Abstract
Background: This paper reviews an experience of surgically treating ossification of the posterior longitudinal ligament (OPLL) with fixation of the involved spinal segments alone, without resorting to any bony or soft tissue decompression or attempts at direct resection of the OPLL. While in the early part of the experience, stabilization of only the involved subaxial cervical spinal segments was done, in the later part of the experience, atlantoaxial fixation was included in the multisegmental spinal fixation construct. This treatment is based on the understanding that spinal instability that includes atlantoaxial instability forms the nodal point of the pathogenesis and development of OPLL, and maturation of the presenting clinical symptoms.
Materials and Methods: Twenty-nine patients were treated in this series. There were 28 males and one female, and their ages ranged from 28 to 75 years (average 57 years). All patients presented with symptoms of neck pain, and progressive and disabling myelopathy-related quadriparesis. In the early part of the series (from 2012 to 2014), 14 patients underwent multilevel subaxial cervical spinal fixation by a transarticular technique of facetal fixation. After November 2014, atlantoaxial lateral mass fixation was included in the fixation construct in the subsequent 15 patients. Clinical assessments were done using a visual analogue scale (VAS), the Japanese Orthopaedic Association (JOA) scale and Goel’s clinical grading scale.
Results: All patients’ clinical symptoms improved in the immediate postoperative period, and the improvement was sustained and progressive in 28 patients.
Conclusion: Atlantoaxial and subaxial spinal instability seems to be the nodal pathogenetic factor in OPLL. Only stabilization of spinal segments that includes the atlantoaxial joint can provide a safe, simple and rational form of treatment.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Ossification of the posterior longitudinal ligament (OPLL) is a disabling cervical spinal disease. The complex structural presentation and potential for devastating postsurgical neurological complications make this disease a surgeon’s nightmare. A number of possible aetiological factors have been implicated in its development, but none has been seen to be convincing or consistent [1,2,3,4,5,6,7,8,9,10]. As the pathogenesis is unclear, the treatment strategy adopted is essentially based on radiological evidence of spinal canal intrusion by the bony mass anterior to the spinal cord, and is focused on restoring spinal canal dimensions that permit an unrestricted and uncompromised traverse of the neural structures.
This paper presents an experience of 29 cases where multiple spinal segment fixation alone was done, without any form of bony or soft tissue decompression or direct resection of the OPLL. In 15 cases in the latter half of the series, the atlantoaxial joint was included in the fixation construct. This treatment strategy is based on the understanding that atlantoaxial and spinal instability forms the basis of the pathogenesis and development of OPLL [11, 12].
Materials and Methods
During the period from June 2012 to April 2016, 29 patients with OPLL were treated with fixation alone as the treatment strategy, aimed at arthrodesis of the spinal segments. This analysis of the subject includes a case experience described in two previous publications [11, 12]. There were 28 males and one female in the series, and their ages ranged from 28 to 75 years (average 57 years). All patients presented with progressive quadriparesis as the primary symptom. Neck and hand pain were also prominent symptoms. Table 1 summarizes the clinical and radiological findings at the time of presentation and prior to surgery [13, 14]. Preoperative imaging included dynamic plain radiographs, computed tomography (CT) scanning and magnetic resonance imaging (MRI) in all patients. During the period from June 2012 to August 2014, 14 patients were treated with multisegmental subaxial cervical spinal fixation. After November 2014, in 15 patients, atlantoaxial fixation was additionally included in the fixation construct. Atlantoaxial instability was diagnosed on the basis of our recently described classification based on facetal malalignment in a neutral head position (type A: when the facet of the atlas is dislocated anterior to the facet of the axis; type B: when the facet of the atlas is dislocated posterior to the facet of the axis) or when facetal instability was identified during direct bone handling and manipulation during surgery (type C) [15]. Eleven patients had type B and four patients had type C atlantoaxial facetal instability. With use of the transarticular screw fixation technique described by Roy Camille and Saillant in 1972, subaxial spinal fixation was done [16]. Atlantoaxial fixation was done with use of the technique described by us in 1994 [17,18,19] (Figs. 1 and 2). For transarticular screw fixation the screws were 14 mm in length and 2.8 mm in diameter [15]. In nine patients, two screws were used for transarticular fixation as this method was considered to be safe and possible. Such ‘double insurance’ transarticular fixation was seen to add significant stability to the implant [20]. For atlantoaxial fixation the atlas and axis screws were 28 mm in length and 2.8 mm in diameter. A bone graft was harvested from the iliac crest and placed in the atlantoaxial joint cavity and in the appropriately prepared host bone of the midline spinal elements, which included the laminae and spinous processes and the lateral gutter. The patients were mobilized within 24 h of surgery and were advised to wear a hard cervical collar for a period of 3 months. After the 3-month healing period and confirmation of bone fusion, all activities and neck movements were permitted. Clinical assessments were done using a visual analogue scale (VAS), the Japanese Orthopaedic Association (JOA) scale and Goel’s clinical grading scale.
Images of a 53-year-old female patient. (a) A T2-weighted magnetic resonance image (MRI) shows ossification of the posterior longitudinal ligament (OPLL) extending from the C3 to C6 cervical levels. (b) A sagittal computed tomography (CT) scan shows the OPLL. (c) A sagittal image shows that the facets of the atlas and axis are in alignment. (d) A postoperative CT scan shows the facetal implant from C1 to T1. (e) A CT scan shows the implant
Images of a 55-year-old male patient. (a) A T2-weighted magnetic resonance image (MRI) shows multisegmental ossification of the posterior longitudinal ligament (OPLL). (b) A sagittal computed tomography (CT) scan shows the OPLL. (c) A sagittal image of the atlantoaxial facets shows marginal type B facetal instability. (d) A CT scan shows the implant
Results
All patients showed ‘remarkable’ immediate postoperative clinical improvement. During the follow-up period, which ranged from 4 to 50 months (average 29 months), the improvement was sustained and progressive in 28 of the 29 patients. Tables 2, 3 and 4 depict the clinical outcome after a minimum follow-up of 3 months. One 45-year-old male who had undergone subaxial spinal fixation in a case of OPLL that extended from C2 to C6 had postoperative improvement, but his neurological condition worsened 3 months after surgery. He then underwent anterior spinal decompression surgery and was subsequently lost to follow-up. The rest of the patients are well, are improved in terms of their clinical symptoms, are independent and active, and have not needed any further surgical treatment.
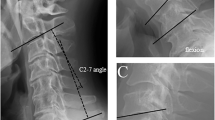
All patients underwent evaluation using static and dynamic cervical spine radiographs, CT scans and MRI. Static neutral lateral radiographs were used to assess cervical sagittal balance, while anteroposterior radiographs were used to exclude abnormal coronal alignment. The lordotic angle was measured using Cobb’s method of measurement [11, 12]. Preoperative assessment suggested that all patients had loss of cervical lordosis, with a lordotic angle ranging from 5° to 15°. After surgery there was a mild decrease in the lordotic angle, with the postoperative angle ranging from 4° to 12°. There was no significant difference between the preoperative and postoperative values. There were no wound infections, implant-related failures or complications. There was restriction of all spinal movements. Although all patients complained of this problem, none were unduly disturbed, considering the improvement in their limbs and their general ability to perform all activities of daily living. Fusion of the spinal segment was defined as the presence of bone formation across the facet joints, with absence of all kinds of motion between spinous processes and intervertebral bodies on flexion–extension, using CT images. According to these criteria, successful bone fusion was observed in all cases at follow-up assessments.
Discussion
OPLL , with its related myelopathy, is a relatively rare clinical event. Although it has been identified throughout the world, the entity has been more frequently reported in Asian countries. OPLL frequently presents in an advanced state, when it has already occupied a significant dimension of the spinal canal. OPLL may be segmental or continuous and can extend over several spinal segments. The additional intrusion of bony elements, which compromise the diameter of the spinal canal and indent into the neural tissues, poses a significant therapeutic challenge. The location of the OPLL anterior to the spinal cord and posterior to the vertebral bodies, and its hard bony consistency, make wide surgical exposure and therapeutic resection difficult and dangerous.
The pathogenesis of OPLL is entirely unclear and has been only speculated about [1, 2, 21]. Dietary, environmental, infective and physical constitution–related factors, apart from a host of other factors, have been incriminated as possible causes [1,2,3,4,5,6,7,8,9,10, 22,23,24]. In general, patients with OPLL are moderately obese, have a relatively thick neck girth and in general have a sedate lifestyle. Although a number of nonsurgical treatment forms have been advocated, the progressive and devastating nature of the clinical symptoms mandates a surgical solution. The surgical treatment is difficult to conceptualize, as the pathogenesis of the disease process is unclear.
The pathology of OPLL involves introduction of additional bony elements into the spinal canal, which traverses between the spinal segments. Considering this issue, the general opinion has been that patients with OPLL have a stable spine or a spine that is more than normally stable [11, 12]. The symptoms of progressive myelopathy and a radiological appearance of severe and long segmental spinal neural compression make the correlation straightforward. Most surgeons dealing with OPLL conceptually favour direct removal of OPLL and consider it the best surgical option, associated with long-term relief from symptoms and a cure for this condition [21, 23, 25,26,27,28,29,30,31,32,33,34,35,36,37]. However, the formidable nature of the surgical procedure, the need for extensive bone removal for wide exposure, the high risk of neural damage during exposure and OPLL dissection off the spinal cord, and the significant risk of cerebrospinal fluid (CSF) leakage make the surgical option of direct resection of OPLL less favourable [38]. Moreover, the devastating nature of the neurological complications following a failed operation makes the entity of OPLL a generally feared disease.
Considering the issues related to direct resection of the OPLL, the majority of surgeons prefer an indirect form of decompression. Some surgeons decide on anterior or posterior decompressive surgery on the basis of the presence or absence of kyphosis or lordosis of the cervical spine. Anterior decompression involves multilevel corpectomies and discoidectomies, and posterior decompression involves a wide and long decompressive laminectomy or various forms of spinal canal–expanding laminoplasty. The issue of spinal instability and the need for bone fusion of the treated spinal segment is considered a consequence of wide and multisegmental bone removal and has been associated with immediate or delayed postoperative spinal instability. Anterior stabilization techniques that include multisegmental metal cage implants with or without associated bone grafting have been identified as a satisfactory mode of spinal stabilization after decompression [23, 25, 26, 29, 30, 34, 36]. Posterior stabilization includes a number of wire/screw and metal rod/loop/ring fixation methods [27, 30, 33, 39]. Essentially, spinal stabilization is a treatment to ward off the possible destabilizing effects of decompressive surgery.
More recently some authors have identified that instability of the spinal segments is associated with OPLL. While neural compression is a static factor of cord compromise, instability is the dynamic factor affecting cord function [2, 11, 12]. On the basis of the premise that the entire pathological sequel of OPLL is related to spinal instability, fixation alone without any form of bony or soft tissue decompression or direct resection of OPLL has been identified as an ideal form of surgical treatment [11, 12]. Despite extensive bone formation in the posterior longitudinal ligament, the articular joints are always functional and active. Direct observation of the facets suggests that the joints not only are functional but actually appear to function excessively and pathologically. In the earlier part of the present study (from June 2012 to August 2014), transarticular screw fixation was done on segments in the vicinity of the OPLL after assessment of their unstable character [12]. Clinical improvement without any decompression and with stabilization alone confirmed that instability was the defining feature in the pathogenesis of OPLL. The observations emphasized that it is not neural compression or deformation but repeated microtrauma related to instability that is the cause of the symptoms [11, 12, 40,41,42,43,44,45,46]. Neural structures have remarkable elasticity, plasticity and capacity to sustain deformation if it is of a long-standing and slowly progressive nature. Such deformation of the spinal cord can be observed in benign spinal tumours and in cases of syringomyelia where the cord substance may be remarkably reduced in girth but the spinal cord still retains its significant functional ability.
As the concept of spinal instability as the cause of the symptoms and pathogenesis of OPLL gained ground, it was identified that atlantoaxial instability was associated with high cervical OPLL, particularly where the OPLL extended above the level of the C3 vertebra [11, 12]. Although the atlantoaxial instability could not be identified on dynamic imaging by assessment of alteration of the atlantodental interval, atlantoaxial facetal instability was identified [15]. Types B and C atlantoaxial facetal instability, as identified in our patients, have been grouped as central or axial instability. In these two types of atlantoaxial instability the atlantodental interval may not be altered and direct compression of the neural structures by the odontoid process is not a hallmark [15]. As cord compression is not a major or primary issue in these cases, the symptoms are subtle and long-standing. It was speculated earlier that instability of the atlantoaxial joint is the primary issue and that musculoskeletal alterations, torticollis, basilar invagination, Chiari malformation type I and syringomyelia are secondary and probably protective bodily responses [47, 48]. It may be that the bone formation in the posterior longitudinal ligament is protective and a response of the body to multisegmental spinal instability [11, 12]. We had speculated earlier that the presence of a retro-odontoid pseudotumour and ossification and osteophyte formation in the degenerative spine are secondary spinal features and a response to spinal instability [49]. It was also suggested that the presence of a retro-odontoid pseudotumour and cervical subaxial osteophytes as indicators of spinal instability suggests the need for stabilization. Direct removal of a retro-odontoid tumour or osteophytes has been considered a counterproductive operative procedure [43, 44, 49]. The understanding that atlantoaxial instability is associated with high OPLL has led to inclusion of atlantoaxial stabilization in the multilevel spinal fixation construct.
With further maturation of our understanding and with increasing experience in the subject, it was realized that atlantoaxial instability is frequently associated with even mid- and low cervical OPLL. In the subsequent part of our series, all 15 patients underwent multilevel spinal fixation that included atlantoaxial fixation. Although it cannot be concluded from the present study, it may be that atlantoaxial instability is the primary nodal point of the pathogenesis of cervical OPLL. While the results regarding the extent of spinal stability in patients treated for OPLL are still under evaluation, it seems that multilevel fixation aimed at arthrodesis of spinal segments including the atlantoaxial joint appears to be a rational form of surgical treatment that addresses the nodal point of the pathogenesis of OPLL.
Compliance with Ethical Standards
No financial support was received for this work.
Competing Interests
The author declares that he has no competing interests.
References
Epstein N. Ossification of the cervical posterior longitudinal ligament: a review. Neurosurg Focus. 2002;13(2):ECP1.
Fujiyoshi T, Yamazaki M, Okawa A, Kawabe J, Hayashi K, Endo T, et al. Static versus dynamic factors for the development of myelopathy in patients with cervical ossification of the posterior longitudinal ligament. J Clin Neurosci. 2010;17:320–4.
Inamasu J, Guiot BH, Sachs DC. Ossification of the posterior longitudinal ligament: an update on its biology, epidemiology, and natural history. Neurosurgery. 2006;58:1027–39.
Kobashi G, Washio M, Okamoto K, Sasaki S, Yokoyama T, Miyake Y, et al. High body mass index after age 20 and diabetes mellitus are independent risk factors for ossification of the posterior longitudinal ligament of the spine in Japanese subjects: a case–control study in multiple hospitals. Spine. 2004;29:1006–10.
Matsunaga S, Sakou T. Ossification of the posterior longitudinal ligament of the cervical spine: etiology and natural history. Spine. 2012;37(5):E309–14.
Matsunaga S, Yamaguchi M, Hayashi K, Sakou T. Genetic analysis of ossification of the posterior longitudinal ligament. Spine. 1999;24:937–9.
Matsunaga S, Kukita M, Hayashi K, Shinkura R, Koriyama C, Sakou T, et al. Pathogenesis of myelopathy in patients with ossification of the posterior longitudinal ligament. J Neurosurg. 2002;96(2 Suppl):168–72.
Ramos-Remus C, Russell AS, Gomez-Vargas AS, Hernandez-Chavez A, Maksymowych WP, Gamez-Nava JI, et al. Ossification of the posterior longitudinal ligament in three geographically and genetically different populations of ankylosing spondylitis and other spondyloarthropathies. Ann Rheum Dis. 1998;57:429–33.
Sakou T, Taketomi E, Matsunaga S, Yamaguchi M, Sonoda S, Yashiki S. Genetic study of ossification of the posterior longitudinal ligament in the cervical spine with human leukocyte antigen haplotype. Spine. 1991;16:1249–52.
Song J, Mizuno J, Hashizume Y, Nakagawa H. Immunohistochemistry of symptomatic hypertrophy of the posterior longitudinal ligament with special reference to ligamentous ossification. Spinal Cord. 2006;44:576–81.
Goel A. Is atlantoaxial instability the cause of “high” cervical ossified posterior longitudinal ligament? Analysis on the basis of surgical treatment of seven patients. J Craniovertebr Junction Spine. 2016;7(1):20–5.
Goel A, Nadkarni T, Shah A, Rai S, Rangarajan V, Kulkarni A. Is only stabilization the ideal treatment for ossified posterior longitudinal ligament? Report of early results with a preliminary experience in 14 patients. World Neurosurg. 2015;84(3):813–9.
Fujiwara A, Kobayashi N, Saiki K, Kitagawa T, Tamai K, Saotome K. Association of the Japanese Orthopaedic Association score with the Oswestry Disability Index, Roland–Morris Disability Questionnaire, and Short-Form 36. Spine. 2003;28:1601–7.
Huskisson EC. Measurement of pain. J Rheumatol. 1982;9:768–9.
Goel A. Goel’s classification of atlantoaxial “facetal” dislocation. J Craniovertebr Junction Spine. 2014;5(1):3–8.
Roy-Camille R, Saillant G. Surgery of the cervical spine. 2. Dislocation. Fracture of the articular processes. J Nouv Presse Med. 1972;1:2484–5.
Goel A, Bhatjiwale M, Desai K. Basilar invagination: a study based on 190 surgically treated cases. J Neurosurg. 1998;88:962–8.
Goel A, Desai K, Muzumdar D. Atlantoaxial fixation using plate and screw method: a report of 160 treated patients. Neurosurgery. 2002;51:1351–7.
Goel A, Laheri VK. Plate and screw fixation for atlanto-axial dislocation. (Technical report). Acta Neurochir (Wien). 1994;129:47–53.
Goel A. Alternative technique of cervical spinal stabilization employing lateral mass plate and screw and intraarticular spacer fixation. J Craniovertebr Junction Spine. 2013;4(2):56–8.
Sugrue PA, McClendon J Jr, Halpin RJ, Liu JC, Koski TR, Ganju A. Surgical management of cervical ossification of the posterior longitudinal ligament: natural history and the role of surgical decompression and stabilization. Neurosurg Focus. 2011;30:E3.
Chiba K, Kato Y, Tsuzuki N, Nagata K, Toyama Y, Iwasaki M, et al. Computer-assisted measurement of the size of ossification in patients with ossification of the posterior longitudinal ligament in the cervical spine. J Orthop Sci. 2005;10:451–6.
Matsunaga S, Sakou T, Taketomi E, Komiya S. Clinical course of patients with ossification of the posterior longitudinal ligament: a minimum 10-year cohort study. J Neurosurg Spine. 2004;100(3 Suppl):245–8.
Resnick D, Guerra J Jr, Robinson CA, Vint VC. Association of diffuse idiopathic skeletal hyperostosis (DISH) and calcification and ossification of the posterior longitudinal ligament. AJR Am J Roentgenol. 1978;131:1049–53.
Abe H, Tsuru M, Ito T, Iwasaki Y, Koiwa M. Anterior decompression for ossification of the posterior longitudinal ligament of the cervical spine. J Neurosurg. 1981;55:108–16.
Belanger TA, Roh JS, Hanks SE, Kang JD, Emery SE, Bohlman HH. Ossification of the posterior longitudinal ligament. Results of anterior cervical decompression and arthrodesis in sixty-one North American patients. J Bone Joint Surg Am. 2005;87:610–5.
Cho WS, Chung CK, Jahng TA, Kim HJ. Post-laminectomy kyphosis in patients with cervical ossification of the posterior longitudinal ligament: does it cause neurological deterioration? J Korean Neurosurg Soc. 2008;43:259–64.
Epstein NE. Circumferential surgery for the management of cervical ossification of the posterior longitudinal ligament. J Spinal Disord. 1998;11:200–7.
Goel A, Pareikh S. Limited oblique corpectomy for treatment of ossified posterior longitudinal ligament. Neurol India. 2005;53(3):280–2.
Iwasaki M, Kawaguchi Y, Kimura T, Yonenobu K. Long-term results of expansive laminoplasty for ossification of the posterior longitudinal ligament of the cervical spine: more than 10 years follow up. J Neurosurg. 2002;96(2 Suppl):180–9.
Iwasaki M, Okuda S, Miyauchi A, Sakaura H, Mukai Y, Yonenobu K, et al. Surgical strategy for cervical myelopathy due to ossification of the posterior longitudinal ligament: part 2: advantages of anterior decompression and fusion over laminoplasty. Spine. 2007;32:654–60.
Kaiser MG, Mummaneni PV, Matz PG, Anderson PA, Groff MW, Heary RF, et al. Radiographic assessment of cervical subaxial fusion. J Neurosurg Spine. 2009;11:221–7.
Kato Y, Iwasaki M, Fuji T, Yonenobu K, Ochi T. Long-term follow-up results of laminectomy for cervical myelopathy caused by ossification of the posterior longitudinal ligament. J Neurosurg. 1998;89:217–23.
Matsuoka T, Yamaura I, Kurosa Y, Nakai O, Shindo S, Shinomiya K. Long-term results of the anterior floating method for cervical myelopathy caused by ossification of the posterior longitudinal ligament. Spine. 2001;26:241–8.
Mizuno J, Nakagawa H. Ossified posterior longitudinal ligament: management strategies and outcomes. Spine J. 2006;6(6 Suppl):282S–8S.
Odom GL, Finney W, Woodhall B. Cervical disc lesions. J Am Med Assoc. 1958;166:23–8.
Onari K, Akiyama N, Kondo S, Toguchi A, Mihara H, Tsuchiya T. Long-term follow-up results of anterior interbody fusion applied for cervical myelopathy due to ossification of the posterior longitudinal ligament. Spine (Phila Pa 1976). 2001;26:488–93.
Joseph V, Kumar GS, Rajshekhar V. Cerebrospinal fluid leak during cervical corpectomy for ossified posterior longitudinal ligament: incidence, management, and outcome. Spine. 2009;34:491–4.
Houten JK, Cooper PR. Laminectomy and posterior cervical plating for multilevel cervical spondylotic myelopathy and ossification of the posterior longitudinal ligament: effects on cervical alignment, spinal cord compression, and neurological outcome. Neurosurgery. 2003;52:1081–8.
Goel A. Facet distraction spacers for treatment of degenerative disease of the spine: rationale and an alternative hypothesis of spinal degeneration. J Craniovertebr Junction Spine. 2010;1(2):65–6.
Goel A. Facet distraction–arthrodesis technique: can it revolutionize spinal stabilization methods? J Craniovertebr Junction Spine. 2011;2(1):1–2.
Goel A. ‘Only fixation’ as rationale treatment for spinal canal stenosis. J Craniovertebr Junction Spine. 2011;2(2):55–6.
Goel A. Is it necessary to resect osteophytes in degenerative spondylotic myelopathy? J Craniovertebr Junction Spine. 2013;4(1):1–2.
Goel A. Letter to Editor: resolution of cystic deterioration of the C1–2 articulation with posterior fusion: treatment implications for asymptomatic patients. Puffer RC, Van Gompel JJ, Morris JM, Krauss WE. World Neurosurg. 2013;79(5–6):773–8.
Goel A, Shah A. Facetal distraction as treatment for single- and multilevel cervical spondylotic radiculopathy and myelopathy: a preliminary report. J Neurosurg Spine. 2011;14(6):689–96.
Goel A, Shah A, Jadhav M, Nama S. Distraction of facets with intraarticular spacers as treatment for lumbar canal stenosis: report on a preliminary experience with 21 cases. J Neurosurg Spine. 2013;19(6):672–7.
Goel A. Chiari malformation—is atlantoaxial instability the cause? Outcome analysis of 65 patients with Chiari malformation treated by atlantoaxial fixation. J Neurosurg Spine. 2015;22(2):116–27.
Goel A. Treatment of basilar invagination by atlantoaxial joint distraction and direct lateral mass fixation. J Neurosurg Spine. 2004;1(3):281–6.
Goel A. Retro-odontoid mass: an evidence of craniovertebral instability. J Craniovertebr Junction Spine. 2015;6(1):6–7.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Goel, A. (2019). Ossification of the Posterior Longitudinal Ligament: Analysis of the Role of Craniovertebral and Spinal Instability. In: Visocchi, M. (eds) New Trends in Craniovertebral Junction Surgery. Acta Neurochirurgica Supplement, vol 125. Springer, Cham. https://doi.org/10.1007/978-3-319-62515-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-62515-7_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-62514-0
Online ISBN: 978-3-319-62515-7
eBook Packages: MedicineMedicine (R0)