Abstract
The regulation of tendon metabolism including the responses to loading is far from being well understood. During the last decade, however, accumulating data show that tendon innervation in addition to afferent functions, via efferent pathways has a regulatory role in tendon homeostasis via a wide range of neuromediators, which coordinate metabolic and neuro-inflammatory pathways.
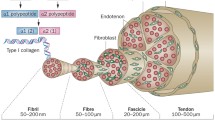
Innervation of intact healthy tendons is localized in the surrounding structures, i.e paratenon, endotenon and epitenon, whereas the tendon proper is practically devoid of neuronal supply. This anatomical finding reflects that the tendon metabolism is regulated from the tendon envelope, i.e. interfascicular matrix (see Chap. 1).
Tendon innervation after injury and during repair, however, is found as extensive nerve ingrowth into the tendon proper, followed by a time-dependent emergence of different neuronal mediators, which amplify and fine-tune inflammatory and metabolic pathways in tendon regeneration. After healing nerve fibers retract to the tendon envelope.
In tendinopathy innervation has been identified to consist of excessive and protracted nerve ingrowth in the tendon proper, suggesting pro-inflammatory, nociceptive and hypertrophic (degenerative) tissue responses.
In metabolic disorders such as eg. diabetes impaired tendon healing has been established to be related to dysregulation of neuronal growth factors.
Targeted approaches to the peripheral nervous system including neuronal mediators and their receptors may prove to be effective therapies for painful, degenerative and traumatic tendon disorders.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Tendon homeostasis is the balance between formation and res orption [1]. Mechanical loading is the most powerf ul and well-k nown extrinsic factor to regulate tendon protein synthesis and degradation [1]. While loading over time leads to a net gain in collagen repeated loading, however, exceeding the tendons capacity of new collagen formation is harmful and may trigger the development of tendinopathy.
Prolonged unloading may additionally be detrimental for the human tendon. Disuse leads to reduced tendon mechanical stiffness [2]. Prolonged unloading post injury also demonstrated negative effects on tendon mechanical properties and production of extracellular matrix molecules [3–5].
Although it is well known both clinically and experimentally that loading improves while unloading impairs tendon protein synthesis, the exact mechanisms and regulatory factors responsible for this mechano-biological transduction are still not fully known. In spite of this gap in knowledge, accumulating data do suggest that the peripheral nervous system including specific mediators and their receptors play an important role in tendon homeostasis and repair as well as in tendinopathy [6].
Innervation of Tendons
Generally, tendons exhibit a low degree of innervation, which may partly explain the slow adaptation to repetitive loading, prolonged healing and vulnerability to chronic injuries [7, 8] (Fig. 4.1). The innervation of tendons originates from neighbouring muscular, cutaneous and peritendinous nerve trunks [9]. From the myotendinous junction nerve fibres cross and enter the endotenon septa. In the paratenon, nerve fibres form rich plexuses and send small branches that penetrate the epitenon. Nerve fibres do not under normal conditions enter the tendon proper, but terminate as nerve endings on the different surfaces of the tendon (paratenon, epitenon, endotenon) [10].
(a–c). Overview of several arranged micrographs of longitudinal sections through the Achilles tendon after incubation with antisera to general nerve marker PGP 9.5. Micrographs depict the proximal half of the Achilles tendon at increasing magnification in figures (a–c). Arrows denote varicosities and nerve terminals. The typical vascular localisation of NPY is depicted in lower left (b), whereas the free nerve endings are typical localisation of SP (c). The immunoreactivity is seen in the paratenon and surrounding loose connective tissue, whereas the proper tensinous tissue, notably, is almost devoid of nerve fibers pt = paratenon (Reproduced with permission from Ackermann et al. [32])
The nerves innervating tendons are composed of a low degree of myelinated, fast transmitting Aα- and Aβ-fibres and a higher degree of unmyelinated, slow transmitting Aγ-, Aδ-, B- and C-fibres [8, 11].
The nerve endings of (1) Aα- and Aβ-fibres are of types I-III and mediate mechanoception. These include Type I or Ruffini corpuscles (pressure and stretching sensors), II or Vater-Pacini corpuscles (pressure sensors, reacting to acceleration and deceleration of movement), type III or Golgi tendon organs (tension receptors) [12, 13]. Tension receptors (Type III) have been found mostly in the myotendinous junction and insertion areas [13].
The nerve endings of (2) Aγ-, Aδ-, and C-fibres are of type IVa, so called nociceptors, mediating deep tissue pain and hyperalgesia that are characteristic features of pain in tendinopathy. The nerve endings of B-fibres, which are autonomic, consist of type IVb fibres that are mainly localised in the walls of small arteries, arterioles, capillaries, and postcapillary veins exerting vasomotor actions [13].
In addition to these classical afferent functions it is now well established that the peripheral nervous system also participates in the efferent regulation of a wide variety of efferent physiological responses, including actions on cell proliferation, expression of cytokines and growth factors, inflammation, immune responses and hormone release. In the last decade, further characterization of tendon innervation related to mediator phenotypes and neuronal regulation of tendon homeostasis has received increasing attention.
In addition to classical neurotransmitters (monoamines, acetylcholine, amino acids) several neuropeptides, which act as chemical messengers in the central and peripheral nervous system, have been identified in tendons [14]. Neuropeptides differ from classical neurotransmitters in several respects [8, 15] since they generally exhibit more long-term effects than the classical neurotransmitters.
The effects of neuropeptides and classical transmitters are also elicited by different receptor mechanisms. While classical transmitters act on ligand-gated ion channels, neuropeptides act by binding to specific plasma membrane receptors, called G-protein coupled receptors [16]. Several G-protein coupled receptors have been identified in tendons and when stimulated they generate various second messengers, which can trigger a wide range of effector mechanisms regulating cellular excitability and function [14] (Table 4.1).
Neuromediators in Healthy Tendon
In general, tendons seem to exhibit neuronal mediators of a similar variety as observed in other organs of the body including those of various sensory including opioid, autonomic, and excitatory neuromediators (Table 4.1). In contrast to most other tissues, however, the tendon proper during normal conditions is devoid of nerve fibers. Innervation is found in the tendon envelope, i.e. the paratenon, endotenon and surrounding loose connective tissue (Fig. 4.1). Another vital feature of tendon neuroanatomy is the presence of counteracting mediators, i.e. pro- and anti-inflammatory peptides [17]. These observations would suggest that the homeostatic regulation of healthy tendon tissue is highly dependent on the balance of neuromediator modulation occurring in the tendon envelope.
Sensory Neuromediators
The sensory nerves Type IVa act principally through release of slowly acting mediators, i.e. neuropeptides and opiods. In tendons, sensory neuropeptides with nociceptive and pro-inflammatory effects (substance P (SP), calcitonin gene related peptide (CGRP) and neurokinin A (NKA)), sensory modulatory neuropeptides with anti-inflammatory actions (galanin (GAL), somatostatin (SOM)), as well as opioid neuropeptides with anti-nociceptive and anti-inflammatory effects (Leu-enkephalin (LE), Met-enkephalin (ME), Met-enkephalin-Arg-Phe (MEAP), Met-enkephalin-Arg-Gly-Leu (MEAGL), nociceptin) have been identified (Table 4.1) (Fig. 4.2a–d) [14].
(a–d) Immunofluorescence micrographs of longitudinal sections through the Achilles tendon after double staining with antisera to SP and CGRP (a), SP and GAL (b) LE and DOR (c) and incubation with antisera to ME (d). A co-existence of SP and CGRP is seen in nerve fibers localised in the paratenon (a), suggesting possible pro-inflammatory actions. Moreover, SP is also co-localised with GAL (b), reflecting anti-inflammatory actions. The immunoreactivity displaying co-existence of LE and DOR is seen as free nerve endings in the paratenon (c), indicating a potential peripheral anti-nociceptive system. ME immunoreaction is localised in a vessel wall (d). t tendon tissue, Pt paratenon; Bar = 50 μm (Reproduced with permission from Ackermann et al. [66, 67])
Abundant amounts of sensory neuromediators have been detected in peri-vascular nerve fibers in the surrounding loose connective tissues, which may reflect an important role in the regulation of blood flow to the tendon structures. Both SP and CGRP have been reported to be potent vasodilators [18] and exert pro-inflammatory effects [19, 20]. The occurrence of sensory neuromediators in free nerve endings not associated with vessels, predominantly seen in the paratenon, may suggest nociceptive, trophic and immune regulatory roles.
In tendons receptors for SP (neurokinin 1, NK1) and CGRP (calcitonin receptor-like receptor, CRLR, and receptor activity-modifying proteins, RAMP-1) have been identified [21] (Table 4.1). These receptors have been found localized on tendon cells, immune cells, blood vessels and on free nerve endings unrelated to vessels. These localizations corroborate the suggested trophic, immune regulatory, vasoregulatory and nociceptive effects of the sensory neuromediators on tendon metabolism.
Autonomic Neuromediators
The sympathetic nervous system regulates inflammatory processes at local and systemic levels through the balanc ed release of sympathetic and parasympathetic mediators. The sympathetic mediator norepinephrine (noradrenaline, NA) together with neuropeptide Y (NPY) are released upon injury or nociceptive input, while parasympathetic mediators acetylcholine (ACh) and vasoactive intestinal polypeptide (VIP) are released by vagus nerve activation called the ‘cholinergic anti-inflammatory pathway’ [17] (Fig. 4.3a–c).
(a–c) Immunofluorescence micrographs of longitudinal sections through the Achilles tendon after incubation with antisera to Noradrenaline (NA) (a), NPY (b) and VIP (c). NA-positive fibers are mainly found as nerve terminals in outer layers of the blood vessel walls. The NPY-positive fibers are arranged as nerve terminals in the vessel walls. VIP-positive nerves are arranged as a “fence”, surrounding the proper tendon, of small varicosities in the paratenon. t tendon tissue, Pt paratenon; Bar = 50 μm (Reproduced with permission from Ackermann et al. [22])
The occurrence of sympathetic NA and NPY as well as parasympathetic mediators ACh and VIP has been demonstrated in tendon [22–27]. The observations of NA and NPY around blood vessels in the loose connective tissue around the main body of the tendon suggest that the sympathetic tendon vasoregulation occurs predominantly in the tendon envelope (Fig. 4.3a–b).
Adrenergic receptors responding to NA (Α-adrenoceptors) and to NPY (Y1) have been identified on tendon cells, blood vessel walls and on nerve fibers [24, 27]. These localizations suggest that adrenergic stimulation of tendons may be involved in proliferation of tenocytes, endothelial cells and possibly nerve cells.
Excitatory Neuromediators
Accumulating data suggest that modulation of glutamate signalling by inhibition of its receptors, ionotropic (NMDA, AMPA, Kainate) and metabotropic (mGlu), may have potential for targeted therapy in several persistent pain conditions [28, 29]. Moreover, glutamate signaling is implicated in programmed cell death, apoptosis.
Recently, glutamate and several of its receptors have been identified in tendon on nerve fibers, blood vessels and cells (Fig. 4.4) [29–31]. These localizations of glutamate have been verified by identification of several glutamate receptors, eg. NMDAR1. The localization of glutamate receptors suggest that glutamate signaling may also be involved in regulating tendon homeostasis [14].
(a–d) Immunofluorescence double-staining micrographs of longitudinal sections through tendinopathic patellar tendons focusing on the tendon proper, after incubation with antisera for PGP9.5 (a, c, thin arrows), N-methyl-D-aspartate receptor type 1 (NMDAR1) (b) and glutamate (d). Thick arrows denote neuronal NMDAR1 and within the tendon proper arrowheads denote neuronal glutamate, which is totally absent in the controls (Scale bar = 25 um) (Reproduced with permission from Schizas et al. [31])
Nerve Ingrowth After Tendon Injury
After tendon injury and during healing the peripheral nervous system responds by nerve ingrowth into the tendon proper, which during healthy conditions is more or less aneuronal and hypovascular (Figs. 4.5–4.6) [32, 33]. Nerve sprouting and growth within the tendon proper is followed by a time dependent expression of neuropeptides during the tendon healing process. After the healing process is finished sprouting nerve fibers within the tendon proper retract to the surrounding structures, ie. the paratenon and loose connective tissue. These observations of early nerve regeneration are in line with observations on bone, ligament and skin healing indicating that nerve ingrowth and subsequent retraction are fundamental aspects of tissue repair [34–38].
(a–b) Overview micrographs of longitudinal sections through the Achilles tendon at 2 weeks post rupture. Incubation with antisera to a nerve growth marker, GAP-43. Micrographs depict the proximal half of the Achilles tendon at increasing magnification in figures (a–b). Arrows denote varicosities and nerve terminals. The GAP-positive fibers, indicating new nerve fiber ingrowth, are abundantly observed in the healing proper tendon tissue( Reproduced with permission from Ackermann et al. [32])
Area occupied by nerve fibers (%) immunoreactive to SP, CGRP and GAL in relation to total area, in the mid third of the tendon, over 16 weeks post rupture (mean ± s.e.m.) (Reproduced with permission from Ackermann et al. [33])
Inflammatory Healing Phase
At 1 week after tendon injury increased occurrence of SP- and CGRP-positive nerve fibers has been demonstrated to be predominantly located in blood vessel walls surrounded by inflammatory cells in the l oose connective tissue [33] (Fig. 4.7a). The findings comply with the nociceptive role of sensory neuropeptides, but also with a pro-inflammatory role [39].
(a–b) Immunofluorescence micrograph of longitudinal sections through healing Achilles tendon 1- (a) and 2- (b) weeks post rupture after incubation with antisera to CGRP. Nerve fibers immunoreactive to CGRP at week 1 are seen as vascular and free nerve endings in the loose connective tissue (a). At week 2, CGRP- immunoreactivity occurs mainly in the healing tendinous tissue as sprouting free nerve fibers (b). v = blood vessel; lct = loose connective tissue; t = proper tendon tissue; Bar = 50 μm (Reproduced with permission from Ackermann et al. [33])
During the first week post tendon injury, increased levels of glutamatergic signaling molecules have been found both experimentally [40] and in vivo on patients with Achilles tendon ruptures [41]. Increased glutamate levels during healing [41] seem to persist until at least week 6 [unpublished data], suggesting essential actions in tendon healing as has also been shown in maintenance of bone tissue [40].
Proliferative Healing Phase
From 1 to 6 weeks post rupture, a striking shift in neuronal occurrence has been demonstrated to occur from the surrounding loose connective tissue into the proper tendinous tissue [33]. This suggests the transition of a predominantly inflammatory into a proliferative repair phase [42].
During weeks 2 to 6 post injury, the expression of SP and CGRP peaked at the rupture site of the proper tendon (Fig. 4.6). SP and CGRP was observed in sprouting free nerve endings among fibroblasts in the healing tendinous tissue, suggesting a stimulatory role of sensory neuropeptides on cell proliferation and stem cell recruitment (Fig. 4.7b) (see Chap. 5) [43, 44]. SP and CGRP are also known to stimulate proliferation of endothelial cells [45, 46], indicating that SP- and CGRP fibers around newly formed blood vessels in the rupture site would comply with a role in angiogenesis (Fig. 4.7).
Additionally the expression of sensory neuropeptide receptors, SP (NK1) and CGRP (CRLR and RAMP-1), in the healing tendon is significantly up-regulated at 2 weeks, but not at 1 week, post tendon rupture [21].
Remodeling Healing Phase
During weeks 6 to 16 post tendon ru pture it has been demonstrated that the nerve fibers retract from the proper tendon tissue to their normal location in the paratenon and surrounding loose connective tissue [33] (Fig. 4.6). This process appears to end simultaneously with the completion of paratenon repair.
Interestingly, between weeks 4 and 6, corresponding to the transition of the proliferative into the remodeling phase, a dramatic increase in the expression of the autonomic neuropeptides VIP and NPY has been demonstrated [33]. Subsequent to the elevated expression of autonomic neuropeptides, a significantly decreased expression of SP and CGRP was observed in the healing tendon [33] (Fig. 4.6). Hypothetically, an autonomic modulation may be required to end the inflammatory and reparative processes, thereby facilitating entry to and maintenance of the remodelling phase .
Protracted Nerve Ingrowth in Tendinopathy
The underlying histology in tendinopathies with chronic pain is often characterized as reflecting a failed healing response. The innervation pattern in tendinopathic tissue resembles that observed during the proliferative phase of healing after tendon injury (Table 4.2).
Sensory Neuromediators
Chronic painful tendons with tendinopathy exhibit new ingrowth of sensory nerve fibers (Fig. 4.8) [47–49] (49, 50, 98), correspondingly to what is also observed during tissue proliferation in healing t endons [32]. The observation of increased ingrowth of sensory nerves into the painful tendon proper, seen as sprouting free nerve endings, possibly represents nociceptors responding to mechanical stimuli by initiating pain signalling.
(a–b) Immunofluorescence micrographs of longitudinal sections from biopsies of healthy Achilles tendon (a) and tendinosis tissue (b) after immunostaining for SP. Arrows denote varicosities and nerve terminals. The micrograph illustrates SP-positive nerve fibres in close vicinity to a proliferated vessel (b). v = blood vessel. Bar = 50 μm (Reproduced with permission from Lian et al. [47])
The neuronal dysregulation in tendinopathy, characterized by aberrant increase of sprouting sensory nerves and increased expression of SP, p ossibly triggers pain signalling and also the hyperproliferative/degenerative changes associated with tendinopathy [47, 48, 50, 51] (Fig. 4.9).
(a–b) Hematoxylin and eosin mi crographs of longitudinal sections from biopsies of healthy patellar tendon (a) and painful tendinopathy (b). Arrows denote tenocytes. The healthy tendon is homogeneous, with organized parallel collagen structure and thin, elongated tenocytes (a). The tendinopathy, on the other hand, is marked by collagen disorganization, increased cell count, activated tenocytes, and vascular ingrowth in the tendon proper (b). V = blood vessel. Bar = 50 μm (Reproduced with permission from Lian et al. [47])
Autonomic Neuromediators
Interestingly, tendinopathic pati ents exhibit a decreased occurrence of sympathetic nerve fibers, immunoreactive to noradrenaline (Fig. 4.10) (see Chap. 7) [47]. The reduction in vasoregulatory noradrenaline would seem to comply with an altered blood flow and a suppressed anti-nociceptive function [52, 53].
(a–b) Immunofluorescence micrographs of longitudinal sections through the patellar tendon of healthy control (a) and painful tendinopathy (b) stained for TH (a marker for noradrenaline). Arro ws denotes nerve fibres. In the healthy tendon, a strong relation is seen between blood vessels and TH positive nerves (a). In painful tendinopathy, a decreased number of TH positive nerves, which are blood vessel related is seen. V = blood vessel. Bar = 50 μm (Reproduced with permission from Lian et al. [47])
Excitatory Neuromediators
Elevated levels of glutamate have, similarly to the findings of early healing, been detected in patients with tendinopathy by microdialysis and by immunohisthochemistry [31, 54]. The specific localization for the increased glutamate levels in tendinopathic patients is observed in morphologically altered tenocytes, in the endothelial and adventitial layers of blood vessel walls and in nerve fibers (Fig. 4.11) [29–31].
(a–d) Micrographs of longitudinal sections through patellar tendon biopsies after incubation with antisera to phospho-NMDAR1 (activated NMDA) in patients with patellar tendinosis (a, b) and controls (d). Micrographs of longitudinal sections through patellar tendon biopsies st ained for PGP 9.5 (a general nerve marker) in patients with patellar tendinosis are shown in (c). Phospho-NMDAR1 immunoreactivity in the tendon proper was exclusively observed in tendinosis as immunoreactive cells (a, arrowheads) as well as penetrating nerve fibres (b, arrows). The controls did not exhibit phospho-NMDAR1 immunoreactivity within the tendon proper (d). The occurrence of PGP 9.5 within the tendon proper depicted extensive nerve ingrowth in tendinopathy not seen in the controls (c, thin arrows; bar = 100 mm) (Reproduced with permission from Aubdool and Brain [39])
Several glutamate receptors have also been identified in tendinopathy patients, eg. ionotropic (NMDAR1 and activated NMDAR (Phospho-NMDAR)), metabotropic mGluR 5, which increases NMDA excitability and mGluR 6,7 that decrease NMDA excitability. Of the identified glutamate receptors, a significant up-regulation has been demonstrated of most receptors (Table 4.2).
Closer analysis has demonstrated the specific localization of the different glutamate receptors. NMDAR-1 and phospho-NMDAR1 were detected on sprouting nerve fibers, newly formed blood vessels and transformed tenocytes [29]. These localizations suggest involvement of glutamate receptors in tendinopathy regulating excitability of tenocytes, endothelial cells and nerves.
Recent reports on glutamatergic signaling in tendinopathic patients demonstrated that elevated glutamate co-existed with its up-regulated receptor NMDA1 in nerve fibers, morphologically altered tenocytes and blood vessels, which may reflect cell hyperexcitation involved in cell proliferation/differentiation. None of the controls, however, exhibited neuronal coexistence of glutamate and NMDAR1 [29, 31].
One of the recent findings established a possible mechanism responsible for activating NMDAR-1 in tendinopathy. It was demonstrated that the elevated occurrence of NMDAR-1 was correlated to that of SP (r2 = 0.54, p = 0.03) in tendinopathic tendons, while their occurrence in controls exhibited no correlation [29]. These data suggest that SP may be involved in the up-regulation of NMDAR1. In fact, SP is known to activate NMDAR1 by removing the magnesium block [55].
Effect of Hampered Neuronal Supply
Accumulating experimental evidence and clinical observations suggest that reduced neuronal supply leads to impairment of the mechanical properties of connective tendon tissue [14]. Thus, several studies have indicated that denervation impairs the mechanical properties of both normal and injured tendons.
Sensory Neuromediators
A selective denervation of sensory neuromediators by the use of Spanish pepper (capsaicin) has in experimental tendon healing reduced the concentrations of SP by approximately 60 % [56] (Fig. 4.12). The study demonstrated that higher residual SP levels correlated with increased mechanical properties of the transverse area, ultimate tensile strength, and stress at failure (r = 0.39, p = 0.036; r = 0.53, p = 0.005; and r = 0.43, p = 0.023, respectively).
(a–b) Immunoflu orescence micrographs of sections of tissue from the right hind paw of denervated (b) and controls (a) after incubation with antisera to SP. Arrows denotes varicosities and nerve terminals. A marked reduction of SP immunoreactivity is seen in the denervated group (a–b) (Reproduced with permission from Bring et al. [56])
Moreover, femoral nerve transection has been demonstrated to impair healing of the medial collateral ligament in rabbits [57]. In one study, blood flow, angiogenesis and mechanical strength of the ligament scar were all significantly decreased in denervated limbs compared to normally innervated limbs, 6 weeks after injury [57].
Autonomic Neuromediators
Surgical sympathectomy reduced failure loads of healing MCLs by 50 % compared to normally innervated healing MCLs, at 2 weeks after injury [58].
Chemical sympathectomy by systemic administration of guanethidine leads to degradation of the mechanical properties of the intact medial collateral ligament (MCL) of the knee joint in rats after only 10 days of treatment [59]. Ligaments from guanethidine treated animals exhibited a larger cross-sectional area, a higher wet weight, a decreased modulus of elasticity and a decreased stress at failure. These structural changes may to some extent be explained by the significantly increased mRNA levels for the matrix degrading enzymes MMP-13 and cathepsin K, and increased ligament blood flow induced by chemical sympathectomy.
Observations in Diabetes Mellitus
It has recently been demonstrated that patients with metabolic disorders such as diabetes mellitus are at greater risk of developing various musculoskeletal disorders [60]. Thus, diabetics often exhibit neuropathy and also decreased levels of sensory neuropeptides, which may be associated with defective tissue healing [61]. Diabetes is associated with impaired connective tissue healing and reduced biomechanical properties, correlated to down-regulated extracellular matrix proteins (see Chaps. 16, 17, 18, and 19) [62].
In injured tendons of diabetic rats there are lower mRNA and protein levels of nerve growth factor (NGF) as well as of its receptor (TrkA) as compared to injured healthy controls. Hence, neuronal mediators as well as neurotrophic factors such as NGF may be potential targets for novel regenerative approaches in tendon disorders, eg. tendinopathy.
Taken together these above mentioned studies strongly support the idea that nerve derived factors have a powerful metabolic control on the structure, function and healing capacity of tendon tissue.
Neuronal Effects of Tendon Loading
Physical activity has been demonstrated to accelerate the neuronal plasticity in tendon repair [63]. It has moreover b een demonstrated that exercise leads to increased levels of various neuromediators and their receptors, including SP and CGRP, which may be involved in regulating the healing response [21, 64, 65].
Maybe the most fascinating example of mechano-neuro transduction is the upregulation by exercise of neuromediator receptors (Fig. 4.13). Thus, mRNA-levels of the SP- and CGRP-receptors in mobilized tendons are significantly increased compared to immobilized controls at 17 days post tendon injury [21]. It may prove that enhanced tendon repair after early loading is related to an increased peripheral sensitivity to nerve factor stimulation, as a result of a local up-regulation of neuronal rece ptors.
(a–b) Normalized express ion of mRNA for the SP- (NK1) (a) and CGRP- (CRLR (b) and RAMP-1 (c) receptors in the healing area, in the Achilles tendon of rats subjected to two different levels of physical activity (freely mobilized versus plaster immobilized) at 8 and 17 days postrupture (mean + SD). * = p < 0.05; n.s. = p > 0.05. Between 8 and 17 days, there was an immense increase of the receptor expression in the mobile healing group, while the expression in the immobilized healing group fell back to levels comparable to the intact tendon control group (Reproduced with permission from Bring et al. [5])
Conclusion
Aggregated knowledge acquired during the last decade demonstrates that neuronal regulation plays an essential role in tendon metabolism, repair, and also pathology. This conception supports the idea that neuronal mediators effectuate crucial, but as yet incompletely defined roles in mechanically active connective tissues such as tendons.
Dysregulation of various neuromediators and their receptors as has been observed experimentally and clinically in eg. diabetes and tendinopathy, leads to a loss of normal tendon homeostasis resulting in chronic pain and gradual degeneration.
These novel findings of neuronal plasticity modulating tendon homeostasis and capable of responding to dysregulation in pathology should stimulate the development of targeted pharmacological and tissue engineering approaches to improve healing and treat painful tendon disorders.
References
Magnusson SP, Langberg H, Kjaer M (2010) The pathogenesis of tendinopathy: balancing the response to loading. Nat Rev Rheumatol 6(5):262–268
Reeves ND, Maganaris CN, Narici MV (2003) Effect of strength training on human patella tendon mechanical properties of older individuals. J Physiol 548(Pt 3):971–981
Schizas N, Andersson T, Fahlgren A, Aspenberg P, Ahmed M, Ackermann P (2010) Compression therapy promotes proliferative repair during rat Achilles tendon immobilization. J Orthop Res; Jan 7. [Epub ahead of print]
Bring D, Reno C, Renstrom P, Salo P, Hart D, Ackermann P (2009) Prolonged immobilization compromises up-regulation of repair genes after tendon rupture in a rat model. Scand J Med Sci Sports 20(3):411–417
Bring DKI, Reno C, Renstrom P, Salo P, Hart DA, Ackermann PW (2009) Joint immobilization reduces the expression of sensory neuropeptide receptors and impairs healing after tendon rupture in a rat model. J Orthop Res 27(2):274–280
Ackermann PW (2014) Healing and repair mechanism. In: Karlsson J, Calder J, van Diek N (eds) Achilles tendon disorders. Current conceps, 2nd edn. DJO Publications, pp 17–26
Ackermann PW, Salo PT, Hart DA (2009) Neuronal pathways in tendon healing. Front Biosci 14:5165–5187
Ackermann PW (2001) Peptidergic innervation of periarticular tissue
Stilwell DL Jr (1957) The innervation of tendons and aponeuroses. Am J Anat 100(3):289–317
Ackermann PW (2014) Tendinopathies in sports: from basic research to the field. In: Doral MN, Karlsson J (ed) Sports injuries. Springer, Berlin⁄Heidelberg, pp 1–15
Hogervorst T, Brand RA (1998) Mechanoreceptors in joint function. J Bone Joint Surg Am 80(9):1365–1378
Strasmann T, Weihe E, Halata Z (1990) CGRP-like immunoreactivity in sensory nerve endings of the Golgi tendon organ. A light- and electron-microscopic study in the grey short-tailed opossum (Monodelphis domestica). Acta Anat 137(3):278–281
Jozsa L, Kannus P (1997) Human tendons. Anatomy, physiology, and pathology. Human Kinetics, Champaign
Ackermann PW, Franklin SL, Dean BJ, Carr AJ, Salo PT, Hart DA (2014) Neuronal pathways in tendon healing and tendinopathy–update. Front Biosci 19:1251–1278
Hokfelt T, Johansson O, Ljungdahl A, Lundberg JM, Schultzberg M (1980) Peptidergic neurones. Nature 284(5756):515–521
Audet M, Bouvier M (2012) Restructuring G-protein- coupled receptor activation. Cell 151(1):14–23
Tracey KJ (2002) The inflammatory reflex. Nature 420:853–859
Brain SD, Williams TJ, Tippins JR, Morris HR, MacIntyre I (1985) Calcitonin gene-related peptide is a potent vasodilator. Nature 313(5997):54–56
Brain SD, Williams TJ (1985) Inflammatory oedema induced by synergism between calcitonin gene-related peptide (CGRP) and mediators of increased vascular permeability. Br J Pharmacol 86(4):855–860
Maggi CA (1995) Tachykinins and calcitonin gene-related peptide (CGRP) as co-transmitters released from peripheral endings of sensory nerves. Prog Neurobiol 45(1):1–98
Bring D, Reno C, Renstrom P, Salo P, Hart D, Ackermann P (2010) Prolonged immobilization compromises up-regulation of repair genes after tendon rupture in a rat model. Scand J Med Sci Sports 20(3):411–417
Ackermann PW, Li J, Finn A, Ahmed M, Kreicbergs A (2001) Autonomic innervation of tendons, ligaments and joint capsules. A morphologic and quantitative study in the rat. J Orthop Res 19(3):372–378
Danielson P, Alfredson H, Forsgren S (2006) Immunohistochemical and histochemical findings favoring the occurrence of autocrine/paracrine as well as nerve-related cholinergic effects in chronic painful patellar tendon tendinosis. Microsc Res Tech 69(10):808–819
Danielson P, Alfredson H, Forsgren S (2007) In situ hybridization studies confirming recent findings of the existence of a local nonneuronal catecholamine production in human patellar tendinosis. Microsc Res Tech 70(10):908–911
Danielson P, Alfredson H, Forsgren S (2007) Studies on the importance of sympathetic innervation, adrenergic receptors, and a possible local catecholamine production in the development of patellar tendinopathy (tendinosis) in man. Microsc Res Tech 70(4):310–324
Ljung BO, Forsgren S, Friden J (1999) Sympathetic and sensory innervations are heterogeneously distributed in relation to the blood vessels at the extensor carpi radialis brevis muscle origin of man. Cells Tissues Organs 165(1):45–54
Wall ME, Faber JE, Yang X, Tsuzaki M, Banes AJ (2004) Norepinephrine-induced calcium signaling and expression of adrenoceptors in avian tendon cells. Am J Physiol 287(4):C912–C918
Wozniak KM, Rojas C, Wu Y, Slusher BS (2012) The role of glutamate signaling in pain processes and its regulation by GCP II inhibition. Curr Med Chem 19(9):1323–1334
Schizas N, Weiss R, Lian O, Frihagen F, Bahr R, Ackermann PW (2012) Glutamate receptors in tendinopathic patients. J Orthop Res 30(9):1447–1452
Scott A, Alfredson H, Forsgren S (2007) VGluT2 expression in painful Achilles and patellar tendinosis: Evidence of local glutamate release by tenocytes. J Orthop Res
Schizas N, Lian O, Frihagen F, Engebretsen L, Bahr R, Ackermann PW (2010) Coexistence of up-regulated NMDA receptor 1 and glutamate on nerves, vessels and transformed tenocytes in tendinopathy. Scand J Med Sci Sports 20(2):208–215
Ackermann PW, Ahmed M, Kreicbergs A (2002) Early nerve regeneration after achilles tendon rupture–a prerequisite for healing? A study in the rat. J Orthop Res 20(4):849–856
Ackermann PW, Li J, Lundeberg T, Kreicbergs A (2003) Neuronal plasticity in relation to nociception and healing of rat achilles tendon. J Orthop Res 21(3):432–441
Hukkanen M, Konttinen YT, Santavirta S, Paavolainen P, Gu XH, Terenghi G et al (1993) Rapid proliferation of calcitonin gene-related peptide-immunoreactive nerves during healing of rat tibial fracture suggests neural involvement in bone growth and remodelling. Neuroscience 54(4):969–979
Kishimoto S (1984) The regeneration of substance P-containing nerve fibers in the process of burn wound healing in the guinea pig skin. J Investig Dermatol 83(3):219–223
Li J, Ahmad T, Spetea M, Ahmed M, Kreicbergs A (2001) Bone reinnervation after fracture: a study in the rat. J Bone Miner Res 16(8):1505–1510
Martin P (1997) Wound healing–aiming for perfect skin regeneration. Science (New York) 276(5309):75–81
Salo PT, Beye JA, Seerattan RA, Leonard CA, Ivie TJ, Bray RC (2008) Plasticity of peptidergic innervation in healing rabbit medial collateral ligament. Can J Surg 51(3):167–172
Aubdool AA, Brain SD (2011) Neurovascular aspects of skin neurogenic inflammation. The journal of investigative dermatology Symposium proceedings/The Society for Investigative Dermatology, Inc [and] European Society for Dermatological Research 15(1):33–39
Molloy TJ, Wang Y, Horner A, Skerry TM, Murrell GA (2006) Microarray analysis of healing rat Achilles tendon: evidence for glutamate signaling mechanisms and embryonic gene expression in healing tendon tissue. J Orthop Res 24(4):842–855
Greve K, Domeij-Arverud E, Labruto F, Edman G, Bring D, Nilsson G et al (2012) Metabolic activity in early tendon repair can be enhanced by intermittent pneumatic compression. Scand J Med Sci Sports 22(4):e55–e63
Ackermann PW, Domeij-Arverud E, Leclerc P, Amoudrouz P, Nader GA (2012) Anti-inflammatory cytokine profile in early human tendon repair. Knee Surg Sports Traumatol Arthrosc
Nilsson J, von Euler AM, Dalsgaard CJ (1985) Stimulation of connective tissue cell growth by substance P and substance K. Nature 315(6014):61–63
Hong HS, Lee J, Lee E, Kwon YS, Lee E, Ahn W et al (2009) A new role of substance P as an injury-inducible messenger for mobilization of CD29(+) stromal-like cells. Nat Med 15(4):425–435
Haegerstrand A, Dalsgaard CJ, Jonzon B, Larsson O, Nilsson J (1990) Calcitonin gene-related peptide stimulates proliferation of human endothelial cells. Proc Natl Acad Sci U S A 87(9):3299–3303
Ziche M, Morbidelli L, Pacini M, Geppetti P, Alessandri G, Maggi CA (1990) Substance P stimulates neovascularization in vivo and proliferation of cultured endothelial cells. Microvasc Res 40(2):264–278
Lian O, Dahl J, Ackermann PW, Frihagen F, Engebretsen L, Bahr R (2006) Pronociceptive and antinociceptive neuromediators in patellar tendinopathy. Am J Sports Med 34(11):1801–1808
Schubert TE, Weidler C, Lerch K, Hofstadter F, Straub RH (2005) Achilles tendinosis is associated with sprouting of substance P positive nerve fibres. Ann Rheum Dis 64(7):1083–1086
Sanchis-Alfonso V, Rosello-Sastre E, Subias-Lopez A (2001) Neuroanatomic basis for pain in patellar tendinosis (“jumper’s knee”): a neuroimmunohistochemical study. Am J Knee Surg 14(3):174–177
Andersson G, Backman LJ, Scott A, Lorentzon R, Forsgren S, Danielson P (2011) Substance P accelerates hypercellularity and angiogenesis in tendon tissue and enhances paratendinitis in response to Achilles tendon overuse in a tendinopathy model. Br J Sports Med 45(13):1017–1022
Backman LJ, Eriksson DE, Danielson P (2014) Substance P reduces TNF-alpha-induced apoptosis in human tenocytes through NK-1 receptor stimulation. Br J Sports Med 48(19):1414–1420
Kager I, Mousa SA, Sieper J, Stein C, Pipam W, Likar R (2011) Blockade of intra-articular adrenergic receptors increases analgesic demands for pain relief after knee surgery. Rheumatol Int 31(10):1299–1306
Klatt S, Fassold A, Straub RH (2012) Sympathetic nerve fiber repulsion: testing norepinephrine, dopamine, and 17beta-estradiol in a primary murine sympathetic neurite outgrowth assay. Ann N Y Acad Sci 1261:26–33
Alfredson H, Forsgren S, Thorsen K, Lorentzon R (2001) In vivo microdialysis and immunohistochemical analyses of tendon tissue demonstrated high amounts of free glutamate and glutamate NMDAR1 receptors, but no signs of inflammation, in Jumper's knee. J Orthop Res 19(5):881–886
Madden DR (2002) The structure and function of glutamate receptor ion channels. Nat Rev Neurosci 3(2):91–101
Bring DK, Paulson K, Renstrom P, Salo P, Hart DA, Ackermann PW (2012) Residual substance P levels after capsaicin treatment correlate with tendon repair. Wound repair and regeneration : official publication of the Wound Healing Society [and] the European Tissue Repair. Society 20(1):50–60
Ivie TJ, Bray RC, Salo PT (2002) Denervation impairs healing of the rabbit medial collateral ligament. J Orthop Res 20(5):990–995
Grorud KW, Jensen KT, Provenzano PP, Vanderby R Jr (2007) Adjuvant neuropeptides can improve neuropathic ligament healing in a rat model. J Orthop Res 25(6):703–712
Dwyer KW, Provenzano PP, Muir P, Valhmu WB, Vanderby R Jr (2004) Blockade of the sympathetic nervous system degrades ligament in a rat MCL model. J Appl Physiol 96(2):711–718
Ramchurn N, Mashamba C, Leitch E, Arutchelvam V, Narayanan K, Weaver J et al (2009) Upper limb musculoskeletal abnormalities and poor metabolic control in diabetes. Eur J Int Med 20(7):718–721
Pradhan L, Nabzdyk C, Andersen ND, LoGerfo FW, Veves A (2009) Inflammation and neuropeptides: the connection in diabetic wound healing. Exp Rev Mol Med 11, e2
Ahmed AS, Schizas N, Li J, Ahmed M, Ostenson CG, Salo P et al (2012) Type 2 diabetes impairs tendon repair after injury in a rat model. J Appl Physiol 113(11):1784–1791
Bring DK, Kreicbergs A, Renstrom PA, Ackermann PW (2007) Physical activity modulates nerve plasticity and stimulates repair after Achilles tendon rupture. J Orthop Res 25(2):164–172
Ytteborg E, Torgersen JS, Pedersen ME, Helland SJ, Grisdale-Helland B, Takle H (2013) Exercise induced mechano-sensing and Substance P mediated bone modeling in Atlantic salmon. Bone 53(1):259–268
Jonsdottir IH (2000) Special feature for the Olympics: effects of exercise on the immune system: neuropeptides and their interaction with exercise and immune function. Immunol Cell Biol 78(5):562–570
Ackermann PW, Finn A, Ahmed M (1999) Sensory neuropeptidergic pattern in tendon, ligament and joint capsule. A study in the rat. Neuroreport 10(10):2055–2060
Ackermann PW, Spetea M, Nylander I, Ploj K, Ahmed M, Kreicbergs A (2001) An opioid system in connective tissue: a study of achilles tendon in the rat. J Histochem Cytochem 49(11):1387–1395
Funding Source
Studies from the author’s laboratories were supported by the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet (project nr. SLL 20110177), and the Swedish National Centre for Research in Sports, as well as the Swedish Medical Research Council (2012–3510) (PWA).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Ackermann, P.W., Salo, P., Hart, D.A. (2016). Tendon Innervation. In: Ackermann, P., Hart, D. (eds) Metabolic Influences on Risk for Tendon Disorders. Advances in Experimental Medicine and Biology, vol 920. Springer, Cham. https://doi.org/10.1007/978-3-319-33943-6_4
Download citation
DOI: https://doi.org/10.1007/978-3-319-33943-6_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33941-2
Online ISBN: 978-3-319-33943-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)