Abstract
Perhaps the most significant change in plant electrophysiological studies that began some 25 years ago was a shift in focus from more basic electrical and biophysical properties of plant membranes to pursuing the understanding of the plant physiological and cell biological functions of individual plant ion channel types. In the 1990s, ion channels were characterized as targets of upstream signal transduction mechanisms, and in the later 1990s powerful combined molecular genetics, patch clamp, and plant physiological response analyses further manifested the importance of ion channels for many biological and stress responses of plants. Essential metals and ions in the intracellular and intraorganellar spaces of plant cells contribute to the activities of regulatory proteins, signal transduction, and to the maintenance of turgor pressure, osmoregulation, toxic metal chelation, and membrane potential control. A large number of studies on mineral nutrition have sustained the profitable cultivation of plant growth and development, and provided important knowledge on plant physiological mechanism of absorption of minerals from soils. Abiotic stress and biotic stresses are a global problem for plant growth in agricultural and noncultivated lands. Ion channels in plant cells play crucial functions in adapting to and overcoming abiotic and biotic stresses. Plant membrane transport systems play an important role not only in the uptake of nutrients from the soil but also in the adaptation to stress and environmental change.
A revolution has taken place in the understanding of cell physiological, biophysical, molecular, and interaction network properties of plant ion channels and transporters as summarized here. However, many exciting and stimulating questions remain open to discovery, promising that research on plant ion channels will continue to be a vibrant area of research for many years to come.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Among plant nutrients, potassium or K+, is the most abundant cellular cation controlling cellular homeostasis, plant movements, cell expansion, guard cell turgor, membrane potentials, and many other processes. Potassium ions also counteract toxic effects of cations such as sodium (Na+). Potassium transport properties have served as a classical model for understanding mechanisms of plant ion transport (Epstein et al. 1963). Studies indeed show that principles learned from K+ transport and K+ channel analyses can be applied to other transport systems.
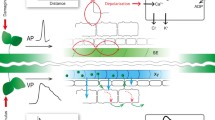
Characterization of ion channel functions in plant stress responses led to the formulation of models of how multiple ion channels and transporters can function together in mediating a response. Studies in guard cells led to an early model for the interplay of a network of ion channels and proton ATPases in mediating stomatal opening and closing (Shimazaki et al. 1986; Schroeder and Hagiwara 1989; Schroeder and Hedrich 1989; Thiel et al. 1992; Lemtiri-Chlieh and MacRobbie 1994; Ward and Schroeder 1994; Davies and Sanders 1995; Blatt et al. 1999). Remarkably, studies of rapid changes in plant pathogen responses and other rapid stimulus-responses show ion transport behaviors that, at least in general terms, show similarity to those mediating stomatal closing (Nurnberger et al. 1994; Jabs et al. 1997). In guard cells, cytosolic Ca2+ activates anion efflux channels and inhibits K+ uptake channels (Schroeder and Hagiwara 1989), which together with Ca2+ inhibition of plasma membrane proton pumps (Kinoshita et al. 1995) causes anion and K+ efflux and depolarization of the plasma membrane to reduce the turgor pressure of guard cells.
Calcium (Ca2+) concentrations are tightly controlled at low submicromolar concentrations in the cytosol. Increases in Ca2+ concentrations and stimulus-induced enhancement in Ca2+ sensitivity (Young et al. 2006) function as an effective signal which modulates calcium-binding proteins thus transmitting signals in signal transduction pathways. Ion channels that mediate Ca2+ influx into the cytosol from the extracellular space and from organelles have been characterized in electrophysiological studies (Miller et al. 1990). However, the genes encoding these ion channels still remain mostly uncharacterized in plant cells, probably due to the presence of large gene families with overlapping functions (Shimazaki et al. 1986; Blatt 2000; Ward et al. 2009). Anion channels in the plasma membrane have also emerged as major mechanisms regulating signal transduction and ion transport. Two types of anion channel currents (slow (S)-type and rapid (R)-type) have been characterized extensively in guard cells and in hypocotyl cells (Keller et al. 1989; Schroeder and Hagiwara 1989; Marten et al. 1992; Colcombet et al. 2001), and genes encoding the anion conducting subunits of slow-type anion channels have been identified using Arabidopsis mutants (Negi et al. 2008; Vahisalu et al. 2008). Recent genetic approaches have led to identification of two additional gene families that encode anion conducting channel subunits that play major roles in aluminum resistance (Sasaki et al. 2004; Furukawa et al. 2007; Magalhaes et al. 2007). Yet another class of proteins exists, which shows similarity to mammalian chloride channels, AtCLCs. Functional characterization of the AtCLCa membrane protein showed that it encodes a nitrate-proton exchanger in the vacuolar membrane, rather than an anion channel (De Angeli et al. 2006) and additional CLC proteins are targeted to other organelle membranes (Marmagne et al. 2007).
In this chapter, we will provide an overview of the classes of different ion channels that have been characterized and their underlying gene families. In several cases, we discuss examples of their physiological functions in guard cells as well as in other cell and tissue types. The relevance of these channels in stress responses in many plant cell types is discussed throughout this book. Figures 1 and 2 summarize progress over the past 25 years in the identification of plant ion channel classes, technical advances, and major genes encoding plant membrane transport systems. Figure 1 exemplifies the accelerating pace of discovery in this thriving field.
Simplified model for mechanisms of Na+ absorption, recirculation, and extrusion by different classes of Na+ channels/transporters, including Na+ loaded into xylem vessel by non-selective outwardly rectifying cation conductance, NORC (Wegner and De Boer 1997), Na+ influx mediated by HKT transporters (Uozumi et al. 2000; Mäser et al. 2002a; Sunarpi et al. 2005), plasma membrane Na+ extrusion via SOS1 antiporters (Shi et al. 2000), and tonoplast Na+ sequestration by NHX antiporters (Apes et al. 1999). AtHKT1;1, and OsHKT1;5 are present in the plasma membrane of xylem parenchyma cells, and mediate unloading of Na+ from xylem vessels into xylem parenchyma cells, thus protecting leaves from Na+ overaccumulation and Na+ damage (leaf Na+ exclusion) (Berthomieu et al. 2003; Sunarpi et al. 2005; Ren et al. 2005). In the case of K+ starvation in soils, rice roots take up Na+ at low extracellular Na+ levels via OsHKT2;1 (Horie et al. 2007). Na+ is sequestered in vacuoles by AtNHX1. Excessive Na+ in the cytosol is transported out of cells by SOS1
2 Plasma Membrane K+ Channels in Guard Cells
The first characterizations of single plant ion channels were reported in 1984 in the analyses of leaf cells (Moran et al. 1984) and guard cells (Schroeder et al. 1984). These successful applications of patch clamp techniques for the measurement of plant ion channels opened the door to electrophysiological characterizations of ion channels in plant membranes of land plant cells, which are usually orders of magnitude smaller than the classically analyzed giant algae cells (Curtis and Cole 1938; Tazawa 1968, 1972). They also reported the measurement of sodium, potassium, and chloride ions in protoplasm of algal cells, which may be more difficult to measure than plant cells. Two major classes of voltage-dependent K+ channels were characterized in guard cells; hyperpolarization-activated “inward-rectifying” K+ channels and depolarization-activated “outward-rectifying” K+ channels (Schroeder et al. 1984, 1987; Blatt 1988). Inward-rectifying K+ channels are activated by hyperpolarization via electrogenic proton pumps controlled by blue light signals (Assmann et al. 1985; Shimazaki et al. 1986). The opening of stomatal pores is regulated by the accumulation of K+ in guard cells. Both inward- and outward-rectifying K+ channels were proposed to contribute to the physiological transport of K+ into and out of guard cells during stomatal movements (Schroeder et al. 1987). Subsequent studies in many different plant cell types including coleoptiles, root hair cells, aleurone, root cortex, and xylem parenchyma cells showed that these types of K+ channels are widely distributed and were proposed to have important functions in K+ transport and membrane potential control (Bush et al. 1988; Kourie and Goldsmith 1992; Gassmann and Schroeder 1994; Wegner and Raschke 1994; Maathuis et al. 1997; de Boer and Volkov 2003).
2.1 Characterization of K+ Channel and Transporter cDNAs
In 1992, two distinct K+ channel genes, KAT1 and AKT1, were isolated from Arabidopsis thaliana by complementation of K+ uptake deficient yeast mutants (Anderson et al. 1992; Sentenac et al. 1992). Both genes encode six putative transmembrane regions and a predicted voltage sensor domain, and resemble Shaker K+ channels in Drosophila neurons. For the isolation of these genes, both groups used yeast mutants which are unable to grow at low concentrations of K+ in the medium. The use of yeast expression systems provides a powerful method for isolation of channel and transporter cDNAs and for structure-function analyses of these transporters (Frommer and Ninnemann 1995; Uozumi et al. 1995; Hoth et al. 1997; Nakamura et al. 1997).
Electrophysiological characterization of the KAT1-encoded protein in Xenopus oocytes showed that KAT1 functions as a heperpolarization-activated K+ channel (Schachtman et al. 1992). Thus these studies led to the first isolation and characterization of eukaryotic inward-rectifying K+ channel genes (Anderson et al. 1992; Schachtman et al. 1992; Sentenac et al. 1992), as hyperpolarization-activated K+ channels genes had not yet been identified in animal genomes (Kubo et al. 1993; Ward et al. 2009).
AKT1 expression in Xenopus oocytes failed to show ion channel activities, but insect cells (Sf9 cell line) expressing AKT1 displayed an inwardly rectifying K+ conductance (Gaymard et al. 1996). Other types of Arabidopsis K+ channel genes have been isolated after this; a weakly inward-rectifying K+ channel, AKT2 (Cao et al. 1995; Ketchum and Slayman 1996), depolarization-activated K+ channels, SKOR and GORK (Gaymard et al. 1998; Ache et al. 2000), and a silent channel, AtKC1 which is likely to modulate other K+ channels (Dreyer et al. 1997; Reintanz et al. 2002). The role of the silent regulatory subunit has been confirmed for the carrot AtKC1 homolog, KDC1 (Bregante et al. 2008). The cytosolic regulatory components, calcineurin B-like proteins (CBLs), and CBL-interacting protein kinases (CIPKs) are closely associated with several ion channels and transporters that function in adaptation to salinity or ion stress in plant cells. The complex of CBL1/CIPK23 directly controls AKT1-mediated K+ uptake in roots and enhances K+ uptake when ambient K+ becomes deficient (Li et al. 2006; Xu et al. 2006).
Interestingly, Escherichia coli was shown to be another heterologous expression system suitable for functional expression of both plasma membrane-located and organelle membrane-located plant channels/transporters (Uozumi 2001). Using this system, K+ uptake activities of KAT1, AKT2, HKT-type transporters, and KUP-type transporters were measured (Kim et al. 1998; Uozumi et al. 2000; Uozumi 2001). Moreover, the transmembrane topologies of the Shaker-type K+ channel KAT1 and the Na+/K+ transporter, HKT1 (TaHKT2;1) were determined by means of a bacterial alkaline phosphatase fusion approarch (Kim et al. 1998; Uozumi et al. 1998, 2000; Kato et al. 2001; Uozumi 2001).
KUP/HAK/KT genes encode a separate class of important plant K+ uptake transport proteins and were isolated after earlier genomic EST sequencing showed plant isoforms with homology to E. coli Kup and yeast HAK transporters (Quintero and Blatt 1997; Santa-Maria et al. 1997; Fu and Luan 1998; Kim et al. 1998). The Arabidopsis genome sequence shows the presence of 13 genes KUP/HAK/KT genes in the Arabidopsis thaliana genome (Mäser et al. 2001; Ahn et al. 2004), and the physiological role of AtKUP4 and AtHAK5 has been reported (Rigas et al. 2001; Gierth et al. 2005). AKT1 and AtHAK5 likely together mediate K+ uptake from soil. The transport mechanism by which these KUP/HAK/KTs mediate K+ uptake into plants cells remains unknown (Maathuis and Sanders 1994). An important question for future research will be the characterization of the interplay of several different K+ transporter/channel classes in mediating K+ transport.
3 Critical Roles of Plasma Membrane Anion Channels in Plant Stress Responses
Stomatal closing is mediated by the release of ions and organic solutes from guard cells. Electrophysiological studies led to a model for the mechanisms that can drive K+ release from guard cells. Electrophysiological research on outward-rectifying K+ channels indicated that inhibition of proton pumps would not suffice for depolarization-activation of K+ channels (Schroeder et al. 1987; Schroeder 1988). Elevation of the cytosolic Ca2+ concentration in guard cells led to the activation of a novel class of plant ion channels – S-type anion channels (Schroeder and Hagiwara 1989). Due to the electrochemical gradient of anions across the plasma membrane of guard cells, activation of anion channels causes anion efflux leading to depolarization. Anion channels were therefore proposed as drivers of ion efflux, thus controlling stomatal closing (Schroeder and Hagiwara 1989). Further research revealed additional types of anion channel in guard cells with properties different from those of S-type anion channels (Keller et al. 1989). These so-called R-type anion channels can also mediate anion efflux leading to stomatal closing.
Anion channels in guard cells are permeable to chloride, nitrate, sulfate, and malate (Keller et al. 1989; Schroeder and Hagiwara 1989; Schmidt and Schroeder 1994). Patch clamp analyses of the plasma membrane of Vicia faba guard cells revealed that these two types of anion channel conductances coexist in the membrane (Schroeder and Keller 1992). R-type anion channels are characterized as rapidly activating with kinetics that are time- and voltage-dependent and that show inactivation (Keller et al. 1989; Hedrich et al. 1990). The other class of depolarization activated anion channels exhibits extremely slow voltage dependent activation and deactivation properties – the S-type anion channels (Schroeder and Hagiwara 1989; Schroeder and Keller 1992). It has been proposed that R-type and S-type anion channels may be encoded by the same channel protein (Linder and Raschke 1992), despite their relatively significant differences in some biophysical and regulatory properties. The plant hormone abscisic acid, which is induced in response to drought stress, activates both S-type and R-type anion channels (Grabov et al. 1997; Pei et al. 1997, 1998; Raschke 2003; Raschke et al. 2003; Roelfsema et al. 2004). S-type and R-type anion channels have also been characterized in hypocotyl cells of Arabidopsis and were also shown to co-exist in the same cells (Colcombet et al. 2005). Studies in the Arabidopsis hypocotyls also suggested that these two anion channels can be clearly distinguished in these cells (Colcombet et al. 2005). Nevertheless, it is possible that these two very different anion currents share molecular components (Raschke 2003).
4 Roles of Anion Channels in Stress Responses and Identification of Anion Channel Gene Families
SLAC1 (slow anion channel-associated 1) encodes a homologue of bacterial dicarboxylate/malic acid (C4-dicarboxylate) transport proteins and was identified as an S-type slow anion channel (Vahisalu et al. 2008). The plasma membrane protein SLAC1 plays an essential role in stomatal closure in response to CO2, ABA, ozone, darkness, humidity reduction, Ca ions, hydrogen peroxide, and nitric oxide (Negi et al. 2008; Vahisalu et al. 2008). Loss-of-function mutations in SLAC1 are accompanied by an overaccumulation of osmoregulatory anions in guard cell protoplasts (Negi et al. 2008). T-DNA insertion and point mutations in the SLAC1 gene led to abrogation of S-type anion channels in guard cells (Vahisalu et al. 2008). Interestingly however, R-type anion channels were intact in slac1 mutant guard cells. SLAC1 shows homology to a yeast and a bacterial malate transporter. The permeability of S-type anion channels to anions and the increased trapping of malate in slac1 guard cells suggest that SLAC1 encodes the anion conducting subunit of S-type anion channels (Negi et al. 2008; Vahisalu et al. 2008). Slac1 mutants provide strong evidence for the model that anion channels represent central mechanisms in mediating stomatal closing. Interestingly, a different type of malate transporter, AtABC14 has been identified as a malate import protein mediating malate uptake from the cell wall into guard cells (Lee et al. 2008) and thus distinct channels and transporters are now known that mediate anion efflux and uptake in guard cells.
Aluminum is the third most abundant element in the Earth’s crust. In acidic soils aluminum (Al3+) is solubilized and Al3+ is toxic to plants. However, plants release organic acids, including malate and citrate from their roots, to chelate free aluminum (Al3+) in acidic soil (Ma et al. 2001; Kochian et al. 2004). Al3+ activates anion channels in the plasma membrane of wheat roots (Ryan et al. 1997). Genes were identified in genetic studies and named ALMTs for Al3+-activated malate transporters, since they play important roles in this Al3+ resistance response (Sasaki et al. 2004). ALMT expression in Xenopus oocytes is sufficient for Al3+-activated anion channels, showing that ALMTs appear to function as a type of Al3+ receptor (Pineros et al. 2008). TaALMT1 mediates transport of malate, and to a lesser extent nitrate/chloride based on electrophysiological measurements (Pineros et al. 2008; Zhang et al. 2008).
Furthermore, Al3+-activated citrate transporters (HvAACT1) (Furukawa et al. 2007) and (SbMATE) (Magalhaes et al. 2007) belong to the multidrug resistance transporter family and also function in aluminum tolerance in acid soils. The Al3+ resistance-associated anion transporters show no homology to the above SLAC1 anion channel from guard cells.
In animals, chloride channels of the ClC family have been characterized. Bacterial CLC homologues however function as 2 Cl−/1H+ exchangers (Accardi and Miller 2004; Picollo and Pusch 2005; Miller 2006). The functions of the homologous genes in Arabidopsis and tobacco have largely remained unknown (Hechenberger et al. 1996; Lurin et al. 1996). However, in 2006 the AtCLCa transporter was characterized as a NO3-/H+ exchanger in the vacuolar membrane of Arabidopsis cells (De Angeli et al. 2006). Atclca knockout mutants provide evidence that AtCLCa functions in nitrate accumulation into vacuoles in Arabidopsis thaliana (Geelen et al. 2000). AtCLCd and AtCLCe are targeted to the thylakoid membranes in chloroplasts and AtCLCf was localized in Golgi membranes (Marmagne et al. 2007). Further studies on the subcellular localizations of AtCLCs may illuminate intracellular anion transport mechanisms in plant cells.
5 Ca2+ Channels and Intracellular Ca2+ Elevations
Stimulus-induced changes in the Ca2+ concentration in the cytoplasm of plant cells are triggered by many diverse stimuli (Hetherington and Brownlee 2004). Intracellular Ca2+ concentration changes in guard cells were identified using fluorescent Ca2+ indicators, Fura-2 (McAinsh et al. 1990; Schroeder and Hagiwara 1990), and Fluo-3 (Gilroy et al. 1990). Patch clamp analyses showed the presence of Ca2+-permeable channels in the plasma membrane of guard cells (Schroeder and Hagiwara 1990; Hamilton et al. 2000; Pei et al. 2000). ABA-induced intracellular Ca2+ elevations have been extensively studied (Allan et al. 1994; Grabov and Blatt 1998; Allen et al. 1999a; Staxen et al. 1999). The pH-independent, green florescent protein-based Ca2+ indicators yellow cameleon 2.1 and 3.6 were applied for monitoring cytoplasmic free Ca2+, [Ca2+]cyt, in Arabidopsis thaliana (Allen et al. 1999b; Miyawaki et al. 1999; Yang et al. 2008). Studies using low concentration cameleon or fura2-based Ca2+ reporters have revealed that repetitive spontaneous Ca2+ transients occur in plant cells (Grabov and Blatt 1998; Allen et al. 1999a; Staxen et al. 1999; Wais et al. 2000; Young et al. 2006; Yang et al. 2008). Furthermore, experimentally imposing Ca2+ oscillations, by repetitive depolarizations and hyperpolarizations of the plasma membrane, showed that independent of the Ca2+ elevation pattern, Ca2+-induced a rapid stomatal closure which was named the “Ca2+ reactive” stomatal closing response (Allen et al. 2001). In addition to this Ca2+ reactive response, it was revealed that the pattern of experimentally-induced [Ca2+]cyt elevations controls the ability of stomata to re-open after the initial stomatal closing response, even when the [Ca2+]cyt elevations are terminated (Allen et al. 2001; Li et al. 2004). This long-term Ca2+ pattern inhibition of re-opening of stomatal pores, was named the “Ca2+ programmed” response and is impaired in glutamate receptor overexpressing guard cells (Cho et al. 2009). Thus [Ca2+]cyt oscillation kinetics in guard cells can function in maintaining steady-state stomatal closing. Organelles in plant cells serve as intracellular stores for Ca2+. A Ca2+ sensing receptor, CAS, was isolated via a functional expression screening approach using heterologous expression (Han et al. 2003). Recent work shows that CAS1 is localized in thylakoid membranes (Nomura et al. 2008; Weinl et al. 2008) and functions in extracellular Ca2+-induced, transient cytosolic Ca2+ increases, which lead to stomatal closure (Han et al. 2003; Nomura et al. 2008; Weinl et al. 2008).
6 Gene Candidates for Plasma Membrane Ca2+ Channels
Several classes of Ca2+ permeable channels have been characterized in the plasma membrane of plant cells, including depolarization-activated Ca2+ channels (Thuleau et al. 1994a, b; Miedema et al. 2008) and hyperpolarization-activated Ca2+ influx channels (Gelli and Blumwald 1997; Hamilton et al. 2000; Pei et al. 2000; Demidchik et al. 2002). In general, plant Ca2+ channels are not entirely Ca2+ selective but also show permeabilities to other cations (Schroeder and Hagiwara 1990; Thuleau et al. 1994a, b; Pei et al. 2000; Demidchik et al. 2002). However, the genes encoding plasma membrane Ca2+ channels remain less well-clarified. Two gene families are likely to provide possible candidates. One family includes 20 genes in the Arabidopsis genome and encodes homologs to “ionotropic” glutamate receptors, which encode receptor ion channels in animal systems (Lam et al. 1998; Kim et al. 2001). Research has shown that glutamate application to roots causes [Ca2+]i elevations that are disrupted in knock-out mutants in the Glr3.3, glutamate receptor gene (Qi et al. 2006). A second candidate family of plant Ca2+ permeable channels is cyclic nucleotide-gated channel homologs. In Arabidopsis, 20 different cyclic nucleotide-gated channel genes (CNGCs) are present, and several individual channels have been analyzed. Voltage dependent K+ channels, including KAT1 and AKT1 have corresponding cyclic nucleotide binding sites in the C-terminal regions (Hoshi 1995). However, CNGC channels do not include the typical “GYG” K+ selectivity signature sequence of K+ channels (Ward et al. 2009). Studies analyzing CNGC functions after heterologous expression in yeast indicate that they may encode Ca2+ permeable channels (Kohler et al. 1999; Leng et al. 1999), although this may not apply to all members of the CNGC family. Genetic analysis showed that both AtCNGC11 and AtCNGC12 are positive mediators of resistance signaling pathways activated by pathogen infection (Yoshioka et al. 2006). Future research into the physiological functions of this large gene family may reveal new and unexpected ion channel functions.
7 Properties of Vacuolar Cation Channels
Plant vacuoles often take up more than 90% of the cell volume, and thus the channels mediating K+ transport across the vacuolar membrane (tonoplast) may be of relevance to cell volume regulation and storage of this nutrient. Three classes of cation channel, SV (Slow Vacuolar), VK (Vacuolar K), and FV (Fast Vacuolar), have been named based on the endogenous K+ channel activities identified by patch clamp studies. FV channels mediate K+ transport at very low concentrations of cytosolic Ca2+ (Hedrich and Neher 1987; Allen and Sanders 1996). SV channels are activated by elevation in the cytosolic Ca2+ concentration (Hedrich and Neher 1987; Pei et al. 1999). SV channels were initially reported to be anion permeable channels (Hedrich et al. 1986). However, later studies revealed that SV channels are Ca2+ permeable cation channels that do not significantly conduct anions (Ward and Schroeder 1994; Ward et al. 1995; Allen and Sanders 1996). A third class of vacuolar cation channels are the Ca2+-activated channels, named VK channels, which are highly K+ selective channels (Ward and Schroeder 1994). The determination of genome sequences of Arabidopsis and reverse genetic approaches have led to the identification of the genes encoding SV channels (Peiter et al. 2005) and VK channels (Gobert et al. 2007). The AtTPC1 protein is targeted to the vacuolar membrane and these proteins encode SV channels (Peiter et al. 2005). The genes encoding two-pore K+ channels (TPKs) include two repeats of membrane-pore-membrane domains (Czempinski et al. 1997, 2002; Kaplan et al. 2007). AtTPK1, 2, 3, and 5 are tonoplast K+ channels (Voelker et al. 2006), whereas AtTPK4 is located in the plasma membrane (Becker et al. 2004). AtTPK1 was shown to encode the VK channel (Gobert et al. 2007). Functional characterization of NtTPK1, located in tobacco tonoplasts, shows K+ currents induced by cytosolic acidification, indicating the presence of other types of vacuolar K+ channels that differ from the above vacuolar channel types (Hamamoto et al. 2008).
8 Sodium Transport Systems in Plants
Sodium (Na+) is not categorized as an essential nutrient in higher plants, and excessive Na+ leads to detrimental effects on plant growth. Several distinct classes of Na+ transporters mediate Na+ homeostasis (Fig. 3). After Na+ entry into the cytoplasm of root cells, Na+ is loaded into the xylem (de Boer 1999). The presence of a Na+/H+ exchange activity at the xylem/symplast interface of soybean roots (Lacan and Durand 1996) and Na+-permeable nonselective ion channels in the plasma membrane of barley root xylem parenchyma cells (NORC) (Wegner and De Boer 1997) and in wheat and Arabidopsis root cortex and epidermis (NSC) (Tyerman et al. 1997; Buschmann et al. 2000; Davenport and Tester 2000; Demidchik and Tester 2002) have been reported.
The exclusion of Na+ from plant cells and the sequestration of Na+ in vacuoles alleviate sodium stress under saline conditions. The plasma membrane Na+/H+ antiporter named SOS1 (Shi et al. 2000), was identified in an Arabidopsis mutant, sos1, that shows a salt oversensitive phenotype (Wu et al. 1996). SOS1-mediated Na+/H+ transport activity is modulated by a Ca2+ sensor/protein kinase complex CBL4 (SOS3)/CIPK24 (SOS2) (Wu et al. 1996; Shi et al. 2002; Zhu 2002). Na+/H+ antiporters were also identified which are targeted to the vacuole-membrane. The first functionally-characterized member of this gene family, AtNHX1, contributes to Na+ and monovalent cation sequestration in plant vacuoles. Overexpression of AtNHX1 was shown to increase salt tolerance in Arabidopsis (Apse et al. 1999).
In contrast to these Na+ transporters that remove Na+ from the cytoplasm, molecular identification of plasma membrane Na+ influx systems into plant cells has also been achieved. Na+ uptake transporters in wheat HKT1 also named, TaHKT1 (TaHKT2;1) (Schachtman and Schroeder 1994; Rubio et al. 1995; Gassmann et al. 1996) and in Arabidopsis thaliana AtHKT1 (AtHKT1;1) were identified (Uozumi et al. 2000). The first HKT gene, TaHKT1 (TaHKT2;1), was originally cloned from wheat and shown to mediate K+ and Na+ co-transport in yeast and Xenopus oocytes (Schachtman and Schroeder 1994; Rubio et al. 1995; Gassmann et al. 1996). Further extensive studies on HKT structure and function demonstrated that HKTs include 4 domains that resemble the K+ permeation pore of a K+ channel tetramer (Durell et al. 1999; Kato et al. 2001; Mäser et al. 2002a; Tholema et al. 2005; Gambale and Uozumi 2006) and HKT transporters have indeed been proposed to mediate channel-like transport (Gassmann et al. 1996; Corratge et al. 2007). Note that the term, transporter or channel has been used interchangeably for HKT transporters, and HKTs provide an interesting model to explore the shrinking distinctions between co-transporters and ion channels. Whereas some HKT transporters change their K+ and Na+ selectivities depending on the ionic conditions, similar to multi-ion channel pores (Schachtman and Schroeder 1994; Rubio et al. 1995; Gassmann et al. 1996; Horie et al. 2001), the only HKT transporter encoded in the Arabidopsis genome, AtHKT1, was found to be more Na+ selective (Uozumi et al. 2000). Further studies showed that HKT transporters fall into either of these two cation selectivity HKT subfamilies (Horie et al. 2001, 2006). Research identified an amino acid residue that contributes to the distinction of these two cation selectivities of HKT transporters: AtHKT1;1 has a Ser instead of Gly in the first pore loop region which reduces K+ selectivity. In contrast, TaHKT1 lacks this residue and is more Na+ selective (Durell et al. 1999; Mäser et al. 2002a; Tholema et al. 2005; Gambale and Uozumi 2006). The nomenclature of HKT transporters cloned from various plants has been divided into two distinct groups, which also largely separate these subfamilies by their Ser or Gly in the selectivity filter, with the exception of OsHKT2;1 (Horie et al. 2001). Bacterial HKT homologs, Trk, or Ktr transporters, function as major K+ uptake systems (Gaber et al. 1988; Ko et al. 1990; Schlosser et al. 1995; Nakamura et al. 1998; Matsuda et al. 2004). K+ uptake is stimulated by Na+ in the cyanobacterial Ktr homologues of this family and significantly contributes to adaptation to hyperosmolar shock (Matsuda et al. 2004).
The question why plants express Na+ selective Na+ influx transporters such as AtHKT1;1 remained. Null mutations or those that reduce activity in the Na+ transporter AtHKT1;1 (Mäser et al. 2002b; Gong et al. 2004; Berthomieu et al. 2003) resulted in Na+ overaccumulation in leaves of these plants. The AtHKT1;1 transporter was immuno-localized in the plasma membrane of xylem parenchyma cells (Sunarpi et al. 2005). The Na+ hypersensitive phenotype of Athkt1;1 mutants (Mäser et al. 2002b) is due to the lack of Na+ retrieval from xylem vessels by AtHKT1;1, leading to toxic Na+ overaccumulation in leaves (Sunarpi et al. 2005). Mapping of a salt tolerance quantitative trait locus (QTL) from rice led to the isolation of OsHKT1;5, which is expressed in xylem parenchyma cells (Ren et al. 2005) and thus AtHKT1;1 and OsHKT1;5 have analogous functions in Na+ retrieval from the xylem sap (Ren et al. 2005; Sunarpi et al. 2005). Interestingly, this HKT transporter-mediated exclusion of Na+ accumulation in Arabidopsis and rice leaves via Na+ removal from the xylem, has more recently been found to be the underlying mechanism of three major salinity tolerance QTLs in wheat (Byrt et al. 2007), providing an example of transfer of knowledge from model plants such as Arabidopsis and rice (Uozumi et al. 2000; Mäser et al. 2002b; Ren et al. 2005; Sunarpi et al. 2005), to applications in the field.
In contrast to the above discussed sodium toxicity at high Na+ concentrations, low concentrations of Na+ (e.g. < 5 mM) support growth of many plant species when K+ is deficient. The Na+ transporter OsHKT2;1 (previously named OsHKT1) is strongly induced in rice roots in response to K+ starvation (Horie et al. 2001). Three loss of function mutant lines in OsHKT2;1 exhibited substantial reduction in Na+ influx into plant roots, showing that rice plants use Na+ as a nutrient in the medium for their survival and growth under K+ starvation and low Na+ conditions (Horie et al. 2007). Thus several classes of Na+ transporters and exchangers exist in plants and each class has unique roles in mediating sodium tolerance.
9 Future Prospects
Starting 25 years ago the study of plant transport moved into the era of identifying and characterizing individual ion channels and transporters. Such studies have benefited from several independent technical innovations including patch clamping, heterologous expression in yeast, oocytes, E coli and animal cells, ion sensitive fluorophores for imaging, biophysical structure-function analyses, forward and reverse genetic analyses, and the sequencing of reference plant genomes. However, the genes encoding some of the known channels/transporters remain to be identified. Additional approaches will aid in their identification including genetic studies of natural variation, systems biology, in silico analyses and proteomics. Abiotic stress and biotic stress continuously influence the plant body. Plants have developed an adaptive response to them; for example, reactive oxygen species have been used as intracellular and extracellular signals, which regulate membrane transport system, and coregulate Ca2+ signaling (McAinsh et al. 1996; Pei et al. 2000; Foreman et al. 2003; Demidchik et al. 2007).
Interestingly, almost every characterized plant ion channel and transporter class was found to have unique and intriguing properties, which have required new concepts and interdisciplinary analyses for their characterizations. These unique properties are often intimately related to their physiological functions and remain a basis for further analyses in the future. These advances are also contributing to the derivation of fundamental principles on the relationship of channels and transporters in all organisms. Moreover, many of the identified plant ion channels and transporters are linked to major environmental stresses that are directly relevant for the challenges facing humanity in the present century, including drought resistance, desiccation avoidance, salt tolerance, aluminum resistance, pathogen responses, and water use efficiency. These pressing global needs will require further creative, interactive, and dynamic research efforts by the community of plant ion transport researchers. In particular, new knowledge will lead to the selection and generation of elite crops.
References
Accardi A, Miller C (2004) Secondary active transport mediated by a prokaryotic homologue of ClC Cl-channels. Nature 427:803–807
Ache P, Becker D, Ivashikina N, Dietrich P, Roelfsema MR, Hedrich R (2000) GORK, a delayed outward rectifier expressed in guard cells of Arabidopsis thaliana, is a K+-selective, K+-sensing ion channel. FEBS Lett 486:93–98
Ahn SJ, Shin R, Schachtman DP (2004) Expression of KT/KUP genes in Arabidopsis and the role of root hairs in K+ uptake. Plant Physiol 134:1135–1145
Allan AC, Fricker MD, Ward JL, Beale MH, Trewavas AJ (1994) Two transduction pathways mediate rapid effects of abscisic acid in Commelina guard cells. Plant Cell 6:1319–1328
Allen GJ, Chu SP, Harrington CL, Schumacher K, Hoffmann T, Tang YY, Grill E, Schroeder JI (2001) A defined range of guard cell calcium oscillation parameters encodes stomatal movements. Nature 411:1053–1057
Allen GJ, Kuchitsu K, Chu SP, Murata Y, Schroeder JI (1999a) Arabidopsis abi1–1 and abi2–1 phosphatase mutations reduce abscisic acid-induced cytoplasmic calcium rises in guard cells. Plant Cell 11:1785–1798
Allen GJ, Kwak JM, Chu SP, Llopis J, Tsien RY, Harper JF, Schroeder JI (1999b) Cameleon calcium indicator reports cytoplasmic calcium dynamics in Arabidopsis guard cells. Plant J 19:735–747
Allen GJ, Sanders D (1996) Control of ionic currents in guard cell vacuoles by cytosolic and luminal calcium. Plant J 10:1055–1069
Anderson JA, Huprikar SS, Kochian LV, Lucas WJ, Gaber RF (1992) Functional expression of a probable Arabidopsis thaliana potassium channel in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 89:3736–3740
Apse MP, Aharon GS, Snedden WA, Blumwald E (1999) Salt tolerance conferred by overexpression of a vacuolar Na+/H+ antiport in Arabidopsis. Science 285:1256–1258
Assmann SM, Simoncini L, Schroeder JI (1985) Blue light activates electrogenic ion pumping in guard cell protoplasts of Visia faba. Nature 318:3
Becker D, Geiger D, Dunkel M, Roller A, Bertl A, Latz A, Carpaneto A, Dietrich P, Roelfsema MR, Voelker C, Schmidt D, Mueller-Roeber B, Czempinski K, Hedrich R (2004) AtTPK4, an Arabidopsis tandem-pore K+ channel, poised to control the pollen membrane voltage in a pH- and Ca2+-dependent manner. Proc Natl Acad Sci USA 101:15621–15626
Berthomieu P, Conejero G, Nublat A, Brackenbury WJ, Lambert C, Savio C, Uozumi N, Oiki S, Yamada K, Cellier F, Gosti F, Simonneau T, Essah PA, Tester M, Very AA, Sentenac H, Casse F (2003) Functional analysis of AtHKT1 in Arabidopsis shows that Na+ recirculation by the phloem is crucial for salt tolerance. EMBO J 22:2004–2014
Blatt MR (1988) Potassiium-dependent, bipolar gating of K+ channels in guard cells. J Membr Biol 102:235–246
Blatt MR (2000) Cellular signaling and volume control in stomatal movements in plants. Annu Rev Cell Dev Biol 16:221–241
Blatt MR, Grabov A, Brearley J, Hammond-Kosack K, Jones JD (1999) K+ channels of Cf-9 transgenic tobacco guard cells as targets for Cladosporium fulvum Avr9 elicitor-dependent signal transduction. Plant J 19:453–462
Bregante M, Yang Y, Formentin E, Carpaneto A, Schroeder JI, Gambale F, Lo Schiavo F, Costa A (2008) KDC1, a carrot Shaker-like potassium channel, reveals its role as a silent regulatory subunit when expressed in plant cells. Plant Mol Biol 66:61–72
Buschmann PH, Vaidyanathan R, Gassmann W, Schroeder JI (2000) Enhancement of Na+ uptake currents, time-dependent inward-rectifying K+ channel currents, and K+ channel transcripts by K+ starvation in wheat root cells. Plant Physiol 122:1387–1397
Bush DS, Hedrich R, Schroeder JI, Jones RL (1988) Channel-mediated K+ flux in barley aleurone protoplasts. Planta 176:368–377
Byrt CS, Platten JD, Spielmeyer W, James RA, Lagudah ES, Dennis ES, Tester M, Munns R (2007) HKT1;5-like cation transporters linked to Na+ exclusion loci in wheat, Nax2 and Kna1. Plant Physiol 143:1918–1928
Cao Y, Ward JM, Kelly WB, Ichida AM, Gaber RF, Anderson JA, Uozumi N, Schroeder JI, Crawford NM (1995) Multiple genes, tissue specificity, and expression-dependent modulationcontribute to the functional diversity of potassium channels in Arabidopsis thaliana. Plant Physiol 109:1093–1106
Cho D, Kim SA, Murata Y, Lee S, Jae SK, Nam HG, Kwak JM (2009) Deregulated expression of the plant glutamate receptor homolog AtGLR3.1 impairs long-term Ca-programmed stomatal closure. Plant J 58:437–449
Colcombet J, Lelievre F, Thomine S, Barbier-Brygoo H, Frachisse JM (2005) Distinct pH regulation of slow and rapid anion channels at the plasma membrane of Arabidopsis thaliana hypocotyl cells. J Exp Bot 56:1897–1903
Colcombet J, Thomine S, Guern J, Frachisse JM, Barbier-Brygoo H (2001) Nucleotides provide a voltage-sensitive gate for the rapid anion channel of Arabidopsis hypocotyl cells. J Biol Chem 276:36139–36145
Corratge C, Zimmermann S, Lambilliotte R, Plassard C, Marmeisse R, Thibaud JB, Lacombe B, Sentenac H (2007) Molecular and functional characterization of a Na+–K+ transporter from the Trk family in the ectomycorrhizal fungus Hebeloma cylindrosporum. J Biol Chem 282:26057–26066
Curtis HJ, Cole KS (1938) Transverse electric impedance of the squid giant axon. J Gen Physiol 21:757–765
Czempinski K, Frachisse JM, Maurel C, Barbier-Brygoo H, Mueller-Roeber B (2002) Vacuolar membrane localization of the Arabidopsis ‘two-pore’ K+ channel KCO1. Plant J 29:809–820
Czempinski K, Zimmermann S, Ehrhardt T, Muller-Rober B (1997) New structure and function in plant K+ channels: KCO1, an outward rectifier with a steep Ca2+ dependency. EMBO J 16:2565–2575
Davenport RJ, Tester M (2000) A weakly voltage-dependent, nonselective cation channel mediates toxic sodium influx in wheat. Plant Physiol 122:823–834
Davies JM, Sanders D (1995) ATP, pH and Mg2+ modulate a cation current in Beta vulgaris vacuoles: a possible shunt conductance for the vacuolar H+-ATPase. J Membr Biol 145:75–86
De Angeli A, Monachello D, Ephritikhine G, Frachisse JM, Thomine S, Gambale F, Barbier-Brygoo H (2006) The nitrate/proton antiporter AtCLCa mediates nitrate accumulation in plant vacuoles. Nature 442:939–942
de Boer AH (1999) Potassium translocation into the root xylem. Plant Biol 1:36–45
de Boer AH, Volkov V (2003) Logistics of water and salt transport through the plant: strucutre and functiong of the xylem. Plant Cell Environ 26:87–101
Demidchik V, Bowen HC, Maathuis FJ, Shabala SN, Tester MA, White PJ, Davies JM (2002) Arabidopsis thaliana root non-selective cation channels mediate calcium uptake and are involved in growth. Plant J 32:799–808
Demidchik V, Shabala SN, Davies JM (2007) Spatial variation in H2O2 response of Arabidopsis thaliana root epidermal Ca2+ flux and plasma membrane Ca2+ channels. Plant J 49:377–386
Demidchik V, Tester M (2002) Sodium fluxes through nonselective cation channels in the plasma membrane of protoplasts from Arabidopsis roots. Plant Physiol 128:379–387
Dreyer I, Antunes S, Hoshi T, Muller-Rober B, Palme K, Pongs O, Reintanz B, Hedrich R (1997) Plant K+ channel alpha-subunits assemble indiscriminately. Biophys J 72:2143–2150
Durell SR, Hao Y, Nakamura T, Bakker EP, Guy HR (1999) Evolutionary relationship between K+ channels and symporters. Biophys J 77:775–788
Epstein E, Rains DW, Elzam OE (1963) Resolution of dual mechanisms of potassium absorption by barley roots. Proc Natl Acad Sci USA 49:684–692
Foreman J, Demidchik V, Bothwell JH, Mylona P, Miedema H, Torres MA, Linstead P, Costa S, Brownlee C, Jones JD, Davies JM, Dolan L (2003) Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422:442–446
Frommer WB, Ninnemann O (1995) Heterologous expression of genes in bacterial, fungal, animal, and plant cells. Annu Rev Plant Physiol Plant Mol Biol 46:419–444
Fu HH, Luan S (1998) AtKuP1: a dual-affinity K+ transporter from Arabidopsis. Plant Cell 10:63–73
Furukawa J, Yamaji N, Wang H, Mitani N, Murata Y, Sato K, Katsuhara M, Takeda K, Ma JF (2007) An aluminum-activated citrate transporter in barley. Plant Cell Physiol 48:1081–1091
Gaber RF, Styles CA, Fink GR (1988) TRK1 encodes a plasma membrane protein required for high-affinity potassium transport in Saccharomyces cerevisiae. Mol Cell Biol 8:2848–2859
Gambale F, Uozumi N (2006) Properties of shaker-type potassium channels in higher plants. J Membr Biol 210:1–19
Gassmann W, Rubio F, Schroeder JI (1996) Alkali cation selectivity of the wheat root high-affinity potassium transporter HKT1. Plant J 10:869–852
Gassmann W, Schroeder JI (1994) Inward-rectifying K+ channels in root hairs of wheat (a mechanism for aluminum-sensitive low-affinity K+ uptake and membrane potential control). Plant Physiol 105:1399–1408
Gaymard F, Cerutti M, Horeau C, Lemaillet G, Urbach S, Ravallec M, Devauchelle G, Sentenac H, Thibaud JB (1996) The baculovirus/insect cell system as an alternative to Xenopus oocytes. First characterization of the AKT1 K+ channel from Arabidopsis thaliana. J Biol Chem 271:22863–22870
Gaymard F, Pilot G, Lacombe B, Bouchez D, Bruneau D, Boucherez J, Michaux-Ferriere N, Thibaud JB, Sentenac H (1998) Identification and disruption of a plant shaker-like outward channel involved in K+ release into the xylem sap. Cell 94:647–655
Geelen D, Lurin C, Bouchez D, Frachisse JM, Lelievre F, Courtial B, Barbier-Brygoo H, Maurel C (2000) Disruption of putative anion channel gene AtCLC-a in Arabidopsis suggests a role in the regulation of nitrate content. Plant J 21:259–267
Gelli A, Blumwald E (1997) Hyperpolarization-activated Ca2+-permeable channels in the plasma membrane of tomato cells. J Membr Biol 155:35–45
Gierth M, Maser P, Schroeder JI (2005) The potassium transporter AtHAK5 functions in K+ deprivation-induced high-affinity K+ uptake and AKT1 K+ channel contribution to K+ uptake kinetics in Arabidopsis roots. Plant Physiol 137:1105–1114
Gilroy S, Read ND, Trewavas AJ (1990) Elevation of cytoplasmic calcium by caged calcium or caged inositol triphosphate initiates stomatal closure. Nature 346:769–771
Gobert A, Isayenkov S, Voelker C, Czempinski K, Maathuis FJ (2007) The two-pore channel TPK1 gene encodes the vacuolar K+ conductance and plays a role in K+ homeostasis. Proc Natl Acad Sci USA 104:10726–10731
Gong JM, Waner DA, Horie T, Li SL, Horie R, Abid KB, Schroeder JI (2004) Microarray-based rapid cloning of an ion accumulation deletion mutant in Arabidopsis thaliana. Proc Natl Acad Sci USA 101:15404–15409
Grabov A, Blatt MR (1998) Membrane voltage initiates Ca2+ waves and potentiates Ca2+ increases with abscisic acid in stomatal guard cells. Proc Natl Acad Sci USA 95:4778–4783
Grabov A, Leung J, Giraudat J, Blatt MR (1997) Alteration of anion channel kinetics in wild-type and abi1–1 transgenic Nicotiana benthamiana guard cells by abscisic acid. Plant J 12:203–213
Hamamoto S, Marui J, Matsuoka K, Higashi K, Igarashi K, Nakagawa T, Kuroda T, Mori Y, Murata Y, Nakanishi Y, Maeshima M, Yabe I, Uozumi N (2008) Characterization of a tobacco TPK-type K+ channel as a novel tonoplast K+ channel using yeast tonoplasts. J Biol Chem 283:1911–1920
Hamilton DW, Hills A, Kohler B, Blatt MR (2000) Ca2+ channels at the plasma membrane of stomatal guard cells are activated by hyperpolarization and abscisic acid. Proc Natl Acad Sci USA 97:4967–4972
Han S, Tang R, Anderson LK, Woerner TE, Pei ZM (2003) A cell surface receptor mediates extracellular Ca2+ sensing in guard cells. Nature 425:196–200
Hechenberger M, Schwappach B, Fischer WN, Frommer WB, Jentsch TJ, Steinmeyer K (1996) A family of putative chloride channels from Arabidopsis and functional complementation of a yeast strain with a CLC gene disruption. J Biol Chem 271:33632–33638
Hedrich R, Busch H, Raschke K (1990) Ca2+ and nucleotide dependent regulation of voltage dependent anion channels in the plasma membrane of guard cells. EMBO J 9:3889–3892
Hedrich R, Flügge UI, Fernandez JM (1986) Patch-clamp studies of ion transport in isolated plant vacuoles. FEBS Lett 22:228–232
Hedrich R, Neher E (1987) Cytoplasmic calcium regulates voltage-dependent ion channels in plant vacuoles. Nature 329:833–837
Hetherington AM, Brownlee C (2004) The generation of Ca2+ signals in plants. Annu Rev Plant Biol 55:401–427
Horie T, Costa A, Kim TH, Han MJ, Horie R, Leung HY, Miyao A, Hirochika H, An G, Schroeder JI (2007) Rice OsHKT2;1 transporter mediates large Na+ influx component into K+-starved roots for growth. EMBO J 26:3003–3014
Horie T, Horie R, Chan WY, Leung HY, Schroeder JI (2006) Calcium regulation of sodium hypersensitivities of sos3 and athkt1 mutants. Plant Cell Physiol 47:622–633
Horie T, Yoshida K, Nakayama H, Yamada K, Oiki S, Shinmyo A (2001) Two types of HKT transporters with different properties of Na+ and K+ transport in Oryza sativa. Plant J 27:129–138
Hoshi T (1995) Regulation of voltage dependence of the KAT1 channel by intracellular factors. J Gen Physiol 105:309–328
Hoth S, Dreyer I, Dietrich P, Becker D, Muller-Rober B, Hedrich R (1997) Molecular basis of plant-specific acid activation of K+ uptake channels. Proc Natl Acad Sci USA 94:4806–4810
Jabs T, Tschope M, Colling C, Hahlbrock K, Scheel D (1997) Elicitor-stimulated ion fluxes and O2- from the oxidative burst are essential components in triggering defense gene activation and phytoalexin synthesis in parsley. Proc Natl Acad Sci USA 94:4800–4805
Kaplan B, Sherman T, Fromm H (2007) Cyclic nucleotide-gated channels in plants. FEBS Lett 581:2237–2246
Kato Y, Sakaguchi M, Mori Y, Saito K, Nakamura T, Bakker EP, Sato Y, Goshima S, Uozumi N (2001) Evidence in support of a four transmembrane-pore-transmembrane topology model for the Arabidopsis thaliana Na+/K+ translocating AtHKT1 protein, a member of the superfamily of K+ transporters. Proc Natl Acad Sci USA 98:6488–6493
Keller BU, Hedrich R, Raschke K (1989) Voltage-dependent anion channels in the plasma membrane of guard cells 450. Nature 341:450–453
Ketchum KA, Slayman CW (1996) Isolation of an ion channel gene from Arabidopsis thaliana using the H5 signature sequence from voltage-dependent K+ channels. FEBS Lett 378:19–26
Kim EJ, Kwak JM, Uozumi N, Schroeder JI (1998) AtKUP1: an Arabidopsis gene encoding high-affinity potassium transport activity. Plant Cell 10:51–62
Kim SA, Kwak JM, Jae SK, Wang MH, Nam HG (2001) Overexpression of the AtGluR2 gene encoding an Arabidopsis homolog of mammalian glutamate receptors impairs calcium utilization and sensitivity to ionic stress in transgenic plants. Plant Cell Physiol 42:74–84
Kinoshita T, Nishimura M, Shimazaki K (1995) Cytosolic Concentration of Ca2+ regulates the plasma membrane H+-ATPase in guard cells of fava bean. Plant Cell 7:1333–1342
Ko CH, Buckley AM, Gaber RF (1990) TRK2 is required for low affinity K+ transport in Saccharomyces cerevisiae. Genetics 125:305–312
Kochian LV, Hoekenga OA, Pineros MA (2004) How do crop plants tolerate acid soils? Mechanisms of aluminum tolerance and phosphorous efficiency. Annu Rev Plant Biol 55:459–493
Kohler C, Merkle T, Neuhaus G (1999) Characterisation of a novel gene family of putative cyclic nucleotide- and calmodulin-regulated ion channels in Arabidopsis thaliana. Plant J 18:97–104
Kourie J, Goldsmith MH (1992) K+ channels are responsible for an inwardly rectifying current in the plasma membrane of mesophyll protoplasts of Avena sativa. Plant Physiol 98:1087–1097
Kubo Y, Baldwin TJ, Jan YN, Jan LY (1993) Primary structure and functional expression of a mouse inward rectifier potassium channel. Nature 362:127–133
Lacan D, Durand M (1996) Na+–K+ exchange at the xylem/symplast boundary (Its significance in the salt sensitivity of soybean). Plant Physiol 110:705–711
Lam HM, Chiu J, Hsieh MH, Meisel L, Oliveira IC, Shin M, Coruzzi G (1998) Glutamate-receptor genes in plants. Nature 396:125–126
Lee M, Choi Y, Burla B, Kim YY, Jeon B, Maeshima M, Yoo JY, Martinoia E, Lee Y (2008) The ABC transporter AtABCB14 is a malate importer and modulates stomatal response to CO2. Nat Cell Biol 10:1217–1223
Lemtiri-Chlieh F, MacRobbie EA (1994) Role of calcium in the modulation of Vicia guard cell potassium channels by abscisic acid: a patch-clamp study. J Membr Biol 137:99–107
Leng Q, Mercier RW, Yao W, Berkowitz GA (1999) Cloning and first functional characterization of a plant cyclic nucleotide-gated cation channel. Plant Physiol 121:753–761
Li L, Kim BG, Cheong YH, Pandey GK, Luan S (2006) A Ca2+ signaling pathway regulates a K+ channel for low-K response in Arabidopsis. Proc Natl Acad Sci USA 103:12625–12630
Li Y, Wang G-X, Xin M, Yang H-M, Wu X-J, Li T (2004) The parameters of guard cell calcium oscillation encodes stomatal oscillation and closure in Vicia faba. Plant Science 166:415–421
Linder B, Raschke K (1992) A slow anion channel in guard cells, activating at large hyperpolarization, may be principal for stomatal closing. FEBS Lett 313:27–30
Lurin C, Geelen D, Barbier-Brygoo H, Guern J, Maurel C (1996) Cloning and functional expression of a plant voltage-dependent chloride channel. Plant Cell 8:701–711
Mäser P, Eckelman B, Vaidyanathan R, Horie T, Fairbairn DJ, Kubo M, Yamagami M, Yamaguchi K, Nishimura M, Uozumi N, Robertson W, Sussman MR, Schroeder JI (2002a) Altered shoot/root Na+ distribution and bifurcating salt sensitivity in Arabidopsis by genetic disruption of the Na+ transporter AtHKT1. FEBS Lett 531:157–161
Mäser P, Hosoo Y, Goshima S, Horie T, Eckelman B, Yamada K, Yoshida K, Bakker EP, Shinmyo A, Oiki S, Schroeder JI, Uozumi N (2002b) Glycine residues in potassium channel-like selectivity filters determine potassium selectivity in four-loop-per-subunit HKT transporters from plants. Proc Natl Acad Sci USA 99:6428–6433
Mäser P, Thomine S, Schroeder JI, Ward JM, Hirschi K, Sze H, Talke IN, Amtmann A, Maathuis FJ, Sanders D, Harper JF, Tchieu J, Gribskov M, Persans MW, Salt DE, Kim SA, Guerinot ML (2001) Phylogenetic relationships within cation transporter families of Arabidopsis. Plant Physiol 126:1646–1667
Ma JF, Ryan PR, Delhaize E (2001) Aluminium tolerance in plants and the complexing role of organic acids. Trends Plant Sci 6:273–278
Maathuis FJ, Ichida AM, Sanders D, Schroeder JI (1997) Roles of higher plant K+ channels. Plant Physiol 114:1141–1149
Maathuis FJ, Sanders D (1994) Mechanism of high-affinity potassium uptake in roots of Arabidopsis thaliana. Proc Natl Acad Sci USA 91:9272–9276
Magalhaes JV, Liu J, Guimaraes CT, Lana UG, Alves VM, Wang YH, Schaffert RE, Hoekenga OA, Pineros MA, Shaff JE, Klein PE, Carneiro NP, Coelho CM, Trick HN, Kochian LV (2007) A gene in the multidrug and toxic compound extrusion (MATE) family confers aluminum tolerance in sorghum. Nat Genet 39:1156–1161
Marmagne A, Vinauger-Douard M, Monachello D, de Longevialle AF, Charon C, Allot M, Rappaport F, Wollman FA, Barbier-Brygoo H, Ephritikhine G (2007) Two members of the Arabidopsis CLC (chloride channel) family, AtCLCe and AtCLCf, are associated with thylakoid and Golgi membranes, respectively. J Exp Bot 58:3385–3393
Marten I, Zeilinger C, Redhead C, Landry DW, al-Awqati Q, Hedrich R (1992) Identification and modulation of a voltage-dependent anion channel in the plasma membrane of guard cells by high-affinity ligands. EMBO J 11:3569–3575
Matsuda N, Kobayashi H, Katoh H, Ogawa T, Futatsugi L, Nakamura T, Bakker EP, Uozumi N (2004) Na+-dependent K+ uptake Ktr system from the cyanobacterium Synechocystis sp. PCC 6803 and its role in the early phases of cell adaptation to hyperosmotic shock. J Biol Chem 279:54952–54962
McAinsh MR, Brownlee C, Hetherington AM (1990) Abscisic acid-induced elevation of guard cell cytosolic Ca2+ precedes stomatal closure. Nature 343:186–188
McAinsh MR, Clayton H, Mansfield TA, Hetherington AM (1996) Changes in ctomatal behavior and guard cell cytosolic free calcium in response to oxidative stress. Plant Physiol 111:1031–1042
Miedema H, Demidchik V, Very AA, Bothwell JH, Brownlee C, Davies JM (2008) Two voltage-dependent calcium channels co-exist in the apical plasma membrane of Arabidopsis thaliana root hairs. New Phytol 179:378–385
Miller AJ, Vogg G, Sanders D (1990) Cytosolic calcium homeostasis in fungi: roles of plasma membrane transport and intracellular sequestration of calcium. Proc Natl Acad Sci USA 87:9348–9352
Miller C (2006) ClC chloride channels viewed through a transporter lens. Nature 440:484–489
Miyawaki A, Griesbeck O, Heim R, Tsien RY (1999) Dynamic and quantitative Ca2+ measurements using improved cameleons. Proc Natl Acad Sci USA 96:2135–2140
Moran N, Ehrenstein G, Iwasa K, Bare C, Mischke C (1984) Ion channels in plasmalemma of wheat protoplasts. Science 226:835–838
Nakamura RL, Anderson JA, Gaber RF (1997) Determination of key structural requirements of a K+ channel pore. J Biol Chem 272:1011–1018
Nakamura T, Yuda R, Unemoto T, Bakker EP (1998) KtrAB, a new type of bacterial K+-uptake system from Vibrio alginolyticus. J Bacteriol 180:3491–3494
Negi J, Matsuda O, Nagasawa T, Oba Y, Takahashi H, Kawai-Yamada M, Uchimiya H, Hashimoto M, Iba K (2008) CO2 regulator SLAC1 and its homologues are essential for anion homeostasis in plant cells. Nature 452:483–486
Nomura H, Komori T, Kobori M, Nakahira Y, Shiina T (2008) Evidence for chloroplast control of external Ca2+-induced cytosolic Ca2+ transients and stomatal closure. Plant J 53:988–998
Nurnberger T, Nennstiel D, Jabs T, Sacks WR, Hahlbrock K, Scheel D (1994) High affinity binding of a fungal oligopeptide elicitor to parsley plasma membranes triggers multiple defense responses. Cell 78:449–460
Pei ZM, Ghassemian M, Kwak CM, McCourt P, Schroeder JI (1998) Role of farnesyltransferase in ABA regulation of guard cell anion channels and plant water loss. Science 282:287–290
Pei ZM, Kuchitsu K, Ward JM, Schwarz M, Schroeder JI (1997) Differential abscisic acid regulation of guard cell slow anion channels in Arabidopsis wild-type and abi1 and abi2 mutants. Plant Cell 9:409–423
Pei ZM, Murata Y, Benning G, Thomine S, Klusener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406:731–734
Pei ZM, Ward JM, Schroeder JI (1999) Magnesium sensitizes slow vacuolar channels to physiological cytosolic calcium and inhibits fast vacuolar channels in fava bean guard cell vacuoles. Plant Physiol 121:977–986
Peiter E, Maathuis FJ, Mills LN, Knight H, Pelloux J, Hetherington AM, Sanders D (2005) The vacuolar Ca2+-activated channel TPC1 regulates germination and stomatal movement. Nature 434:404–408
Picollo A, Pusch M (2005) Chloride/proton antiporter activity of mammalian CLC proteins ClC-4 and ClC-5. Nature 436:420–423
Pineros MA, Cancado GM, Kochian LV (2008) Novel properties of the wheat aluminum tolerance organic acid transporter (TaALMT1) revealed by electrophysiological characterization in Xenopus oocytes: functional and structural implications. Plant Physiol 147:2131–2146
Qi Z, Stephens NR, Spalding EP (2006) Calcium entry mediated by GLR3.3, an Arabidopsis glutamate receptor with a broad agonist profile. Plant Physiol 142:963–971
Quintero FJ, Blatt MR (1997) A new family of K+ transporters from Arabidopsis that are conserved across phyla. FEBS Lett 415:206–211
Raschke K (2003) Alternation of the slow with the quick anion conductance in whole guard cells effected by external malate. Planta 217:651–657
Raschke K, Shabahang M, Wolf R (2003) The slow and the quick anion conductance in whole guard cells: their voltage-dependent alternation, and the modulation of their activities by abscisic acid and CO2. Planta 217:639–650
Reintanz B, Szyroki A, Ivashikina N, Ache P, Godde M, Becker D, Palme K, Hedrich R (2002) AtKC1, a silent Arabidopsis potassium channel α-subunit modulates root hair K+ influx. Proc Natl Acad Sci USA 99:4079–4084
Ren ZH, Gao JP, Li LG, Cai XL, Huang W, Chao DY, Zhu MZ, Wang ZY, Luan S, Lin HX (2005) A rice quantitative trait locus for salt tolerance encodes a sodium transporter. Nat Genet 37:1141–1146
Rigas S, Debrosses G, Haralampidis K, Vicente-Agullo F, Feldmann KA, Grabov A, Dolan L, Hatzopoulos P (2001) TRH1 encodes a potassium transporter required for tip growth in Arabidopsis root hairs. Plant Cell 13:139–151
Roelfsema MR, Levchenko V, Hedrich R (2004) ABA depolarizes guard cells in intact plants, through a transient activation of R- and S-type anion channels. Plant J 37:578–588
Rubio F, Gassmann W, Schroeder JI (1995) Sodium-driven potassium uptake by the plant potassium transporter HKT1 and mutations conferring salt tolerance. Science 270:1660–1663
Ryan PR, Skerrett M, Findlay GP, Delhaize E, Tyerman SD (1997) Aluminum activates an anion channel in the apical cells of wheat roots. Proc Natl Acad Sci USA 94:6547–6552
Santa-Maria GE, Rubio F, Dubcovsky J, Rodriguez-Navarro A (1997) The HAK1 gene of barley is a member of a large gene family and encodes a high-affinity potassium transporter. Plant Cell 9:2281–2289
Sasaki T, Yamamoto Y, Ezaki B, Katsuhara M, Ahn SJ, Ryan PR, Delhaize E, Matsumoto H (2004) A wheat gene encoding an aluminum-activated malate transporter. Plant J 37:645–653
Schachtman DP, Schroeder JI (1994) Structure and transport mechanism of a high-affinity potassium uptake transporter from higher plants. Nature 370:655–658
Schachtman DP, Schroeder JI, Lucas WJ, Anderson JA, Gaber RF (1992) Expression of an inward-rectifying potassium channel by the Arabidopsis KAT1 cDNA. Science 258:1654–1658
Schlosser A, Meldorf M, Stumpe S, Bakker EP, Epstein W (1995) TrkH and its homolog, TrkG, determine the specificity and kinetics of cation transport by the Trk system of Escherichia coli. J Bacteriol 177:1908–1910
Schmidt C, Schroeder JI (1994) Anion selectivity of slow anion channels in the plasma membrane of guard cells (large nitrate permeability). Plant Physiol 106:383–391
Schroeder JI (1988) K+ transport properties of K+ channels in the plasma membrane of Vicia faba guard cells. J Gen Physiol 92:667–683
Schroeder JI, Hagiwara S (1989) Cytosolic calcium regulates ion channels in the plasma membrane of VIsia faba guard cells. Nature 338:427–430
Schroeder JI, Hagiwara S (1990) Repetitive increases in cytosolic Ca2+ of guard cells by abscisic acid activation of nonselective Ca2+ permeable channels. Proc Natl Acad Sci USA 87:9305–9309
Schroeder JI, Hedrich R (1989) Involvement of ion channels and active transport in osmoregulation and signaling of higher plant cells. Trends Biochem Sci 14:187–192
Schroeder JI, Hedrich R, Fernandez M (1984) Potassium-selective single channels in guard cell protoplasts of Vicia faba. Nature 312:2–3
Schroeder JI, Keller BU (1992) Two types of anion channel currents in guard cells with distinct voltage regulation. Proc Natl Acad Sci USA 89:5025–5029
Schroeder JI, Raschke K, Neher E (1987) Voltage dependence of K channels in guard-cell protoplasts. Proc Natl Acad Sci USA 84:4108–4112
Sentenac H, Bonneaud N, Minet M, Lacroute F, Salmon JM, Gaymard F, Grignon C (1992) Cloning and expression in yeast of a plant potassium ion transport system. Science 256:663–665
Shi H, Ishitani M, Kim C, Zhu JK (2000) The Arabidopsis thaliana salt tolerance gene SOS1 encodes a putative Na+/H+ antiporter. Proc Natl Acad Sci USA 97:6896–6901
Shi H, Quintero FJ, Pardo JM, Zhu JK (2002) The putative plasma membrane Na+/H+ antiporter SOS1 controls long-distance Na+ transport in plants. Plant Cell 14:465–477
Shimazaki K, Iino M, Zeiger E (1986) Blue light-dependent proton extrusion by guard-cell protoplasts of Vicia faba. Nature 319:324–326
Staxen I, Pical C, Montgomery LT, Gray JE, Hetherington AM, McAinsh MR (1999) Abscisic acid induces oscillations in guard-cell cytosolic free calcium that involve phosphoinositide-specific phospholipase C. Proc Natl Acad Sci USA 96:1779–1784
Sunarpi HT, Motoda J, Kubo M, Yang H, Yoda K, Horie R, Chan WY, Leung HY, Hattori K, Konomi M, Osumi M, Yamagami M, Schroeder JI, Uozumi N (2005) Enhanced salt tolerance mediated by AtHKT1 transporter-induced Na unloading from xylem vessels to xylem parenchyma cells. Plant J 44:928–938
Tazawa M (1968) Motive force of the cytoplasmic streaming in Nitella. Protoplasma 65:207–222
Tazawa M (1972) Membrane characteristics as revealed by water and ionic relations of algal cells. Protoplasma 75:427–460
Thiel G, MacRobbie EA, Blatt MR (1992) Membrane transport in stomatal guard cells: the importance of voltage control. J Membr Biol 126:1–18
Tholema N, Vor der Bruggen M, Maser P, Nakamura T, Schroeder JI, Kobayashi H, Uozumi N, Bakker EP (2005) All four putative selectivity filter glycine residues in KtrB are essential for high affinity and selective K+ uptake by the KtrAB system from Vibrio alginolyticus. J Biol Chem 280:41146–41154
Thuleau P, Moreau M, Schroeder JI, Ranjeva R (1994a) Recruitment of plasma membrane voltage-dependent calcium-permeable channels in carrot cells. EMBO J 13:5843–5847
Thuleau P, Ward JM, Ranjeva R, Schroeder JI (1994b) Voltage-dependent calcium-permeable channels in the plasma membrane of a higher plant cell. EMBO J 13:2970–2975
Tyerman SD, Skerrett M, Garill A, Findlay GP, Leigh RA (1997) Pathways for the permeation of Na+ and Cl− into protoplasts derived from the cortex of wheat roots. J Exp Bot 48:459–480
Uozumi N (2001) Escherichia coli as an expression system for K+ transport systems from plants. Am J Physiol 281:733–739
Uozumi N, Gassmann W, Cao Y, Schroeder JI (1995) Identification of strong modifications in cation selectivity in an Arabidopsis inward rectifying potassium channel by mutant selection in yeast. J Biol Chem 270:24276–24281
Uozumi N, Kim EJ, Rubio F, Yamaguchi T, Muto S, Tsuboi A, Bakker EP, Nakamura T, Schroeder JI (2000) The Arabidopsis HKT1 gene homolog mediates inward Na+ currents in Xenopus laevis oocytes and Na+ uptake in Saccharomyces cerevisiae. Plant Physiol 122:1249–1259
Uozumi N, Nakamura T, Schroeder JI, Muto S (1998) Determination of transmembrane topology of an inward-rectifying potassium channel from Arabidopsis thaliana based on functional expression in Escherichia coli. Proc Natl Acad Sci USA 95:9773–9778
Vahisalu T, Kollist H, Wang YF, Nishimura N, Chan WY, Valerio G, Lamminmaki A, Brosche M, Moldau H, Desikan R, Schroeder JI, Kangasjarvi J (2008) SLAC1 is required for plant guard cell S-type anion channel function in stomatal signalling. Nature 452:487–491
Voelker C, Schmidt D, Mueller-Roeber B, Czempinski K (2006) Members of the Arabidopsis AtTPK/KCO family form homomeric vacuolar channels in planta. Plant J 48:296–306
Wais RJ, Galera C, Oldroyd G, Catoira R, Penmetsa RV, Cook D, Gough C, Denarie J, Long SR (2000) Genetic analysis of calcium spiking responses in nodulation mutants of medicago truncatula. Proc Natl Acad Sci USA 97:13407–13412
Ward JM, Maser P, Schroeder JI (2009) Plant ion channels: gene families, physiology, and functional genomics analysis. Annu Rev Physiol 71:59–82
Ward JM, Pei ZM, Schroeder JI (1995) Roles of ion channels in initiation of signal transduction in higher plants. Plant Cell 7:833–844
Ward JM, Schroeder JI (1994) Calcium-activated K+ channels and calcium-Induced calcium release by slow vacuolar ion channels in guard cell vacuoles implicated in the control of stomatal closure. Plant Cell 6:669–683
Wegner LH, De Boer AH (1997) Properties of two outward-rectifying channels in root xylem parenchyma cells suggest a role in K+ homeostasis and long-distance signaling. Plant Physiol 115:1707–1719
Wegner LH, Raschke K (1994) Ion channels in the xylem parenchyma of barley roots. A procedure to isolate protoplasts from this tissue and a patch-clamp exploration of salt passageways into xylem vessels. Plant Physiol 105:799–813
Weinl S, Held K, Schlucking K, Steinhorst L, Kuhlgert S, Hippler M, Kudla J (2008) A plastid protein crucial for Ca2+-regulated stomatal responses. New Phytol 179:675–686
Wu SJ, Ding L, Zhu JK (1996) SOS1, a genetic locus essential for salt tolerance and potassium acquisition. Plant Cell 8:617–627
Xu J, Li HD, Chen LQ, Wang Y, Liu LL, He L, Wu WH (2006) A protein kinase, interacting with two calcineurin B-like proteins, regulates K+ transporter AKT1 in Arabidopsis. Cell 125:1347–1360
Yang Y, Costa A, Leonhardt N, Siegel RS, Schroeder JI (2008) Isolation of a strong Arabidopsis guard cell promoter and its potential as a research tool. Plant Meth 4:6
Yoshioka K, Moeder W, Kang HG, Kachroo P, Masmoudi K, Berkowitz G, Klessig DF (2006) The chimeric Arabidopsis Cyclic Nucleotide-Gated Ion Channel11/12 activates multiple pathogen resistance responses. Plant Cell 18:747–763
Young JJ, Mehta S, Israelsson M, Godoski J, Grill E, Schroeder JI (2006) CO2 signaling in guard cells: calcium sensitivity response modulation, a Ca2+-independent phase, and CO2 insensitivity of the gca2 mutant. Proc Natl Acad Sci USA 103:7506–7511
Zhang WH, Ryan PR, Sasaki T, Yamamoto Y, Sullivan W, Tyerman SD (2008) Characterization of the TaALMT1 protein as an Al3+-activated anion channel in transformed tobacco (Nicotiana tabacum L.) cells. Plant Cell Physiol 49:1316–1330
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Uozumi, N., Schroeder, J.I. (2010). Ion Channels and Plant Stress: Past, Present, and Future. In: Demidchik, V., Maathuis, F. (eds) Ion Channels and Plant Stress Responses. Signaling and Communication in Plants. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-10494-7_1
Download citation
DOI: https://doi.org/10.1007/978-3-642-10494-7_1
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-10493-0
Online ISBN: 978-3-642-10494-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)