfir M (2000) Effects of bryophytes and lichens on seedling emergence of alvar plants: evidence from greenhouse experiments. Oikos 88:603–611
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Chiapusio, G., Jassey, V.E.J., Hussain, M.I., Binet, P. (2013). Evidences of Bryophyte Allelochemical Interactions: The Case of Sphagnum . In: Cheema, Z., Farooq, M., Wahid, A. (eds) Allelopathy. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-30595-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-642-30595-5_3
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-30594-8
Online ISBN: 978-3-642-30595-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)