Abstract
Contrast-enhanced MRI is currently the method of choice for the diagnosis of diffuse low-grade gliomas and provides an excellent depiction of structural changes in the brain. Nevertheless, the delineation of the tumor from normal brain tissue and non-specific abnormalities on MRI such as edema or treatment-related changes can be difficult. Positron-Emission-Tomography (PET) provides additional information on tumor metabolism and is helpful in many clinical situations. In particular, PET using radiolabeled amino acids has a wide range of applications and helps to solve a number of clinical issues. At initial diagnosis, amino acid PET may be helpful to estimate the prognosis of a low-grade glioma and to optimize patient counseling. Furthermore, the method improves targeting of biopsy and provides additional information of tumor extent, which is also helpful for resection planning and radiotherapy. In the further course of the disease, amino acid PET allows a sensitive monitoring of treatment response, the early detection of tumor recurrence, and an improved differentiation of tumor recurrence from treatment-related changes. In the past, the method had only limited availability due to the low number of PET scanners and the use of radiopharmaceuticals with a short half-life. In recent years, however, the number of PET scanners in hospitals has increased considerably. Furthermore, novel amino acid tracers labeled with positron emitters with a longer half-life have been developed and clinically validated which allow a more efficient and cost-effective application. These developments and the well-documented diagnostic performance of PET using radiolabeled amino acids suggest that its application continues to spread and that the method may be available as a routine diagnostic technique for certain indications in the near future.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
MRI with its excellent soft tissue contrast, the high spatial resolution, and its multiplanar reconstruction capabilities is currently the method of first choice for the diagnosis of cerebral gliomas. Despite these unsurpassed properties of MRI, many problems in the diagnostic assessment of low-grade gliomas remain and a number of pivotal questions concerning the management of low-grade gliomas cannot be answered satisfactorily. Thus, at initial diagnosis, diffuse low-grade gliomas may exhibit only minimal changes in the brain tissue, which can hardly be distinguished from benign lesions. In larger tumors, the differentiation of glioma tissue from surrounding edema may be difficult, particularly when the tumor is not sharply demarcated from normal brain tissue due to diffuse tumor cell infiltration. Although commonly assigned as low-grade gliomas the course is clinically diverse and for some patients the disease has a benign course, whereas others experience rapid progression [1]. In the further course of the disease, the tumors may exhibit regional malignant progression, which is difficult to detect, especially when the blood-brain barrier (BBB) remains intact. In these patients with a heterogeneous tumor and intact BBB, biopsy guidance may be especially difficult. Monitoring of treatment response is another important factor to optimize individual treatment strategy where volume changes in MRI are just a very late sign. After treatment, postoperative or radiogenic changes in peritumoral brain tissue may result in contrast-enhancement on MRI that cannot be reliably distinguished from vital tumor tissue of recurrent glioma [2, 3].
Therefore, alternative imaging methods reflecting metabolic features of the tumor tissue have attracted the interest of neuro-oncologists for many years in order to facilitate the process of clinical decision-making in this challenging tumor entity. PET is a powerful method in Nuclear Medicine that has shown great potential for the diagnostic assessment of malignant tumors. The most widely used tracer for PET is 18F-Fluorodeoxglucose (FDG), which is accumulated in the majority of tumors due to an increased energy demand and consequently an increased glucose metabolism. FDG has been used for the evaluation of brain tumors since the early days of PET and a relationship of FDG uptake and tumor grade of gliomas and prognosis of cerebral gliomas has been reported in numerous studies [4]. In low-grade gliomas, however, FDG uptake is generally low and there is high FDG uptake in the surrounding normal brain tissue. Therefore, the usefulness of this tracer for low-grade gliomas is limited. In this chapter, the most promising PET tracers for the diagnostic assessment of low-grade gliomas will be reviewed. The chapter deals mainly with the application of radiolabeled amino acids owing to the ability of these substrates to pass the intact BBB and to depict brain tumors with a high tumor-to-background contrast. These tracers are well investigated, allow decisive diagnostic information in cerebral gliomas with respect to many clinical aspects, and are quite close to be established in routine clinical diagnosis (Table 15.1) [5,6,7,8,9,10,11,12].
2 Radiopharmaceuticals for PET in Low-Grade Gliomas
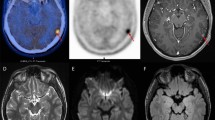
Today, the most widely used application of PET is the measurement of glucose metabolism with FDG in various types of cancer. In cerebral gliomas, FDG uptake is correlated with the degree of malignancy of the tumor (WHO grading) and with the patient’s outcome [4, 13, 14]. Due to the high rate of glucose metabolism especially in the grey matter of the brain, however, it is difficult to distinguish glioma tissue from normal brain tissue by FDG-PET. While most high-grade gliomas WHO grade III and nearly all grade IV glioblastomas show an increased FDG uptake compared to the white matter, low-grade gliomas WHO grade II usually exhibit an indifferent or even a decreased FDG uptake (see Fig. 15.1). Therefore, FDG PET is not useful to delineate low-grade gliomas from the surrounding brain tissue.
Astrocytoma WHO Grade II in the left hemisphere. The T1-weighted MRI after application of Gd-DTPA shows no contrast enhancement indicating an intact BBB and depiction of the tumor in the T2 weighted MRI is similar. FDG PET shows hypometabolism and is not helpful to guide biopsy. FET PET identifies a hot spot within the tumor and detects an optimal biopsy site
Nevertheless, FDG PET has been shown to be useful to detect malignant transformation in low-grade gliomas and may therefore be useful for follow-up in low-grade gliomas [4]. The use of proliferation markers such as [18F]3′-deoxy-3′-fluorothymidine (FLT) showed even a better correlation with the grade of malignancy and prognosis of cerebral gliomas than FDG uptake or MR spectroscopy [15, 16]. An image-guided biopsy study demonstrated that FLT is a useful marker of cellular proliferation that correlates with regional variation in cellular proliferation, but was unable to identify the margin of gliomas [17]. This is caused by the fact that FLT is not able to pass the intact BBB and accumulates usually in areas with contrast enhancement on MRI only [15, 18,19,20]. Therefore, portions of the tumor with an intact blood-brain barrier (BBB)—frequently present in low-grade gliomas—cannot be detected with FLT-PET. Furthermore, 11C-Choline or 18F-Fluoro-choline (FCH) has been used as a marker of cell membrane phospholipids in brain tumors and shows a significant correlation of uptake with the degree of malignancy in gliomas [21, 22]. Tracer uptake in areas with intact BBB is generally low but some studies have reported that FCH might be helpful to detect recurrent LGG in brain areas showing no contrast enhancement in MRI [23, 24].
Another interesting approach is to investigate the presence of intratumoral hypoxia using 18F-Fluoromisonidazole [25,26,27,28]. Hypoxia in tumors is a pathophysiological consequence of structurally and functionally disturbed angiogenesis along with deterioration in the inability of oxygen to diffuse through tissues. A PET study in patients with cerebral gliomas demonstrated areas of hypoxia in glioblastomas, but all investigated low-grade gliomas showed low uptake of 18F-Fluoromisonidazole [29]. This is not unexpected since tumor growth and angiogenesis in low-grade gliomas are still in a balance so that this approach is particularly attractive for the evaluation of high-grade gliomas.
A promising new target for brain tumor imaging is the mitochondrial translocator protein (TSPO), which is a component of the mitochondrial permeability transition pore and is strongly expressed by glioma cell lines [30]. In the recent past, PET imaging using TSPO ligands such as 11C-(R)PK11195 focused mainly on inflammatory brain diseases as an indicator of microglial activation but recent studies suggest a role of this method in the assessment of brain tumors [31,32,33]. A recent study has shown that TSPO expression may extend beyond the tumor margins in MRI and amino acid PET indicating an infiltration zone that exhibited tumor progression in the further follow-up of the patients [34].
At present, the best established PET tracers for the investigation of low-grade gliomas are radiolabeled amino acids of the class of large neutral amino acids such as [Methyl-11C]-L-methionine (MET), O-(2-[18F]fluorethyl)-L-tyrosine (FET) and 3,4-dihydroxy-6-[18F]fluoro-phenylalanine (FDOPA) [5, 6, 9,10,11,12, 35, 36]. Because the uptake of these amino acids by both, the white and grey matter of normal brain tissue is relatively low, cerebral gliomas can be distinguished from the surrounding normal tissue with high contrast. It was long assumed that increased uptake of MET in brain tumors reflects an increased protein synthesis rate. Experiments in mice, however, demonstrated that an inhibition of protein synthesis did not influence the uptake of radiolabeled MET in tumors and brain [37] suggesting that alterations of amino acid transport rather than increased protein synthesis caused increased uptake in tumors. Furthermore, the predominant role of transport phenomena for increased amino acid uptake in gliomas is confirmed by the observation that PET using radiolabeled amino acids such as FET which are not incorporated into protein exhibit nearly identical results concerning brain tumor imaging as MET PET or FDOPA PET. Thus, a number of studies have shown that imaging of cerebral gliomas with MET, FET and FDOPA is rather similar [38,39,40,41,42,43]. Since FDOPA is a precursor of dopamine it shows also uptake in the striatum and can be used to trace the dopaminergic pathway in the nigrostriatal region to evaluate the presynaptic function in patients with neurodegenerative and movement disorders [44]. This property may cause problems in the delineation of gliomas affecting the striatum [12, 45].
The increased uptake of amino acids such as MET, FET and FDOPA by cerebral glioma tissue appears to be caused predominantly by increased transport via the transport system L for large neutral amino acids namely the subtypes LAT1 and LAT2 [46,47,48,49,50]. A recent study suggested that the trapping of FET within the cells is caused by the asymmetry of its intra- and extracellular recognition by LAT1 [48]. Nevertheless, there appear to be some differences in transport characteristics of MET, FET and FDOPA. FET shows different patterns of time-activity-curves in low-grade and high-grade gliomas [51,52,53,54,55] which could not be observed with MET or FDOPA [43, 56].
Since large neutral amino acids also enter normal brain tissue, a disruption of the BBB, i.e., enhancement of contrast media on MRI scans, is not a prerequisite for intratumoral accumulation of MET, FET and FDOPA (see Fig. 15.1). Consequently, uptake of these tracers has been reported in many low-grade gliomas without BBB leakage [5, 35, 57,58,59,60].
Most PET studies of cerebral gliomas have been performed with the amino acid MET [5], although the short half-life of 11C (20 min) limits the use of this technique to the few centers that are equipped with an in-house cyclotron facility. In contrast to MET, 18F-labelled amino acids (half-life, 109 min) such as FET and FDOPA can be transported from a cyclotron unit to multiple external PET centers. This enables a wider application of amino acid PET in clinical diagnosis. One of the best-established 18F-labelled amino acids is FET that can be produced in large amounts for clinical purposes like the widely used FDG [36, 61, 62].
Animal experiments have shown that FET, in contrast to MET, exhibits no uptake in inflammatory cells and in inflammatory lymph nodes but false positive uptake has been observed for MET, FET and FDOPA in human brain abscesses, demyelinating processes, near cerebral ischemia and hematomas [12, 63,64,65]. Therefore, increased uptake of the tracers is not specific for cerebral gliomas although increased amino acid uptake has a high positive predictive value for cerebral gliomas [66]. The report is focused on the clinical experiences with MET, FET and FDOPA, which are at present the best-validated amino acid tracers for PET.
3 Clinical Applications of PET in Diffuse Low-Grade Gliomas
3.1 Detection of Low-Grade Gliomas and Differential Diagnosis
The diagnostic potential of amino acid PET to detect low-grade gliomas is limited since MET and FET exhibit increased uptake only in a fraction of about 60–80% of low-grade gliomas [6, 35, 57, 58, 60, 63, 65, 67,68,69]. The specificity of MET, FET and FDOPA PET for neoplastic lesions and especially in the case of low-grade gliomas is generally affected by possible tracer uptake in the area of benign processes such as hematoma, ischemia, traumatic brain injury, and acute inflammatory processes [5, 63,64,65, 70,71,72,73]. In the largest study to date evaluating MET PET in a consecutive series of 196 patients with suspected brain tumors, differentiation between gliomas and non-neoplastic lesions was correct in 79% using a threshold of the tumor/brain ratio of 1.47. Exclusion of high-grade gliomas (99 low-grade gliomas versus 24 non-neoplastic lesions) yielded a sensitivity of 67% and specificity of 72% for distinguishing low-grade gliomas from non-neoplastic brain lesions [6, 68]. The diagnostic performance of FET PET has been evaluated in a series of 174 newly diagnosed cerebral lesions with suspicion of glioma, which included 72 histologically confirmed diffuse low-grade gliomas [58]. In that study, the mean tumor to brain ratio (TBR) was 1.8 ± 0.5 in low-grade gliomas versus 1.4 ± 0.4 in nonneoplastic lesions yielding a sensitivity of 79% and specificity of 48% for distinguishing low-grade gliomas from non-neoplastic brain lesions. These results are similar to the observations in other publications in which low-grade gliomas, with the exception of oligodendrogliomas, presented with mean TBR in the range of inflammatory and other active (e.g., ischemic, traumatic) brain lesions [63, 65, 67]. Higher amino acid uptake in the subgroup of low-grade oligodendrogliomas and oligoastrocytomas according to the WHO classification 2007 [74] has been reported in several studies and is most probably related to the increased cellular and vascular density in this glioma subtype [52, 60, 75,76,77].
Taken together, the diagnostic accuracy of amino acid PET to detect low-grade gliomas and to differentiate suspicious lesions from non-specific uptake in non-neoplastic lesions is limited. Therefore, a histological evaluation of suspicious brain lesions by biopsy remains necessary under most circumstances.
3.2 Identification of an Optimal Site for Biopsy
An important aspect of the diagnostic assessment of low-grade gliomas is the definition of areas with the highest cellular proliferation rates. Since the tumor biology is dominated by the most aggressive glioma parts, representative tissue samples are vitally important for histological tumor diagnosis, prognostication, and treatment planning. The ability of MRI to show the most rapidly proliferating portions of diffuse low-grade gliomas is limited, particularly when the tumor shows no contrast enhancement in MRI, which occurs frequently in low-grade gliomas. FDG and FLT PET are usually negative in low-grade gliomas and provide no information on regional heterogeneity of metabolic activity in these tumors. Radiolabeled amino acids exhibit increased uptake in the majority of diffuse low-grade gliomas and are helpful to optimize the targeting of biopsies and prevent the problem of non-diagnostic biopsies from non-specifically altered tissue (Figs. 15.1 and 15.2). Biopsy controlled studies have shown that MET and FET uptake correlate with microvessel and cell density in non-contrast enhancing gliomas [77,78,79]. Vascular density is a frequently described feature linked to early malignant transformation in gliomas [80]. A number of studies have compared the diagnostic potential of PET using FDG and MET or FET to identify metabolic hot spots in cerebral gliomas to guide biopsy [67, 81, 82]. These studies consistently report that regionally increased FDG uptake, if present, is congruent with that of increased MET or FET uptake but amino acid PET is generally more sensitive than FDG PET. A number of studies have evaluated the role of amino acid PET for biopsy guidance in the subgroup of low-grade gliomas. In a study with 32 patients that included 10 low-grade gliomas MET PET allowed the definition of a biopsy target in all low-grade gliomas while FDG showed increased uptake in only one of these tumors [83]. In a patient series of 22 histologically confirmed low-grade gliomas, FET PET identified a local maximum for biopsy guidance in 16 of the tumors (72%), while FDG identified a metabolic spot in only 2 (9%) of the low-grade gliomas [67]. Another study including 72 histologically confirmed diffuse low-grade gliomas, FET PET identified a local maximum in 79% of the tumors [58]. Other studies emphasize the role of kinetic analyses of FET uptake in low grade glioma [51, 54, 59, 69, 84]. Areas with an early peak in FET uptake followed by a descending time activity curve were associated with areas of malignant transformation and poor prognosis. Interestingly, a “malignant curve pattern” was also predictive for poor outcome if FET uptake in the suspicious brain lesion was low [51, 59, 69]. These data suggest that amino acid PET is a useful tool for identifying metabolic hot spots in low-grade gliomas to target biopsies. Furthermore, dynamic FET PET appears to provide important additional information on the aggressiveness of the tumors independent of the degree of tracer uptake. Nevertheless, it is not yet proven beyond doubt that the maximum concentration of amino acid uptake in low-grade gliomas corresponds to the most aggressive part of the tumor and further studies are needed to investigate this aspect (Fig. 15.3).
Astrocytoma WHO Grade II in the frontal lobe. T1-weighted MRI after application of Gd-DTPA shows no pathological contrast-enhancement and a tumor cannot be clearly delineated. T2-weighted MRI shows widespread abnormalities within the complete frontal lobe and is not helpful to depict the tumor. FET PET identifies a clear tumor with high tracer uptake in the lower frontal lobe
MRI and FET PET and of a patient with newly diagnosed histologically confirmed diffuse astrocytoma WHO grade II in right temporal lobe (upper row). T1 weighted MRI shows no contrast enhancement (left) and hyperintensity on T2-weighted image (middle) and slightly increased FET uptake (left). Time–activity curve on the right shows constantly increasing FET uptake. Twenty-nine months later (lower row), patient presented with malignant progression to WHO grade III astrocytoma associated with significant increase in FET uptake, discrete contrast enhancement and enlargement of hyperintensity on T2-weighted image. Dynamic evaluation of FET uptake (right) shows a “malignant curve pattern” with an early peak of FET uptake followed by declining time activity curve
3.3 Delineation of Tumor Extent for Treatment Planning
Multiple studies in which the radiological findings were compared with the histological findings in tissue samples obtained by biopsy or open surgery have provided clear evidence that PET using radiolabeled amino acid detects the solid tumor mass of cerebral glioma tissue more reliably than either CT or MRI [81, 83, 85,86,87,88]. This is especially relevant for the non-enhancing parts of gliomas in MRI, which predominantly occur in low-grade gliomas. In a study exploring the potential of FET PET to image the extent of cerebral gliomas, 52 neuronavigated biopsies were taken from cerebral gliomas of 31 patients. Neoplastic tissue was found in 94% of biopsies in FET-PET positive areas, but only in 53% of the suspicious areas identified by MRI [87]. In that study, 12 biopsies yielded the histopathological diagnosis of a diffuse low-grade glioma and FET uptake was increased in all but one of the areas from which the biopsies were taken. In contrast, none of these areas showed contrast-enhancement on MRI. Another study investigated the role of FET-PET as a surrogate marker for accumulation of 5-aminolevulinic acid (5-ALA), which is used as a metabolic marker of malignant glioma cells for fluorescence-guided resection [89]. In that study, patients with 17 low-grade gliomas were included. FET was positive in 7 of the tumors while 5-ALA was observed in only one of the low-grade gliomas, which showed corresponding contrast enhancement. These data indicate that amino acid uptake in PET is a more sensitive indicator of low-grade glioma than 5-ALA fluorescence.
Since amino acid PET appears to be a valuable instrument to detect the solid tumor mass of cerebral gliomas, this technique has been used for resection planning. In a study evaluating integrated MET PET and MRI guided resection of 103 brain tumors, a large fraction of low-grade gliomas was included [90]. Resection planning in 59 low-grade gliomas demonstrated that the PET volume did not match the MR volume and improved the tumor volume definition in 88% of the cases. Similar results were reported in other studies for MET and FET PET which mainly included high-grade gliomas [91,92,93].
These data suggest that resection of low-grade gliomas guided by amino acid PET may increase the resection extent and thus the patients’ survival. It needs, however, to be considered that MET, FET and FDOPA show increased uptake in only 60–80% of low-grade gliomas and that the resection of the tumors with low amino acid uptake cannot be improved.
The improved imaging of glioma tissue using amino acid PET has also been applied to improve planning of radiation treatment of brain tumors [94]. A number of centers have started to integrate amino acid imaging into CT- and MRI-based radiotherapy planning, particularly in high-grade gliomas and when high-precision radiotherapy is to be given or in the setting of dose escalation studies or for the re-irradiation of recurrent tumors [95,96,97,98,99,100,101,102,103]. Experiences with amino acid PET radiotherapy planning of low-grade gliomas are limited but indicate improved sensitivity in detecting postoperative residual tumor and a benefit for radiotherapy planning in patients with inconclusive MRI findings [104]. Improved outcome of the patients with radiotherapy planning by amino acid imaging compared with conventional therapy planning, however, has not yet been proven.
3.4 Glioma Grading and Prognosis
FDG PET is considered as a relative accurate predictor of tumor grade and prognosis of cerebral gliomas and the detection of foci with increased FDG uptake in low-grade glioma is highly suspicious for malignant progression [13, 105]. The combination of FDG PET and multiparametric MRI may further improve the diagnostic accuracy to differentiate high-grade and low-grade glioma [14]. Most PET studies employing amino acids have shown that gliomas of different WHO grades overlap in their degree of amino acid uptake, so that the tumor grade cannot be reliably predicted with this technique [5, 19, 41, 52, 58, 87, 106]. A high potential to differentiate high-grade and low-grade gliomas has also been claimed for FLT, but FLT uptake goes along with BBB disruption and there is a high fraction of anaplastic astrocytoma without significant contrast enhancement on MRI which consecutively are negative in FLT PET [19, 20].
Using FET PET a number of studies have demonstrated that the evaluation of tracer kinetics in the tumors may be helpful to differentiate between high-grade and low-grade gliomas [52, 53, 55, 59, 107]. High-grade gliomas are characterized by an early peak of the time-activity curve around 10–15 min after tracer injection followed by a decrease of FET uptake. In contrast, time–activity curves slightly and steadily increase in low-grade gliomas of WHO grade II. Using dynamic evaluation of selected regions of the tumor, high-grade and low-grade gliomas could be distinguished with an accuracy of 70–90% in primary tumors as well as in recurrent tumors [52, 53, 59, 84, 107]. Furthermore, a recent study suggested that early static scans of FET uptake have a higher diagnostic accuracy for grading of gliomas than the standard 20–40 min scans but this approach did not reach the accuracy of dynamic FET imaging [108]. Studies using MET and FDOPA demonstrated that unlike FET PET, the time-activity curves of tracer uptake do not allow the classification of low- and high-grade gliomas [43, 56].
Considering gliomas of all WHO grades the prognostic significance of amino acid uptake remains a matter of controversy. Some studies seem to show that lower amino acid uptake especially in astrocytic glioma is associated with a better prognosis, but there is generally high uptake in oligodendroglioma despite their apparently better prognosis [5, 60, 75, 76, 109]. Recent studies suggest that dynamic FET PET [110], the “biological tumor volume” (BTV) as assessed by amino acid PET at primary diagnosis [111, 112] or textural parameters considering tumor heterogeneity may be important predictors of prognosis [113].
For patients with low-grade gliomas prognostication is an important factor for disease management. Some of these patients have a stable course with an excellent quality of life for many years or decades even without treatment, while others experience rapid tumor progression with malignant transformation to a high-grade glioma and a poor prognosis. A better identification of individuals with either a poor or a favorable prognosis is highly desirable to optimize patient counseling. A study with MET PET showed that these patients benefit from a surgical procedure only when increased amino acid uptake can be demonstrated [114]. In a series of 24 patients with low-grade gliomas, patients with a tumor/brain ratio >2.2 had a significantly shorter survival time than the patients with a tumor/brain ratio <2.2 [115]. Similarly, in a series of 50 patients with low-grade gliomas a SUV of FDOPA uptake >1.75 was an independent predictor of disease progression [35]. Another study indicated that the combined evaluation of FET-PET and MR morphology was a statistically significant prognostic predictor for patients with newly diagnosed low-grade gliomas [57]. Within a 7-year period, a group of 33 consecutive patients with previously untreated non-enhancing WHO grade II glioma were included in a prospective study. A baseline, both MRI and FET-PET were performed before histology in all patients on tissue samples by biopsy and a “watch and wait” strategy without further treatment was started. During the follow-up it turned out, that baseline FET uptake and a circumscribed versus a diffuse growth pattern on MRI were highly significant predictors for patients course and outcome: Those low-grade gliomas that were well delineated on MRI and showed no FET uptake had an excellent prognosis with long progression-free intervals, good clinical condition and late malignant transformation. In contrast, patients with low-grade gliomas with diffuse tumor margins on T2-weighted MRI and FET uptake had a poor outcome with early progression in combination with malignant transformation to a HGG, rapid clinical deterioration, and die earlier. A recent study in 54 gliomas of WHO grade II observed no correlation between FET uptake and progression-free survival but that analysis included 16 patients with recurrent tumors and comparison with other studies is difficult [60]. In any case, also low-grade gliomas with low FET uptake should be monitored carefully because also tumors without tracer uptake can harbor high-grade glioma tissue [59].
Recent studies have emphasized the role of kinetic analyses of FET uptake in the evaluation and follow-up of low-grade glioma [51, 54, 59, 69, 84, 116]. Areas with an early peak of FET uptake followed by a descending time activity curve in suspected low-grade gliomas were associated with areas of malignant transformation and poor prognosis. Interestingly, a “malignant curve pattern” is also predictive for poor outcome if FET uptake in the suspicious brain lesion is low [59, 69].
3.5 The Diagnostic Assessment of Recurrent Tumors
Early detection of recurrent tumor is of particular interest. It is difficult to distinguish recurrent glioma from nonspecific post-therapeutic changes with conventional MRI alone, because pathological contrast enhancement may reflect either re-growth of tumor or reactive changes after radio- or chemotherapy [2, 117]. Furthermore, contrast-enhancement is usually missing in recurrent low-grade gliomas and MRI cannot differentiate between tumor, edema and nonspecific treatment-related changes, unless a mass effect or distinct bloating of cortex or other grey matter structures is seen [68]. Unfortunately, most publications in the literature have evaluated the role of PET in the detection of recurrent gliomas in groups with mixed WHO grades. The potential of FDG-PET in differentiating tumor recurrence from radionecrosis in high grade gliomas is limited because of the higher frequency of non-specific uptake [118] and the performance of FLT PET is also limited [119]. Multiple studies have shown that MET-PET is highly sensitive to detect recurrent gliomas but the specificity for the differentiation of vital tumor tissue from non-neoplastic changes is not optimal and in the range of 70–75% [5, 120, 121]. The specificity of FET-PET for the differentiation of recurrent gliomas from non-neoplastic changes appears to be higher than that of MET-PET. In a study involving 45 patients (including 11 low-grade gliomas), the sensitivity and specificity of FET-PET for the detection of recurrent gliomas were 100% and 93%, respectively, compared with 93% and 50%, respectively for MRI [122]. A recent study in 124 glioma patients including 55 patients with low-grade gliomas demonstrated that the combined use of static and dynamic FET PET parameters differentiate progressive or recurrent glioma from treatment-related non neoplastic changes with an accuracy of 93% [123]. One study that focused on the role of FET PET as a diagnostic tool for detection of malignant progression in patients with low-grade gliomas reported that the combined analysis of FET PET parameters (i.e., changes of TBRmax, TTP, or time–activity curve pattern) yielded a significantly higher diagnostic accuracy for the detection of malignant progression than changes of contrast enhancement in MR imaging (accuracy, 81 vs. 63%; P = 0.003) [54]. Thus, especially FET-PET is considered as a valuable tool in differentiating recurrent tumor from non-neoplastic changes.
3.6 Monitoring of Radio- and Chemotherapy
Imaging for radiological response assessment in low-grade gliomas is based on serial measurements of T1- and T2-weighted MRI. Low-grade gliomas usually show no contrast enhancement on MRI due to an intact BBB, and the diffusely infiltrative nature of these tumors makes the assessment of tumor boundaries difficult. Changes in apparent tumor size that are seen in MRI are taken as indicators of the response to therapy but this approach is limited by the difficulty in distinguishing vital tumor tissue and unspecific treatment effects. The feasibility and usefulness of PET for treatment assessment and follow-up in cerebral gliomas of all grades of malignancy after radiotherapy and chemotherapy have been explored in multiple studies and the diagnostic accuracy compared with conventional MRI is considered to be very efficient.
The current experience concerning treatment monitoring in brain tumors with PET is based mainly on the data obtained in patients with high-grade gliomas. Several studies evaluated the role of amino acid PET using MET, FDOPA and FET in patients with high-grade gliomas to monitor external fractionated radiation therapy [112], treatment effects during standard chemotherapy regimen, i.e., adjuvant temozolomide [124] or chemotherapy with procarbazine, CCNU, and vincristine (PCV) [125], dose-intensified chemotherapy with temozolomide [126], and experimental treatment such as intracavitary radioimmunotherapy [127], convection-enhanced delivery of paclitaxel [128], tyrosine kinase inhibitor treatment [129], brachytherapy [130] and antiangiogenic treatment with bevacizumab [131,132,133].
The currently available data suggest that a reduction of the tumor-to-brain ratio of amino acid uptake and the metabolic volume of high-grade glioma is a sign of a response to treatment.
Besides cytoreductive surgery, local radiotherapy is an important treatment option especially in patients with an astrocytoma WHO grade II. The possibility of late side effects of radiotherapy (e.g., neurotoxicity) in this group of patients with a much longer life expectancy makes it necessary to identify groups that benefit from early radiotherapy compared with those in whom radiotherapy should be delayed until the time of tumor progression. The role of MET PET has been evaluated in a small number of retrospective studies, mostly in comparison to FDG PET. Roelcke and co-workers evaluated the effects of postoperative external fractionated radiotherapy using MET and FDG PET in patients with an astrocytoma WHO grade II [134]. Tracer uptake was assessed by tumor to brain ratios during follow-up and at the time of first tumor progression, and was not significantly different in patients who received external radiotherapy after tumor resection (n = 13) in comparison to patients treated with surgery alone (n = 17). Different results could be observed in brachytherapy after implantation of 125I seeds. One year after seed implantation, FDG uptake did not change in patients with low-grade glioma, but a significant decline of MET uptake was detected [135, 136] indicating that MET PET may provide more information on therapeutic effects than FDG following brachytherapy. The different results of these studies may be explained by different follow-up times and radiotherapy modalities used in the study protocols.
In order to assess the response to chemotherapy using amino acid PET in patients with low-grade gliomas FET and MET PET was used in a prospective study to evaluate the response to an intensified temozolomide regimen in a series of 33 patients with low-grade glioma. Reduction of metabolically active tumor volumes, but not reduction of PET uptake ratios or MRI tumor volumes, correlated with improved seizure control following chemotherapy. A decrease of the active tumor volume of ≥80.5% predicted a progression free survival of ≥60 months and a decrease of ≥64.5% a progression free survival of ≥48 months. [137, 138].
In patients who showed a clinical response, a reduction of the metabolically active tumor volume after initiation of treatment could be observed in FET PET earlier than volume reductions on FLAIR MRI sequences (see Fig. 15.4). In a retrospective study the effect of PCV chemotherapy was examined using MET PET in seven patients with an oligodendroglioma WHO grade II [139]. Similar to the above-mentioned study, changes of tumor volume in MRI FLAIR sequences and metabolically active tumor volume derived from MET PET were monitored. MRI-FLAIR and MET PET provided concordant information on tumor to PCV treatment, but MET PET was found to be more sensitive for the assessment of PCV responsiveness.
Patient with an oligoastrocytoma WHO grade II during chemotherapy with temozolomide. FET PET identifies response to treatment at an early stage of disease, while T2-weighted MRI remains ambiguous. With kind permission from Springer Science + Business Media: J Neurooncol: Early metabolic responses in temozolomide treated low-grade glioma patients, Vol. 95, 2009, Wyss et al. Fig. 2. Licence No. 2834741072095
The findings indicate the sensitivity of amino acid PET for detecting early treatment response in low-grade gliomas. Furthermore, the early identification of non-responders may help to minimize negative impact of chemotherapy on quality of life.
4 Perspectives for PET in the Management of Low-Grade Gliomas
Diagnostic assessment of diffuse low-grade gliomas by PET using radiolabeled amino acids permits a more specific representation of the spatial extent of the tumors than is possible by conventional MRI alone. This has been shown to be advantageous for biopsy planning, tumor resection, and radiotherapy. Valuable prognostic information can be obtained at initial diagnosis, to detect malignant transformation during follow-up and the treatment response can be judged early in the course of treatment. Recurrent tumors can be differentiated from post-therapeutic changes with a high degree of specificity. Advanced MRI methods may also yield metabolic information that is markedly more specific than that obtainable by conventional MRI [140]. A recent meta-analysis has analyzed the role of advanced MR imaging with magnetic resonance spectroscopy (MRS), perfusion weighted imaging (PWI), diffusion weighted (DWI) and diffusion tensor imaging (DTI) in the management of adults with diffuse low-grade glioma [141]. Although these techniques are established and widely available for a longer period of time there is still not enough evidence to recommend the integration of either in standard diagnostic imaging protocols. The fact that amino acid PET is widely used in centers that also have full access to the spectrum of functional and molecular MR methods, emphasizes the additional value of amino acid PET beyond alternative MRI methods. The diagnostic accuracy of these techniques in comparison with amino acid PET remains to be investigated. First studies have demonstrated the potential benefit of integrating fiber tracking by DTI and FET PET [142,143,144]. These studies indicated complementary information and more detailed understanding of peritumoral fiber tract alterations in gliomas, which are more complex as described so far.
A first comparative study of FET PET and PWI in 56 patients with cerebral glioma, showed higher TBRs and larger tumor volumes in FET PET than the maps of regional cerebral blood volume [145]. The spatial congruence of both parameters was poor and the locations of the local hot spots differed considerably. Similar results were observed in another study including 55 patients with cerebral glioma when comparing FDOPA PET and PWI [45].
The future will also be strongly influenced by the integration of PET and MRI in one imaging device [146,147,148]. The advent of hybrid PET-MRI systems offers a multimodal approach for the investigation of brain tumors and improved patient comfort due to a significant reduction in measurement time and improved spatial and temporal co-registration of PET and MRI data.
The scientifically documented utility of amino acid PET of low-grade gliomas seems to justify its introduction as a routine diagnostic technique for certain indications, but it remains to be confirmed that this will improve the overall quality of care. Initial studies have already evaluated the cost effectiveness of amino acid PET for target selection in gliomas and achieved promising results [149]. The guidelines of the European and the German Association of Nuclear Medicine for brain tumor imaging using labelled amino acid analogues have been published in recent years [150, 151]. The Response Assessment in Neuro-Oncology Working Group (RANO) which is an international effort to develop new standardized response criteria for clinical trials in brain tumors has recently recommended the use of amino acid PET in all stages of patient management, i.e., at primary diagnosis especially for the differentiation of equivocal brain lesions in MRI, after diagnosis for the definition of tumor extent for resection, biopsy and radiotherapy planning, in the early course of treatment to differentiate pseudoprogression and early tumor progression, at a later stage for the differentiation of radionecrosis and recurrent tumor and for treatment monitoring [10]. The logistical prerequisites for amino acid PET have become markedly less difficult to achieve in recent years with the introduction of FET PET. In Europe, MET PET has been replaced in many neuro-oncological centers by the more convenient PET tracer FET and the improved availability and high clinical interest in this method has led to more than 10,000 FET PET scans in some centers [152]. As the first country, Switzerland has approved FET PET for brain tumor imaging in 2014 [153].
The benefit of amino acid PET in cerebral gliomas appears to be well justified by its clinical utility since the costs of PET imaging are relatively small in relation to the expenses of local or systemic treatment approaches and, consecutively, the management of possible adverse effects. The information provided by amino acid PET assists to optimize the individual treatment strategy and to minimize negative impact of treatment approaches on quality of life.
References
Wessels PH, Weber WE, Raven G, Ramaekers FC, Hopman AH, Twijnstra A. Supratentorial grade II astrocytoma: biological features and clinical course. Lancet Neurol. 2003;2(7):395–403.
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, et al. Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol. 2010;28:1963–72.
Okada H, Weller M, Huang R, Finocchiaro G, Gilbert MR, Wick W, et al. Immunotherapy response assessment in neuro-oncology: a report of the RANO working group. Lancet Oncol. 2015;16(15):e534–42.
Chen W. Clinical applications of PET in brain tumors. J Nucl Med. 2007;48(9):1468–81.
Singhal T, Narayanan TK, Jain V, Mukherjee J, Mantil J. 11C-L-methionine positron emission tomography in the clinical management of cerebral gliomas. Mol Imaging Biol. 2008;10(1):1–18.
Smits A, Baumert BG. The clinical value of PET with amino acid tracers for gliomas WHO grade II. Int J Mol Imaging. 2011;2011:372509.
Minn H. PET and SPECT in low-grade glioma. Eur J Radiol. 2005;56(2):171–8.
Langen KJ, Tatsch K, Grosu AL, Jacobs AH, Weckesser M, Sabri O. Diagnostics of cerebral gliomas with radiolabeled amino acids. Dsches Arzteblatt Int. 2008;105(4):55–61.
Herholz K, Langen KJ, Schiepers C, Mountz JM. Brain tumors. Semin Nucl Med. 2012;42(6):356–70.
Albert NL, Weller M, Suchorska B, Galldiks N, Soffietti R, Kim MM, et al. Response Assessment in Neuro-Oncology working group and European Association for neuro-oncology recommendations for the clinical use of PET imaging in gliomas. Neuro Oncol. 2016;18(9):1199–208.
Galldiks N, Langen KJ, Pope WB. From the clinician’s point of view – what is the status quo of positron emission tomography in patients with brain tumors? Neuro-Oncology. 2015;17(11):1434–44.
Galldiks N, Langen KJ. Applications of PET imaging of neurological tumors with radiolabeled amino acids. Q J Nucl Med Mol Imaging. 2015;59(1):70–82.
Padma MV, Said S, Jacobs M, Hwang DR, Dunigan K, Satter M, et al. Prediction of pathology and survival by FDG PET in gliomas. J Neuro-Oncol. 2003;64(3):227–37.
Yoon JH, Kim JH, Kang WJ, Sohn CH, Choi SH, Yun TJ, et al. Grading of cerebral glioma with multiparametric MR imaging and 18F-FDG-PET: concordance and accuracy. Eur Radiol. 2014;24(2):380–9.
Chen W, Cloughesy T, Kamdar N, Satyamurthy N, Bergsneider M, Liau L, et al. Imaging proliferation in brain tumors with 18F-FLT PET: comparison with 18F-FDG. J Nucl Med. 2005;46(6):945–52.
Collet S, Valable S, Constans JM, Lechapt-Zalcman E, Roussel S, Delcroix N, et al. [(18)F]-fluoro-L-thymidine PET and advanced MRI for preoperative grading of gliomas. NeuroImage Clin. 2015;8:448–54.
Price SJ, Fryer TD, Cleij MC, Dean AF, Joseph J, Salvador R, et al. Imaging regional variation of cellular proliferation in gliomas using 3′-deoxy-3′-[18F]fluorothymidine positron-emission tomography: an image-guided biopsy study. Clin Radiol. 2009;64(1):52–63.
Jacobs AH, Thomas A, Kracht LW, Li H, Dittmar C, Garlip G, et al. 18F-fluoro-L-thymidine and 11C-methylmethionine as markers of increased transport and proliferation in brain tumors. J Nucl Med. 2005;46(12):1948–58.
Hatakeyama T, Kawai N, Nishiyama Y, Yamamoto Y, Sasakawa Y, Ichikawa T, et al. 11C-methionine (MET) and 18F-fluorothymidine (FLT) PET in patients with newly diagnosed glioma. Eur J Nucl Med Mol Imaging. 2008;35(11):2009–17.
Nowosielski M, DiFranco MD, Putzer D, Seiz M, Recheis W, Jacobs AH, et al. An intra-individual comparison of MRI, [18F]-FET and [18F]-FLT PET in patients with high-grade gliomas. PLoS One. 2014;9(4):e95830.
Ohtani T, Kurihara H, Ishiuchi S, Saito N, Oriuchi N, Inoue T, et al. Brain tumour imaging with carbon-11 choline: comparison with FDG PET and gadolinium-enhanced MR imaging. Eur J Nucl Med. 2001;28(11):1664–70.
Sollini M, Sghedoni R, Erba PA, Cavuto S, Froio A, De Berti G et al. Diagnostic performances of [18F]fluorocholine positron emission tomography in brain tumors. Q J Nucl Med Mol Imaging. 2015 [Epub ahead of print] PubMed PMID: 26329494.
Hatazawa J. 18F-Fluorocholine PET/CT as a complementary tool in the follow-up of low-grade glioma. Eur J Nucl Med Mol Imaging. 2015;42(6):885.
Gomez-Rio M, Testart Dardel N, Santiago Chinchilla A, Rodriguez-Fernandez A, Olivares Granados G, Luque Caro R, et al. 18F-Fluorocholine PET/CT as a complementary tool in the follow-up of low-grade glioma: diagnostic accuracy and clinical utility. Eur J Nucl Med Mol Imaging. 2015;42(6):886–95.
Koh WJ, Rasey JS, Evans ML, Grierson JR, Lewellen TK, Graham MM, et al. Imaging of hypoxia in human tumors with [F-18]fluoromisonidazole. Int J Radiat Oncol Biol Phys. 1992;22(1):199–212.
Kobayashi H, Hirata K, Yamaguchi S, Terasaka S, Shiga T, Houkin K. Usefulness of FMISO-PET for glioma analysis. Neurol Med Chir. 2013;53(11):773–8.
Toyonaga T, Hirata K, Yamaguchi S, Hatanaka KC, Yuzawa S, Manabe O, et al. F-fluoromisonidazole positron emission tomography can predict pathological necrosis of brain tumors. Eur J Nucl Med Mol Imaging. 2016.
Gerstner E, Zhang Z, Fink J, Muzi M, Hanna L, Greco E, et al. ACRIN 6684: assessment of tumor hypoxia in newly diagnosed GBM using 18F-FMISO PET and MRI. Clin Cancer Res. 2016;22(20):5079–86.
Cher LM, Murone C, Lawrentschuk N, Ramdave S, Papenfuss A, Hannah A, et al. Correlation of hypoxic cell fraction and angiogenesis with glucose metabolic rate in gliomas using 18F-fluoromisonidazole, 18F-FDG PET, and immunohistochemical studies. J Nucl Med. 2006;47(3):410–8.
Winkeler A, Boisgard R, Awde AR, Dubois A, Theze B, Zheng J, et al. The translocator protein ligand [18F]DPA-714 images glioma and activated microglia in vivo. Eur J Nucl Med Mol Imaging. 2012;39(5):811–23.
Janczar K, Su Z, Raccagni I, Anfosso A, Kelly C, Durrenberger PF, et al. The 18-kDa mitochondrial translocator protein in gliomas: from the bench to bedside. Biochem Soc Trans. 2015;43(4):579–85.
Su Z, Roncaroli F, Durrenberger PF, Coope DJ, Karabatsou K, Hinz R, et al. The 18-kDa mitochondrial translocator protein in human gliomas: an 11C-(R)PK11195 PET imaging and neuropathology study. J Nucl Med. 2015;56(4):512–7.
Roncaroli F, Su Z, Herholz K, Gerhard A, Turkheimer FE. TSPO expression in brain tumours: is TSPO a target for brain tumour imaging? Clin Transl Imaging. 2016;4:145–56.
Jensen P, Feng L, Law I, Svarer C, Knudsen GM, Mikkelsen JD, et al. TSPO imaging in glioblastoma multiforme: a direct comparison between 123I-CLINDE SPECT, 18F-FET PET, and gadolinium-enhanced MR imaging. J Nucl Med. 2015;56(9):1386–90.
Villani V, Carapella CM, Chiaravalloti A, Terrenato I, Piludu F, Vidiri A, et al. The role of PET [18F]FDOPA in evaluating low-grade glioma. Anticancer Res. 2015;35(9):5117–22.
Langen KJ, Hamacher K, Weckesser M, Floeth F, Stoffels G, Bauer D, et al. O-(2-[18F]fluoroethyl)-L-tyrosine: uptake mechanisms and clinical applications. Nucl Med Biol. 2006;33(3):287–94.
Ishiwata K, Kubota K, Murakami M, Kubota R, Sasaki T, Ishii S, et al. Re-evaluation of amino acid PET studies: can the protein synthesis rates in brain and tumor tissues be measured in vivo? J Nucl Med. 1993;34(11):1936–43.
Weber WA, Wester HJ, Grosu AL, Herz M, Dzewas B, Feldmann HJ, et al. O-(2-[18F]fluoroethyl)-L-tyrosine and L-[methyl-11C]methionine uptake in brain tumours: initial results of a comparative study. Eur J Nucl Med. 2000;27(5):542–9.
Langen KJ, Jarosch M, Muhlensiepen H, Hamacher K, Broer S, Jansen P, et al. Comparison of fluorotyrosines and methionine uptake in F98 rat gliomas. Nucl Med Biol. 2003;30(5):501–8.
Grosu AL, Astner ST, Riedel E, Nieder C, Wiedenmann N, Heinemann F, et al. An interindividual comparison of O-(2-[18F]Fluoroethyl)-L-tyrosine (FET)- and L-[methyl-11C]methionine (MET)-PET in patients with brain gliomas and metastases. Int J Radiat Oncol Biol Phys. 2011;81(4):1049–58.
Becherer A, Karanikas G, Szabo M, Zettinig G, Asenbaum S, Marosi C, et al. Brain tumour imaging with PET: a comparison between [18F]fluorodopa and [11C]methionine. Eur J Nucl Med Mol Imaging. 2003;30(11):1561–7.
Lapa C, Linsenmann T, Monoranu CM, Samnick S, Buck AK, Bluemel C, et al. Comparison of the amino acid tracers 18F-FET and 18F-DOPA in high-grade glioma patients. J Nucl Med. 2014;55(10):1611–6.
Kratochwil C, Combs SE, Leotta K, Afshar-Oromieh A, Rieken S, Debus J, et al. Intra-individual comparison of 18F-FET and 18F-DOPA in PET imaging of recurrent brain tumors. Neuro-Oncology. 2014;16(3):434–40.
Sioka C, Fotopoulos A, Kyritsis AP. Recent advances in PET imaging for evaluation of Parkinson’s disease. Eur J Nucl Med Mol Imaging. 2010;37(8):1594–603.
Cicone F, Filss CP, Minniti G, Rossi-Espagnet C, Papa A, Scaringi C, et al. Volumetric assessment of recurrent or progressive gliomas: comparison between F-DOPA PET and perfusion-weighted MRI. Eur J Nucl Med Mol Imaging. 2015;42(6):905–15.
Wiriyasermkul P, Nagamori S, Tominaga H, Oriuchi N, Kaira K, Nakao H, et al. Transport of 3-fluoro-L-alpha-methyl-tyrosine by tumor-upregulated L-type amino acid transporter 1: a cause of the tumor uptake in PET. J Nucl Med. 2012;53(8):1253–61.
Youland RS, Kitange GJ, Peterson TE, Pafundi DH, Ramiscal JA, Pokorny JL, et al. The role of LAT1 in 18F-DOPA uptake in malignant gliomas. J Neuro-Oncol. 2013;111(1):11–8.
Habermeier A, Graf J, Sandhofer BF, Boissel JP, Roesch F, Closs EI. System L amino acid transporter LAT1 accumulates O-(2-fluoroethyl)-L-tyrosine (FET). Amino Acids. 2015;47(2):335–44.
Barollo S, Bertazza L, Watutantrige-Fernando S, Censi S, Cavedon E, Galuppini F, et al. Overexpression of L-type amino acid transporter 1 (LAT1) and 2 (LAT2): novel markers of neuroendocrine tumors. PLoS One. 2016;11(5):e0156044.
Okubo S, Zhen HN, Kawai N, Nishiyama Y, Haba R, Tamiya T. Correlation of L-methyl-11C-methionine (MET) uptake with L-type amino acid transporter 1 in human gliomas. J Neuro-Oncol. 2010;99(2):217–25.
Thon N, Kunz M, Lemke L, Jansen NL, Eigenbrod S, Kreth S, et al. Dynamic 18F-FET PET in suspected WHO grade II gliomas defines distinct biological subgroups with different clinical courses. Int J Cancer. 2015;136(9):2132–45.
Popperl G, Kreth FW, Mehrkens JH, Herms J, Seelos K, Koch W, et al. FET PET for the evaluation of untreated gliomas: correlation of FET uptake and uptake kinetics with tumour grading. Eur J Nucl Med Mol Imaging. 2007;34(12):1933–42.
Calcagni ML, Galli G, Giordano A, Taralli S, Anile C, Niesen A, et al. Dynamic O-(2-[18F]fluoroethyl)-L-tyrosine (F-18 FET) PET for glioma grading: assessment of individual probability of malignancy. Clin Nucl Med. 2011;36(10):841–7.
Galldiks N, Stoffels G, Ruge MI, Rapp M, Sabel M, Reifenberger G, et al. Role of O-(2-18F-fluoroethyl)-L-tyrosine PET as a diagnostic tool for detection of malignant progression in patients with low-grade glioma. J Nucl Med. 2013;54(12):2046–54.
Weckesser M, Langen KJ, Rickert CH, Kloska S, Straeter R, Hamacher K, et al. O-(2-[18F]fluorethyl)-L-tyrosine PET in the clinical evaluation of primary brain tumours. Eur J Nucl Med Mol Imaging. 2005;32(4):422–9.
Moulin-Romsee G, D’Hondt E, de Groot T, Goffin J, Sciot R, Mortelmans L, et al. Non-invasive grading of brain tumours using dynamic amino acid PET imaging: does it work for 11C-methionine? Eur J Nucl Med Mol Imaging. 2007;34(12):2082–7.
Floeth FW, Pauleit D, Sabel M, Stoffels G, Reifenberger G, Riemenschneider MJ, et al. Prognostic value of O-(2-18F-fluoroethyl)-L-tyrosine PET and MRI in low-grade glioma. J Nucl Med. 2007;48(4):519–27.
Rapp M, Heinzel A, Galldiks N, Stoffels G, Felsberg J, Ewelt C, et al. Diagnostic performance of 18F-FET PET in newly diagnosed cerebral lesions suggestive of glioma. J Nucl Med. 2013;54(2):229–35.
Jansen NL, Graute V, Armbruster L, Suchorska B, Lutz J, Eigenbrod S, et al. MRI-suspected low-grade glioma: is there a need to perform dynamic FET PET? Eur J Nucl Med Mol Imaging. 2012;39(6):1021–9.
Bette S, Gempt J, Delbridge C, Kirschke JS, Schlegel J, Foerster S, et al. Prognostic value of O-(2-[18F]-Fluoroethyl)-L-tyrosine-positron emission tomography imaging for histopathologic characteristics and progression-free survival in patients with low-grade glioma. World Neurosurg. 2016;89:230–9.
Wester HJ, Herz M, Weber W, Heiss P, Senekowitsch-Schmidtke R, Schwaiger M, et al. Synthesis and radiopharmacology of O-(2-[18F]fluoroethyl)-L-tyrosine for tumor imaging. J Nucl Med. 1999;40(1):205–12.
Hamacher K, Coenen HH. Efficient routine production of the 18F-labelled amino acid O-2-18F fluoroethyl-L-tyrosine. Appl Radiat Isot. 2002;57(6):853–6.
Pichler R, Dunzinger A, Wurm G, Pichler J, Weis S, Nussbaumer K, et al. Is there a place for FET PET in the initial evaluation of brain lesions with unknown significance? Eur J Nucl Med Mol Imaging. 2010;37(8):1521–8.
Sala Q, Metellus P, Taieb D, Kaphan E, Figarella-Branger D, Guedj E. 18F-DOPA, a clinically available PET tracer to study brain inflammation? Clin Nucl Med. 2014;39(4):e283–5.
Hutterer M, Nowosielski M, Putzer D, Jansen NL, Seiz M, Schocke M, et al. [18F]-fluoro-ethyl-L-tyrosine PET: a valuable diagnostic tool in neuro-oncology, but not all that glitters is glioma. Neuro-Oncology. 2013;15(3):341–51.
Dunet V, Rossier C, Buck A, Stupp R, Prior JO. Performance of 18F-fluoro-ethyl-tyrosine (18F-FET) PET for the differential diagnosis of primary brain tumor: a systematic review and metaanalysis. J Nucl Med. 2012;53(2):207–14.
Pauleit D, Stoffels G, Bachofner A, Floeth FW, Sabel M, Herzog H, et al. Comparison of 18F-FET and (18)F-FDG PET in brain tumors. Nucl Med Biol. 2009;36(7):779–87.
Herholz K, Holzer T, Bauer B, Schroder R, Voges J, Ernestus RI, et al. 11C-methionine PET for differential diagnosis of low-grade gliomas. Neurology. 1998;50(5):1316–22.
Unterrainer M, Schweisthal F, Suchorska B, Wenter V, Schmid-Tannwald C, Fendler WP, et al. Serial 18F-FET PET imaging of primarily 18F-FET-negative glioma – does it make sense? J Nucl Med. 2016.
Salber D, Stoffels G, Oros-Peusquens AM, Shah NJ, Reifenberger G, Hamacher K, et al. Comparison of O-(2-18F-fluoroethyl)-L-tyrosine and L-3H-methionine uptake in cerebral hematomas. J Nucl Med. 2010;51(5):790–7.
Salber D, Stoffels G, Pauleit D, Oros-Peusquens AM, Shah NJ, Klauth P, et al. Differential uptake of O-(2-18F-fluoroethyl)-L-tyrosine, L-3H-methionine, and 3H-deoxyglucose in brain abscesses. J Nucl Med. 2007;48(12):2056–62.
Salber D, Stoffels G, Pauleit D, Reifenberger G, Sabel M, Shah NJ, et al. Differential uptake of [18F]FET and [3H]l-methionine in focal cortical ischemia. Nucl Med Biol. 2006;33(8):1029–35.
Floeth FW, Pauleit D, Sabel M, Reifenberger G, Stoffels G, Stummer W, et al. 18F-FET PET differentiation of ring-enhancing brain lesions. J Nucl Med. 2006;47(5):776–82.
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, et al. The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114(2):97–109.
Manabe O, Hattori N, Yamaguchi S, Hirata K, Kobayashi K, Terasaka S, et al. Oligodendroglial component complicates the prediction of tumour grading with metabolic imaging. Eur J Nucl Med Mol Imaging. 2015;42(6):896–904.
Shinozaki N, Uchino Y, Yoshikawa K, Matsutani T, Hasegawa A, Saeki N, et al. Discrimination between low-grade oligodendrogliomas and diffuse astrocytoma with the aid of 11C-methionine positron emission tomography. J Neurosurg. 2011;114(6):1640–7.
Kracht LW, Friese M, Herholz K, Schroeder R, Bauer B, Jacobs A, et al. Methyl-[11C]- l-methionine uptake as measured by positron emission tomography correlates to microvessel density in patients with glioma. Eur J Nucl Med Mol Imaging. 2003;30(6):868–73.
Stockhammer F, Plotkin M, Amthauer H, van Landeghem FK, Woiciechowsky C. Correlation of F-18-fluoro-ethyl-tyrosin uptake with vascular and cell density in non-contrast-enhancing gliomas. J Neuro-Oncol. 2008;88(2):205–10.
Okita Y, Kinoshita M, Goto T, Kagawa N, Kishima H, Shimosegawa E, et al. (11)C-methionine uptake correlates with tumor cell density rather than with microvessel density in glioma: a stereotactic image-histology comparison. NeuroImage. 2010;49(4):2977–82.
Stiver SI. Angiogenesis and its role in the behavior of astrocytic brain tumors. Front Biosci. 2004;9:3105–23.
Goldman S, Levivier M, Pirotte B, Brucher JM, Wikler D, Damhaut P, et al. Regional methionine and glucose uptake in high-grade gliomas: a comparative study on PET-guided stereotactic biopsy. J Nucl Med. 1997;38(9):1459–62.
Pirotte B, Goldman S, Massager N, David P, Wikler D, Vandesteene A, et al. Comparison of 18F-FDG and 11C-methionine for PET-guided stereotactic brain biopsy of gliomas. J Nucl Med. 2004;45(8):1293–8.
Pirotte B, Goldman S, Massager N, David P, Wikler D, Lipszyc M, et al. Combined use of 18F-fluorodeoxyglucose and 11C-methionine in 45 positron emission tomography-guided stereotactic brain biopsies. J Neurosurg. 2004;101(3):476–83.
Kunz M, Thon N, Eigenbrod S, Hartmann C, Egensperger R, Herms J, et al. Hot spots in dynamic 18FET-PET delineate malignant tumor parts within suspected WHO grade II gliomas. Neuro-Oncology. 2011;13(3):307–16.
Mosskin M, Ericson K, Hindmarsh T, von Holst H, Collins VP, Bergstrom M, et al. Positron emission tomography compared with magnetic resonance imaging and computed tomography in supratentorial gliomas using multiple stereotactic biopsies as reference. Acta Radiol. 1989;30(3):225–32.
Kracht LW, Miletic H, Busch S, Jacobs AH, Voges J, Hoevels M, et al. Delineation of brain tumor extent with [11C]L-methionine positron emission tomography: local comparison with stereotactic histopathology. Clin Cancer Res. 2004;10(21):7163–70.
Pauleit D, Floeth F, Hamacher K, Riemenschneider MJ, Reifenberger G, Muller HW, et al. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain J Neurol. 2005;128(Pt 3):678–87.
Lopez WO, Cordeiro JG, Albicker U, Doostkam S, Nikkhah G, Kirch RD, et al. Correlation of 18F-fluoroethyl tyrosine positron-emission tomography uptake values and histomorphological findings by stereotactic serial biopsy in newly diagnosed brain tumors using a refined software tool. OncoTargets Ther. 2015;8:3803–15.
Floeth FW, Sabel M, Ewelt C, Stummer W, Felsberg J, Reifenberger G, et al. Comparison of 18F-FET PET and 5-ALA fluorescence in cerebral gliomas. Eur J Nucl Med Mol Imaging. 2011;38(4):731–41.
Pirotte B, Goldman S, Dewitte O, Massager N, Wikler D, Lefranc F, et al. Integrated positron emission tomography and magnetic resonance imaging-guided resection of brain tumors: a report of 103 consecutive procedures. J Neurosurg. 2006;104(2):238–53.
Arbizu J, Tejada S, Marti-Climent JM, Diez-Valle R, Prieto E, Quincoces G, et al. Quantitative volumetric analysis of gliomas with sequential MRI and 11C-methionine PET assessment: patterns of integration in therapy planning. Eur J Nucl Med Mol Imaging. 2012;39(5):771–81.
Buchmann N, Klasner B, Gempt J, Bauer JS, Pyka T, Delbridge C, et al. 18F-Fluoroethyl-l-thyrosine positron emission tomography to delineate tumor residuals after glioblastoma resection: a comparison with standard postoperative magnetic resonance imaging. World Neurosurg. 2016;89:420–6.
Klasner B, Buchmann N, Gempt J, Ringel F, Lapa C, Krause BJ. Early [18F]FET-PET in gliomas after surgical resection: comparison with MRI and histopathology. PLoS One. 2015;10(10):e0141153.
Grosu AL, Weber WA. PET for radiation treatment planning of brain tumours. Radiother Oncol. 2010;96(3):325–7.
Levivier M, Massager N, Wikler D, Lorenzoni J, Ruiz S, Devriendt D, et al. Use of stereotactic PET images in dosimetry planning of radiosurgery for brain tumors: clinical experience and proposed classification. J Nucl Med. 2004;45(7):1146–54.
Grosu AL, Weber WA, Franz M, Stark S, Piert M, Thamm R, et al. Reirradiation of recurrent high-grade gliomas using amino acid PET (SPECT)/CT/MRI image fusion to determine gross tumor volume for stereotactic fractionated radiotherapy. Int J Radiat Oncol Biol Phys. 2005;63(2):511–9.
Rickhey M, Koelbl O, Eilles C, Bogner L. A biologically adapted dose-escalation approach, demonstrated for 18F-FET-PET in brain tumors. Strahlenther Onkol. 2008;184(10):536–42.
Weber DC, Zilli T, Buchegger F, Casanova N, Haller G, Rouzaud M, et al. [18F]Fluoroethyltyrosine- positron emission tomography-guided radiotherapy for high-grade glioma. Radiat Oncol. 2008;3:44.
Piroth MD, Pinkawa M, Holy R, Stoffels G, Demirel C, Attieh C, et al. Integrated-boost IMRT or 3-D-CRT using FET-PET based auto-contoured target volume delineation for glioblastoma multiforme—a dosimetric comparison. Radiat Oncol. 2009;4:57.
Munck Af Rosenschold P, Costa J, Engelholm SA, Lundemann MJ, Law I, Ohlhues L, et al. Impact of [18F]-fluoro-ethyl-tyrosine PET imaging on target definition for radiation therapy of high-grade glioma. Neuro-Oncology. 2015;17(5):757–63.
Rieken S, Habermehl D, Giesel FL, Hoffmann C, Burger U, Rief H, et al. Analysis of FET-PET imaging for target volume definition in patients with gliomas treated with conformal radiotherapy. Radiother Oncol. 2013;109(3):487–92.
Piroth MD, Pinkawa M, Holy R, Klotz J, Schaar S, Stoffels G, et al. Integrated boost IMRT with FET-PET-adapted local dose escalation in glioblastomas. Results of a prospective phase II study. Strahlenther Onkol. 2012;188(4):334–9.
Kosztyla R, Chan EK, Hsu F, Wilson D, Ma R, Cheung A, et al. High-grade glioma radiation therapy target volumes and patterns of failure obtained from magnetic resonance imaging and 18F-FDOPA positron emission tomography delineations from multiple observers. Int J Radiat Oncol Biol Phys. 2013;87(5):1100–6.
Nuutinen J, Sonninen P, Lehikoinen P, Sutinen E, Valavaara R, Eronen E, et al. Radiotherapy treatment planning and long-term follow-up with [(11)C]methionine PET in patients with low-grade astrocytoma. Int J Radiat Oncol Biol Phys. 2000;48(1):43–52.
Dunet V, Pomoni A, Hottinger A, Nicod-Lalonde M, Prior JO. Performance of 18F-FET versus 18F-FDG-PET for the diagnosis and grading of brain tumors: systematic review and meta-analysis. Neuro-Oncology. 2016;18(3):426–34.
Janvier L, Olivier P, Blonski M, Morel O, Vignaud JM, Karcher G, et al. Correlation of SUV-derived indices with tumoral aggressiveness of gliomas in static 18F-FDOPA PET: use in clinical practice. Clin Nucl Med. 2015;40(9):e429–35.
Pöpperl G, Kreth FW, Herms J, Koch W, Mehrkens JH, Gildehaus FJ, et al. Analysis of 18F-FET PET for grading of recurrent gliomas: is evaluation of uptake kinetics superior to standard methods? J Nucl Med. 2006;47(3):393–403.
Albert NL, Winkelmann I, Suchorska B, Wenter V, Schmid-Tannwald C, Mille E, et al. Early static 18F-FET-PET scans have a higher accuracy for glioma grading than the standard 20–40 min scans. Eur J Nucl Med Mol Imaging. 2016;43(6):1105–14.
Kaschten B, Stevenaert A, Sadzot B, Deprez M, Degueldre C, Del Fiore G, et al. Preoperative evaluation of 54 gliomas by PET with fluorine-18-fluorodeoxyglucose and/or carbon-11-methionine. J Nucl Med. 1998;39(5):778–85.
Jansen NL, Suchorska B, Wenter V, Schmid-Tannwald C, Todica A, Eigenbrod S, et al. Prognostic significance of dynamic 18F-FET PET in newly diagnosed astrocytic high-grade glioma. J Nucl Med. 2015;56(1):9–15.
Suchorska B, Jansen NL, Linn J, Kretzschmar H, Janssen H, Eigenbrod S, et al. Biological tumor volume in 18FET-PET before radiochemotherapy correlates with survival in GBM. Neurology. 2015;84(7):710–9.
Piroth MD, Pinkawa M, Holy R, Klotz J, Nussen S, Stoffels G, et al. Prognostic value of early [18F]fluoroethyltyrosine positron emission tomography after radiochemotherapy in glioblastoma multiforme. Int J Radiat Oncol Biol Phys. 2011;80(1):176–84.
Pyka T, Gempt J, Hiob D, Ringel F, Schlegel J, Bette S, et al. Textural analysis of pre-therapeutic [18F]-FET-PET and its correlation with tumor grade and patient survival in high-grade gliomas. Eur J Nucl Med Mol Imaging. 2016;43(1):133–41.
Ribom D, Eriksson A, Hartman M, Engler H, Nilsson A, Langstrom B, et al. Positron emission tomography 11C-methionine and survival in patients with low-grade gliomas. Cancer. 2001;92(6):1541–9.
De Witte O, Goldberg I, Wikler D, Rorive S, Damhaut P, Monclus M, et al. Positron emission tomography with injection of methionine as a prognostic factor in glioma. J Neurosurg. 2001;95(5):746–50.
Pyka T, Gempt J, Ringel F, Huttinger S, van Marwick S, Nekolla S, et al. Prediction of glioma recurrence using dynamic 18F-fluoroethyltyrosine PET. AJNR Am J Neuroradiol. 2014;35(10):1924–9.
Brandsma D, van den Bent MJ. Pseudoprogression and pseudoresponse in the treatment of gliomas. Curr Opin Neurol. 2009;22(6):633–8.
Ricci PE, Karis JP, Heiserman JE, Fram EK, Bice AN, Drayer BP. Differentiating recurrent tumor from radiation necrosis: time for re-evaluation of positron emission tomography? AJNR Am J Neuroradiol. 1998;19(3):407–13.
Li Z, Yu Y, Zhang H, Xu G, Chen L. A meta-analysis comparing 18F-FLT PET with 18F-FDG PET for assessment of brain tumor recurrence. Nucl Med Commun. 2015;36(7):695–701.
Terakawa Y, Tsuyuguchi N, Iwai Y, Yamanaka K, Higashiyama S, Takami T, et al. Diagnostic accuracy of 11C-methionine PET for differentiation of recurrent brain tumors from radiation necrosis after radiotherapy. J Nucl Med. 2008;49(5):694–9.
Nihashi T, Dahabreh IJ, Terasawa T. Diagnostic accuracy of PET for recurrent glioma diagnosis: a meta-analysis. AJNR Am J Neuroradiol. 2013;34(5):944–50. S1-11
Rachinger W, Goetz C, Popperl G, Gildehaus FJ, Kreth FW, Holtmannspotter M, et al. Positron emission tomography with O-(2-[18F]fluoroethyl)-l-tyrosine versus magnetic resonance imaging in the diagnosis of recurrent gliomas. Neurosurgery. 2005;57(3):505–11.
Galldiks N, Stoffels G, Filss C, Rapp M, Blau T, Tscherpel C, et al. The use of dynamic O-(2-18F-fluoroethyl)-l-tyrosine PET in the diagnosis of patients with progressive and recurrent glioma. Neuro-Oncology. 2015;17(9):1293–300.
Galldiks N, Kracht LW, Burghaus L, Thomas A, Jacobs AH, Heiss WD, et al. Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur J Nucl Med Mol Imaging. 2006;33(5):516–24.
Herholz K, Kracht LW, Heiss WD. Monitoring the effect of chemotherapy in a mixed glioma by C-11-methionine PET. J Neuroimaging. 2003;13(3):269–71.
Galldiks N, Kracht LW, Burghaus L, Ullrich RT, Backes H, Brunn A, et al. Patient-tailored, imaging-guided, long-term temozolomide chemotherapy in patients with glioblastoma. Mol Imaging. 2010;9:40–6.
Popperl G, Gotz C, Rachinger W, Schnell O, Gildehaus FJ, Tonn JC, et al. Serial O-(2-[18F]fluoroethyl)-L: -tyrosine PET for monitoring the effects of intracavitary radioimmunotherapy in patients with malignant glioma. Eur J Nucl Med Mol Imaging. 2006;33(7):792–800.
Popperl G, Goldbrunner R, Gildehaus FJ, Kreth FW, Tanner P, Holtmannspotter M, et al. O-(2-[18F]fluoroethyl)-L-tyrosine PET for monitoring the effects of convection-enhanced delivery of paclitaxel in patients with recurrent glioblastoma. Eur J Nucl Med Mol Imaging. 2005;32(9):1018–25.
Galldiks N, Ullrich R, Schroeter M, Fink GR, Kracht LW. Imaging biological activity of a glioblastoma treated with an individual patient-tailored, experimental therapy regimen. J Neuro-Oncol. 2009;93:425–30.
Jansen NL, Suchorska B, Schwarz SB, Eigenbrod S, Lutz J, Graute V, et al. [18F]fluoroethyltyrosine-positron emission tomography-based therapy monitoring after stereotactic iodine-125 brachytherapy in patients with recurrent high-grade glioma. Mol Imaging. 2013;12(3):137–47.
Hutterer M, Nowosielski M, Putzer D, Waitz D, Tinkhauser G, Kostron H, et al. O-(2-18F-fluoroethyl)-L-tyrosine PET predicts failure of antiangiogenic treatment in patients with recurrent high-grade glioma. J Nucl Med. 2011;52(6):856–64.
Galldiks N, Rapp M, Stoffels G, Fink GR, Shah NJ, Coenen HH, et al. Response assessment of bevacizumab in patients with recurrent malignant glioma using [18F]Fluoroethyl-L-tyrosine PET in comparison to MRI. Eur J Nucl Med Mol Imaging. 2013;40(1):22–33.
Schwarzenberg J, Czernin J, Cloughesy TF, Ellingson BM, Pope WB, Grogan T, et al. Treatment response evaluation using 18F-FDOPA PET in patients with recurrent malignant glioma on bevacizumab therapy. Clin Cancer Res. 2014;20(13):3550–9.
Roelcke U, von Ammon K, Hausmann O, Kaech DL, Vanloffeld W, Landolt H, et al. Operated low grade astrocytomas: a long term PET study on the effect of radiotherapy. J Neurol Neuosurg Psychiatry. 1999;66:644–7.
Voges J, Herholz K, Holzer T, Würker M, Bauer B, Pietrzyk U, et al. 11C-methionine and 18F-2-fluorodeoxyglucose positron emission tomography: a tool for diagnosis of cerebral glioma and monitoring after brachytherapy with 125I seeds. Stereotact Funct Neurosurg. 1997;69:129–35.
Würker M, Herholz K, Voges J, Pietrzyk U, Treuer H, Bauer B, et al. Glucose consumption and methionine uptake in low-grade gliomas after iodine-125 brachytherapy. Eur J Nucl Med. 1996;23:583–6.
Wyss M, Hofer S, Bruehlmeier M, Hefti M, Uhlmann C, Bartschi E, et al. Early metabolic responses in temozolomide treated low-grade glioma patients. J Neuro-Oncol. 2009;95:87–93.
Roelcke U, Wyss MT, Nowosielski M, Ruda R, Roth P, Hofer S, et al. Amino acid positron emission tomography to monitor chemotherapy response and predict seizure control and progression-free survival in WHO grade II gliomas. Neuro-Oncology. 2015;18(5):744–51.
Tang BN, Sadeghi N, Branle F, De Witte O, Wikler D, Goldman S. Semi-quantification of methionine uptake and flair signal for the evaluation of chemotherapy in low-grade oligodendroglioma. J Neuro-Oncol. 2005;71:161–8.
Herholz K, Coope D, Jackson A. Metabolic and molecular imaging in neuro-oncology. Lancet Neurol. 2007;6(8):711–24.
Fouke SJ, Benzinger T, Gibson D, Ryken TC, Kalkanis SN, Olson JJ. The role of imaging in the management of adults with diffuse low grade glioma: a systematic review and evidence-based clinical practice guideline. J Neuro-Oncol. 2015;125(3):457–79.
Stadlbauer A, Polking E, Prante O, Nimsky C, Buchfelder M, Kuwert T, et al. Detection of tumour invasion into the pyramidal tract in glioma patients with sensorimotor deficits by correlation of 18F-fluoroethyl-L: -tyrosine PET and magnetic resonance diffusion tensor imaging. Acta Neurochir. 2009;151(9):1061–9.
Stadlbauer A, Hammen T, Grummich P, Buchfelder M, Kuwert T, Dorfler A, et al. Classification of peritumoral fiber tract alterations in gliomas using metabolic and structural neuroimaging. J Nucl Med. 2011;52(8):1227–34.
Neuschmelting V, Weiss Lucas C, Stoffels G, Oros-Peusquens AM, Lockau H, Shah NJ, et al. Multimodal imaging in malignant brain tumors: enhancing the preoperative risk evaluation for motor deficits with a combined hybrid MRI-PET and navigated transcranial magnetic stimulation approach. AJNR Am J Neuroradiol. 2016;37(2):266–73.
Filss CP, Galldiks N, Stoffels G, Sabel M, Wittsack HJ, Turowski B, et al. Comparison of 18F-FET PET and perfusion-weighted MR imaging: a PET/MR imaging hybrid study in patients with brain tumors. J Nucl Med. 2014;55(4):540–5.
Herzog H, Langen KJ, Weirich C, Rota Kops E, Kaffanke J, Tellmann L, et al. High resolution brain PET combined with simultaneous MRI. Nuklearmedizin Nucl Med. 2011;50(2):74–82.
Shah NJ, Oros-Peusquens AM, Arrubla J, Zhang K, Warbrick T, Mauler J, et al. Advances in multimodal neuroimaging: hybrid MR-PET and MR-PET-EEG at 3 T and 9.4 T. J Magn Reson. 2013;229:101–15.
Neuner I, Kaffanke JB, Langen KJ, Kops ER, Tellmann L, Stoffels G, et al. Multimodal imaging utilising integrated MR-PET for human brain tumour assessment. Eur Radiol. 2012;22(12):2568–80.
Heinzel A, Stock S, Langen KJ, Muller D. Cost-effectiveness analysis of FET PET-guided target selection for the diagnosis of gliomas. Eur J Nucl Med Mol Imaging. 2012;39(7):1089–96.
Vander Borght T, Asenbaum S, Bartenstein P, Halldin C, Kapucu O, Van Laere K, et al. EANM procedure guidelines for brain tumour imaging using labelled amino acid analogues. Eur J Nucl Med Mol Imaging. 2006;33(11):1374–80.
Langen KJ, Bartenstein P, Boecker H, Brust P, Coenen HH, Drzezga A, et al. German guidelines for brain tumour imaging by PET and SPECT using labelled amino acids. Nuklearmedizin Nucl Med. 2011;50(4):167–73.
Langen KJ, Tonn JC, Weller M, Galldiks N. Letter to the Editor: “The role of imaging in the management of progressive glioblastoma. A systematic review and evidence-based clinical practice guideline” [J. Neurooncol. 2014; 118: 435–460]. J Neuro-Oncol. 2014;120(3):665–6.
Swissmedic. Swiss agency for therapeutic products. J Swissmedic. 2014;13:651.
Misch M, Guggemos A, Driever PH, Koch A, Grosse F, Steffen IG, et al. 18F-FET-PET guided surgical biopsy and resection in children and adolescence with brain tumors. Child’s Nerv Syst. 2015;31(2):261–7.
Ribom D, Smits A. Baseline 11C-methionine PET reflects the natural course of grade 2 oligodendrogliomas. Neurol Res. 2005;27(5):516–21.
Pirotte B, Levivier M, Morelli D, Van Bogaert P, Detemmerman D, David P, et al. Positron emission tomography for the early postsurgical evaluation of pediatric brain tumors. Child’s Nerv Syst. 2005;21(4):294–300.
Popperl G, Gotz C, Rachinger W, Gildehaus FJ, Tonn JC, Tatsch K. Value of O-(2-[18F]fluoroethyl)-L-tyrosine PET for the diagnosis of recurrent glioma. Eur J Nucl Med Mol Imaging. 2004;31(11):1464–70.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer-Verlag London Ltd.
About this chapter
Cite this chapter
Langen, KJ., Rapp, M., Sabel, M., Galldiks, N. (2017). Positron-Emission-Tomography in Diffuse Low-Grade Gliomas. In: Duffau, H. (eds) Diffuse Low-Grade Gliomas in Adults. Springer, Cham. https://doi.org/10.1007/978-3-319-55466-2_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-55466-2_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-55464-8
Online ISBN: 978-3-319-55466-2
eBook Packages: MedicineMedicine (R0)