Abstract
In contrast to conventional echocardiography performed by cardiologists on a consultative basis, critical care echocardiography is performed and interpreted by the intensivist at the bedside of the patient in the intensive care unit. It assists the frontline intensivist in establishing diagnosis and guiding management of patients with hemodynamic or respiratory failure. Since critical care echocardiography is best performed at the time of organ failure, it should be available on a 24-h basis every day of the year. This emphasizes the need of a dedicated equipment in the modern intensive care unit. Critical care echocardiography requires immediate interpretation since its results must be interpreted in light of the medical history of the patient, the clinical context, ventilator settings and ongoing treatments. A background in intensive care medicine is required to incorporate the obtained information in the medical reasoning and diagnostic work-up. The identification of the leading mechanism for hemodynamic compromise and early recognition of overloaded right ventricle in mechanically ventilated patients with respiratory failure avoid using inefficient or harmful therapeutic interventions. In addition, serial examinations allow monitoring both the efficacy and tolerance of echocardiography-guided therapeutic interventions. Transthoracic and transesophageal approaches can be used for this purpose. The level of competence of the intensivist closely depends on previous personal training and acquired cognitive and technical skills.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Intensive Care Unit
- Intensive Care Unit Patient
- Circulatory Failure
- Bedside Ultrasonography
- Hemodynamic Assessment
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
In recent years, bedside ultrasonography has gained wide acceptance for the assessment of critically ill patients. As opposed to “blind” monitoring systems, which frequently rely on invasive techniques (e.g., pulmonary artery catheter), echocardiography Doppler allows real-time imaging of the heart and great vessels, thereby proving unparalleled anatomical and functional information. Because of its versatility, safety, and instantaneous diagnostic capability, bedside ultrasonography is ideally suited for the evaluation of unstable patients in the intensive care unit (ICU). This overview will examine the utility of critical care echocardiography and the current field of use of transthoracic echocardiography (TTE) and transesophageal echocardiography (TEE) in the ICU. Cardiac output monitoring by esophageal Doppler [1, 2] and noncardiac ultrasonography [3] are purposely excluded from this overview. Training issues are discussed elsewhere (see Chap. 21).
1.1 Historical Perspective
Whereas hemodynamic assessment of ICU patients was predominantly invasive in the 1980s and 1990s, the current approach tends to be less invasive, more functional, and to integrate heart-lung interactions in the diagnostic process. During the last decade, right heart catheterization (RHC) has been progressively supplanted by bedside echocardiography in numerous ICUs [4]. Since its introduction in 1970 [5], RHC has been utilized widely by intensivists for the management of patients with circulatory failure. Therapeutic algorithms based upon RHC rely on pressure, flow, and metabolic measurements. Concordant publications clearly showed a lack of benefit and potential harm associated with the routine use of RHC [6–11]. In addition, clinically relevant discrepancies between RHC and bedside echocardiography in the evaluation of patients with circulatory failure have been published [12–20] and attributed to intrinsic limitations of RHC [21]. As a result, some intensivists have come to regard bedside ultrasonography as a superior alternative to RHC for the hemodynamic assessment of ICU patients [22, 23].
The early pioneers of critical care echocardiography used TTE and emphasized the superiority of bedside echocardiography over RHC [24–26]. The development of TEE accelerated the diffusion of ultrasonography into some ICUs since surface echocardiography was often limited by inadequate image quality in the ICU environment. TEE yielded good image quality in virtually all ventilated patients and allowed assessment of deep anatomical structures, which were otherwise not accessible to TTE [27–41]. In addition, TEE became the first-line diagnostic procedure recommended officially for the assessment of circulatory failure complicating the perioperative course or occurring in ICU ventilated patients [42–44]. In the past decade, progress in machine design has allowed the development of smaller, more powerful echocardiography systems with increased TTE diagnostic capabilities (Table 1.1).
1.2 Specificities of Critical Care Echocardiography
As opposed to conventional echocardiography, which is performed by cardiologists on a consultative basis in the echocardiography laboratory, critical care echocardiography is carried out and interpreted by the intensivist at the bedside of the ICU patient. Critical care echocardiography allows the frontline clinician to establish diagnosis and guide management of hemodynamic failure. Critical care echocardiography has specific requirements that cannot be met by consultant cardiologists and that support the need for training intensivists in the use of this technique in the ICU (Table 1.2).
Critically ill patients require nonscheduled management and care. Critical care echocardiography must therefore be available on a 24-h basis. A key requirement is that a capable machine be immediately available to the intensivist in the ICU. By definition, critical care echocardiography is performed personally by the frontline intensivist, the study is interpreted at the bedside of the critically ill patient, and the results are used for immediate diagnosis and management. In contrast to conventional echocardiography performed in stable patients presenting with chronic cardiac disease, critical care echocardiography has optimal diagnostic ability when performed at the time of the clinical deterioration. When performed after the initiation of a treatment that rapidly alters the hemodynamic profile (e.g., diuretics in decompensated congestive heart failure, afterload reduction in mitral regurgitation), echocardiography may be less informative [45].
The current approach to hemodynamic assessment using bedside echocardiography has to integrate heart and lung interactions [46, 47]. This requires a specific background in critical care medicine [48]. Whereas positive-pressure ventilation has long been recognized as a potential confounding factor for hemodynamic assessment using RHC [21, 49], Doppler echocardiography allows a better understanding of the complex effects of volume-controlled ventilation on central hemodynamics [47]. Over the last decade, several echocardiographic indices using heart-lung interactions produced by positive-pressure ventilation in the presence of a clinically relevant hypovolemia have been validated for predicting fluid responsiveness [50–54]. Recently, passive leg-raising has allowed echocardiographic indices to predict responders to a fluid challenge in patients with spontaneous breathing activity [55, 56]. These simple yet robust indices of fluid responsiveness allow intensivists to use echocardiography routinely in deciding when to proceed with volume resuscitation.
Heart-lung interactions are influenced not only by the volume status of the mechanically ventilated patient, but also by the lung compliance and ventilator settings [57]. This is especially relevant in patients who are on ventilatory support for acute respiratory distress syndrome (ARDS). Specifically, inappropriate ventilator settings may directly impede right ventricular ejection and contribute to the development of circulatory failure [58]. Early recognition of the negative effects of mechanical ventilation on central hemodynamics is crucial as circulatory failure is a leading cause of death with ARDS [59].
The findings provided by an imaging modality have to be interpreted in light of the clinical scenario and patient history. Accordingly, critical care echocardiography results always need to be interpreted within the clinical context. The intensivist who is directly in charge of the management of a case is best equipped to do this. Operator experience is crucial in order to integrate echocardiography results correctly with the clinical presentation. The results of critical care echocardiography are immediately interpreted by the attending intensivist, and so they may often substantially alter workup and therapy.
1.3 Indications and Safety of Echocardiography in ICU Patients
1.3.1 Indications for Critical Care Echocardiography
Indications for echocardiography have long been defined in the cardiology community [43, 44]. According to these guidelines, hemodynamically unstable patients and patients sustaining severe chest or multisystem trauma represent the main indications for performing echocardiography in the ICU setting. Societies of anesthesiologists have also defined specific indications of TEE in the perioperative period [42]. In contrast, indications for performing Doppler echocardiography in ICU patients have not yet been established by critical care societies. Recent recommendations describe echocardiography as an alternative to right-heart catheterization for the measurement of cardiac output in septic patients [60]. There is compelling evidence that leads us to propose bedside ultrasonography as the first-line diagnostic tool in the ICU for the assessment of patients presenting with circulatory failure (i.e., hypotension, shock), respiratory failure, or both [12–41, 61, 62].
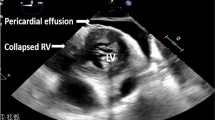
Shock has recently been defined by an international consensus conference as clinical evidence and/or a biological marker of inadequate tissue perfusion – e.g., decreased central venous oxygen saturation (ScvO2) or mixed venous oxygen saturation (SvO2), increased blood lactate, increased base deficit, and low pH – considering that hypotension could be inconsistent [63]. Respiratory failure usually refers to the conjunction of dyspnea and potential muscular fatigue, hypoxemia, and radiographic infiltrates. In these clinical settings, Doppler echocardiography provides rapid and comprehensive evaluation of the hemodynamics (e.g., hypovolemia, left or right ventricular failure, vasoplegia, elevated filling pressures) and also allows for immediate critical diagnoses (e.g., severe valve failure, central pulmonary embolus, thrombus in transit, pericardial effusion). When compared with blind and more invasive traditional monitoring techniques, echocardiography has the unparalleled advantage of directly depicting certain mechanisms of hemodynamic embarrassment that otherwise would be undetectable, including ventricular interdependence, pericardial constraints, regional wall-motion abnormalities, impaired relaxation, dynamic left ventricular outflow obstruction, and acute valvular regurgitation. In addition to the major indication for echocardiography in the ICU, which is cardiopulmonary failure, other standard indications apply, such as assessment of blunt chest trauma patients at high risk of cardiovascular injury, searching for a cardiac embolic source, endocarditis [64], and intracardiac shunt (Table 1.3).
1.3.2 TTE Versus TEE
Traditionally, it is assumed that TTE is the first-line approach owing to its versatility, tolerance, and availability [44, 62]. TTE has certain advantages over TEE, including a better Doppler beam alignment with intracardiac flows and a broader field of examination of relatively superficial anatomical structures (Table 1.4). In general, TEE is appropriately used as an adjunct or subsequent test to TTE when surface examination is nondiagnostic (e.g., poor imaging quality, inaccessibility of deep anatomical structures). ICU patients present challenges when assessed by means of TTE since hyperinflation related to mechanical ventilation or chronic obstructive pulmonary disease, obesity, edema, chest wall wounds or dressings, tubings, or surgical emphysema frequently interfere with ultrasound transmission and result in inadequate image quality [43, 62]. This explains why previous studies conducted in the ICU consistently reported a superior diagnostic capability of TEE compared with TTE [12, 15, 28, 30, 32, 33, 36]. When serial hemodynamic assessment is required to monitor acute therapeutic changes in unstable patients, reproducible tomographic imaging planes are frequently more easily obtained with TEE than with TTE. Although recent-generation miniaturized echocardiography machines have excellent image quality and surface echocardiography may be diagnostic in hemodynamically unstable patients [65], TTE remains difficult to perform in certain patient populations. In these cases, TEE may be required. Visualization of specific anatomical structures for accurate diagnoses and assessment of ventilated patients in perioperative settings are other main indications for performing a primary TEE examination [44, 62] (Table 1.4). In North America, TEE is not yet widely performed by intensivists, whereas it is routinely used by frontline intensivists in several European countries.
1.3.3 Tolerance of TEE
TEE can be performed safely in patients with critical illness. The rate of serious complication is very low. Patients are generally on mechanical ventilatory support, with the airway secured by tracheal intubation. Respiratory complications are therefore very rare. Injury to the esophagus (esophageal abrasion, perforation, or bleeding) is always a possibility. This can be avoided by appropriate patient selection and by minimizing rotational movement of the endoscope tip while it is under flexion. A full history has to be obtained that addresses the risk of esophageal injury from TEE. TEE is contraindicated in the setting of esophageal varices, strictures, bleeding, recent surgery, tumors, diverticuli, or other significant esophageal pathology. Coagulopathy or thrombocytopenia are relative contraindications to TEE. Risks associated with sedation, such as airway compromise and transient hypotension, are similar to those with other endoscopic procedures.
In a summary review of 2,504 TEE studies performed in the ICU, there was a 2.6% overall complication rate (discounting inadvertent dislodgement of a nasogastric tube), and no procedure-related deaths were reported [66]. Unsuccessful TEE probe insertion is rare when using laryngoscopic guidance in adequately sedated patients on ventilatory support. In spontaneously breathing patients, the major risk of TEE is related to the development of an acute respiratory failure secondary to the esophageal intubation [67, 68]. TEE should be discouraged in unstable patients who are not on ventilatory support, especially when a tamponade or a massive pulmonary embolism is suspected, because the procedure may precipitate circulatory or respiratory compromise (Table 1.5). In ambulatory patients examined in the echocardiography laboratory, a large cardiological series of over 10,000 consecutive TEE examinations reported an incidence of complications as low as 0.88% [69] and a rate of hypopharyngeal or esophageal perforation of 0.03% [70]. There are no known mechanical complications with TTE.
1.4 Therapeutic Impact of Critical Care Echocardiography
Owing to its excellent diagnostic capability, TEE has a therapeutic impact that is consistently superior to that of TTE when ventilated ICU patients are evaluated with both procedures [12, 15, 28, 30, 32, 33, 36]. Critical care echocardiography has documented a direct impact on therapy in up to 50% of ICU patients [12, 15, 17, 20, 28, 31, 33–37, 39–41]. TEE has been shown to prompt cardiac surgery in up to 20% of examined patients, according to the type of ICU recruitment [17, 20, 27, 29, 31–35, 37–41]. By accurately identifying the mechanism of shock or acute respiratory failure, critical care echocardiography allows the intensivist to change therapeutic strategy and reduce the risk of inefficient or harmful therapy. It frequently corrects initial diagnosis derived from conventional hemodynamic monitoring [12–20]. In unstable patients, acute therapy (e.g., fluid challenge, initiation of inotropic support, ventilator settings) may result in a rapid variation of the hemodynamic profile. Accordingly, the hemodynamic assessment is best performed early in the course of organ failure and serial echocardiographic examinations provide real-time monitoring of both the efficacy and tolerance of therapeutic interventions in ICU patients. The anticipated efficacy of therapeutic changes related to critical care echocardiography is closely related to the severity of patient presentation, its benefit being maximal in the most unstable patients at the time of examination.
1.5 Conclusions
Echocardiography should be the primary diagnostic tool for the evaluation of ICU patients with circulatory or respiratory failure. Critical care echocardiography requires that a capable machine be immediately available on a 24-h basis in the ICU. The frontline intensivist who uses echocardiography to guide the management of the critically patient must be competent in image acquisition, image interpretation, and application of the results to the clinical situation. Both TTE and TEE offer strong utility in the ICU. TEE may be required where TTE image quality is inadequate to answer the clinical question, but the use of TEE may also be curtailed by its limited availability in some ICUs. TEE has an excellent safety record in the ICU. The intensivist should regard echocardiography as a key area in which to acquire cognitive and technical skills for optimal management of patients with cardiopulmonary failure.
References
Cholley BP, Singer M (2003) Esophageal Doppler: noninvasive cardiac output monitor. Echocardiography 20:763–769
Dark PM, Singer M (2004) The validity of transesophageal Doppler ultrasonography as a measure of cardiac output in critically ill adults. Intensive Care Med 30:2060–2066
Beaulieu Y, Marik PE (2005) Bedside ultrasonography in the ICU (part 2). Chest 128:1766–1781
Vieillard-Baron A, Slama M, Cholley B, Janvier G, Vignon P, for the ECHO-in-ICU group (2008). Echocardiography in the intensive care unit: from evolution to revolution? Intensive Care Med 34:243–249
Swan HJ, Ganz W, Forrester J, Marcus H, Diamond G, Chonette D (1970) Catheterization of the heart in man with use of a flow-directed balloon-tipped catheter. N Engl J Med 283:447–451
Connors AF, Speroff T, Dawson NV, Thomas C, Harrell FE, Wagner D, Desbiens N, Goldman L, Wu AW, Califf RM, Fulkerson WJ, Vidaillet H, Broste S, Bellamy P, Lynn J, Knaus WA (1996) The effectiveness of right heart catheterization in the initial care of critically ill patients. JAMA 276:889–897
Richard C, Warszawski J, Anguel N, Deye N, Combes A, Barnoud D, Boulain T, Lefort Y, Fartoukh M, Baud F, Boyer A, Brochard L, Teboul JL (2003) French Pulmonary Artery Catheter Study Group. Early use of the pulmonary artery catheter and outcomes in patients with shock and acute respiratory distress syndrome: a randomized controlled trial. JAMA 290:2713–2720
Rhodes A, Cusack RJ, Newman PJ, Grounds RM, Benett ED (2002) A randomized, controlled trial of the pulmonary artery catheter in critically ill patients. Intensive Care Med 28:256–264
Sandham JD, Hull RD, Brant RF, Knox L, Pineo GF, Doig CJ, Laporta DP, Viner S, Passerini L, Devitt H, Kirby A, Jacka M (2003) Canadian Critical Care Clinical Trials Group. A randomized, controlled trial of the use of pulmonary-artery catheters in high-risk surgical patients. N Engl J Med 348:5–14
Harvey S, Harrison DA, Singer M, Ashcroft J, Jones CM, Elbourne D, Brampton W, Williams D, Young D, Rowan K (2005) PAC-Man study collaboration. Lancet 366:472–477
Binanay C, Califf RM, Hasselblad V, ÓConnor CM, Shah MR, Sopko G, Stevenson LW, Francis GS, Leier CV, Miller LW, ESCAPE investigators and ESCAPE study coordinators (2005) Evaluation study of congestive heart failure and pulmonary catheterization effectiveness: the ESCAPE trial. JAMA 294:1625–1633
Reichert CLA, Visser CA, Koolen JJ, Van den Brink RBA, Van Wezel HB, Meyne NG, Dunning AJ (1992) Transesophageal echocardiography in hypotensive patients after cardiac operation. J Thorac Cardiovasc Surg 104:321–326
Jardin F, Valtier B, Beauchet A, Dubourg O, Bourdarias JP (1994) Invasive monitoring combined with two-dimensional echocardiographic study in septic shock. Intensive Care Med 20:550–554
Kaul S, Stratienko AA, Pollock SG, Marieb MA, Keller MW, Sabia PJ (1994) Value of two-dimensional echocardiography for determining the basis of hemodynamic compromise in critically ill patients: a prospective study. J Am Soc Echocardiogr 7:598–606
Poelaert JI, Trouerbach J, De Buyzere M, Everaert J, Colardyn FA (1995) Evaluation of transesophageal echocardiography as a diagnostic and therapeutic aid in a critical care setting. Chest 107:774–779
Benjamin E, Griffin K, Leibowitz AB, Manasia A, Oropello JM, Geffroy V, DelGiudice R, Hufanda J, Rosen S, Goldman M (1998) Goal-directed transesophageal echocardiography performed by intensivists to assess left ventricular function: comparison with pulmonary artery catheterization. J Cardiothorac Vasc Anesth 12:10–15
Harris KM, Petrovic O, Davila-Roman VG, Yusen RD, Littenberg B, Barzilai B (1999) Changing patterns of transesophageal echocardiography use in the intensive care unit. Echocardiography 16:559–565
Wake PJ, Ali M, Carroll J, Siu SC, Cheng DCH (2001) Clinical and echocardiographic diagnoses disagree in patients with unexplained hemodynamic instability after surgery. Can J Anesth 48:778–783
Costachescu T, Denault A, Guimond JG, Couture P, Carignan S, Sheridan P, Hellou G, Blair L, Normandin L, Babin D, Allard M, Harel F, Buithieu J (2002) The hemodynamically unstable patient in the intensive care unit: hemodynamic vs. transesophageal echocardiographic monitoring. Crit Care Med 30:1214–1223
Colreavy FB, Donovan K, Lee KY, Weekes J (2002) Transesophageal echocardiography in critically ill patients. Crit Care Med 30:989–996
Jardin F, Bourdarias JP (1995) Right heart catheterization at bedside: a critical view. Intensive Care Med 21:291–295
Vignon P (2005) Hemodynamic assessment of critically-ill patients using echocardiography Doppler. Curr Opin Crit Care 11:227–234
Vieillard-Baron A, Prin S, Chergui K, Dubourg O, Jardin F (2003) Hemodynamic instability in sepsis: bedside assessment by Doppler echocardiography. Am J Respir Crit Care Med 168:1270–1276
Ozier Y, Guéret P, Jardin F, Farcot JC, Bourdarias JP, Margairaz A (1984) Two-dimensional echocardiographic demonstration of acute myocardial depression in septic shock. Crit Care Med 12:596–599
Jardin F, Gueret P, Dubourg O, Farcot JC, Margairaz A, Bourdarias JP (1985) Two-dimensional echocardiographic evaluation of right ventricular size and contractility in acute respiratory failure. Crit Care Med 13:952–956
Jardin F, Brun-Ney D, Auvert B, Beauchet A, Bourdarias JP (1990) Sepsis-related cardiogenic shock. Crit Care Med 18:1055–1060
Oh JK, Seward JB, Khandheria BK, Gersh BJ, Mc Gregor CGA, Freeman WK, Sinak LJ, Tajik AJ (1990) Transesophageal echocardiography in critically ill patients. Am J Cardiol 66:1492–1495
Pavlides GS, Hauser AM, Stewart JR, ÓNeill WW, Timmis GC (1990) Contribution of transesophageal echocardiography to patient diagnosis and treatment: a prospective analysis. Am Heart J 120:910–914
Pearson AC, Castello R, Labovitz AJ (1990) Safety and utility of transesophageal echocardiography in the critically ill patient. Am Heart J 119:1083–1089
Font VE, Obarski TP, Klein AL, Barlett JC, Nemec JJ, Stewart WJ, Salcedo EE (1991). Transesophageal echocardiography in the critical care unit. Cleveland Clin J Med 58:315–322
Foster E, Schiller NB (1992) The role of transesophageal echocardiography in critical care: UCSF experience. J Am Soc Echocardiogr 5:368–374
Hwang JJ, Shyn KG, Chen JJ, Tseng YZ, Kuan P, Lien WP (1993). Usefulness of transesophageal echocardiography in the treatment of critically ill patients. Chest 104:861–866
Vignon P, Mentec H, Terré S, Gastinne H, Guéret P, Lemaire F (1994) Diagnostic accuracy and therapeutic impact of transthoracic and transesophageal echocardiography in mechanically ventilated patients in the ICU. Chest 106:1829–1834
Khoury AF, Afridi I, Quinones MA, Zoghbi WA (1995) Transesophageal echocardiography in critically ill patients: feasibility, safety and impact on management. Am Heart J 127:1363–1371
Sohn DW, Shin GJ, Oh JK, Tajik AJ, Click RL, Miller FA, Seward JB (1995). Role of transesophageal echocardiography in hemodynamically unstable patients. Mayo Clin Proc 70:925–931
Heidenreich PA, Stainback RF, Redberg RF, Schiller NB, Cohen NH, Foster E (1995) Transesophageal echocardiography predicts mortality in critically ill patients with unexplained hypotension. J Am Coll Cardiol 26:152–158
Slama M, Novara A, Van De Putte P, Diebold B, Safavian A, Safar M, Ossart M, Fagon JY (1996) Diagnostic and therapeutic implications of transesophageal echocardiography in medical ICU patients with unexplained shock, hypoxemia, or suspected endocarditis. Intensive Care Med 22:916–922
Alam M (1996) Transesophageal echocardiography in critical care units: Henry Ford hospital experience and review of the literature. Prog Cardiovasc Dis 38:315–328
McLean AS (1998) Transesophageal echocardiography in the intensive care unit. Anaesth Intensive Care 26:22–25
Schmidlin D, Schuepbach R, Bernard E, Ecknauer E, Jenni R, Schmid ER (2001) Indications and impact of postoperative transesophageal echocardiography in cardiac surgical patients. Crit Care Med 29:2143–2148
Bruch C, Comber M, Schmermund A, Eggebrecht H, Bartel T, Erbel R (2003) Diagnostic usefulness and impact on management of transesophageal echocardiography in surgical intensive care units. Am J Cardiol 91:510–513
Practice guidelines for perioperative transesophageal echocardiography (1996) A report by the American Society of Anesthesiologists and the Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiography. Anesthesiology 84:986–1006
ACC/AHA guidelines for the clinical application of echocardiography (1997) A report of the American College of Cardiology/American Heart Association Task Force on Practice guidelines (Committee on Clinical Application of Echocardiography). Circulation 95:1686–1744
Douglas PS, Khandheria B, Stainback RF, Weissman NJ (2007) ACCF/ASE/ACEP/ASNC/SCAL/SCCT/SCMR 2007 appropriateness criteria for transthoracic and transesophageal echocardiography. J Am Coll Cardiol 50:187–204
Vignon P (2008) Assessment of critically-ill patients with acute heart failure syndrome using echocardiography Doppler. In: Mebazaa A, Gheorghiade M, Zannad FM, Parillo JE (eds) Aute heart failure. Springer, London, pp 424–445
Vignon P (2004) Evaluation of fluid responsiveness in ventilated septic patients: back to venous return [Editorial]. Intensive Care Med 30:1699–1701
Vieillard-Baron A, Chergui K, Augarde R, Prin S, Page B, Beauchet A, Jardin F (2003) Cyclic changes in arterial pulse during respiratory support revisited by Doppler echocardiography. Am J Respir Crit Care Med 168:671–676
Mayo PH, Beaulieu Y, Doelken P, Feller-Kopman D, Harrod C, Kaplan A, Oropello J, Vieillard-Baron A, Axler O, Lichtenstein D, Maury E, Slama M, Vignon P (2009) American College of Chest Physicians/La Société de Réanimation de Langue Française statement on competence in critical care ultrasonography. Chest 135:1050–1060
Raper R, Sibbald WJ (1986) Misled by the wedge? The Swan-Ganz catheter and left ventricular preload. Chest 89:427–434
Feissel M, Michard F, Mangin I, Ruyer O, Faller JP, Teboul JL (2001). Respiratory changes in aortic blood velocity as an indicator of fluid responsiveness in ventilated patients with septic shock. Chest 119:867–873
Slama M, Masson H, Teboul JL, Arnout ML, Susic D, Frohlich E, Andrejak M (2002) Respiratory variations of aortic VTI: a new index of hypovolemia and fluid responsiveness. Am J Physiol Heart Circ Physiol 283:H1729–H1733
Vieillard-Baron A, Chergui K, Rabiller A, Peyrouset O, Page B, Beauchet A, Jardin F (2004) Superior vena cava collapsibility as a gauge of volume status in ventilated septic patients. Intensive Care Med 30:1734–1739
Barbier C, Loubières Y, Schmit C, Hayon J, Ricôme JL, Jardin F, Vieillard-Baron A (2004) Respiratory changes in inferior vena cava diameter are helpful in predicting fluid responsiveness in ventilated septic patients. Intensive Care Med 30:1740–1746
Feissel M, Michard F, Faller JP, Teboul JL (2004) The respiratory variation in inferior vena cava diameter as a guide to fluid therapy. Intensive Care Med 30:1834–1837
Lamia B, Ochagavia A, Monnet X, Chemla D, Richard C, Teboul JL (2007) Echocardiographic prediction of volume responsiveness in critically ill patients with spontaneously breathing activity. Intensive Care Med 33:1125–1132
Maizel J, Airapetian N, Lorne E, Tribouilloy C, Massy Z, Slama M (2007) Diagnosis of central hypovolemia using passive leg rising. Intensive Care Med 33:1133–1138
Reuter DA, Bayerlein J, Goepfert MS, Weis FC, Kilger E, Lamm P, Goetz AE (2003) Influence of tidal volume on left ventricular stroke volume variation measured by pulse contour analysis in mechanically ventilated patients. Intensive Care Med 29:476–480
Vieillard-Baron A, Jardin F (2003) Why protect the right ventricle in patients with acute respiratory distress syndrome? Curr Opin Crit Care 9:15–21
Monchi M, Bellenfant F, Cariou A, Joly LM, Thebert D, Laurent I, Dhainaut JF, Brunet F (1998) Early predictive factors of survival in the acute respiratory distress syndrome. Am J Respir Crit Care Med 158:1076–1081
Hollenberg SM, Ahrens TS, Annane D, Astiz ME, Chalfin DB, Dasta JF, Heard SO, Martin C, Napolitano LM, Susla GM, Totaro R, Vincent JL, Zanotti-Cavazzoni S (2004) Practice parameters for hemodynamic support of sepsis in adult patients: 2004 update. Crit Care Med 32:1928–1948
Price S, Nicol E, Gibson DG, Evans TW (2006) Echocardiography in the critically ill: current and potential roles. Intensive Care Med 32:48–59
Beaulieu Y, Marik PE (2005) Bedside ultrasonography in the ICU (part 1). Chest 128:881–895
Antonelli M, Levy M, Andrews PJD, Chastre J, Hudson LD, Manthous C, Meduri GU, Moreno RP, Putensen C, Stewart T, Torres A (2007) Hemodynamic monitoring in shock and implications for management. Intensive Care Med 33:575–590, International consensus conference, Paris, France, 27–28 Apr 2006
Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG, Ryan T, Bashore T, Corey GR (2000) Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 30:633–638
Joseph MX, Disney PJS, Da Costa R, Hutchison SJ (2004) Transthoracic echocardiography to identify or exclude cardiac cause of shock. Chest 126:1592–1597
Hüttemann E, Schelenz C, Kara F, Chatzinikolaou K, Reinhart K (2004) The use and safety of transoesophageal echocardiography in the general ICU – a minireview. Acta Anaesthesiol Scand 48:827–836
Gendreau MA, Triner WR, Bartfield J (1999) Complications of transesophageal echocardiography in the ED. Am J Emerg Med 17:248–251
Vignon P, Mentec H, François B, Gastinne H, Gay R (1996) Tolérance de l’échocardiographie transoesophagienne en réanimation. Réan Urg 5:3–8
Daniel WG, Erbel R, Kasper W, Visser CA, Engberding R, Sutherland GR, Grube E, Hanrath P, Maish B, Dennig K, Schartl M, Kremer P, Angermann C, Illiceto S, Curtius JM, Mugge A (1991) Safety of transesophageal echocardiography. A multicenter survey of 10, 419 examinations. Circulation 83:817–821
Min JK, Spencer KT, Furlong KT, DeCara JM, Suseng L, Ward RP, Lang RM (2005) Clinical features of complications from transesophageal echocardiography: a single-center case series of 10,000 consecutive examinations. J Am Soc Echocardiogr 18:925–929
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Berlin Heidelberg
About this chapter
Cite this chapter
Vignon, P., Mayo, P. (2011). Echocardiography in the Critically Ill: An Overview. In: de Backer, D., Cholley, B., Slama, M., Vieillard-Baron, A., Vignon, P. (eds) Hemodynamic Monitoring Using Echocardiography in the Critically Ill. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-87956-5_1
Download citation
DOI: https://doi.org/10.1007/978-3-540-87956-5_1
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-87954-1
Online ISBN: 978-3-540-87956-5
eBook Packages: MedicineMedicine (R0)