Abstract
Stilbenoids are a class of plant phenolics containing C6–C2–C6 unit in their structures and classified into five groups, covering stilbenes, oligostilbenes, bibenzyls, bisbibenzyls, and phenanthrenes. They have been a hot research topic for their intricate structures and diverse biological activities. Resveratrol and combretastatin A-4 are the star compounds due to their potent cardioprotective, chemopreventive, and antitumor properties and have the potential to be developed as new drugs. The stilbenoids do not enjoy a wide distribution and are only found in special genus. Although the constituent unit is simple, the structures of stilbenoids highlight the chemical diversity by different substitutes and various oligomeric styles. In a biogenesis viewpoint, they are formed by a branch of the flavonoid biosynthetic pathway. This chapter provides a summary of the occurrence, phytochemisty, biosynthesis, and biological aspects of the stilbenoids.
Access provided by Autonomous University of Puebla. Download reference work entry PDF
Similar content being viewed by others
Keywords
- Bibenzyls
- bioactivities
- biosynthesis
- bisbibenzyls
- occurrence
- oligostilbenes
- phenanthrenes
- phytochemistry
- stilbenes
- stilbenoids
1 Introduction
The term “stilbenoids” was proposed by Gorham in 1980 [1, 2], which refers to a class of plant phenolics with 1,2-diphenylethylene or 1,2-diphenylethane nucleus in their structures. Stilbenoids are regarded as plant phytoalexins and have been a hot research topic for their intricate structures and diverse biological activities. The phytochemical research concerning the stilbenoids developed quickly in recent years. More than 1,000 compounds belonging to this group have been discovered, compared with just over 100 listed in 1980 and about 300 in 1995 [2]. Recent advances in analytical and spectroscopic techniques, especially the NMR methods, speed up the discovery and elucidation of the intricate structures of stilbenoids. The intricate structures and stereochemistry of oligostilbenes and bisbibenzyls were established based on the modern techniques. Furthermore, these compounds demonstrated diverse biological activities, including antitumor, antimicrobial, antioxidant effects, antiplatelet aggregation, phytotoxicity, etc. These bioactive compounds and their derivatives are of great interest for drug research and development as a result of their potential in therapeutic or preventive applications, exemplified by resveratrol and combretastatin A-4. In this chapter, we will give an overview of structural features, occurrence, phytochemical aspects, biosynthesis, and biological activities of the stilbenoids.
2 Phytochemical Aspects
According to their structural characteristics, stilbenoids are mainly divided into five categories, stilbenes, oligostilbenes, bibenzyls, bisbibenzyls, and phenanthrenes. In the section of phytochemical aspects, the structural characteristic, distribution, typical representatives, and their structures of each group are introduced.
2.1 Stilbenes
Stilbenes possess a skeleton with two aromatic rings joined by a methylene bridge. The simple stilbene nucleus is generally substituted by different groups of hydroxyl, methyl, methoxy, prenyl, geranyl, etc., and combined with sugars to form glycosides. The double bonds in naturally occurring stilbenes are usually E-configuration, but stilbenes with Z-configuration are also observed. The compounds of this group highlight the chemical structural diversity through the modification of above styles on the nucleus. About 125 new stilbenes have been discovered between the year of 1995 and 2008 [3]. They mainly occur in the families of Aceraceae, Anchinoidae, Asteraceae, Bombycidae, Burseraceae, Combretaceae, Cyperaceae, Dipterocarpaceae, Euphorbiaceae, Gnetaceae, Hepaticae, Iridaceae, Leguminosae, Lejeuneaceae, Liliaceae, Meliaceae, Moraceae, Ophioglossaceae, Orchidaceae, Polygonaceae, Rosaceae, Stemonaceae, Vitaceae, and Zingiberaceae.
Resveratrol 1 is the most famous representative of this group and occurs in Polygonum cuspidatum root and Vitis species. It is a phytotoxin produced by several plants in response to infection or other stresses and attracted attention for its cardioprotective effect in red wine. In addition, it is the most important unit for the construction of oligomeric stilbenes.
Combretastatins, a series of bioactive stilbenes (combretastatin A series), bibenzyls (B series), phenanthrenes (C series), and macrocyclic lactone (D series), were obtained from the African willow tree Combretum caffrum (Combretaceae). Among them, the A series of combretastatins belonging to stilbenes, including combretastatins A-1 to A-6 2–7, are found to be tubulin polymerization inhibitors.
Typical stilbenes substituted with hydroxyls, methyl, methoxy, menthane groups, and their origin were listed as follows. Trans-4-[2-(3,5-dimethoxyphenyl)ethenyl]-1,2-benzenediol 8 was isolated from Sphaerophysa salsula (Leguminosae) [4]. Thunalbene 9 was obtained from Thunia alba (Orchidaceae), and the structure was designated as 3,3′-dihydroxy-5-methoxystilbene [5]. The Phragmipedium species produced three new stilbenes including 2,3′-dihydroxy-5′-methoxystilbene 10, 2,3-dihydroxy-3′,5′-dimethoxystilbene 11, and 2,3′-dihydroxy-5,5′-dimethoxystilbene 12 [6]. Phoyunbenes A-D 13–16 were found in Pholidota yunnanensis (Orchidaceae) [7]. 5,4′-Dihydroxy-3-methoxystilbene 17, 3,5-dihydroxy-4′-methoxystilbene 18, and (E)-3,3′-dimethoxy-4,4′-dihydroxystilbene 19 were isolated from Rumex bucephalophorus and Leuzea carthamoides [8, 9], respectively. Two Z-type stilbenes named (Z)-3-methoxy-5-hydroxystilbene 20 and (Z)-3,5-dihydroxystilbene 21, together with a menthane-substituted stilbene (E)-1-(1-terpinen-4-olyl)-3-methoxystilbene 22, were obtained from aerial parts of Alpinia katsumadai (Zingiberaceae) [10, 11].
There are two stilbene representatives isolated from special origins. Bryophytes are characterized by the production of bisbibenzyls, and no stilbene has been obtained before the isolation of 3,4-dihydroxy-3′-methoxystilbene 23 from Marchesina bongardiana (Lejeuneaceae) [12]. Kirkpatrickia variolosa, a kind of Antarctic red sponge of Anchinoidae family, yielded a triacetate derivative 3,4,5-triacetoxystilbene 24 which was the only marine natural stilbene [13].
Two stilbene glycosides, named (E)-3,4′-dimethoxyl-5-rutinosyl stilbene 25 and 3,5-dimethoxy-4′-O-(β-rhamnopyranosyl-(1 → 6)-β-glucopyranoside)stilbene 26, were isolated from Guibourtia tessmanii (Leguminosae) [14, 15]. Acer mono (Aceraceae), a Korean folk medicine for hemostasis, produced two new stilbene glycosides 5-O-methyl-(E)-resveratrol 3-O-β-d-glucopyranoside 27 and 5-O-methyl-(E)-resveratrol 3-O-β-d-apiofuranosyl-(1 → 6)-β-d-glucopyranoside 28 [16].


Prenyl substitution is a conventional derivatization style in stilbenes, exemplified by artoindonesianin N 29 from Artocarpus gomezianus [17]. The prenyl groups in stilbenes cyclize to form new derivatives. 4-Hydroxy-5′-methoxy-6″,6″-dimethylpyran[2″,3″:3′,4′]stilbene 30, 3,5′-dimethoxy-4-hydroxy-6″,6″-dimethylpyran[2″,3″:3′,4′]stilbene 31, and 3,4,5-trimethoxy-6″,6″-dimethylpyran[2″,3″:3′,4′]stilbene 32, with dimethylchromene ring in their structures, have been obtained from Lonchocarpus utilis (Leguminosae) [18]. Furthermore, schweinfurthins A-C 33–35 from the leaves of Macaranga schweinfurthii (Euphorbiaceae) are typical samples of the prenylated stilbenes [19].

Arylbenzofuran derivatives are a group of special stilbenes formed by C7–O–C7 linkage, for instance, gnetofurans B 36 and C 37 from Gnetum klossii [20]. In addition, schoenoside 38, a phenylbenzofuran glucoside discovered from Schoenocaulon officinale (Liliaceae), as well as stemofurans A-C 39–41 from Stemona collinsae belongs to this group [21, 22].

2.2 Oligostilbenes
The structures of oligostilbenes are produced by coupling between homogeneous or heterogeneous monomeric stilbenes, leading to the construction of dimer, trimer, and even the octamer. They do not enjoy a wide distribution in plant kingdom and have been found in the family of Agavaceae, Apiaceae, Arecaceae, Celastraceae, Cyperaceae, Dipterocarpaceae, Gnetaceae, Haemodoraceae, Iridaceae, Leguminosae, Moraceae, Musaceae, Orchidaceae, Pinaceae, Polygonaceae, Ranunculaceae, Vitaceae, and Welwitschiaceae. Thereinto, Vitaceae, Leguminosae, Gnetaceae, and Dipterocarpaceae are particular rich resource of this group.
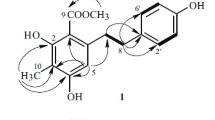
Oligostilbenes are constructed by C–C or C–O–C linkage of various stilbene units with diverse coupling patterns and producing structures with diverse skeletons, complex configurations, and different degrees of oligomerization. The most common monomeric stilbene units which comprised the oligostilbenes are resveratrol, isorhapontigenin, piceatannol, oxyresveratrol, etc. (Fig. 62.1). Therefore, oligostilbenes are classified into six groups which are resveratrol oligomers, isorhapontigenin oligomers, piceatannol oligomers, oxyresveratrol oligomers, resveratrol and oxyresveratrol oligomers, and finally miscellaneous oligomers [3].
2.2.1 Resveratrol Oligomers
The group of resveratrol oligomers comprises the largest number of oligostilbenes and is characterized by the polymerization of two to eight resveratrols. About 180 constituents of this group covering dimer to octamers have been reported, which is produced by diverse polymeric styles.
Vitisinol A 42 isolated from Vitis thunbergii is a dimer linked by four C–C or C–O–C bonds [23]. Two dimeric stilbene glycosides 43 and 44 were reported from Polygonum cuspidatum (Polygonaceae) [24]. Thereinto, 44 is a symmetrical molecule and possesses a novel four-membered ring which is very rare in natural products. Isoampelopsin F 45 linked by three C–C bonds and heimiol A 46 with seven-member ring were isolated from Parthenocissus tricuspidata and Neobalanocarpus heimii [25]. A novel resveratrol dimer with a five-membered lactone ring, namely, shorealactone 47, was obtained from Shorea hemsleyana [26]. Schneide reported the isolation of anigopreissin A 48 from the Anigozanthos preissii (Haemodoraceae) and Musa cavendish (Musaceae), which is the first dimer containing unsaturated benzofuan moiety [27]. Moreover, an aldehyde-substituted derivative, (−)-viniferal 49, was isolated from Vitis vinifera [28].
Two trimers containing rare tribenzobicyclo[3.3.2]decatriene system were isolated from Vatica rassak and V. pauciflora, named vaticanol G 50 and vaticaside D 51 [29, 30]. Caragaphenol A 52, with a nine-membered ring in the molecules, was found in Caragana stenophylla [31]. Three isomers which have bicyclo[5.3.0]decane ring system were obtained including amurensin G 53 from Vitis amurensis [32] and suffruticosols A 54 and B 55 from Paeonia suffruticosa (Ranunculaceae) [33].
Two new resveratrol pentamers, named amurensins E 56 and F 57, have been isolated from Vitis amurensis [34]. The isolation and structural elucidation of a hexamer vaticanol D 58 and a heptamer vaticanol J 59 from Vatica rassak have been reported [29]. An octamer vateriaphenol A 60 from Vateria indica was reported by Ito and coworkers, and it is the largest molecules of stilbenoids [35].




2.2.2 Isorhapontigenin Oligomers
Oligostilbenes of this group mainly occur in the genus of Gnetum. The representatives comprise gnetuhainins P 61 and I 62 from G. hainanense [36, 37], dimeric stilbene epimers gnetifolins M 63 and N 64 from G. montanum [38], gnemonol M 65 from G. gnemon [39], gneafricanin F 66 found in G. africanum [40], as well as bisisorhaphontigenin B 67 obtained from G. africanum [41]. With the exception of Gnetum species, Salacia lehmbachii produced three isorhapontigenin dimers, named lehmbachols A-C 68–70 [42]. The only two isorhapontigenin trimers, gnetuhainins N 71 and O 72, which are stereoisomers have been found in Gnetum hainanense [43]. A tetramer named gnetuhainin R 73 was obtained from the same species (G. hainanense) [44].


2.2.3 Piceatannol Oligomers
Only piceatannol dimers have been discovered from the plant species. Longusol C 74 and gneafricanin C 75 were isolated from Cyperus longus and Gnetum africanum [40, 45]. Tibeticanol 76 was obtained from Caragana tibetica [46]. Two piceatannol dimer glycosides named piceasides A–B 77–78 were isolated from Norway spruce Picea abies as a mixture in a ration of 1:1 [47].

2.2.4 Oxyresveratrol Oligomers
Andalasin A 79 and artogomezianol 80 were isolated from Artocarpus gomezianus (Moraceae) [48]. Structures 79 and 80 are possible intermediates in the biogenesis of oxyresveratrol dimers. Parvifolol C 81 and gnetumontanin A 82 were discovered in two Gnetum species G. parvifolium and G. montanum [49, 50].

2.2.5 Resveratrol and Oxyresveratrol Oligomers
The resveratrol and oxyresveratrol oligomers are only obtained from the genus Gnetum (Gnetaceae) and polymerize by oxidative coupling between resveratrol and oxyresveratrol. About 24 compounds of this group have been elucidated. A dimer containing a benzofuran and a dihydrobenzofuran moiety, named gnemonol G 83, was isolated from G. gnemon [51]. Four stilbene dimers, gnetuhainins A 84, B 85, D 86 and E 87, were obtained from G. hainanense [52]. Gnemonoside J 88, a diglucoside of 84, was isolated from G. africanum [53]. Three stereoisomers including parvifolols A 89 and B 90 and gnetuhainin S 91 have been founded from G. parvifolium and G. hainanense [44, 49, 54]. Gnemonol A 92 and its stereoisomers and gnemonol I 93 composed of two resveratrol units and one oxyresveratrol unit were discovered in G. gnemon [51, 55]. Gnemonol J 94 from G. gnemon possesses the same skeleton of 92 and 93; however, it is coupled by one resveratrol unit and two oxyresveratrol units [51]. Gnemonol C 95 from G. gnemonoides is a stilbene tetramer constructed by three resveratrol and one oxyresveratrol units [55].


2.2.6 Miscellaneous Oligomers
Oligostilbenes polymerize from different stilbene units with the exception of resveratrol and oxyresveratrol oligomers, and containing miscellaneous structural skeleton will be classified into this group. Longusol B 96 from Cyperus longus [45] is a stilbene dimer composed of resveratrol and piceatannol units. Gnetuhainin Q 97, an isorhapontigenin and resveratrol dimer, was found in Gnetum hainanense [36]. The first isorhapontigenin and oxyresveratrol dimer named gnetuhainin J 98 was isolated from G. hainanense [37]. An isorhapontigenin and piceatannol dimer, gneafricanin B 99 was discovered in Gnetum africanum [41]. Two isorhapontigenin and 2-hydroxyisorhapontigenin dimers were obtained from G hainanense and named gnetuhainin G 100. An isorhapontigenin and gnetol dimer gnetuhainin K 101 was isolated from G. hainanense [36].


2.3 Bibenzyls
The bibenzyls are characterized by the presence of one 1,2-diphenylethane structure in their molecules. Similar with the stilbene structures, there are hydroxyls, methyl, methoxy, prenyl, geranyl, etc., located in the structures of bibenzyls. Bibenzyls have been mainly isolated from bryophytes. In addition, a few compounds of this group were reported from the genera of Stemona, Dendrobium, and Polygonum.
According to the suggestion given by Gorm et al. [56], bibenzyl compounds are classified into four groups. However, a few groups of bibenzyls are new addition to the growing list of naturally occurring bibenzyls, such as tyrolobibenzyls. In addition, bibenzyls containing isoprene units, regardless of branched or heterocyclic ring-forming isoprene units, are regarded as the same group. Therefore, the bibenzyls are reclassified into five groups according to their substitute patterns on both the benzene nucleus and ethylidene bridge.
2.3.1 Group A: Simple Bibenzyls
Bibenzyls having halogenated, hydroxylated, methyled, methoxylated, carboxylate, benzoyl, and/or methylenedioxy substitutes constitute the group of simple bibenzyls. The representatives are 2′,6′-bis(p-hydroxybenzyl)-3,3′-dihydroxy-5-methoxybibenzyl 102 from Bletilla formosana [57], bulbophyllum 103 from Bulbophyllum protractum [58], 2-carboxy-4-hydroxy-3,4′-dimethoxybibenzyl 104 from Plagiochila species [59], 2-chloro-3-hydroxybibenzyl 105 from Riccardia marginata [60], 1-(3′,5′-dihydroxyphenyl)-2-(4′-hydroxyphenyl)-ethane-1,2-diol 106 from Polygonum cuspidatum [61], methyl 4-hydroxy-4′-O-methyllunularate 107 from Plagiochila spinulosa [62], tragopogonic acid 108 from Tragopogon porrifolius [63], and 2,4,6-trichloro-3-hydroxybibenzyl 109 from Riccardia marginata [60].

2.3.2 Group B: Isoprene Unit–Substituted Bibenzyls
Bibenzyls of this group are characterized by the presence of prenyl, geranyl, and/or farnesyl substitutes. Isoprene units in the structures may be branched and/or form five- to seven-member rings. The typical compounds of this group are bauhinols A 110 and B 111 from Bauhinia saccocalyx [64], 2-carboxy-3-methoxy-4,6-di-(3-methyl-2-butenyl)-5,4′-dihydroxy-bibenzyl 112 from Lethocolea glossophylla [65], 2-isopropenyl-6-hydro-4-(2-phenylethyl)dihydrobenzoluran 113 from Radula perrottetii [66], and 3,5,4′-trihydroxy-4-(3,7,11-trimethyl-2,6,10-dodecatrienyl)bibenzyl 114 from Radula species [67].


2.3.3 Group C: Glycosylated Bibenzyls
Bibenzyls conjugated with glycosyl substitutes on the aromatic rings or benzylic methylenes (excluding tyrolobibenzyls) belong to this group. The representatives of this group include 2′-carboxy-4,3′-dihydroxybibenzyl-3-O-β-d-glucopyranoside 115 from Ricciocarpus natans [68], 2-carboxyl-3,4′-dihydroxy-5-β-d-xylopyranosyloxybibenzyl 116 and 5,4′-dihydroxy-3-α-l-rhamnopyranosyl-(1 → 3)-β-d-xylopyranosyloxybibenzyl 117 from Tragopogon porrifolius [63], and combretastatin B-1,2′-β-d-glucoside 118 from Combretum erythrophyllum [69].

2.3.4 Group D: Tyrolobibenzyls
Tyrolobibenzyls are a new class of naturally occurring bibenzyl derivatives possessing a unique phenylethyl-benzofuran skeleton. Tyrolobibenzyls A 119, B 120, D 121, and F 122 from Scorzonera humilis L. (Asteraceae) are the typical constituents of this group [70–72].

2.3.5 Group E: Other Bibenzyls
The hydrogenated bibenzyls 123–124 from Plagiochila longispina [73], dihydrocoumarin-type bibenzyl 125 from Vittaria anguste-elongata [74], cannabinoid-type bibenzyl 126 from Radula marginata [75], and dihydrophenanthrene hybrids such as shancilin 127 from Pleione bulbocodioides, and terpenoid hybrid spinuloplagin A from Plagiochila spinulosa 128 are classified into this group.

2.4 Bisbibenzyls
Bisbibenzyls are dimeric bibenzyls and chemically constructed by two lunularin moieties with diarylether and/or biphenyl linkages, and producing cyclic and acyclic four aromatic rings system. They are usually distributed in liverworts and rarely found in other plant species. The first bisbibenzyl not obtained from liverworts is perrottetin H 149, which was isolated from a pteridophyte Hymenophyllum barbatum [2]. It is also believed that the distribution of bisbibenzyls in both pteridophytes and liverwort is an important marker of determining the evolutionary ladder of terrestrial spore-forming plants.
Bisbibenzyls are classified into ten types of cyclic bisbibenzyls and six types of acyclic bisbibenzyls on the basis of basic bisbibenzyls skeletons (see Table 62.1). Over 100 cyclic and acyclic bisbibenzyls have been obtained from plant kingdom to date.
2.4.1 Macrocyclic Bisbibenzyls
According to the constitution of the macrocyclic ring, these bisbibenzyls can be divided into three main groups, those with two diarylether bonds (marchantins, isomarchantins, neomarchantins, and riccardins II), those with two biphenyl bonds (isoplagiochin II), or those with one diarylether bond and one biphenyl bond (riccardin I, isoriccardin C, plagiochins, isoplagiochin I, and planusin A) (see Table 62.1). The range of macrocyclic structures is extended ultimately and derived from additional functions (e.g., carbonyl, hydroxyl, and methoxyl) on both the benzene nucleus and ethylidene bridge and the ways in which aromatic rings are linked.
Asakawa and his coworkers reported the isolation of a marchantin-type macrocyclic bisbibenzyl marchantin P 129 from the liverwort Marchantia chenopoda collected in Venezuela [76]. Three isomarchantin-type macrocyclic bisbibenzyls were isolated from the liverwort Ptychantus striatus, belonging to the Lejeuneaceae, and designated them as ptychantols A-C 130–132 [77]. Neomarchantins A and B 133–134 were obtained from Schistochila glaucescens [78].
Two riccardin I-type compounds, riccardins F 135 and H 136, were isolated from Blasia pusilla [79] and Marchantia polymorpha [80], respectively. Riccardin B 137 from Preissia quadrata belongs to the riccardin II-type bisbibenzyls. Isoriccardin C 138, a compound of isoriccardin group, was obtained from Plagiochila sciophila [81].
Plagiochins A-B 139 and 140 were plagiochin-type constituents from Plagiochila fruticosa [81]. Isoplagiochins E-F 141–142, belonging to the isoplagiochin I-type bisbibenzyls, have been isolated from several Plagiochila species. Isoplagiochins C 143 and D 144 with two biphenyl linkages, which are different from isolagiochin I-type bisbibenzyls, represent isolagiochin II-type of macrocyclic bisbibenzyls. Planusin A 145 with a cis -stilbene moiety was discovered from cultured cells of the liverwort Heteroscyphus planus and classified into planusin-type bisbibenzyls [82].


2.4.2 Acyclic Bisbibenzyls
Compared with cyclic bisbibenzyls, acyclic bisbibenzyls receive less attention due to their small number and poor structural diversity. Some novel acyclic bisbibenzyls, however, have been obtained in the past few years. These bisbibenzyls can be divided into two main groups, those with one diarylether bond (perrottetins and paleatins) or those with one biphenyl bond (isoperrottetin A, plagilins, isoplagilin, plagiolin, artogomezianol, and andalasin). The linkages occurred between two aromatic rings or between one benzene nucleus and ethylidene bridge (see Table 62.1).
Perrottetins and isoperrottetins represent the most frequently encountered skeletal types of acyclic bisbibenzyls. They are of much interest for the investigation of biogenesis of macrocyclic bisbibenzyls, for example, the derivatives of riccardin, plagiochin, ptychantol, and isoplagiochin types. Examples of the perrottetin-type compounds are perrottetins E-H 146–149. They have been found in different liverwort species and a fern Hymenophyllum barbatum [83]. Isoperrottetin A 150 and its chlorinated derivative 151 belong to isoperrottetin-type compounds [66, 84], which contain one biphenyl bond between aromatic rings instead of one diarylether bond for perrottetins.
Plagilin 152, isoplagilin 153, and plagiolin 154 were obtained from a neotropical Plagiochila species [85]. Another plagilin-type bisbibenzyl, vittarin-E 155, has been isolated from the whole plant of Vittaria anguste-elongata [74]. This is one more evidence for the occurrence of acyclic bisbibenzyls in the pteridophytes.
Paleatins A 156 and B 157 were isolated from the methanol extract of Marchantia paleacea var. diptera [86]. These phenolic compounds are of interest because they are the linear analogues of the macrocyclic bisbibenzyl ethers and possible biogenetic precursors of the plagiochins and riccardins.

2.5 Phenanthrenes
The phenanthrenes are a rather uncommon group of aromatic constituents formed by oxidative coupling of the aromatic rings of stilbene precursors and existed in the form of monomers, dimers, and even trimers [87]. A large number of phenanthrenes have been isolated from higher plants (mainly in the Orchidaceae family) and covering 49 species. The genera Dendrobium, Bulbophyllum, Eria, Maxillaria, Bletilla, Coelogyna, Cymbidium, Ephemerantha, and Epidendrum were particularly rich resources of phenanthrenes. In addition, a few phenanthrenes have been discovered in the family of Dioscoreaceae, Combretaceae and Betulaceae, and the Hepaticae class. The greatest number of phenanthrenes has been obtained from the Juncus species.
2.5.1 Monomeric Phenanthrenes
Most of the phenanthrenes are present in the form of monomers, containing about 210 compounds. Hydroxyl and methyloxyl are the most common substitutes located in the phenanthrene skeletons and occupy about 50% of all monomers. For instance, coeloginanthrin 158 from Coelogyne cristata [88], 4-methoxyphenanthrene-2,3,7-triol 159 and 4-methoxyphenanthrene-2,3,6,7-tetrol 160 from Bulbophyllum vaginatum [89], as well as 2-methoxy-3,4,7-trihydroxy-phenanthrene 161 from B. inconsipicum [90] are hydroxyl- and/or methyloxyl-substituted ones.
With the exception of hydroxy and methyloxy groups, methyl-, hydroxymethyl-, carboxy-, formyl-, prenyl-, and vinyl-substituted compounds are observed, with stemanthrenes A-D 162–165, 166, 167, dehydroeffusal 168, and gancaonin U 169 as the representatives [91–94]. Furthermore, glycosides were isolated from the plants of Juncus effusus, Epimedium koreanum, Dendrobium chrysanthum, and Bulbophyllum striata, for example, effuside I 170, epimedoicarisoside A 171, and denchryside A 172 [95–97].
Phenanthraquinones are special phenanthrenes with quinone group in the structures. Ephemeranthoquionone 173 from Dendrobium plicatile, cymbinodin A 174 from C. aloifolium, and moniliformin 175 from D. moniliforme were typical constituents of this group [98–100].


2.5.2 Dimeric Phenanthrenes
The dimeric phenanthrenes are commonly constructed by 1-1′ linkage of monomers, and the dimers with 1-3′, 1-8′, and 3-3′ link patterns also existed. Nearly 40 dimeric phenanthrenes have been found in the plants [87]. The representatives are cirrhopetalanthrin 176 from Cremastra maculosum [101], blestrianol A 177 from Bletilla striata [102], blestriarene B 178 from Bletilla formosana [57], and spiranthesol 179 from Spiranthes sinensis [103].

2.5.3 Triphenanthrene
Hitherto, only one triphenanthrene 180 has been reported from the tubers of an orchidaceous plant Cremastra appendiculata [104].

3 Biosynthesis
3.1 Biosynthesis of Stilbenes
The biosynthesis of simple stilbenes has been found out, and it shared a similar biosynthetic pathway with the flavonoids. Taking resveratrol for example, it starts from a cinnamoyl-CoA unit and extended the chain with three malonyl-CoA molecules (Scheme 62.1) [105]. Then, the resveratrol structure is produced by aldol reaction with the presence of stilbene synthase. Nevertheless, the flavonoids are formed depending on chalcone synthase and Claisen reaction.

Scheme 62.1
3.2 Biosynthesis of Oligostilbenes
Sotheeswaran has mentioned that the oligostilbenes with dihydrobenzofuran moiety are biosynthesized through an important intermediate trans-ε-viniferin [106]. Combined with the reported work of oligostilbenes, the biosynthesis of oligostilbenes is summarized. For instance, the dimers named (+)-ampelopsin B, (−)-ampelopsin D, and (+)-ampelopsin F are produced by isomerization and/or rearrangement of trans-ε-viniferin (Scheme 62.2). The differences in their structures are caused by the different protonation position at the initial stage of reaction. Furthermore, trans-ε-viniferin is able to transform to the isomers (Scheme 62.3) and then forming the tetramers, (+)-vitisin A, (−)-vitisin B, (+)-hopeaphenol, and (+)-viniferol A, etc., by oxidative coupling (Scheme 62.4) [2, 107].

Scheme 62.2

Scheme 62.3


Scheme 62.4
3.3 Biosynthesis of Bibenzyls and Bisbibenzyls
The biosynthesis of marchantins A and C has been certified by a C-labeled precursor feeding experiment. It shows that rings A and C of the marchantin molecules are derived from the benzene ring of l-phenylalanine. The bibenzyl lunularic acid is biosynthesized through dihydro-p-coumaric acid coupling with three malonyl-CoA units and then coupled in typical ways to form bisbibenzyls marchantins A and C (Scheme 62.5) [108].

Scheme 62.5
Evidences have confirmed that bisbibenzyls can be produced by the coupling of two phenolic systems by means of free-radical reactions. These reactions can be mediated by oxidase enzymes. C–C bonds involving positions ortho or para to the original phenols, or ether linkages, may be formed in coupling of two of these bibenzyl structures [105]. A previous hypothesis for the biogenesis of marchantins, riccardins, and plagiochins considers cyclization of open-ring precursors, such as perrottetin E 146, through intramolecular phenol oxidation, accompanied by C–O or C–C linkage of the terminal m-hydroxyphenyl units [109]. Likewise, isoplagiochins A-D might be biosynthesized from isoperrottetin A 150 (Scheme 62.6) [110].

Scheme 62.6
3.4 Biosynthesis of Phenanthrenes
Bibenzyls are regarded as the bicyclic intermediates of 9,10-dihydrophenanthrenes, and the biosynthetic pathway of 9,10-dihydrophenanthrenes was proposed as showed in Scheme 62.7. Oxidative coupling of the bibenzyl intermediate leads to the formation of phenanthrene [2].

Scheme 62.7
4 Biological Activities
4.1 Antitumor Activity
4.1.1 Stilbenes and Oligostilbenes
Resveratrol, as a representative compound of stilbene, possesses diverse pharmacological activities. The antitumor property has taken the spotlight for its cancer preventive effect on skin cancer in a mice model. Subsequently, plenty of in vivo experiments targeting different tumor model were carried out to evaluate its therapeutic effects on tumors [111]. The results definitely show that resveratrol is able to inhibit or possess chemopreventive functions on different tumors, including breast cancer, liver cancer, gastric cancer, colorectal cancer, prostate cancer, leukemia, lung cancer, neuroblastoma, etc. In these experiments, the incidences, tumor volume, and metastasis are improved. Clinical trials of investigating resveratrol’s effects on colon cancer and melanoma (skin cancer) are intending to launch.
Combretastatins attract a lot of interests for their potent antitumor properties by inhibiting tubulin polymerization and disrupting the formation of tumoral vasculature. Combretastatin A-4 5 was proved to be the most potent candidate of combretastatins with GI50 of 3.20 nM in an antitumor screening project against the NCI-60 human tumor cell lines, followed by combretastatins A-1 2 and A-2 3 [112]. Further studies focused on the antitumor mechanism of combretastatins, and the results suggested that compounds 2, 3, and 5 possessed potent antimitotic effect through binding to the tubulin at colchicine site [113]. Combretastatin A-4, its prodrug combretastatin A-4 phosphate (CA-4-P), and other analogues are currently being investigated in the clinical trials. CA-4-P, being developed as vascular targeting agents, in combination with carboplatin has entered into phase III clinical trial for the treatment of anaplastic thyroid cancer.
The seeds of Iris halophila (Iridaceae) produced halophilol A, which possesses moderate cytotoxicity against KB cells and human mammary epithelial cells (HMECs) with IC50 values of 17.28 and 22.47 μM [114], respectively. The prenyl stilbenes 30–32 with dimethylchromene ring exhibit cytotoxic activity against Hepa-1clc7 cells with IC50 values of 8.5, 13.0, and 7.0 μM [18], respectively. Tested in the NCI-60 cell line human cancer screen, schweinfurthins A 33 and B 34 show significant cytotoxic activity with mean panel GI50 of 0.36 and 0.81 μM. Schweinfurthins E-H were isolated from M. alnifolia [115] and display potent antiproliferative effect against A2780 human ovarian cancer cell line with IC50 values ranging from 0.26 to 5.0 μM, respectively. Lakoochins A and B also possess cytotoxic activity against breast cancer cell line (6.1 and 3.1 μg/mL) and nasopharyngeal carcinoma cell line (20 and 6.1 μg/mL) [116].
4.1.2 Oligostilbenes
Three trimers nepalensinols A, C, and D and three tetramers nepalensinols B, G, and F were obtained from Kobresia nepalensis (Cyperaceae) [117, 118]. The inhibitory effect of the above six oligomers against the decatenation activity of topoisomerase II on kinetoplast DNA is evaluated with IC50 values ranging from 0.02 to 10.8 μg mL−1. Among them, nepalensinol B exhibits the strongest activity with an IC50 of 0.02 μg mL−1, much better than the clinical antitumor drugs daunorubicin (IC50 4.8 μg mL−1) and etoposide (IC50 70.0 μg mL−1). Vaticanol C is a resveratrol tetramer with dibenzobicyclo[3.2.1]octadiene moiety, is widely distributed in Dipterocarpaceae species, and shows potent growth suppressive activity with IC50 values of 5.9 μM against HL60 cells. Upunaphenol A is a hexamer obtained from Upuna borneensis and was found to suppress cell growth in HL60 cells through induction of apoptosis with IC50 at 9.2 μM [119].
4.1.3 Bibenzyls and Bisbibenzyls
Erianin (also named dihydrocombretastatin A-4), a dihydro derivative of combretastatin A-4 which has been initiated phase II clinical trial as antitumor agent, possessed potent cytotoxicity toward diverse cancer cell lines. It was evaluated against A-549 lung carcinoma, MCF-7 breast carcinoma, HT-29 colon adenocarcinoma, SKMEL-5 melanoma, and MLM melanoma cell lines with ED50 ranging from 0.002 to 0.33 μM, respectively [120]. It also showed potent inhibitory activity on the proliferation of HL-60 cells (IC50 38 nM) and was able to alter expression of bcl-2 and bax genes in HL-60 cells [121].
Bauhinols A 110 and B 111 exhibit significant cytotoxicity against NCI-H187 (small-cell lung cancer) and BC (breast cancer) cell lines with IC50 values ranging from 1.1 to 9.7 μg/mL. In addition, bauhinol A 110 is active toward KB cells (IC50 4.5 μg/mL) [64]. 3,5-Dihydroxy-4-methylbibenzyl shows cytotoxic activity, which is able to inhibit the growth rate of P-388 leukemia and hepatoma cell lines by 99.7% and 83.6% at 10 μg/mL, respectively [122].
The bisbibenzyls neomarchantins A 133 and B 134, marchantin C, and Glaucescens Bis Bibenzyl A and B possess moderate cytotoxicity against P-388 leukemia cells with IC50 ranging from 8 to 18 μg/mL [123]. Riccardin C and pusilatins B-C display moderate cytotoxicity against KB cells with ED50 of 7.1 to 16.4 μg/mL [79].
4.1.4 Phenanthrenes
The cytotoxic activities both in vitro and in vivo of lusianthridin and denbinobin isolated from Dendrobium nobile are evaluated. Both of them exhibit potent antitumor effects against A549 human lung carcinoma, SK-OV-3 human ovary adenocarcinoma, and HL-60 human promyelocytic leukemia with EC50 values ranging from 0.11 to 9.8 μg/mL [124].
Dimeric phenanthrenes denthyrsinol, denthyrsinone, and monomer denthirsinin demonstrate potent cytotoxicity against cervix adenocarcinoma HeLa, K-562, and MCF-7 cells with IC50 values from 1.6 to 9.9 μM [125]. A series of phenanthrenes, including 7-hydroxy-2,3,4,8-tetramethoxyphenanthrene, 3-hydroxy-2,4,-dimethoxy-7,8-methylenedioxyphenanthrene, 2-hydroxy-3,5,7-trimethoxyphenanthrene, 2-hydroxy-3,5,7-trimethoxy-9,10-dihydrophenanthrene, and confusarin are evaluated on their antitumor properties against HeLa cell line with IC50 values of 0.97–14.21 μM. [126, 127].
3,6-Dihydroxy-l,7-dimethyl-9-methoxyphenanthrene 181 and 3,6-dihydroxy-l-hydroxymethyl-9-methoxy-7-methylphenanthrene 182 are found to demonstrate significant cytotoxic responses against several tumor cell lines (Table 62.2). Compound 181 is more active against drug-resistant KB cells, while 182 is active against HT (fibrosarcoma) and U373 (glioma) cell lines [128].
4.2 Antioxidant Activity
4.2.1 Stilbenes and Oligostilbenes
Lespedezavirgatol was obtained from Lespedeza virgata and shows potent antioxidant property. Its oxygen radical absorbance capacity (ORAC) value for Trolox equivalents is 762.96 at 1.5 μM, much better than 164.56 of vitamin C. Inhibitory effects of lespedezavirgatol against lipid peroxidation toward malondialdehyde levels in rat kidney homogenate and plasma are also evaluated with IC50 values of 0.16 and 0.18 mM, better than the control vitamin C, with IC50 values of 5.54 and 3.05 mM. The above results suggest that lespedezavirgatol is a potent candidate for antioxidants [129]. Tibeticanol 76 was obtained from Caragana tibetica and exhibits strong superoxide anion scavenging activity with an IC50 of 1.33 μM [46].
A series of oligostilbenes isolated from Vitis thunbergii are evaluated on their antioxidant properties based on the radical scavenging effect of the stable ABTS•+ free radical. The results are shown in Table 62.3. All of the tested compounds are more active than the positive control Trolox and display free-radical scavenging activity with EC50 values from 2.8 to 13.8 μM. Among them, (+)-ε-viniferin shows the most potent radical scavenging potency with EC50 of 2.8 μM [23].
4.2.2 Bibenzyls and Bisbibenzyls
Marchantin H is able to inhibit nonenzymatic iron-induced lipid peroxidation in rat brain homogenates and NADPH-dependent microsomal lipid peroxidation with an IC50 of 0.51 and 0.32 μM [130]. It also possesses inhibitory effects of copper-catalyzed oxidation of human low-density lipoprotein. Marchantinquinone exhibits inhibitory effects of Fe2+-induced lipid peroxidation in rat brain homogenates (IC50 15.3 μM) and displays radical scavenging activity [131]. Schwartner et al. reported the antioxidative potential of three macrocyclic bisbibenzyls [marchantins A, B, and D], one acyclic bisbibenzyl (paleatin B) and a prenylated bibenzyl (perrottetin D) by pulse-radiolytic and EPR-spectroscopic techniques. The results confirm that these compounds are effective antioxidants [132]. Isoamoenylin and 5,4′-dihydroxy-3-α-l-rhamnopyranosyl-(1 → 3)-β-d- xylopyranosyloxybibenzyl showed radical scavenging activity, comparable with the control vitamin C and ascorbic acid [63, 133]. Five prenylated dihydrostilbenes, α,α-dihydro-3,5,3,4-tetrahydroxy-4,5-diisopentenylstilbene, α,α-dihydro-3,5,3,4-tetrahydroxy-5-isopentenylstilbene 183, α,α-dihydro-3,5,4-trihydroxy-4,5-diisopentenylstilbene, α,α-dihydro-3,5,4-trihydroxy-5-isopentenyl stilbene, and α,α-dihydro-3,5,3-trihydroxy-4-methoxy-5-isopentenylstilbene 184, from Glycyrrhiza glabra were tested for antioxidant effects by measuring the absolute inhibition rate constant (k inh) of the oxidation process. Compounds 183 and 184 display potent antioxidant properties with k inh values of 1.1 × 104 and 0.9 × 104 M−1 s−1 [134].
4.3 Antiplatelet Activity
Resveratrol prevents platelet aggregation and thrombus formation in vitro. In a hypercholesterolaemic diet-induced rabbit model, administration of resveratrol inhibited the platelet aggregation. This effect was also verified by reducing the atherosclerotic area and the size of the thrombus generated by laser-induced damage to the endothelium in mice.
Vitis thunbergii (Vitaceae), a folk medicine in Taiwan, produced several resveratrol oligomers, including vitisinol C with a tropilene structure in molecule, vitisinols B and D, and some known compounds (−)-viniferal, (+)-ε-viniferin, and (+)-vitisin C, etc. The isolated compounds were evaluated for antiplatelet aggregation activities induced by arachidonic acid (AA) and 9,11-dideoxy-11α,9α-epoxy-methanoprostaglandin F2α (U46619). Most of the tested oligomers demonstrate potent antiplatelet aggregation property with IC50 < 10 μM, more positive than aspirin (EC50 32.7 μM). In the above bioassay, (−)-viniferal and (+)-vitisin C are most effective against aggregation induced by AA and U46619, with IC50 values of 5.7 and 3.1 μM, respectively [23]. The results are summarized in Table 62.4.
Marchantinquinone displays potent inhibitory activity on the aggregation of washed rabbit platelets induced by thrombin, arachidonic acid (AA), collagen, U46619, and platelet-activating factor (PAF) [135, 136]. It inhibits thromboxane B2 (TxB2) formation induced by thrombin, PAF, and collagen. In addition, marchantinquinone is able to inhibit the rising intracellular Ca2+ concentration stimulated by five inducers mentioned above [136]. Gigantol exhibits antiplatelet aggregation activity on SD-rat platelet aggregation [137]. 3-Methylgigantol possesses significant inhibitory effects against aggregation induced by AA, collagen, and PAF [138]. Among them, 3-methylgigantol is most potent effective against AA-induced aggregation (IC50 30 μM). Moscatilin is able to inhibit AA- and collagen-induced platelet aggregations [139]. Perrottetin E 146 shows inhibitory activity of thrombin (IC50 18 μM) [140].
The phenanthrenes, erianthridin, and denbinobin display potent antiplatelet activity on washed rabbit platelets against aggregation induced by either thrombin, arachidonic acid (AA), collagen, or PAF at a dose of 100 μg/mL. Erianthridin is proved to be the most potent compound with an IC50 of 9 μM against AA-induced aggregation [138].
4.4 Antidiabetic Activity
5,4′-Dihydroxystilbene-3-O-α-arabinopyranoside, named rumexoid, was found in Rumex bucephalophorus (Polygonaceae). This compound and resveratrol display potent α-glucosidase inhibitory activity even better than the commercial antidiabetic agent acarbose [141]. 2′-O-Demethylbidwillol B and addisofurans A-B were prenyl-substituted arylbenzofurans isolated from Erythrina addisoniae. Those compounds are inhibitors of type II diabetes target protein tyrosine phosphatase 1B with IC50 values of 13.6–15.7 μM. The linear prenyl chain was responsible for its inhibitory activity, and the cyclization of prenyl group decreased this effect [142]. In a bioassay-guided fractionation against α-glucosidase, 13-hydroxykompasinol A and scirpusin C were obtained from the seeds of Syagrus romanzoffiana and possess potent inhibitory activity against α-glucosidase type IV from Bacillus stearothermophilus with the IC50 value of 6.5 and 4.9 μM [143].
4.5 Antimicrobial Activity
4.5.1 Stilbenes and Oligostilbenes
Machaeriol B, a compound with hexahydro-6H-benzo[c]chromene system, was obtained from Machaerium multiflorum (Leguminosae), and it demonstrates potent antimalarial activity (IC50 0.12 μg/mL) against Plasmodium falciparum [144]. Preracemosols A and B exhibit moderate antimalarial activity with EC50 of 18.0 and 3.0 mg/mL, respectively [145]. Trans-4-isopentenyl-3,5,2′,4′-tetrahydroxystilbene was discovered in Artocarpus integer (Moraceae) and possesses antimalarial activity against Plasmodium falciparum (EC50 = 1.7 μg mL−1) [146]. This is the first report of antimalarial activity of stilbenes.
A series of arylbenzofuran-type stilbenes, guided by bioautographic assay for antifungal activity against Cladosporium herbarum, have been isolated from the root of Stemona collinsae (Stemonaceae) and tested in microwells against another four microfungi Alternaria citri, Fusarium avenaceum, Pyricularia grisea, and Botrytis cinerea [22]. Among them, stemofuran B shows the highest antifungal potency against above four parasitic fungi with EC50 values of 1.4 μg/mL. Stemofuran E exhibited antifungal property against C. herbarum with EC50 of 0.09 μg/mL.
Stilbene derivatives were obtained from Calligonum leucocladum, and the structures were determined as (E)-resveratrol 3-(6-galloyl)-O-β-d-glucopyranoside. Although ineffective when tested alone, it is able to restore oxacillin’s effectiveness against oxacillin/methicillin-resistant Staphylococcus aureus when used in combination. The galloyl group may play a role in this synergistic activity [147].
Hopeanolin was obtained from the stem bark of Hopea exalata and exhibits potent antifungal properties against six types of pathogenic fungi Alternaria attenata, Alternaria solani, Colletotrichum lagenarium, Fusarium oxysporum f. sp. vasinfectum, Pyricularia oryzae, and Valsa mali with MIC values ranging from 0.10 to 22.5 μg mL−1 [148].
4.5.2 Bibenzyls and Bisbibenzyls
Bryophytes normally grow in humid habitats; however, they are seldom damaged by fungi. It indicates that bryophytes are able to elaborate constitutive and inducible antifungal natural products against adverse effects. A large amount of bibenzyls and bisbibenzyls with antifungal activity have been found in plants, primarily in bryophytes (Table 62.5), which provided important sources for research and development of antifungal agents. The antifungal effects of bibenzyls and bisbibenzyls were summarized in Table 62.5.
The bibenzyls 4-hydroxy-3′-methoxybibenzyl, 2,4,6-trichloro-3-hydroxybibenzyl, 2, 4-dichloro-3-hydroxylbibenzyl, 2-chloro-3-hydroxybibenzyl, together with bisbibenzyls neomarchantins A 133 and B 134, and marchantin C show antimicrobial activity against the gram-positive bacterium Bacillus subtilis [60, 123, 150]. In addition, 4-hydroxy-3′-methoxybibenzyl is active toward Escherichia coli [150].
4.6 Anti-inflammatory Activity
Anti-inflammatory properties of resveratrol have been confirmed, which is an effective inhibitor of cyclooxygenase (COX) in vitro. It is also found that resveratrol significantly reduces acute and chronic chemically induced edema, lipopolysaccharide-induced airway inflammation, and osteoarthritis. Resveratrol could also present as an alternative, instead of aspirin, for treatments of chronic inflammation because of the latter’s side effect on the stomach. Resveratrol derivatives displayed similar anti-inflammatory effects. For instance, resveratrol (E)-dehydrodimer 11-O-β-d-glucopyranoside and resveratrol (E)-dehydrodimer from Vitis vinifera show significant inhibitory activity against cyclooxygenase-1 (COX-1) with IC50 of 5.2 and 4.3 μM and against cyclooxygenase-2 (COX-2) with IC50 of 7.5 and 3.7 μM. From the above results, these two compounds seem to be the worthy candidates for further research to find application in anti-inflammatory treatment [151].
(+)-Vitisifuran A and heyneanol A were found in Vitis genus and display potent inhibition on biosynthesis of LTB4 with inhibitory rate of 72% and 76% at a concentration of 10 μM [152]. A tetramer named gnetuhainin R 73 was obtained from the same species (G. hainanense) and shows potent histamine receptor antagonism (IC50 0.1 μM) [44]. Aiphanol exhibited significant inhibitory activities against COX-1 (IC50 1.9 μM) and COX-2 (9.9 μM) [153].
Stemofurans B, D, G, and J and stilbostemin G were reported possessing anti-inflammatory effects with IC50 values ranging from 3.7 to 26.3 μM by inhibiting leukotriene formation [154]. The inhibition of lipopolysaccharide-induced nitric oxide synthase (NOS) by 19 bisbibenzyls in RAW 264.7 macrophages has been reported, and marchantin A is most effective with IC50 values of 1.44 μM. The structure-activity relationship (SAR) is discussed, and the phenolic hydroxyl groups and diarylether bonds play important roles in its inhibitory effect [155]. Pusilatins B-C exhibit selective DNA polymerase-β inhibitory activity with IC50 of 13.0 and 5.16 μM [79]. Bauhinol B 111 and 3,5-dihydroxy-2-(3-methyl-2-butenyl)bibenzyl are potent inhibitors of COX-1 and COX-2 with IC50 ranging from 1.3 to 9.0 μg/mL [134].
Phenanthrenes obtained from the Stemona species were evaluated on their leukotriene biosynthesis inhibition property using human neutrophile granulocytes in vivo. Stemanthrenes A 162 and D 165 display inhibitory activity in a dose-dependent manner, with IC50 values of 8.5 and 4.8 μM. Stemanthrenes B 163 and C 164 possess 100% inhibition against leukotriene biosynthesis at 25 μM. The phenanthrenes might be responsible for the anti-inflammatory and antiasthmatic principles of the Stemona species [154]. Denbinobin, a phenanthraquinone from Dendrobium moniliforme, shows in vitro anti-inflammatory activity. It inhibits the formation of tumor necrosis factor and prostaglandin E2 induced by lipopolysaccharide in RAW 264.7 and N9 cells at a dose of 1 μM [100].
4.7 Neuroprotective Activity
In vivo pharmacological studies have indicated that resveratrol has a neuroprotective effect, including reduced lipid, peroxidation and neurological cell destruction, attenuation of induced lesion areas, induced tolerance to brain injury, reduced frequency of seizures, impairment of motor coordination, and enhancement of learning [111].
Stilbostemin B 3′-β-d-glucopyranoside, stilbostemin H 3′-β-d-glucopyranoside, and stilbostemin I 2″-β-d-glucopyranoside possess significant neuroprotective activity against 6-hydroxydopamine-induced neurotoxicity in human neuroblastoma SH-SY5Y cells [156]. Hopeahainol A from Hopea hainanensis contains an unprecedented carbon skeleton and shows potent acetylcholinesterase inhibitory effect with an IC50 value of 4.33 μM, comparable even to that of huperzine A (IC50 1.6 μM) [157]. The tetramer neohopeaphenol A from the same species also displays significant inhibitory action against AChE with an IC50 value of 7.66 μM [158].
4.8 Hepatoprotective Activity
Acer mono (Aceraceae), a Korean folk medicine for hemostasis, produces two potent hepatoprotective stilbene glycosides, 5-O-methyl-(E)-resveratrol 3-O-β-d-glucopyranoside and 5-O-methyl-(E)-resveratrol 3-O-β-d-apiofuranosyl-(1 → 6)-β-d-glucopyranoside [16]. Those two compounds significantly prevent the depletion of glutathione (GSH) in H2O2-injured primary cultured rat hepatocytes and potently restore the level of GSH depleted by buthionine sulfoximine or diethylmaleate in the presence or absence of H2O2. In addition, they preserve the effects of antioxidant enzymes such as superoxide dismutase, glutathione reductase, and glutathione peroxidase reduced by H2O2 insults. Therefore, it is concluded that both compounds exerted significant hepatoprotective effects against H2O2-induced hepatotoxicity by maintaining the antioxidant defense system [159].
4.9 Cardioprotective Activity
It is well known that drinking wine and grape juices will reduce cardiovascular, cerebrovascular, and peripheral vascular risks due to the presence of resveratrol. As a natural antioxidant, resveratrol is able to prevent LDL oxidation, scavenge intracellular reactive oxygen species, lower the oxidative stress, and induce NO synthesis. Resveratrol modulates various aspects of cardiovascular diseases and is effective against atherosclerosis, hypertension, ischemia reperfusion injury and heart failure, and many other cardiac dysfunctions [111].
4.10 Phytotoxicity
The phenanthrenes, ephemeranthol-A and fimbriol A from Epidendrum rigidum, together with erianthridin from M. densa demonstrated phytotoxicity against Amaranthus hypochondriacus with IC50 values of 0.12, 5.9, and 58.2 μM [160]. The phenanthrenes from the Juncus genus show growth inhibitory effects against the green alga Selenastrum capricornutum with IC50 values ranging from 11.1 to 19.9 μM [161].
Bibenzyls are elaborated to confer the producing plants’ selective advantage against the competition from the other plants and microbial attack. Gigantol, batatasin III, 2,3-dimethoxy-9,10-dihydrophenathrene-4,7-diol, and 3,4,9-trimethoxyphenanthrene-2,5-diol from the orchid Epidendrum rigidum inhibit radicle growth of Amaranthus hypochondriacus with IC50 of 0.65, 0.1, 0.12, and 5.9 μM [160].
5 Conclusion
Stilbenoids represent a group of important natural products in the plant kingdom. The developments of modern analytical methods accelerate the discovery of these compounds, and to this day more than 1,000 stilbenoids have been isolated. For the stilbenes, bibenzyls, and phenanthrenes, these three groups shared the feature of their nucleus with hydroxyls, methyl, methoxy, prenyl, geranyl, glycosyl, etc., substituents. The oligostilbenes and bisbibenzyls are formed by polymerization of stilbene and bibenzyl units, and the diverse polymerized patterns produced their diverse structures. They display diverse biological activities and have the potential to be developed as new drugs, especially in the field of antitumor, anti-inflammation, and cardioprotective drug research. The representative compounds resveratrol and combretastatin A-4 phosphate are currently being evaluated as drugs for the treatment of Alzheimer’s disease and tumors in clinical trials and have shown satisfactory therapeutic effects. Furthermore, resveratrol displays developing prospects as a cardioprotective drug.
There are problems that need to be noted. Concerning the oligostilbenes, they commonly possess large molecules, intricate structures, and complex stereochemistry, and these characteristics cause troubles in structure identification and chemical total synthesis, therefore, limiting the probability to be developed to new drug. The bisbibenzyls are mainly distributed in the bryophytes, which are very small terrestrial spore-forming green plants, and different for collections. Therefore, getting enough plant materials for phytochemical investigation and new drug research is really a Gordian knot and resorts to the chemical synthesis for resolving the resource problems.
References
Gorham J (1980) The stilbenoids. In: Harbome JB, Swain T, Reinhold L (eds) Progress in phytochemistry. Pergamon Press, Oxford
Xiao K, Zhang HJ, Xuan LJ, Zhang J, Xu YM, Bai DL (2008) Stilbenoids: chemistry bioactivities. In: Atta-ur-Rahman (ed) Studies in natural products chemistry. Elsevier, Amsterdam
Shen T, Wang XN, Lou HX (2009) Natural stilbenes: an overview. Nat Prod Rep 26:916–935
Ma ZJ, Li X, Li N, Wang JH (2002) Stilbenes from Sphaerophysa salsula. Fitoterapia 73:313–315
Majumder PL, Roychowdhury M, Chakraborty S (1998) Thunalbene, a stilbene derivative from the orchid Thunia alba. Phytochemistry 49:2375–2378
Garo E, Hu JF, Goering M, Hough G, O’Neil-Johnson M, Eldridge G (2007) Stilbenes from the orchid Phragmipedium sp. J Nat Prod 70:968–973
Guo XY, Wang J, Wang NL, Kitanaka S, Liu HW, Yao XS (2006) New stilbenoids from Pholidota yunnanensis and their inhibitory effects on nitric oxide production. Chem Pharm Bull 54:21–25
Kerem Z, Regev-Shoshani G, Flaishman MA, Sivan L (2003) Resveratrol and two monomethylated stilbenes from Israeli Rumex bucephalophorus and their antioxidant potential. J Nat Prod 66:1270–1272
Hajdú Z, Varga E, Hohmann J, Kálmán A, Argay G, Günther G (1998) A stilbene from the roots of Leuzea carthamoides. J Nat Prod 61:1298–1299
Ngo KS, Brown GD (1998) Amino acid-catalyzed asymmetric α-amination of carbonyls. Phytochemistry 47:1117–1119
Asakawa Y, Toyota M, Tori M, Hashimoto T (2000) Chemical structures of macrocyclic bis(bibenzyls) isolated from liverworts (Hepaticae). Spectroscopy 14:149–175
Speicher A, Schoeneborn R (1997) 3,4-dihydroxy-3′-methoxystilbene, the first monomeric stilbene derivative from bryophytes. Phytochemistry 45:1613–1615
Jayatilake GS, Baker BJ, Mcclintoc JB (1995) Isolation and identification of a stilbene derivative from the Antarctic sponge Kirkpatrickia variolosa. J Nat Prod 58:1958–1960
Nyemba AM, Mpondo TN, Kimbu SF, Connolly JD (1995) Stilbene glycosides from Guibourtia tessmannii. Phytochemistry 39:895–898
Fuendjiep V, Wji J, Tillequin F, Mulholl DA, Budzikiewicz H, Fomum ZT, Nyemba AM, Koch M (2002) Chalconoid and stilbenoid glycosides from Guibourtia tessmanii. Phytochemistry 60:803–806
Yang H, Sung SH, Kim YC (2005) Two new hepatoprotective stilbene glycosides from Acer mono leaves. J Nat Prod 68:101–103
Hakim EH, Ulinnuha UZ, Syah YM, Ghisalbertib EL (2002) Artoindonesianins N and O, new prenylated stilbene and prenylated arylbenzofuran derivatives from Artocarpus gomezianus. Fitoterapia 73:597–603
Fang N, Casida JE (1999) New bioactive flavonoids and stilbenes in cubé resin insecticide. J Nat Prod 62:205–210
Beutler JA, Shoemaker RH, Johnson T, Boyd MR (1998) Cytotoxic geranyl stilbenes from Macaranga schweinfurthii. J Nat Prod 61:1509–1512
Ali Z, Tanaka T, Iliya I, Iinuma M, Furusawa M, Ito T, Nakaya K, Murata J, Darnaedi D (2003) Phenolic constituents of Gnetum klossii. J Nat Prod 66:558–560
Kanchanapoom T, Suga K, Kasai R, Yamasaki K, Kamel MS, Mohamed MH (2002) Stilbene and 2-arylbenzofuran glucosides from the rhizomes of Schoenocaulon officinale. Chem Pharm Bull 50:863–865
Pacher T, Seger C, Engelmeier D, Vajrodaya S, Hofer O, Greger H (2002) Antifungal stilbenoids from Stemona collinsae. J Nat Prod 65:820–827
Huang YL, Tsai WJ, Shen CC, Chen CC (2005) Resveratrol derivatives from the roots of Vitis thunbergii. J Nat Prod 68:217–220
Xiao K, Xuan LJ, Xu YM, Bai DL, Zhong DX, Wu HM, Wang ZH, Zhang NX (2002) Dimeric stilbene glycosides from Polygonum cuspidatum. European J Org Chem 3:564–568
Tanaka T, Ohyama M, Morimoto K, Asai F, Iinuma M (1998) A resveratrol dimer from Parthenocissus tricuspidata. Phytochemistry 48:1241–1243
Ito T, Tanaka T, Iinuma M, Nakaya K, Takahasic Y, Nakamura Y, Naganawa H, Risw S (2003) Helv Chim Acta 86:3394–3401
Hölscher D, Schneider B (1996) A resveratrol dimer from Anigozanthos preissii and Musa cavendish. Phytochemistry 43:471–473
Ito J, Niwa M (1996) Absolute structures of new hydroxystilbenoids, vitisin C and viniferal, from Vitis vinifera ‘Kyohou’. Tetrahedron 52:9991–9998
Ito T, Tanaka T, Nakaya K, Iinuma M, Takahashi Y, Naganawa H, Ohyama M, Nakanishi Y, Bastow KF, Lee KH (2001) A novel bridged stilbenoid trimer and four highly condensed stilbenoid oligomers in Vatica rassak. Tetrahedron 57:7309–7321
Ito T, Tanaka T, Iinuma M, Iliya I, Nakaya K, Ali Z, Takahashi Y, Sawa R, Shirataki Y, Murata J, Darnaedi D (2003) New resveratrol oligomers in the stem bark of Vatica pauciflora. Tetrahedron 59:5347–5363
Liu HX, Lin WH, Yang JS (2004) Oligomeric stilbenes from the root of Caragana stenophylla. Chem Pharm Bull 52:1339–1341
Huang KS, Lin M, Yu LN, Kong M (1999) A new oligostilbene from the roots of Vitis amurensis. Chin Chem Lett 9:775–776
Sarker SD, Whiting P, Dinan L (1999) Identification and ecdysteroid antagonist activity of three resveratrol trimers (suffruticosols A, B and C) from Paeonia suffruticosa. Tetrahedron 55:513–524
Huang KS, Lin M, Yu LN, Kong M (2000) Four novel oligostilbenes from the roots of Vitis amurensis. Tetrahedron 56:1321–1329
Ito T, Tanaka T, Iinuma M, Nakaya K, Takahashi Y, Sawa R, Naganawa H, Chelladurai V (2003) Two new oligostilbenes with dihydrobenzofuran from the stem bark of Vateria indica. Tetrahedron 59:1255–1264
Wang YH, Huang KS, Lin M (2001) Four new stilbene dimers from the lianas of Gnetum hainanense. J Asian Nat Prod Res 3:169–176
Huang KS, Wang YH, Li RL, Lin M (2000) Stilbene dimers from the lianas of Gnetum hainanense. Phytochemistry 54:875–881
Chen H, Lin M (1998) A pair of dimeric stilbene epimers from Gnetum montanum. Chin Chem Lett 11:1013–1015
Iliya I, Ali Z, Tanaka T, Iinuma M, Furusawa M, Nakaya K, Murata J, Darnaedi D, Matsuura N, Ubukata M (2003) Stilbene derivatives from Gnetum gnemon Linn. Phytochemistry 62:601–606
Iliya I, Tanaka T, Iinuma M, Ali Z, Furasawa M, Nakaya K, Matsuura N, Ubukata M (2002) Four dimeric stilbenes in stem lianas of Gnetum africanum. Heterocycles 57:1507–1512
Iliya I, Tanaka T, Iinuma M, Ali Z, Furasawa M, Nakaya K (2002) Dimeric stilbenes from stem lianas of Gnetum africanum. Heterocycles 57:1057–1062
Kawazoe K, Shimogai N, Takaishi Y, Rao KS, Imakura Y (1997) Four stilbenes from Salacia lehmbachii. Phytochemistry 44:1569–1573
Huang KS, Li RL, Wang YH, Lin M (2001) Three new stilbene trimers from the lianas of Gnetum hainanense. Planta Med 67:61–64
Huang KS, Zhou S, Lin M, Wang YH (2002) An isorhapontigenin tetramer and a novel stilbene dimer from Gnetum hainanense. Planta Med 68:916–920
Morikawa T, Xu F, Matsuda H, Yoshikawa M (2002) Heterocycles 57:1983
Xiang T, Uno T, Ogino F, Ai C, Duo J, Sankawa U (2005) Antioxidant constituents of Caragana tibetica. Chem Pharm Bull 53:1204–1206
Li S-H, Niu X-M, Zahn S, Gershenzon J, Weston J, Schneider B (2008) Diastereomeric stilbene glucoside dimers from the bark of Norway spruce (Picea abies). Phytochemistry 69:772–782
Likhitwitayawuid K, Sritularak B (2001) A new dimeric stilbene with tyrosinase inhibitiory activity from Artocarpus gomezianus. J Nat Prod 64:1457–1459
Tanaka T, Iliya I, Ito T, Furusawa M, Nakaya K, Iinuma M, Shitataki Y, Matsuura N, Ubukata M, Murata J, Simozono F, Hirai K (2001) Stilbenoids in lianas of Gnetum parvifolium. Chem Pharm Bull 49:858–862
Li XM, Lin M, Wang YH, Liu X (2004) Four new stilbenoids from the lianas of Gnetum montanum f. megalocarpum. Planta Med 70:160–165
Iliya I, Alib Z, Tanaka T, Iinuma M, Furusawa M, Nakaya K, Murata J, Darnaedi D (2002) Four new stilbene oligomers in the root of Gnetum gnemon. Helv Chim Acta 85:2538–2546
Huang KS, Wang YH, Li RL, Lin M (2000) Five new stilbene dimers from the lianas of Gnetum hainanense. J Nat Prod 63:86–89
Iliya I, Tanaka T, Iinuma M, Furusawa M, Alib Z, Nakaya K, Murata J, Darnaedi D (2002) Five stilbene glucosides from Gnetum gnemonoides and Gnetum africanum. Helv Chim Acta 85:2394–2402
Zhou S, Wang YH, Lin M (2002) Gnetuhainin S, a new resveratrol dimer from Gnetum hainanense. Chin Chem Lett 13:549–550
Iliya I, Tanaka T, Iinuma M, Ali Z, Furasawa M, Nakaya K, Shirataki Y, Murata J, Darnaedi D (2002) Stilbene derivatives from two species of Gnetaceae. Chem Pharm Bull 50:796–801
Gorham J, Tori M, Asakawa Y (1995) The biochemistry of the stilbenoids. Chapman & Hall, London
Lin Y-L, Chen W-P, Macabalang AD (2005) Dihydrophenanthrenes from Bletilla formosana. Chem Pharm Bull 53:1111–1113
Majumder PL, Roychowdhury M, Chakraborty S (1997) Bibenzyl derivatives from the orchid Bulbophyllum protractum. Phytochemistry 44:167–172
Anton H, Kraut L, Mues R, Morales ZMI (1997) Phenanthrenes and bibenzyls from a Plagiochila species. Phytochemistry 46:1069–1075
Baek S-H, Phipps RK, Perry NB (2004) Antimicrobial chlorinated bibenzyls from the liverwort Riccardia marginata. J Nat Prod 67:718–720
Xiao K, Xuan L, XuY BD, Zhong D (2002) Constituents from Polygonum cuspidatum. Chem Pharm Bull 50:605–608
Connolly JD, Rycroft DS, Srivastava DL, Cole WJ, Ifeadike P, Kimbu SF, Singh J, Hughes M, Thom C, Gerhard U, Organ AJ, Smith RJ, Harrison LJ (1999) Aromatic compounds from the liverwort Plagiochila spinulosa. Phytochemsitry 50:1159–1165
Zidorn C, Lohwasser U, Pschorr S, Salvenmoser D, Ongania KH, Ellmerer EP, Borner A, Stuppner H (2005) Bibenzyls and dihydroisocoumarins from white salsify (Tragopogon porrifolius subsp. porrifolius). Phytochemistry 66:1691–1697
Apisantiyakom S, Kittakoop P, Manyum T, Kirtikara K, Bremner JB, Thebtaranonth Y (2004) Novel biologically active bibenzyls from Bauhinia saccocalyx Pierre. Chem Biodivers 1:1694–1701
Kraut L, Mues R, Zinsmeister HD (1997) Prenylated bibenzyl derivatives from Lethocolea glossophylla and Radula voluta. Phytochemistry 45:1249–1255
Toyota M, Kinugawa T, Asakawa Y (1994) Bibenzyl cannabinoid and bisbibenzyl derivative from the liverwort Radula perrottetii. Phytochemistry 37:859–862
Nagashima F, Kuba Y, Asakawa Y (2006) Diterpenoids and aromatic compounds from the three New Zealand liverworts Jamesoniella kirkii, Balantiopsis rosea, and Radula species. Chem Pharm Bull 54:902–906
Kunz S, Becker H (1992) Bibenzyl glycosides from the liverwort Ricciocarpus natans. Phytochemistry 31:3981–3983
Schwikkard S, Zhou BN, Glass TE, Sharp JL, Mattern MR, Johnson RK, Kingston DG (2000) Bioactive compounds from Combretum erythrophyllum. J Nat Prod 63:457–460
Zidorn C, Ellmerer-Müller EP, Stuppner H (2000) Tyrolobibenzyls – novel secondary metabolites from Scorzonera humilis. Helv Chim Acta 83:2920–2925
Zidorn C, Spitaler R, Ellmerer-Müller EP, Perry NB, Gerhäuser C, Stuppner HZ (2002) Z Naturforsch C 57C:614
Zidorn C, Ellmerer-Müller EP, Sturm S, Stuppner H (2003) Tyrolobibenzyls E and F from Scorzonera humilis and distribution of caffeic acid derivatives, lignans and tyrolobibenzyls in European taxa of the subtribe Scorzonerinae (Lactuceae, Asteraceae). Phytochemistry 63:61–67
Heinrichs J, Anton H, Gradstein SR, Mues R (2000) Systematics of Plagiochila sect. Glaucescentes Carl (Hepaticae) from tropical America: a morphological and chemotaxonomical approach. Plant Syst Evol 220:115–138
Wu P-L, Hsu Y-L, Zao C-W, Damu AG, Wu T-S (2005) Constituents of Vittaria anguste-elongata and their biological activities. J Nat Prod 68:1180–1184
Toyota M, Shimamura T, Ishii H, Renner M, Braggins J, Asakawa Y (2002) New bibenzyl cannabinoid from the New Zealand liverwort Radula marginata. Chem Pharm Bull 50:1390–1392
Tori M, Aoki M, Asakawa Y (1994) Chenopodene, marchantin P and riccardin G from the liverwort Marchantia chenopoda. Phytochemistry 36:73–76
Hashimoto T, Ikeda H, Takaoka S, Tanaka M, Asakawa Y (1999) Ptychantols A–C, macrocyclic bis(bibenzyls), possessing a trans-stilbene structure from the liverwort Ptychanthus striatus. Phytochemistry 52:501–509
Asakawa Y (2004) Chemosystematics of the Hepaticae. Phytochemistry 65:623–669
Yoshida T, Hashimoto T, Takaoka S, Kan Y, Tori M, Asakawa Y (1996) Tetrahedron 52:14487
Niu C, Qu J-B, Lou H-X (2006) Antifungal bis[bibenzyls] from the Chinese liverwort Marchantia polymorpha L. Chem Biodivers 3:34–40
Hashimoto T, Tori M, Asakawa Y (1987) Sacculaplagin, a new highly oxidized sacculatane diterpenoid hemiacetal from the liverwort Plagiochila acanthophylla subsp. japonica. Tetrahedron Lett 28:6293–6294
Nabeta K, Ohkubo S, Hozumi R, Katoh K (1998) Macrocyclic bisbibenzyls in cultured cells of the liverwort, Heteroscyphus planus. Phytochemistry 49:1941–1943
Oiso Y, Toyota M, Asakawa Y (1999) Occurrence of a bis-bibenzyl derivative in the Japanese fern Hymenophyllum barbatum: first isolation and identification of Perrottetin H from the Pteridophytes. Chem Pharm Bull 47:297–298
Hertewich UM, Zapp J, Becker H (2003) Secondary metabolites from the liverwort Jamesoniella colorata. Phytochemistry 63:227–233
Anton H, Schoeneborn R, Mues R (1999) Bibenzyls and bisbibenzyls from a neotropical Plagiochila species. Phytochemistry 52:1639–1645
Hashimoto T, Kanayama S, Kan Y, Tori M, Asakawa Y (1994) Structures and total synthesis of two novel bis(bibenzyls), paleatins A and B, from the liverwort Marchantia paleacea var diptera. Chem Pharm Bull 42:1376–1378
Kovács A, Vasas A, Hohmann J (2008) Natural phenanthrenes and their biological activity. Phytochemistry 69:1084–1110
Majumder PL, Sen S, Majumder S (2001) Phenanthrene derivatives from the orchid Coelogyne cristata. Phytochemistry 58:581–586
Leong Y-W, Harrison LJ, Powell AD (1999) Phenanthrene and other aromatic constituents of Bulbophyllum vaginatum. Phytochemistry 50:1237–1241
Tan G-S, Sun L, Cao J-G, Wang X-M, Li F-S, Xu K-P, Deng Y-H, Yan D-L, Zhang W, Zhou Y-J, Li Y-J (2006) Cytotoxic constituents from Bulbuphyllum inconsipicum. You ji hua xue 26:372–374
Kostecki K, Engelmeier D, Pacher T, Hofer O, Vajrodaya S, Greger H (2004) Dihydrophenanthrenes and other antifungal stilbenoids from Stemona cf. pierrei. Phytochemistry 65:99–106
DellaGreca M, Isidori M, Lavorgna M, Monaco P, Previtera L, Zarrelli A (2004) Bioactivity of phenanthrenes from Juncus acutus on Selenastrum capricornutum. J Chem Ecol 30:867–879
Shima K, Toyota M, Asakawa Y (1991) Phenanthrene derivatives from the medullae of Juncus effusus. Phytochemistry 30:3149–3151
Fukai T, Wang Q-H, Nomura T (1991) Six prenylated phenols from Glycyrrhiza uralensis. Phytochemistry 30:1245–1250
DellaGreca M, Fiorentino A, Monaco P, Previtera L, Zarrelli A (1995) Effusides I-V: 9,10-dihydrophenanthrene glucosides from Juncus effusus. Phytochemistry 40:533–535
Li W-K, Pan J-Q, Lu M-J, Zhang R-Y, Xiao P-G (1995) A 9,10-dihydrophenanthrene derivate from Epimedium koreanum. Phytochemistry 39:231–233
Ye Q-H, Zhao W-M, Qin G-W (2003) New fluorenone and phenanthrene derivatives from Dendrobium chrysanthum. Nat Prod Res 17:201–205
Yamaki M, Honda C (1996) The stilbenoids from Dendrobium plicatile. Phytochemistry 43:207–208
Barua AK, Ghosh BB, Ray S, Patra A (1990) Cymbinodin-A, a phenanthraquinone from Cymbidium aloifolium. Phytochemistry 29:3046–3047
Lin T-H, Chang S-J, Chen C-C, Wang J-P, Tsao L-T (2001) Two phenanthraquinones from Dendrobium moniliforme. J Nat Prod 64:1084–1086
Majumder PL, Pal A, Joardar M (1990) Cirrhopetalanthrin, a dimeric phenanthrene derivative from the orchid Cirrhopetalum maculosum. Phytochemistry 29:271–274
Bai L, Kato T, Inoue K, Yamaki M, Takagi S (1991) Blestrianol A, B and C, biphenanthrenes from Bletilla striata. Phytochemistry 30:2733–2735
Tezuka Y, Ji L, Hirano H, Ueda M, Nagashima K, Kikuchi T (1990) Studies on the constituents of orchidaceous plants. IX: Constituents of Spiranthes sinensis (PERS.) AMES var. amoena (M. BIEBERSON) HARA. (2). Structures of Spirantheosol, Spiranthoquinone, Spiranthol-C, and Spirasineol-B, new isopentenyldihydrophenanthrenes. Chem Pharm Bull 38:629–635
Xue Z, Li S, Wang S, Wang Y, Yang Y, Shi J, He L (2006) Mono-, Bi-, and triphenanthrenes from the tubers of Cremastra appendiculata. J Nat Prod 69:907–913
Dewick PM (2002) Medicinal natural products. Wiley, Chichester
Sotheeswaran S, Pasupathy V (1993) Distribution of resveratrol oligomers in plants. Phytochemistry 32:1083–1092
Lin M, Yao C-S (2006) Natural oligostilbenes. In: Atta-ur-Rahman (ed) Studies in natural products chemistry. Elsevier, Amsterdam
Friederich S, Rueffer M, Asakawa Y, Zenk MH (1999) Cytochromes P-450 catalyze the formation of marchantins A and C in Marchantia polymorpha. Phytochemistry 52:1195–1202
Zinsmeister HD, Becker H, Eiche T (1991) Bryophytes, a source of biologically active, naturally occurring material? Angew Chem Int Ed Engl 30:130–147
Hashimoto T, Kananyama S, Kan Y, Tori M, Asakawa Y (1996) Isoplagiochins C and D, new type of macrocyclic bis(bibenzyls), having two biphenyl linkages from the liverwort Plagiochila fruticosa. Chem Lett 25:741
Vang O, Ahmad N, Baile CA, Baur JA, Brown K, Csiszar A, Das DK, Delmas D, Gottfried C, Lin H-Y, Ma Q-Y, Mukhopadhyay P, Nalini N, Pezzuto JM, Richard T, Shukla Y, Y-JSurh ST, Szkudelski T, Walle T, Wu JM (2011) What is new for an old molecule? Systematic review and recommendations on the use of resveratrol. PLoS One 6:e19881
Pettit GR, Singh SB, Boyd MR, Hamel E, Pettit RK, Schmidt JM, Hogan F (1995) Antineoplastic agents. 291. Isolation and synthesis of combretastatins A-4, A-5, and A-6. J Med Chem 38:1666–1672
Lin CM, Ho HH, Pettit GR, Hamel E (1989) Antimitotic natural products combretastatin A-4 and combretastatin A-2: studies on the mechanism of their inhibition of the binding of colchicine to tubulin. Biochemistry 28:6984–6991
Wang YQ, Tan JJ, Tan CH, Jiang SH, Zhu DY (2003) Halophilols A and B, two new stilbenes from Iris halophila. Planta Med 69:779–781
Yoder BJ, Cao S, Norris A, Miller JS, Ratovoson F, Razafitsalama J, riantsiferana R, Rasamison VE, Kingston DGI (2007) Antiproliferative prenylated stilbenes and flavonoids from Macaranga alnifolia from the Madagascar rainforest. J Nat Prod 70:342–346
Puntumchai A, Kittakoop P, Rajviroongit S, Vimuttipong S, Likhitwitayawuid K, Thebtaranonth Y (2004) Lakoochins A and B, new antimycobacterial stilbene derivatives from Artocarpus lakoocha. J Nat Prod 67:485–486
Ito T, Tanaka T, Ali Z, Akao Y, Nozawa Y, Takahashi Y, Sawa R, Nakaya K, Murata J, Darnaedi D, Iinuma M (2004) A new resveratrol hexamer from Upuna borneensis. Heterocycles 63:129–136
Yamada M, Hayashi K, Hayashi H, Ikeda S, Hoshino T, Tsutsui K, Tsutsui K, Iinuma M, Nozaki H (2006) Stilbenoids of Kobresia nepalensis (Cyperaceae) exhibiting DNA topoisomerase II inhibition. Phytochemistry 67:307–313
Yamada M, Hayashi K, Hayashi H, Tsuji R, Kakumoto K, Ikeda S, Hoshino T, Tsutsui K, Ito T, Iinuma M, Nozaki H (2006) Nepalensinols D-G, new resveratrol oligomers from Kobresia nepalensis (Cyperaceae) as potent inhibitors of DNA topoisomerase II. Chem Pharm Bull 54:354–358
Cushman M, Nagarathnam D, Gopal D, Chakraborti AK, Lin CM, Hamel E (1991) Synthesis and evaluation of stilbene and dihydrostilbene derivatives as potential anticancer agents that inhibit tubulin polymerization. J Med Chem 34:2579–2588
Li YM, Wang HY, Liu GQ (2001) Erianin induces apoptosis in human leukemia HL-60 cells. Acta Pharmacol Sin 22:1018–1022
Zhao W, Qin G, Ye Y, Xu R, Le X (1995) Bibenzyls from Stemona tuberosa. Phytochemistry 38:711–713
Scher JM, Burgess EJ, Lorimerb SD, Perry NB (2002) A cytotoxic sesquiterpene and unprecedented sesquiterpene-bisbibenzyl compounds from the liverwort Schistochila glaucescens. Tetrahedron 58:7875–7882
Lee YH, Park JD, Baek NI, Kim SI, Ahn BZ (1995) In vitro and in vivo antitumoral phenanthrenes from the aerial parts of Dendrobium nobile. Planta Med 61:178–180
Zhang G-N, Zhong L-Y, Bligh SWA, Guo Y-L, Zhang C-F, Zhang M, Wang Z-T, Xu L-S (2005) Bi-bicyclic and bi-tricyclic compounds from Dendrobium thyrsiflorum. Phytochemistry 66:1113–1120
Vasas A, Falkay G, Hohmann J, Forgo P, Zupkó I, Réthy B, Kovács A, (2006) Cytotoxic phenanthrenes from the rhizomes of Tamus communis. Planta Med 72:767–770
Kovács A, Forgo P, Zupkó I, Réthy B, Falkay GY, Szabó P, Hohmann J (2007) Steroids from Amalocalyx yunnanensis. Phytochemistry 33:687–692
Long L, Lee SK, Chai H-B, Rasoanaivo P, Gao Q, Navarro H, Wall ME, Wani MC, Farnsworth NR, Cordell GA, Pezzuto JM, Kinghorn AD (1997) Novel bioactive phenanthrene derivatives from Domohinea perrieri. Tetrahedron 53:15663–15670
Chen Y, Wei X, Xie H, Deng H (2008) Antioxidant 2-phenylbenzofurans and a coumestan from Lespedeza virgata. J Nat Prod 71:929–932
Hsiao G, Teng C-M, Wu C-L, Ko F-N (1996) Marchantin H as a natural antioxidant and free radical scavenger. Arch Biochem Biophys 334:18–26
Ko F-N, Liao C-H, Wu C-L (1995) Marchantinquinone, isolated from Reboulia hemisphaerica, as inhibitor of lipid peroxidation and as free radical scavenger. Chem Biol Interact 98:131–143
Schwartner C, Michel C, Stettmaier K, Wagner H, Bors W (1996) Marchantins and related polyphenols from liverwort: physico-chemical studies of their radical-scavenging properties. Free Radic Biol Med 20:237–244
Venkateswarlu S, Raju MSS, Subbaraju GV (2002) Synthesis and biological activity of isoamoenylin, a metabolite of Dendrobium amoenum. Biosci Biotechnol Biochem 66:2236–2238
Biondi DM, Rocco C, Ruberto G (2003) New dihydrostilbene derivatives from the leaves of Glycyrrhiza glabra and evaluation of their antioxidant activity. J Nat Prod 66:477–480
Wei HC, Ma SJ, Wu CL (1995) Sesquiterpenoids and cyclic bisbibenzyls from the liverwort Reboulia hemisphaerica. Phytochemistry 39:91–97
Liao C-H, Ko F-N, Wu C-L, Teng C-M (2000) Antiplatelet effect of marchantinquinone, isolated from Reboulia hemisphaerica, in rabbit washed platelets. J Pharm Pharmacol 52:353–359
Miyazawa M, Shimamura H, Nakamura S, Kameoka H (1997) Antimutagenic activity of gigantol from Dendrobium nobile. J Agric Food Chem 45:2849–2853
Chen C-C, Huang Y-L, Teng C-M (2000) Antiplatelet aggregation principles from Ephemerantha lonchophylla. Planta Med 66:372–374
Chen C-C (1994) Antiplatelet aggregation principles of Dendrobium loddigesii. J Nat Prod 57:1271–1274
Nagashima F, Momosaki S, Watanabe Y, Toyota M, Huneck S, Asakawa Y (1996) Terpenoids and aromatic compounds from six liverworts. Phytochemistry 41:207–211
Kerem Z, Bilkis I, Flaishman MA, Sivan L (2006) Antioxidant activity and inhibition of α-glucosidase by trans-resveratrol, piceid, and a novel trans-stilbene from the roots of Israeli Rumex bucephalophorus L. J Agric Food Chem 54:1243–1247
Na M, Hoang DM, Njamen D, Mbafor JT, Fomum ZT, Thuong PT, Ahn JS, Oh WK (2007) Inhibitory effect of 2-arylbenzofurans from Erythrina addisoniae on protein tyrosine phosphatase-1B. Bioorg Med Chem Lett 17:3868–3871
Lam SH, Chen JM, Kang CJ, Chen CH, Lee SS (2008) α-Glucosidase inhibitors from the seeds of Syagrus romanzoffiana. Phytochemistry 69:1173–1178
Muhammad I, Li XC, Dunbar DC, ElSohly MA, Khan IA (2001) Antimalarial (+)-trans-hexahydrodibenzopyran derivatives from Machaerium multiflorum. J Nat Prod 64:1322–1325
Kittakoop P, Kirtikara K, Tanticharoen M, Thebtaranonth Y (2000) Antimalarial preracemosols A and B, possible biogenetic precursors of racemosol from Bauhinia malabarica Roxb. Phytochemistry 55:349–352
Boonlaksiri C, Oonanant W, Kongsaeree P, Kittakoop P, Tanticharoen M, Thebtaranonth Y (2000) An antimalarial stilbene from Artocarpus integer. Phytochemistry 54:415–417
Okasaka M, Takaishi Y, Kogure K, Fukuzawa K, Shibata H, Higuti T, Honda G, Ito M, Kodzhimatov OK, Ashurmetov O (2004) New stilbene derivatives from Calligonum leucocladum. J Nat Prod 67:1044–1046
Ge HM, Huang B, Tan SH, Shi DH, Song YC, Tan RX (2006) Bioactive oligostilbenoids from the stem bark of Hopea exalata. J Nat Prod 69:1800–1802
Scher JM, Speakman J-B, Zapp J, Becker H (2004) Bioactivity guided isolation of antifungal compounds from the liverwort Bazzania trilobata (L.) S.F. Gray. Phytochemistry 65:2583–2588
Lorimer SD, Perry NB (1993) An antifungal bibenzyl from the New Zealand liverwort, Plagiochila stephensoniana. Bioactivity-directed isolation, synthesis, and analysis. J Nat Prod 56:1444–1450
Waffo-Teguo P, Lee D, Cuendet M, Merillon JM, Pezzuto JM, Kinghorn AD (2001) Two new stilbene dimer glucosides from grape (Vitis vinifera) cell cultures. J Nat Prod 64:136–138
Huang KS, Lin M, Cheng GF (2001) Anti-inflammatory tetramers of resveratrol from the roots of Vitis amurensis and the conformations of the seven-membered ring in some oligostilbenes. Phytochemistry 58:357–362
Lee D, Cuendet M, Vigo JS, Graham JG, Cabieses F, Fong HHS, Pezzuto JM, Kinghorn AD (2001) A novel cyclooxygenase-inhibitory stilbenolignan from the Seeds of Aiphanes aculeata. Org Lett 3:2169
Adams M, Pacher T, Greger H, Bauer R (2005) Inhibition of leukotriene biosynthesis by stilbenoids from Stemona species. J Nat Prod 68:83–85
Harinantenaina L, Quang DN, Takeshi N, Hashimoto T, Kohchi C, Soma G-I, Asakawa Y (2005) Bis(bibenzyls) from liverworts inhibit lipopolysaccharide-induced inducible NOS in RAW 264.7 cells: a study of structure – activity relationships and molecular mechanism. J Nat Prod 68:1779–1781
Lee KY, Sung SH, Kim YC (2006) Neuroprotective bibenzyl glycosides of Stemona tuberosa roots. J Nat Prod 69:679–681
Ge HM, Zhu CH, Shi DH, Zhang LD, Xie DQ, Yang J, Ng SW, Tan RX (2008) Hopeahainol A: an acetylcholinesterase inhibitor from Hopea hainanensis. Chem Eur J 14:376–381
Liu JY, Ye YH, Wang L, Shi DH, Tan RX (2005) New resveratrol oligomers from the stem bark of Hopea hainanensis. Helv Chim Acta 88:2910–2917
Yang H, Lee MK, Kim YC (2005) Protective activities of stilbene glycosides from Acer mono leaves against H2O2-induced oxidative damage in primary cultured rat hepatocytes. J Agric Food Chem 53:4182–4186
Hernez-Romero Y, Acevedo L, Sanchez M, Shier WT, Abbas HK, Mata R (2005) Phytotoxic activity of bibenzyl derivatives from the orchid Epidendrum rigidum. J Agric Food Chem 53:6276–6280
Della Greca M, Fiorentino A, Monaco P, Pinto G, Previtera L, Zarrelli A (2001) Synthesis and antialgal activity of dihydrophenanthrenes and phenanthrenes 11: mimics of naturally occurring compounds in Juncus effusus. J Chem Ecol 27:257–271
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this entry
Cite this entry
Shen, T., Xie, CF., Wang, XN., Lou, HX. (2013). Stilbenoids. In: Ramawat, K., Mérillon, JM. (eds) Natural Products. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-22144-6_63
Download citation
DOI: https://doi.org/10.1007/978-3-642-22144-6_63
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-22143-9
Online ISBN: 978-3-642-22144-6
eBook Packages: Chemistry and Materials ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics