Abstract
Antarctic krill has been fished commercially in the Southern Ocean since the 1970s and has been consistently the largest fishery, by tonnage, in the region since then. The fishery has seen changes in the nations involved, with early catches dominated by vessels from the USSR, Japanese vessels in the middle years and, more recently, most of the catch has been taken by vessels from Norway. A variety of products have emerged from the fishery with early efforts aimed at human consumption but latterly, the bulk of the catch has been used as high-end aquaculture feed with a small but valuable fraction being used to produce krill oil. The fishery has been managed by the international Commission for the Conservation of Antarctic Marine Living Resources which recognised the potential threat to the marine ecosystem through krill harvesting and which has implemented a precautionary approach to management of the fishery. Currently the fishery catches approximately 300,000 tonnes annually, all from the South Atlantic, where the precautionary catch limit has been set at 5.6 million tonnes. The fishery and its management regime will face challenges in the future with the emergence of new technologies, increased catches by new entrants and environmental changes.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
11.1 Introduction
The fishery for Antarctic krill ( Euphausia superba ) has been operating in the Southern Ocean since the 1970s, and the development of the fishery has been regularly documented (Budzinksi et al. 1985; Nicol and Foster 2003). Following the initiation of the fishery the scientific community recognised the poor level of information concerning krill and the Southern Ocean ecosystem and an ambitious international scientific program (BIOMASS – Biological Investigations on Marine Antarctic Systems and Stocks) was established to provide information that might be used to regulate the fishery. The remit of the BIOMASS Program was to “gain a deeper understanding of the structure and dynamic functioning of the Antarctic marine ecosystem as a basis for the future management of potential living resources” (El-Sayed 1977, 1994). This initiative was an important predecessor to the negotiation in the late 1970s of an international regime tasked with managing the harvest of krill and other Southern Ocean marine resources (CCAMLR – the Convention on the Conservation of Antarctic Marine Living Resources) (Frank 1983; Edwards and Heap 1981). The main concern in the development of CCAMLR was the effect that a dramatic reduction in the size of krill stocks would have on populations of krill predators , most notably, penguins , seals , whales , fish and seabirds (Howard 1989). The Convention on the Conservation of Antarctic Marine Living Resources (CCAMLR) was adopted in 1980 and it entered into force 2 years later.
CCAMLR was initially known as the “krill convention” (Fernholm and Rudback 1989; Nicol 1991) because of its predominant focus on ensuring the ecosystem-based management of Southern Ocean krill stocks. However, it was not until the early 1990s that CCAMLR began the process of regulating the krill fishery (Nicol 1992). Since then there has been a slow accumulation of management measures that have collectively given effect to many aspects of the ecosystem approach to management of the krill fishery that was originally envisaged in the Convention. Because the krill fishery did not grow in size as quickly as was originally predicted there was little pressure to develop a comprehensive management regime, resulting in the Commission allowing the fishery to develop slowly as CCAMLR focussed on other emerging issues.
In this chapter, we will concentrate mainly on recent developments in the fishery and its management but we provide an outline of the early years and references to published works that deal comprehensively with the early history of the fishery. We describe the current state of the fishery: where it operates, who is involved, how much is caught and the seasonal nature of the operation. We then deal with the nature of the fishery itself: how it operates, the products derived from the catch and the trends in technology and product types. Finally we address the management of the fishery and the scientific and environmental issues that will make future management of this large stock a challenge.
11.2 The Development of the Antarctic Krill Fishery
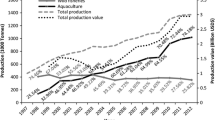
The commercial krill fishery has operated in the Southern Ocean since the mid-1970s and several publications have recorded the history and development of the fishery (Table 11.1). There have been a number of phases in the development of the fishery (Fig. 11.1). The early years of the fishery were characterised by low catches, primarily aimed towards research and product development (Grantham 1977) with vessels from the Soviet Union (from the 1961/1962 season) and Japan (from the 1972/1973 season) conducting exploratory fisheries. In the late 1970s and early 1980s there was a rapid increase in fishing effort, with total catches reaching levels not seen since. This period of high catch rates was primarily a result of fishing by vessels from the USSR, but also included catches by vessels from Japan, Poland, former East Germany and Chile (Table 11.2). This era marked the move of the fishery from an exploratory/experimental phase to a developmental/commercial phase (Budzinski et al. 1985; Nicol and Endo 1997).
Catches of krill in the Southern Ocean from 1973 to 2014 (CCAMLR 2014)
There was a sharp decline in catches in the fishery in the mid-1980s, which has been attributed to technical difficulties (Nicol and Endo 1997; Budzinski et al. 1985). Further development of processing techniques was undertaken and catches quickly recovered before once again declining rapidly in 1991 coinciding with the break-up of the Soviet Union (Nicol and Endo 1997). From the mid-1990s, the krill fishery remained low reaching a catch level of 125,823 tonnes in 2009. The catches during this period were dominated by Japanese and Korean vessels. Since 2009 the catch has increased more rapidly reaching a level of nearly 300,000 tonnes in 2013/2014. This phase has been associated with the entry into the fishery of Norwegian companies and in recent years, from increased catches by vessels from Korea and by the emergence of China as a fishing nation (SC-CCAMLR 2014).
The catch of the early exploratory fishery was aimed at developing harvesting and processing techniques (Eddie 1977; Nicol and Endo 1997). Once the fishery began to develop commercially, catches were used in products for human consumption and meal for animal feed (Eddie 1977). Into the 1980s the scope for use of krill broadened with interest in krill as a source of: enzymes , other proteins, lipids , chitin (Budzinski et al. 1985). Since the 1990s the catch has been used mainly for the production of aquaculture feed, either as meal or as whole frozen krill, and more recently, krill oil as a human health supplement (Nicol et al. 2000, 2012; Nicol and Foster 2003).
11.2.1 Spatial and Temporal Trends
Although the krill fishery was initially circumpolar, it has been concentrated in the South Atlantic (CCAMLR Area 48) since the mid-1990s (Fig. 11.2). The fishery first targeted the South Indian Ocean but over the decades this area became less important and the South Atlantic became the dominant fishing ground. Today the catch is mainly taken from the waters surrounding the Antarctic Peninsula and South Shetland Islands (subarea 48.1), the South Orkneys (subarea 48.2) and from the South Georgia area (subarea 48.3). This concentration of operations in the South Atlantic is primarily operational as the South Atlantic fishing grounds are closer to port services and are increasingly free of ice for longer periods than the rest of the Southern Ocean (Kawaguchi et al. 2009). Additionally, the Southwest Atlantic is estimated to contain 40 % of the global krill stock (Atkinson et al. 2009). The fishing grounds in the South Atlantic have also changed over the history of the fishery, with the importance of subarea 48.3 declining and that of subarea 48.1 increasing. The krill fishery has also changed its peak season of operation. In the early years of the fishery, most krill were taken in summer and autumn, with lowest catches being taken in spring (Fig. 11.3). In recent years krill catches have shown a reversal in this historic trend with lowest catches occurring over summer, peaking in late autumn, with very little fishing activity in spring (Fig. 11.3).
Some of these temporal and spatial shifts in the fishery over time have been attributed to reductions in winter sea-ice extent in the south of the Scotia Sea which allow winter fishing in subareas 48.1 and 48.2 (Kawaguchi et al. 2009). There has also been a recent tendency of the fishing vessels to avoid catching krill that are actively feeding on algae because this adversely affects the type of product that can be manufactured (Kawaguchi et al. 1999; Ichii 2000; Nicol et al. 2000). The production of krill oil has also led to vessels concentrating on fishing in autumn and winter when krill are richest in lipids (Kim et al. 2014). Additionally, since 1993, vessels fishing in the waters around South Georgia – historically the only ice free krill fishing grounds – have been subject to a license fee imposed by the Government of South Georgia (Agnew 2004) which may act as an incentive for operators to fish in other areas.
11.2.2 Changes in Fishing Technologies
The krill fishery has traditionally used large midwater trawls with a mesh liner towed by large (80+ m) freezer trawlers (Budzinski et al. 1985). These trawls can have a horizontal opening of 60 m and a vertical opening of 50 m. Trawls are generally towed for up to an hour and landed catches are of the order of 10 tonnes. Some of these trawls have been emptied by a pumping system but more usually the catch is landed on deck or into a storage tank. The catch per hour of vessels using conventional trawls has increased recently and this may be a result of changes in fishing behaviour or processing technology.
One of the Norwegian companies that entered the fishery in 2009, Aker Biomarine, has developed a patented “eco-harvesting technology” (patent WO2005004593) that allows much higher catch rates than conventional trawls (800 tonnes/day compared to 400 tonnes per day) (Fig. 11.4). This technology lands krill on the vessel alive and in good condition rendering them suitable for the manufacture of a wider range of products. The eco-harvesting technology uses a hose attached to the cod-end of a small (20 × 20 m mouth opening) net that is lowered into a krill swarm. Air is pumped into the cod-end and this lifts water and krill to the surface and onto the ship’s conveyor that leads to the factory (Fig. 11.5). The net can be left in the water for weeks at a time and the fishing operation proceeds continuously with the catch rate being determined by the ability of the ship’s factory to process the catch. It is also claimed that the continuous fishing system results in less waste of landed catch, allows for accurate estimates of tonnage caught, reduces bycatch and may reduce escape mortality of krill. Escape mortality refers to the krill that are killed by fishing equipment that do not end up in the catch. There has been concern within CCAMLR that large trawls may inflict considerable mortality on the krill population that passes through the wider mesh panels on the wings of the net or through being struck by the towing lines (SC-CCAMLR 2011 para 3.12). Modelling studies and some experimental research has failed to clarify the magnitude of this problem (CCAMLR-WG-EMM 2014, paragraphs 2.23–2.24).
Daily catch (tonnes) of individual fishing vessel in the krill fishery. Black is conventional trawls and Grey is the continuous fishing system. In the boxplot the notch indicates the median with the filled bars over the 25–75 percentile, the max and min (bars) and any values that lie outside 1.5 times the IQR (Plot and data supplied by CCAMLR Secretariat)
11.2.3 Estimating the Total Removal of Krill
A second factor that affects the estimate of mortality of krill attributable to the fishery is the uncertainty surrounding the quantity of krill landed on fishing vessels. The krill catch is generally estimated from the quantity of product from each vessel. It has become apparent that the conversion factors used to relate the weight of product to the tonnage of the catch can vary widely between vessels. Additionally, with the entry into the krill fishery of a different harvesting technology, CCAMLR has focussed on the issue of more accurate assessment of the total catch (SC-CCAMLR 2014, paragraphs 3.4 and 3.5).
11.3 Products from the Krill Fishery
11.3.1 Early Developments
Krill are a difficult raw material; they are easily crushed in nets and need to be rapidly processed once caught because they spoil rapidly once on deck. Krill need to be processed within 1 h of landing if they are to be used for human consumption and within 3 h if they are destined for animal feed (Budzinski et al. 1985; Nicol and Endo 1999). With the initial interest in the commercial fishery in the 1970s the FAO commissioned a series of Fisheries Technical Papers (Budzinski et al. 1985; Eddie 1977; Grantham 1977) addressing the harvesting, utilisation and possibilities of processing and marketing products made from Antarctic krill. In the mid-1970s small catches were being used for product and processing development, with the nutritional value of the products being a high priority (Sidhu et al. 1970). These products included krill meal for animal feed, krill protein paste, frozen cooked whole krill, dried krill, minced muscle and tail meats, all for human consumption (Lyubimova et al. 1973; Siebert et al. 1980; Oehlenschläger and Schreiber 1981; Eddie 1977).
Product development continued throughout the 1980s and early 1990s. However, in the mid-1980s product development research stalled for several years when the industry came to terms with the discovery of high fluoride content of krill (Nicol and Endo 1997; Budzinski et al. 1985). High levels of fluoride that exist in the exoskeleton , particularly in the carapace (Soevik and Braekkan 1979) impeded the use of krill in animal feeds (Yoshitomi et al. 2007) and human products due to health concerns associated with the accumulation of fluoride in the bones and tissues which can be toxic at high levels (Adelung et al. 1987). This problem was eventually overcome with research undertaken into ways to decrease and/or mitigate the quantity of fluorine that would be included in the end product as a result of the inclusion of krill as an ingredient (Tenuto-Filho and Alvarenga 1999; Tenuto-Filho 1993; Budzinski et al. 1985; Wang et al. 2011). Research continues into methods to reduce fluoride in krill products so that they can be utilised for humans and domestic animals (Xie et al. 2012; Wang and Yin 2012; Zhang et al. 2014). Because many marine vertebrates are tolerant of high fluoride levels in their prey a greater focus on the use of krill as an aquaculture feed developed (Grave 1981; Julshamn et al. 2004) and low fluoride aquaculture feeds have been developed and successfully trialled (Yoshitomi et al. 2007).
The mid-1990s shift into the development of higher value, lower yield products, was predicted to occur in the FAO Technical Papers of the late 1970s and mid-1980s. Grantham (1977) suggested that the initial phase of the krill fishery would see competition for a place in the market, not for the resource, a situation which is highly unusual in commercial fisheries. This prediction was based on the high estimated abundance of krill (biomass estimates at the time ranged from 60 to 5000 million tonnes) (Voronia 1983; Lyubimova et al. 1973) and the low demand for the product in the marketplace at that time (Grantham 1977). The demise of the Soviet Union also meant that the fishery had to move towards a more economic mode of operation. To increase revenues from the fishery, the industry has examined producing a variety of both high value products as well as bulk commodities that are destined for animal feed.
As part of the notification system that CCAMLR has in place, companies provide an indication of the mix of products that they expect to produce. For the 2014/2015 season the frequency of product inclusion in notifications , from the 20 notifications for the 2014/2015 krill fishery was: whole frozen 18, boiled 3, meal 15, oil 3 , other product 5 (frozen krill meat, hydrolysate, lipid complex, paste, peeled) (information supplied by the CCAMLR Secretariat). The krill fishery is still dominated by the bulk production of frozen krill and meal; however, the production of smaller quantities of more specialised products probably increases the economic yield from the fishery considerably. The krill fishery operates in waters distant from most ports and in extremely difficult environmental conditions. It is not a fishery that is entered on a whim and because of its relatively low returns and high costs is one that will reward companies with a long term investment in the industry.
Despite the difficulties involved in fishing for krill and of krill as a raw product there is continuing commercial interest in the krill fishery. An index of this interest is the level of patent activity and 1040 krill-related patents have been lodged between 1976 and 2014 (Foster et al. 2011, updated 2014). The most active period has been between 2006 and 2014 when 34 % (a total of 355) of all krill-related patents were lodged (Fig. 11.6). Most recent patents relating to processing (45 % of all patents 2010–2014) were lodged for methods and processes for extracting high-quality krill products including low-fluoride , high quality oils and krill meals that are suited to use in medical applications. Medical uses comprise 29 % of recent patents indicating the industry’s focus on products for the high value pharma and nutraceutical sectors.
Number of krill-related patents lodged from 1976 to 2014 (Data from the krill patent database presented in Foster et al. (2011) updated in March 2014)
There has also been a shift in the Nations represented in the lodgement of krill-related patents over the past several years. Norwegian interest in the krill fishery has been accompanied by an increase in patent activity. From 1976 to 2008 Norway only represented 2 % of all krill-related-patents lodged but between 2010 and 2014, this increased to 8 %. From 2012 to 2014 China lodged a total of 129 krill-related patents , representing 70 % of all patents lodged. Patents from China have related primarily to new processing technologies for the extraction of high quality products. This level of activity indicates the serious Chinese interest in krill fishing.
11.3.2 Krill for Human Consumption
Since the commencement of the krill fishery there has been an emphasis on producing high quality protein for human consumption , although there have been difficulties in producing a product that is marketable, safe and economically viable (Budzinski et al. 1985; Nicol and Endo 1997; Nicol et al. 2000). Currently very little of the krill caught is destined for use as human food with the bulk going to aquaculture and to produce krill oils which are used as a health supplement. There is still considerable research being carried out into krill for human consumption (Tou et al. 2007; Chi et al. 2013) but the future of krill as a human food will depend largely on finding an economically effective method of removing fluoride (Jung et al. 2013).
11.3.3 Krill as Fishing Bait and Aquarium Feed
A large proportion (~34 %) of the Japanese krill catch in the 1990s and early 2000s was used to attract fish in recreational fishery and freeze dried krill is still used as a commercially available feed for aquarium fish (Nicol et al. 2000). With the Japanese fishing fleet now absent from the krill fishery it is difficult to estimate the current proportion of the catch that is used for these purposes.
11.3.4 Krill Meal for Aquaculture
Production of carnivorous fish through aquaculture has increased dramatically over the last three decades and this has led to an increase in demand for fish meals and marine byproducts. Because of declining fish stocks, the aquaculture industry is investigating the supplementation of currently used proteins and additives with lower cost alternatives that are readily available (Yoshitomi et al. 2007; Davis and Arnold 2000). Some research has focused on determining the properties of krill meal compared to commonly used fish meals (Giogios et al. 2009). Because fishing for krill is an expensive activity krill meal is also expensive and is most commonly used as a high value source of protein, feeding enhancing agents, colouration, growth promoters and sources of Omega-3 oils to aquaculture feeds (Floreto et al. 2001) rather than the primary ingredient. Krill meal has a nutritional value equal to, or surpassing, that of regular fish meals (Yoshitomi et al. 2007; Karlsen et al. 2006; Opstad et al. 2006; Gaber 2005) when used as a substitute in the diets of various farmed species including Atlantic cod, Atlantic salmon and Pacific white shrimp. Due to the increasing price of fish meals, producers are constantly striving to improve the uptake of feeds to minimise feed wastage and provide better economic returns (Smith et al. 2005). Krill oil and chitosan have also been shown to increase the nutritional value and shelf life of various fish fillet products (Duan et al. 2010).
11.3.5 Chitin
Krill contain a number of valuable chemicals that could be extracted as part of the post-capture manufacturing process. The shell of krill contains chitin (up to 40 % dry weight ) and this is a chemical for which there is a developing market (Yanase 1981) which could be a potentially plentiful by-product from the fishery. In the traditional processing of rolling krill for a meat product, the exoskeleton has usually been a waste product but is increasingly seen as valuable commodity in its own right, with research into the quality of the waste product being undertaken by Polish researchers in the early 1980s (Naczk et al. 1981) and more recently (Tou et al. 2007).
Chitin is the main source of production of chitosan , which is used in a number of applications, such as a flocculating agent, a wound healing agent and a delivery vehicle for various pharmaceuticals . A number of publications have focussed on the utilisation of krill for these purposes (Bustos et al. 2003). Due to their high biomass and current underutilisation, krill are major producers of chitin in the world’s oceans and the fishery for Antarctic krill is potentially a large source of chitin for industry (Nicol and Hosie 1993).
11.3.6 Pigments
The characteristic red colouration of Antarctic krill comes from the pigment astaxanthin, and other carotenoid pigments in the exoskeleton (Nicol et al. 2000). There has been considerable research into the addition of krill-derived pigments to aquaculture feed to improve colouration in farmed species (Savage and Foulds 1987; Kalinowski et al. 2007; Suontama et al. 2007; Moretti et al. 2006; Floreto et al. 2001). Synthetic colourants have generally been used for pigmentation of aquaculture species but consumer demand for more natural products is increasing (Moreira et al. 2006).
11.3.7 Krill Oils
As early as 1970 it was recognised that, as well as being an excellent source of protein, krill could also be a source of oil for consumption by humans and animals (Sidhu et al. 1970). There is an increasing demand for diet supplements from marine oils rich in Omega-3 fatty acids (Arthi 2009; Shahidi and Wanasundara 1998; Hill 2013). Fish oils have been subject to large amounts of R&D in fish processing, feed, functional food and nutra- and pharmaceutical markets over the past 15 years. The Omega-3 and Omega-6 properties of seafood have long been recognised as having health benefits (Fereidoon and Wanasundara 1998) including prevention and treatment of arthritic conditions, cardiovascular disease, PMS and facial appearance.
The high fatty acid properties of krill oil make it effective in the treatment of autoimmune murine lupus (Chandrasekar, et al. 1996), cardiovascular disease (Batetta et al. 2009; Bunea et al. 2004) and arthritis (Deutsch 2007). Other research indicates that properties of krill, including its enzymes , antioxidant astaxanthin and chitin , make it effective in the treatment of liver disease (Tandy et al. 2009) and maintenance of general health and wellbeing (Bridges et al. 2010). It has been suggested that the increased bioavailability of the Omega-3 and Omega-6 fatty acids may make krill oil a superior treatment for cardiovascular disease and other health conditions, compared to regular fish oils (Kidd 2009), however, research continues into whether krill oil is clinically superior to fish oils (Backes and Howard 2014).
In 2004 the European market for Omega-3 fatty acid products was valued at approximately US$194 million with more than three-quarters of these oils being from marine based sources (Nichols and Nelson 2007). However, whilst the market for the use of fish oils in nutraceutical products is high in value, it remains relatively small in volume, estimated to represent only 5–6 % of total world fish oil production (Nichols 2007). Krill oil production, currently at less than 1000 tonnes per year, uses only ~10 % of the tonnage of krill landed. Even if the market of krill oil increases greatly in the near future the extra demand could readily be met from existing catches.
11.3.8 Enzymes
The suite of proteolytic enzymes contained within krill’s digestive gland was initially a significant problem for the fishery because they led to rapid spoilage of catch (Nicol et al. 2000). However, these hydrolytic enzymes , in particular the proteases, carbohydrases, nucleases and phospholipases, have wide ranging uses in different medical applications (Nicol et al. 2000). Applications include treatment of spinal injuries (Melrose et al. 1995) and necrotic wounds (Karlstam et al. 1991) and in clinical drug applications (Nicol et al. 2000).
11.4 Management of the Krill Fishery
11.4.1 Background
Antarctic krill is classified by the FAO as an underexploited fish stock (stocks that are currently harvested but could withstand an increased level of harvest) (Maguire et al. 2006). Only 2 % of stocks monitored by the FAO are classed as underexploited (FAO 2008). The current krill catch in Antarctic waters is ~300,000 tonnes a year which is only 3.5 % of estimated precautionary catch level of some 8.6 million tonnes (CCAMLR figures) so the potential for increase is considerable. The careful management of the krill fishery into the future is essential given its key role ecological as the cornerstone of the Antarctic food chain.
Management of the krill fishery is the responsibility of the Commission for the Conservation of Antarctic Marine Living Resources (CCAMLR ). CCAMLR first met in 1982 and currently has 25 Members and 9 acceding States. CCAMLR is responsible for the conservation of living resources (with the exception of seals and whales which are managed by other instruments), both targeted and dependent and associated species, in the Southern Ocean (CCAMLR 2009b), (the Southern Ocean is defined for this purpose as the 32.9 million square kilometres of ocean between the Antarctic Polar Front and the continent) (CCAMLR 2009a; Nicol and Endo 1997). The CCAMLR Area is further divided for management purposes into areas based on the FAO Statistical Areas . (Fig. 11.7).
CCAMLR has adopted both a ‘precautionary’ and ‘ecosystem approach ’ to fisheries management that attempts to combine conservation and “rational use” (CCAMLR 2009b; Miller 2002). CCAMLR ’s precautionary approach requires that management decisions should have a low risk of long-term adverse effects, an important factor when working with scientific information that has a degree of uncertainty (Hewitt and Linen Low 2000). The ecosystem approach differs from conventional single species fisheries management approaches by managing target species whilst taking into account the needs of dependent and related species. Article II (3) of the Convention set outs the remit for taking an ecosystem approach to the management of Southern Ocean resources:
3. Any harvesting and associated activities in the area to which this Convention applies shall be conducted in accordance with the provisions of this Convention and with the following principles of conservation :
(a) prevention of decrease in the size of any harvested population to levels below those which ensure its stable recruitment. For this purpose its size should not be allowed to fall below a level close to that which ensures the greatest net annual increment;
(b) maintenance of the ecological relationships between harvested, dependent and related populations of Antarctic marine living resources and the restoration of depleted populations to the levels defined in subparagraph (a) above; and
(c) prevention of changes or minimisation of the risk of changes in the marine ecosystem which are not potentially reversible over two or three decades, taking into account the state of available knowledge of the direct and indirect impact of harvesting, the effect of the introduction of alien species, the effects of associated activities on the marine ecosystem and of the effects of environmental changes , with the aim of making possible the sustained conservation of Antarctic marine living resources.
CCAMLR has two primary objectives in managing the krill fishery. Firstly, ensuring that overall krill biomass is not compromised and secondly, ensuring that krill biomass is adequate to meet the needs of land-based krill predators (Hewitt and Low 2000). The phrase “dependent and related species ” has not been further elaborated by CCAMLR but it refers to those animals such as whales , seals and seabirds that depend on krill for food and those species that coexist in the ecosystem such as other species of krill.
The Commission implements management directives through Conservation Measures (CMs) , which are adopted by consensus. Currently, the krill fishery is managed through a series of such Conservation Measures relating to aspects of operation of the fishery including: precautionary catch limits , gear restrictions, data reporting, notification of intent to fish, minimisation of incidental mortality , observer deployment as well as measures regulating exploratory fisheries. Although there has not been a systematic approach to incorporating ecosystem considerations and precaution when managing the krill fishery, the Commission has addressed a range of issues and the overall result of this is to give some practical effect to the principles in Article II of the Convention (Table 11.3). These measures include rules that are specifically aimed at ensuring both ecosystem -based and precautionary management of the krill fishery as well as several measures which are commonly implemented by fisheries management regimes to collect basic fishery data including catch and effort statistics. Some of these measures are specific to the krill fishery whilst others apply across all fisheries operating in the Area.
11.4.2 Precautionary Catch Limits
The first Conservation Measure to specifically regulate the krill fishery (Conservation Measure 51-01 originally numbered 32/X setting precautionary catch limits on krill in Area 48) was adopted in 1991 some 10 years after the Convention was signed (Nicol 1991, 1992). This measure has been updated several times, most notably following the collection of new data from a synoptic survey in 2000 (Hewitt et al. 2004a) (see Chap. 2, Siegel and Watkins 2016). Precautionary catch limits have subsequently been established for Division 58.4.2 in 1992 (revised using new data collected in 2006), and for Division 58.4.1 in 1996 (Table 11.4). These precautionary catch limits are periodically updated using data from new biomass surveys and as a result of changes in the method by which the biomass is calculated from acoustic survey results. The adoption of the first precautionary catch limits for the krill fishery in Area 48, in 1991 (as Conservation Measure 32X) is heralded as a significant achievement in line with the precautionary approach (Fabra and Gascón 2008).
Precautionary catch limits for the krill fishery use data on the krill biomass which have been derived from dedicated acoustic surveys within the relevant CCAMLR statistical area . The original precautionary catch limits in Area 48 and Division 58.4.2 used the results of the BIOMASS surveys of the early 1980s which were the only acoustic surveys at an appropriate scale (Hewitt et al. 2004a). Because the BIOMASS data was not collected using CCAMLR -specific protocols there was a need to obtain more systematic acoustic data to ensure that precautionary catch limits were based on the most reliable information (Hewitt et al. 2004a). There have been three large-scale surveys (see Chap. 2, Siegel and Watkins 2016) that have been conducted for the purposes of setting precautionary catch limits : the BROKE survey of Division 58.4.1 in 1996 (Nicol et al. 2000), the CCAMLR 2000 survey in 2000 (Hewitt et al. 2004a) and the BROKE West survey of Division 58.4.2 in 2006 (Nicol et al. 2010).
Precautionary catch limits are calculated using an estimate of the potential yield of the krill population derived from the regional krill biomass estimates , data on natural mortality , individual growth rates and estimates of inter-annual recruitment variability . These estimates were calculated using the Krill Yield Model and later the Generalised Yield Model (Kock et al. 2007). These models take into account predator requirements through the mortality term and through a three part decision rule. This decision rule was developed for determining the proportion of the krill population (γ) which could be harvested each year with minimal risk, thus addressing Article II , 3 of the Convention (Constable et al. 2000; Miller 2002; Croxall and Nicol 2004).
(iii) Choose γ1 such that the probability of the median krill spawning stock biomass declining below 20 % of its pre-exploitation median level over a 20-year harvesting period is 10 %.
(ii) Choose γ2 so that the median krill spawning stock biomass after over 20 years of fishing is 75 % of the pre-exploitation median level.
(iii) Select the lower value of γ1 and γ2 as the level for γ for the calculation of krill yield.
The decision rules take explicit account of the needs of predators by allowing a krill escapement of 75 %. The revised yield model and the three-part decision rule were adopted by the Commission in 1994. These rules provided the first example of specifying the objectives of CCAMLR in scientifically interpretable and measurable terms (Constable et al. 2000).
A further level of precaution was added when CCAMLR introduced trigger levels in 1993. A trigger level is a level of catch (below the precautionary catch limit) which must not be exceeded until a mechanism to subdivide the overall catch limit amongst smaller management units has been agreed. Trigger levels have been employed to ensure that large krill catches are not taken from restricted areas. The trigger level of 620,000 tonnes set in CM 51-01, was derived from the aggregate of the highest annual commercial catches taken from each of the Subareas in Area 48 (SC-CCAMLR 2008, paragraph 3.33). Those in Division 58.4.2 were calculated on a pro rata basis using the proportions from Area 48 (CCAMLR 2008). These trigger levels are in place to ensure that the fishery develops in an orderly manner, with no irreversible impacts on predator species. In 2009 Conservation Measure 51-07 was adopted, providing for the interim distribution of the trigger level catch across Subareas 48.1, 48.2, 48.3 and 48.4. This measure was adopted to avoid the trigger level catch being taken from small areas, alleviating potential pressure on predator populations. The trigger level in Area 48 is set at a level that is only 1 % of the estimated biomass for this area of 60.3 million tonnes, (SC-CCAMLR 2010). The fishery, even if it is operating at the trigger level , would be unlikely to be a significant competitor for krill with the major consumers in the ecosystem (see Chap. 9, Trathan and Hill 2016).
In 2009, the adoption of two Conservation Measures – 51-04 and 51-05 (Table 11.3) served to further control the development of new krill fisheries in the CCAMLR Area. Essentially, the krill fishery now cannot expand into an unsurveyed area without an approved data collection plan and the catch in any unsurveyed subarea or Division is limited to 15,000 tonnes. Further, in these areas no more than 75 % of the catch limit can be taken within 60 n miles of known breeding colonies of land-based krill-dependent predators including seals , birds and penguins . The adoption of Conservation Measures 51-04 and 51-05 mean that there are limits on the krill fishery throughout the Convention Area either as a result of scientifically estimated precautionary catch limits or through regulations in un-surveyed areas . Prior to 1991, the krill fishery could operate in all regions of the CCAMLR area without limitation, by 1996 all the major fishing grounds were covered by precautionary catch limits and by 2009 krill fishing in the entire Convention Area was subject to catch limits or restrictions. CCAMLR has made considerable progress in managing the rational use of its largest fishery whilst allowing for the conservation of the world’s largest unexploited marine biomass.
11.4.3 Limiting By-Catch
CCAMLR ’s ecosystem approach to management of the krill fishery requires that the effects of fishing on dependent and related species are taken into account. This means that the direct effects of fishing on other species – bycatch and incidental mortality – are taken into account. A number of Conservation Measures have been adopted which aim to reduce the level of by-catch taken in krill fisheries, as well as to reduce the incidental mortality of seabirds and marine mammals in krill fishing operations (Table 11.3). There is still uncertainty whether the mortality of young and larval fish is a significant issue in the krill fishery and whether such mortality varies with space or time (Okuda and Kiyota 2012). Incidental mortality of seals , penguins and seabirds has never constituted a serious problem in the krill fishery but a number of Conservation Measures have been adopted to minimise fisheries impacts on birds and seals . Assessing larval fish mortality and recording incidental mortality of seals and seabirds are major tasks for scientific observers on krill fishing vessels (Sabourenkov and Appleyard 2005).
11.4.4 CCAMLR’s International Observer Scheme
The CCAMLR Scheme of International Scientific Observation was adopted in 1992 under Article XXIV of the Convention (Sabourenkov and Appleyard 2005). It is an important source of scientific information that is essential for assessing the impact of fishing on the ecosystem , including the status of target populations, as well as those of related and dependent species. The scheme also plays a crucial role in developing approaches to reducing the impact of fishing on the ecosystem by collecting data on the effectiveness of mitigation measures.
All vessels fishing in CCAMLR fisheries are required to carry an observer for some of their fishing operations. At CCAMLR meetings there has been considerable debate over the level of observer coverage in the krill fishery (Agnew et al. 2010) and despite the requirement for 100 % coverage by an international observer in all other CCAMLR fisheries the krill fishery has a target coverage of only 50 % using either international or national observers (CCAMLR 2014).
Scientific observers record information on the gear configuration (including measures to reduce incidental mortality of seabirds and marine mammals), fishing operations (including catch composition), the measurement of green weight and conversion factors and biological measurements of target and by-catch species. The absence of systematically-collected data collected from the krill fishery by recognised scientific observers has meant that it has been difficult to assess the significance of issues such as the bycatch of larval fish in the krill fishery (Agnew et al. 2010).
11.4.5 Krill Fishery Trends
CCAMLR has had persistent difficulties in obtaining information on potential future developments in the krill fishery. The Scientific Committee has repeatedly requested such information (Table 11.5). The absence of publicly available information on krill fishery developments led CCAMLR to formalise the process whereby Members notify their intention to fish for krill in the upcoming season.
11.4.6 Notifications
In 2006 CCAMLR adopted Conservation Measure 21-03 making it mandatory for Members to notify of their intention to fish for krill in the coming season. Member nations have to provide information on: the expected catch level, the area to be fished, details of the vessel and gear types being used to fish and the expected products to be derived from catch. This Measure was to aid the Commission in tracking development of the krill fishery in light of increasing interest in the fishery and in the absence of other tools for tracking the fishery’s development (CCAMLR 2006, paragraph 12.24). From 2003, the Commission had relied on an informal, voluntary system whereby Members would verbally inform the Scientific Committee on their plans for krill fishing in the upcoming season (CCAMLR 2003, paragraphs 4.37–4.39). This voluntary system resulted in little substantial information being submitted to allow the Commission to monitor market interest or fishery trends (Croxall and Nicol 2004).
Since Conservation Measure 21-03 was adopted there has been a marked change in the number and type of notifications being made to the Commission. There has been an increase in the tonnage of krill notified since the scheme was adopted (Fig. 11.8). However, there is a large discrepancy between the level of catch notified by Members and the actual catch that has occurred, with notified catches consistently being considerably higher than actual catches for the duration of this scheme. Although notifications have exceeded the trigger level of 620,000 tonnes for Area 48, actual catches have never reached even half this level in since 1991. In the 14 years prior to mandatory notifications , actual catches were higher than notified catches in 65 % of cases, resulting in an underestimation of the future krill catch. Since 2006, notifications have always been higher than actual catches. Additionally, the notification process has also led to predictions of a larger krill fishing fleet and projections of a larger number of nations being involved in the fishery. It appears that the notification process is being used by Member nations to keep their options open – they cannot fish unless they notify and the number of vessels and the projected catch are over-stated to retain operational flexibility (CCAMLR 2007, paragraph 3.25 and CCAMLR -2008, paragraphs 8.13–8.21). The discrepancy between notified and actual catches could result in an incorrect assumption of if, or when, the trigger level may be reached, as well as an increased workload for the Secretariat in processing inaccurate notifications . To prevent excessive notifications some Members have proposed that a fee be associated with lodging notifications and that a penalty apply for those vessels that notified but don’t fish (CCAMLR 2008, paragraph 8.19).
Krill catch and notification data from 1992 to 2014 (CCAMLR 2014). Note that mandatory notifications data was not required until the 2006 season
The notification process has not allowed CCAMLR to confidently predict the next season’s krill catch, the participants in the fishery nor the size of the fishing fleet. It has however, forced Member and non-Member nations to indicate their interest in the fishery. Further refinement of the process may allow the system to be both predictive as well as indicative.
11.4.7 Economic Data
Investment in products and technology that are specific to the krill fishery is evident in the number and type of krill-related patents being lodged (see Fig. 11.6) (Nicol and Foster 2003; Nicol et al. 2012; Foster et al. 2011). CCAMLR recognised that such information provided an insight into developments in the industry that could lead to future expansion of the krill fishery (SC-CCAMLR 1999, paragraphs 4.9–4.11). Patents and patent databases are increasingly being used for detecting investor interest in emerging technologies (Schiermeier 2010) and for identifying products and technologies relating to particular ingredients (Pilkington 2004). Patented technologies and products represent significant investment by the assignee in the krill industry (to lodge and maintain a patent is a costly process) and therefore can be used as an indicator of likely future activity in the krill marketplace. The CCAMLR Scientific Committee agreed that it would be useful if the Secretariat could maintain a patent database to provide annual updates on these trends (SC-CCAMLR 2009, paragraphs 4.10–4.12).
11.4.8 CCAMLR’s Management Structures
The CCAMLR Commission is the decision-making body that ultimately agrees on management measures relating to conservation and fisheries in the Convention Area. The Commission is advised by a Scientific Committee and these two bodies meet annually. The Scientific Committee, in turn, relies on the advice of specialised Working Groups one of which, the Working Group on Ecosystem Monitoring and Management (WG-EMM) , is concerned with the krill-based ecosystem and the krill fishery. It is at the annual meeting of WG-EMM that the development of management approaches to the krill fishery are proposed and discussed. CCAMLR and its subsidiary bodies are supported by a Secretariat, based in Hobart, Tasmania, which provides a wide range of services including data management and strategic research. The Secretariat produces an annual Krill Fishery Report which is presented at WG-EMM and provides information that includes: krill catches, notifications , scientific observations and bycatch . Information from these reports becomes available through the Reports of the Scientific Committee and on CCAMLR ’s website (www.ccamlr.org) and provides a valuable annual update on the status of the fishery.
11.5 Future Management of the Krill Fishery
11.5.1 Small Scale Management Units
There has also been recognition that management of the fishery needs to move from catch limits established for large scale areas to much smaller scale areas (Constable and Nicol 2002). In 1991, when the first Conservation Measure (32X) allocating a precautionary catch limit for Area 48 was adopted by the Commission, the need to consider distribution of precautionary catch limits across smaller spatial areas was first raised (CCAMLR 2008, paragraph 12.63 and Miller 2002). The trigger level in Area 48 has been subdivided (see above) but there is not yet agreement on a subdivision of the overall catch limit. There have been moves to subdivide the precautionary catch limit down to much smaller scales – small scale management units (SSMUs) – to avoid potential effects that concentrated fishing effort might have on the ecosystem (Hewitt et al. 2004b). Spatial boundaries of 15 SSMUs for Area 48 were established in 2002 (CCAMLR 2002, paragraph 4.5), but a mechanism for the subdivision of the precautionary catch limit across the SSMUs is yet to be agreed upon. A number of candidate schemes have been proposed and discussed by within CCAMLR (Hewitt et al. 2004b):
-
1.
the spatial distribution of historical catches by the krill fishery;
-
2.
the spatial distribution of predator demand;
-
3.
the spatial distribution of krill biomass;
-
4.
the spatial distribution of krill biomass minus predator demand;
-
5.
spatially explicit indices of krill availability that may be monitored or estimated on a regular basis; and
-
6.
structured fishing strategies in which catches are rotated within and between SSMUs .
Because there has been no agreement on a method for subdividing the precautionary catch across the SSMUs , in 2009 the Commission adopted an interim measure which divides the precautionary catch amongst four of the most commonly fished Subareas in Area 48 (Conservation Measure 51-07). The trigger level for each subarea was based on the proportion of the krill biomass found in each subarea in the CCAMLR 2000 survey : subarea 48.1: 155,000 tonnes, subarea 48.2: 279,000 tonnes, Subarea 48.3: 297,000 tonnes and Subarea 48.4: 93,000 tonnes (SC-CCAMLR 2009). This subdivision has resulted in the closure of the krill fishery four times when the subdivided trigger level (155,000 tonnes per year) for subarea 48.1 has been reached. CM 51-07 was renewed in 2011 and for a further two years in 2014.
11.5.2 Feedback Management
The current management regime for the krill fishery has always been viewed as an interim scheme that would remain in place until a more elaborate and comprehensive management approach was developed (Nicol and de la Mare 1993). The long-term aim of CCAMLR has been to the development of a feedback management procedure for the krill fishery (adaptive management whereby management measures are regularly adjusted based on ecosystem monitoring indices) (Nicol and de la Mare 1993; Fabra and Gascón 2008). This system would see management measures regularly changed in response to continually updated information from the ecosystem , aimed at detecting impacts from fishing and responding accordingly, before irreversible change occurs (Constable et al. 2000; Constable 2011; Hill and Canon 2013). The development of a feedback management procedure for a remote fishery such as that for Antarctic krill is a complex process and CCAMLR has recognised that its implementation will be difficult, agreeing to a staged approach (CCAMLR 2014). In the interim, a number of other measures are being explored to ensure that the management process uses the maximum amount of information available.
11.5.3 Integrated Assessments
Catch limits on the krill fishery have been established using the results of very large scale acoustic surveys of krill biomass. Such surveys are extremely labour intensive, expensive and take considerable planning and analysis. CCAMLR is considering a move from such basin-wide biomass surveys to an integrated stock assessment (SC-CCAMLR 2014, paragraph 3.52). An integrated krill stock assessment model could be used to provide regular stock assessments based on data from various sources, including: scientific surveys, fishing vessel surveys, observers and the CCAMLR Ecosystem Monitoring Program (CEMP). Robust estimates of the consumption of krill by predators would help to scale biomass estimates . The model would account for differences in the timing of different data sources, and gear-specific selectivity.
11.5.4 Research from Krill Fishing Vessels
Until very recently, most of the research required for the management of the krill fishery was obtained using fisheries-independent surveys (Nicol et al. 2000, 2010, and Hewitt et al. 2004a). There are few research vessels operating in Antarctic waters at any one time and krill research is only one of a number of priority areas for study. Additionally, research vessels are usually limited to studies of 1–2 months in length. Fishing vessels, on the other hand, operate throughout most of the year and sample krill throughout the day (Kawaguchi and Nicol 2007). An International Scientific Observer scheme has been operated by CCAMLR since its inception but scientific observer coverage on krill fishing vessels has been patchy and no information from this scheme has yet been used for management purposes. Recently, CCAMLR has recognised that obtaining information from krill fishing vessels will be essential for future management of the fishery (Godø et al. 2014). Regular, dedicated acoustic surveys using krill fishing vessels have already commenced (Krafft et al. 2011) but there is far more research that can be conducted from the commercial fleet. Suggestions for future research include studies into basic biology of krill, biochemistry of krill and oceanography of the krill fishing grounds . There are even suggestions for the dedicated use of krill trawlers for research purposes and for the construction of hybrid krill fishing-research vessels (Godø et al. 2014). Information from fishing vessels is seen as crucial in the development of feedback management for the krill fishery (SC-CCAMLR 2014 paragraph 3.34).
11.5.5 Industry-Based Approaches to Management
There has been a recent trend in global fisheries for industry to adopt measures over and above those imposed by management bodies such as CCAMLR (Washington and Ababouch 2011). These approaches include third party certification and initiatives that demonstrate the credentials of the companies or industry in pursuing sustainable harvesting. Fisheries throughout the world are confronted with increased market demands to document conformity in relation to the principles of sustainable harvesting. Scientific knowledge and documentation of the ecosystem effects of fisheries is essential for many management bodies, and also within the market place. The achievements of the Marine Stewardship Council (MSC) certification process for marine products indicates how scientifically-based information and documentation directly affects the product price and market access.
The Norwegian company that currently harvests the greatest tonnage of krill, Aker Biomarine, was awarded MSC certification for their “Eco-Harvesting” krill fishing activities (Aker 2010). This certification is awarded to fisheries that undertake their harvesting operations in an environmentally sustainable manner (MSC 2010). Aker uses this certification as a marketing tool that sets its Qrill™ and Superba™ products apart from competitor’s products as Aker at the time was the only company with the certification for their krill fishing activities. Jacquet and Pauly (2010) have raised concerns that the certification process overlooked reports from the scientific community that krill stocks are declining and ignored the unsustainable use of krill in fishmeal although these concerns were subsequently addressed by the certifying body (MSC 2010). In 2014 the Aker krill fishery was re-assessed and was re-certified (MSC ) with no conditions placed on the company and no adverse comments received on the certification (Schulz 2014). A second Norwegian company, Olympic fisheries, is currently undergoing MSC assessment (MSC 2015). Although the MSC process addresses krill fishing by company and by vessel their assessments do reflect the overall state of the fishery and its management .
In 2010 several krill fishing companies formed the Association of Responsible Krill Fishing Companies (ARK) with the active involvement of the Worldwide Fund for Nature (WWF) . ARK coordinates and cooperates with CCAMLR on the provision of research and information on krill, and the krill fishery and its impact on the ecosystem , with the aim of contributing to CCAMLR’s work on managing the krill fishery sustainably (ARK 2015). This initiative arose out of a recognition that the safety of long-term investment in the krill industry would lie in it being well managed and been seen to be well managed. In 2015 ARK had member companies from Norway (2), Chile and Korea and has been an official observer at CCAMLR meetings since 2011. It has begun the process of involving the krill fishing industry in the development of feedback management approach (SC-CCAMLR 2013, paragraphs 10.12–10.15). ARK members currently catch ~80 % of the krill harvested in the Southern Ocean and ARK is in a position to be influential in the future development of the krill fishery.
11.5.6 Environmental Footprint of Krill Fishery
Distant-water fishing operations are frequently criticised for the resources they consume and the emissions they produce. The Aker Biomarine krill fishery was subjected to a Life Cycle Analysis to quantify and characterize life cycle environmental burdens associated with their fishing and processing operations (Parker and Tyedmers 2012). The Fuel Use Intensity (litres per tonne of landed catch) of Antarctic krill is relatively low when compared to high-value species such as cod, tuna or lobster, but much higher than for other fisheries targeting species for reduction into meal and oil e.g. ten times that of the Peruvian anchovy fishery. The fuel consumption by a resupply vessel used to transport meal and oil from the fishing vessel to port accounts for roughly the same scale of energy use and emissions as fuel consumption on the fishing vessel. This suggests that the distance between the fishing vessel and port plays a particularly important role in the life cycle environmental impacts of krill products.
11.5.7 Climate Change and the Krill Fishery
The life history and population dynamics of Antarctic krill are likely to be impacted by a changing climate that results from increasing levels of CO2 in the atmosphere (Flores et al. 2012), and may result in a reduced habitat range for Antarctic krill (Hill et al. 2013). The reproductive output and recruitment success of krill has been related to the extent, timing and duration of winter sea ice cover (Kawaguchi and Satake 1994, Quetin and Ross 2003). Krill growth has also been observed to decrease above a temperature optimum of 0.5° C (Atkinson et al. 2006). The early developmental stage of krill will be vulnerable to the increased levels of CO2 projected to occur within their habitat range to the year 2100 and beyond (Kawaguchi et al. 2013). Overall, the cumulative impact of climate change is most likely to be negative. Changes in the distribution and abundance of krill as a result of climate change are, obviously, likely to affect the fishery. Already the effect of changes in sea ice extent and seasonality have changed the behaviour of the fishing fleets (Kawaguchi et al. 2009). It is uncertain whether a warming surface layer may affect the vertical distribution of krill (Schmidt et al. 2011) but the fishing fleet is already fishing much deeper in some areas (Kawaguchi et al. 2006).
11.6 Conclusions
The krill fishery has been operating for over 40 years in Antarctic waters with nearly 8 million tonnes being harvested during this period. Despite concerns over the effects of the krill fishery on the Southern Ocean ecosystem , there have been no observations of an effect of the harvesting of krill on dependent or related species. This lack of an effect can be traced to the level of fishing remaining low compared to the large biomass and because of the precautionary management regime implemented by CCAMLR . There is currently unprecedented interest in krill as a source of marine proteins and oils and more nations are involved in the fishery than in the past. The future of the fishery, and of the ecosystem , will be played out within CCAMLR and this organisation will either succeed or fail based on its ability to effectively manage its largest fishery.
References
Adelung D, Buchholz F, Culik B, Keck A (1987) Fluoride in tissues of krill Euphausia superba Dana and Meganyctiphanes norvegica M. Sars in relation to the moult cycle. Polar Biol 7:43–50
Agnew DJ (2004) Fishing south: the history and management of South Georgia fisheries. Government of South Georgia
Agnew DJ, Grove P, Peatman T, Burn R, Edwards CTT (2010) Estimating optimal observer coverage in the Antarctic krill fishery. CCAMLR Sci 17:139–154
Aker (2010) www.akerbiomarine.com. Accessed 23rd July 2010
ARK (2015) www.ark-krill.org/. Accessed Feb 2015
Arthi V (2009) Omega 3 concentrates – window of opportunities. Frost & Sullivan Market Insight, 27 Aug 2009. http://www.frost.com/prod/servlet/market-insight-top.pag?docid=178244151. Accessed 2nd Nov 2009
Atkinson A, Shreeve RS, Hirst AG, Rothery P, Tarling GA, Pond DW, Korb RE, Murphy EJ, Watkins JL (2006) Natural growth rates in Antarctic krill (Euphausia superba): II. Predictive models based on food, temperature, body length, sex, and maturity stage. Limnol Oceanogr 51(2):973–987
Atkinson A, Siegel V, Pakhomov EA, Jessopp MJ, Loeb V (2009) A re-appraisal of the total biomass and annual production of Antarctic krill. Deep-Sea Res I Oceanogr Res Pap 56(5):727–740
Backes JM, Howard PA (2014) Krill oil for cardiovascular risk prevention: is it for real? Hosp Pharm 49(10):907–912
Batetta B, Griinari M, Carta G, Murru E, Ligresti A, Cordeddu L, Giordano E, Sanna F, Bisogno T, Uda S, Collu M, Bruheim I, Di Marzo V, Banni S (2009) Endocannabinoids may mediate the ability of (n-3) fatty acids to reduce ectopic fat and inflammatory mediators in obese zucker rats. J Nutr 139(8):1495–1501
Bridges KM, Gigliotti JC, Altman S, Jaczynski J, Tou JC (2010) Determination of digestibility, tissue deposition and metabolism of the omega-3 fatty acid content of krill protein concentrate in growing rats. J Agric Food Chem 58:2830–2837
Budzinski E, Bykowski P, Dutkiewicz D (1985) Possibilities of processing and marketing of products made from Antarctic krill, FAO fisheries technical paper no. 268. FAO, Rome, p. 46p
Bunea R, Farrah KE, Deutsch L (2004) Evaluation of the effects of Neptune Krill Oil on the clinical course of hyperlipidemia. Altern Med Rev 9(4):420–428
Bustos R, Romo L, Yáñez K, Díaz G, Romo C (2003) Oxidative stability of carotenoid pigments and polyunsaturated fatty acids in microparticulate diets containing krill oil for nutrition of marine fish larvae. J Food Eng 56:289–293
CCAMLR (2002) Report of the twenty-first meeting of the commission. CCAMLR, Hobart
CCAMLR (2003) Report of the twenty-second meeting of the commission. CCAMLR, Hobart
CCAMLR (2006) Report of the twenty-fifth meeting of the commission. CCAMLR, Hobart
CCAMLR (2007) Report of the twenty-sixth meeting of the commission. CCAMLR, Hobart
CCAMLR (2008) Report of the twenty-seventh meeting of the commission. CCAMLR, Hobart
CCAMLR (2009a) www.ccamlr.org. Accessed 1st Oct 2009
CCAMLR (2009b) Report of the twenty-eighth meeting of the commission. CCAMLR, Hobart
CCAMLR (2014) Report of the thirty-third meeting of the commission. CCAMLR, Hobart
CCAMLR (2015) Stat Bull, 26 http://www.ccamlr.org/en/data/statistical-bulletin
CCAMLR-WG-EMM (2014) Report of the working group on ecosystem monitoring and management (Punta Arenas, Chile, 7 to 18 July 2014)
Chandrasekar B, Troyer DA, Venkatraman JT, Fernandes G (1996) Tissue specific regulation of transforming growth factors beta by omega-3 lipid-rich krill oil in autoimmune murine lupus. Nutr Res 16(3):489–503
Chi H, Li X, Yang X (2013) Processing status and utilisation strategies of Antarctic krill (Euphausia superba) in China. World J Fish Mar Sci 5(3):275–281
Constable AJ (2011) Lessons from CCAMLR on the implementation of the ecosystem approach to managing to fisheries. Fish Fish 12(2):138–151
Constable AJ, Nicol S (2002) Defining smaller-scale management units to further develop the ecosystem approach in managing large-scale pelagic krill fisheries in Antarctica. CCAMLR Sci 9:117–131
Constable AJ, de la Mare WK, Agnew DJ, Everson I, Miller D (2000) Managing fisheries to conserve the Antarctic marine ecosystem: practical implementation of the Convention on the Conservation of Antarctic Marine Living Resources (CCAMLR). ICES J Mar Sci 57:778–791
Croxall JP, Nicol S (2004) Management of Southern Ocean fisheries: global forces and future sustainability. Antarct Sci 16(4):569–584
Davis DA, Arnold CR (2000) Replacement of fish meal in practical diets for the pacific white shrimp, Liptopenaeus vannamei. Aquaculture 185:291–298
Deutsch L (2007) Evaluation of the effect of Neptune Krill Oil on chronic inflammation and arthritic symptoms. J Am Coll Nutr 26(1):39–48
Duan J, Jiang Y, Cherian G, Zhao Y (2010) Effect of combined chitosan-krill oil coating and modified atmosphere packaging on the storability of cold-stored lingcod (Ophiodon elongates). Food Chem 122(4):1035–1042
Eddie GC (1977) The harvesting of krill. Southern Ocean Fisheries Survey Programme. FAO, Rome, GLO/SO/77/2, 76p
Edwards DM, Heap JA (1981) Convention on the conservation of Antarctic marine living resources: a commentary. Polar Rec 20(127):353–362
El-Sayed SZ (ed) (1977) Biological investigations of marine Antarctic systems and stocks (BIOMASS), vol 1. Scott Polar Research Institute, Cambridge
El-Sayed SZ (ed) (1994) Southern Ocean ecology: the BIOMASS perspective. Cambridge University Press, Cambridge
Everson I (1977) The living resources of the Southern Ocean. Southern Ocean Fisheries Survey Programme. FAO, Rome, GLO/SO/77/1, 156p
Fabra A, Gascón V (2008) The Convention on the Conservation of Antarctic Marine Living Resources (CCAMLR) and the ecosystem approach. Int J Mar Coast Law 23:567–598
FAO (2008) The state of world fisheries and aquaculture. FAO Fisheries and Aquaculture Department, Rome. 178p
Fereidoon S, Wanasundara UN (1998) Omega-3 fatty acid concentrates: nutritional aspects and production technologies. Trends Food Sci Technol 9:230–240
Fernholm B, Rudback GT (1989) Marine resource management of the Antarctic. Ambio 18(1):68–70
Flores H, Atkinson A, Kawaguchi S, Krafft BA, Milinevsky G, Nicol S, Reiss C, Tarling GA, Werner R, Bravo Rebolledo E, Cirelli V, Cuzin-Roudy J, Fielding S, Groeneveld JJ, Haraldsson M, Lombana A, Marschoff E, Meyer B, Pakhomov EA, Rombolá E, Schmidt K, Siegel V, Teschke M, Tonkes H, Toullec JY, Trathan PN, Tremblay N, Van de Putte AP, van Franeker JA, Werner T (2012) Impact of climate change on Antarctic krill. Mar Ecol Prog Ser 458:1–19
Floreto EAT, Brown PB, Bayer RC (2001) The effects of krill hydrolysate-supplemented soy-bean based diets on the growth, colouration, amino and fatty acids profiles of juvenile American lobster, Homarus americanus. Aquac Nutr 7:33–43
Foster J, Nicol S, Kawaguchi S (2011) The use of patent databases to detect trends in the krill fishery. CCAMLR Sci 18:135–144
Frank RF (1983) The convention on the conservation of Antarctic marine living resources. Ocean Dev Int Law J 13(3):291–345
Gaber MMA (2005) The effect of different levels of krill meal supplementation of soybean-based diets on feed intake, digestibility, and chemical composition of juvenile Nile Tilapia Oreochromis niloticus, L. J World Aquacult Soc 36(3):346–353
Giogios I, Grigorakis K, Nengas I, Papasolomontos S, Papaioannou N, Alexis MN (2009) Fatty acid composition and volatile compounds of selected marine oils and meals. J Sci Food Agric 89:88–100
Godø OR, Reiss C, Siegel V, Watkins JL (2014) Commercial fishing vessels as research vessels in the Antarctic – requirements and solutions exemplified with a new vessel. CCAMLR Sci 21:11–1
Grantham GJ (1977) The utilization of Krill. Southern Ocean Fisheries Survey Programme. FAO, Rome: GLO/SO/77/3 61p
Grave H (1981) Fluoride content of salmonids fed on Antarctic krill (Euphausia superba). Aquaculture 24:191–196
Hewitt RP, Low EHL (2000) The fishery on Antarctic Krill: defining an ecosystem approach to management. Rev Fish Sci 8(3):235–298
Hewitt RP, Watkins J, Naganobu M, Sushin V, Brierley AS, Demer D, Kasatkina S, Takao Y, Goss C, Malyshko A, Brandon M, Kawaguchi S, Siegel V, Trathan P, Emery J, Everson I, Miller D (2004a) Biomass of Antarctic krill in the Scotia Sea in January/February 2000 and its use in revising an estimate of precautionary yield. Deep-Sea Res II Top Stud Oceanogr 51(12):1215–1236
Hewitt RP, Watters G, Trathan PN, Croxall JP, Goebel ME, Ramm D, Reid K, Trivelpiece WZ, Watkins JL (2004b) Options for allocating the precautionary catch limit of krill among small-scale management units in the Scotia Sea. CCAMLR Sci 11:81–97
Hill SL (2013) Prospects for a sustainable increase in the availability of long chain omega 3s: lessons from the Antarctic Krill fishery. In: Omega-6/3 fatty acids. Humana Press, New York, pp 267–296
Hill SL, Cannon M (2013) A potential feedback approach to ecosystem-based management: model predictive control of the Antarctic krill fishery. CCAMLR Sci 20:119–137
Hill SL, Phillips T, Atkinson A (2013) Potential climate change effects on the habitat of Antarctic krill in the Weddell quadrant of the Southern Ocean. PLoS One 8(8):e72246. doi:10.1371/journal.pone.0072246
Howard M (1989) The convention on the conservation of Antarctic marine living resources: a five-year review. Int Comp Law Q 38:104–149
Ichii T (2000) Krill harvesting. In: Everson I (ed) Krill biology, ecology and fisheries. Blackwell Science, Oxford, pp 228–262
Jacquet J, Pauly D (2010) Seafood stewardship in crisis. Nature 467(2):28–29
Julsham K, Malde MK, Bjorvatn K, Krogedal P (2004) Fluoride retention of Atlantic salmon (Salmo salar) fed krill meal. Aquac Nutr 10(1):9–13
Jung HR, Kim MA, Seo YS, Lee YB, Chun BS, Kim SB (2013) Decreasing effect of fluoride content in Antarctic krill (Euphausia superba) by chemical treatments. Int J Food Sci Technol 46(6):1252–1259
Kalinowski CT, Izquierdo MS, Schuchardt LE (2007) Dietary supplementation time with shrimp shell meal on red porgy (Pagrus pagrus) skin colour and carotenoid concentration. Aquaculture 272:451–457
Karlsen Ø, Suontama J, Olsen RE (2006) Effect if Antarctic krillmeal on quality of farmed Atlantic cod (Gadus morhua L.). Aquac Res 37(16):1676–1684
Karlstam B, Vincen J, Johansson B, Bryno C (1991) A simple purification method of squeezed krill for obtaining high levels of hydrolytic enzymes. Prep Biochem 21:237–256
Kawaguchi S, Nicol S (2007) Learning about Antarctic krill from the fishery. Antarct Sci 19(2):219–230
Kawaguchi S, Satake M (1994) Relationship between recruitment of the Antarctic Krill and the degree of ice cover near the South Shetland Islands. Fish Sci 60(1):123–124
Kawaguchi S, Ichii T, Naganobu M (1999) Green krill, the indicator of micro-and nano-size phytoplankton availability to krill. Polar Biol 22(2):133–136
Kawaguchi S, Nicol S, Taki K, Naganobu M (2006) Fishing ground selection in the Antarctic krill fishery: trends in patterns across years, seasons and nations. CCAMLR Sci 13:117–141
Kawaguchi S, Nicol S, Press AJ (2009) Direct effects of climate change on the Antarctic krill fishery. Fish Manag Ecol 16:424–427
Kawaguchi S, Ishida A, King R, Raymond B, Waller N, Constable A, Nicol S, Wakita M, Ishimatsu A (2013) Risk maps for Antarctic krill under projected Southern Ocean acidification. Nat Clim Chang 3(9):843–847
Kidd PM (2009) Integrated brain restoration after ischemic stroke – medical management, risk factors, nutrients, and other interventions for managing inflammation and enhancing brain plasticity. Altern Med Rev 14(1):14–35
Kim M-A, Jung H-R, Lee Y-B, Chun B-S, Kim SB (2014) Monthly variations in the nutritional composition of Antarctic krill Euphausia superba. Fish Aquat Sci 17(4):409–419
Kock K-H, Reid K, Croxall J, Nicol S (2007) Fisheries in the Southern Ocean: an ecosystem approach. Philos Trans R Soc B 362:2333–2349
Krafft BA, Skaret G, Calise L (2011) Antarctic krill and apex predators in the South Orkney Islands area 2011, surveyed with the commercial fishing vessel Saga Sea. Rapport fra Havforskningen; Nr. 6–2011
Lyubimova TC, Naumov AG, Lagunov LL (1973) Prospects of the utilisation of krill and other unconventional resources of the world ocean. J Fish Res Board Can 30(12/2):2196–2201
Maguire J-J, Sissenwine M, Csirke J, Grainger R, Garcia S (2006) The state of world highly migratory, straddling and other high seas fishery resources and associated species, FAO fisheries technical paper no. 495. FAO, Rome. 84p
McElroy JK (1984) Antarctic fisheries. History and prospects. Mar Policy 8(3):239–259
Melrose J, Hall A, Macpherson C, Bellenger CR, Ghosh P (1995) Evaluation of digestive proteinases from the Antarctic krill Euphausia superba as potential chemonucleolytic agents. In vitro and in vivo studies. Arch Orthop Trauma Surg 114:145–152
Miller DGM (1991) Exploitation of Antarctic marine living resources: a brief history and a possible approach to managing the krill fishery. S Afr J Mar Sci 10:321–339
Miller DGM (2002) Antarctic krill and ecosystem management—from Seattle to Siena. CCAMLR Sci 9:175–2002
Miller D, Agnew D (2000) Management of krill fisheries in the Southern Ocean. In: Everson I (ed) Krill biology, ecology and fisheries. Blackwell Science, Oxford, pp 300–337
Moreira AC, Muller ACA, Pereira N, Antunes A (2006) Pharmaceutical patents on plant derived materials in Brazil: policy, law and statistics. World Patent Inf 28:34–42
Moretti VM, Mentasti T, Bellagamba F, Luzzana U, Caprino GM, Giani I, Valfrè F (2006) Determination of astaxanthin stereoisomers and colour attributes in flesh of rainbow trout (Oncorhynchus mykiss) as a tool to distinguish the dietary pigmentation source. Food Addit Contam 23(11):1056–1063
MSC (2010) www.msc.org. Marine Stewardship Council. Accessed 3 Aug 2010
MSC (2015) www.msc.org/track-a-fishery/fisheries-in-the-program/in-assessment/southern-ocean/olympic-seafood-antarctic-krill. Accessed Feb 2015
Naczk M, Synowiecki J, Sikorski ZE (1981) The gross chemical composition of Antarctic krill waste. Food Chem 7:175–179
Nichols PD (2007) Fish oil sources. In: Breivik H (ed) Long-chain omega-3 speciality oils. The Oily Press, Cambridge, p 314
Nichols PD, Nelson MM (2007) Marine oils in Australasia. In: O’Connor, J (Chairman) Handbook of Australasian edible oils. Oils and Fats Specialist Group of NZIC. Auckland, p 297
Nicol S (1991) CCAMLR and its approaches to management of the krill fishery. Polar Rec 27(162):229–236
Nicol S (1992) Management of the krill fishery; was CCAMLR slow to act? Polar Rec 28(165):155–157
Nicol S, De la Mare W (1993) Ecosystem management and the Antarctic krill. Am Sci 81(1):36
Nicol S, Endo Y (1997) Krill fisheries of the world, FAO fisheries technical paper no. 367. FAO, Rome, p 81
Nicol S, Endo Y (1999) Krill fisheries – their development, management and ecosystem implications. Aquat Living Resour 12(2):105–120
Nicol S, Foster J (2003) Recent trends in the fishery for Antarctic krill. Aquat Living Resour 16:42–45
Nicol S, Hosie GW (1993) Chitin production by Krill. Biochem Syst Ecol 21(2):181–184
Nicol S, Pauly T, Bindoff NL, Strutton PG (2000) “BROKE” a biological/oceanographic survey off the coast of East Antarctica (80–150°E) carried out in January–March 1996. Deep- Sea Res II 47(12/13):2281–2297
Nicol S, Meiners K, Raymond B (2010) Editorial. BROKE-West, a large ecosystem survey of the South West Indian Ocean sector of the Southern Ocean 30–80°E (CCAMLR Division 58.4.2). Deep-Sea Res II 57(9–10):693–700
Nicol S, Foster J, Kawaguchi S (2012) The fishery for Antarctic krill – recent developments. Fish Fish 13(1):30–40
Oehlenschläger J, Schreiber W (1981) A functional protein concentrate (FKPC) from Antarctic Krill (Euphausia superba, Dana 1850). Z Lebensm Unters Forsch 172:393–398
Okuda T, Kiyota M (2012) Analysis of variability of krill size and fish by-catch in the Japanese krill fishery based on scientific observer data. CCAMLR Sci 19:31–47
Opstad I, Suontama J, Langmyhr E, Olsen RE (2006) Growth, survival and development of Atlantic cod (Gadus morhua L.) weaned onto diets containing various sources of marine protein. ICES J Mar Sci 63:320–325
Parker RW, Tyedmers PH (2012) Life cycle environmental impacts of three products derived from wild-caught Antarctic krill (Euphausia superba). Environ Sci Technol 46(9):4958–4965
Pilkington A (2004) Technology portfolio alignment as an indicator of commercialisation: an investigation of fuel cell patenting. Technovation 24:761–771
Quetin LB, Ross RM (2003) Episodic recruitment in Antarctic krill Euphausia superba in the Palmer LTER study region. Mar Ecol Prog Ser 259:185–200
Sabourenkov EN, Appleyard E (2005) Scientific observations in CCAMLR fisheries – past, present and future. CCAMLR Sci 12:81–98
Savage GP, Foulds MJ (1987) Chemical composition and nutritive value of Antarctic krill (Euphausia superba) and southern blue whiting (Micromesistius australis). N Z J Mar Freshw Res 21:599–604
SC-CCAMLR (1999) Report of the eighteenth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2000) Report of the nineteenth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2001) Report of the twentieth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2002) Report of the twenty-first meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2004) Report of the twenty-third meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2005) Report of the twenty-fourth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2008) Report of the twenty-seventh meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2009) Report of the twenty-eighth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2010) Report of the twenty-ninth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2011) Report of the thirtieth meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2013) Report of the thirty-second meeting of the scientific committee. CCAMLR, Hobart
SC-CCAMLR (2014) Report of the thirty-third meeting of the scientific committee. CCAMLR, Hobart
Schiermeier Q (2010) Green patents corralled. Nature 465(6):21
Schmidt K, Atkinson A, Steigenberger S, Fielding S, Lindsay MCM, Pond DW, Tarling GA, Klevjer TA, Allen CS, Nicol S, Achterberg EP (2011) Seabed foraging by Antarctic krill: implications for stock assessment, bentho-pelagic coupling, and the vertical transfer of iron. Limnol Oceanogr 56(4):1411–1428
Schulz H (2014) Nutra-Ingrediants USA.com. Aker says MSC recertification eliminates any lingering doubts on sustainability of krill. http://www.nutraingredients-usa.com/Suppliers2/Aker-says-MSC-recertification-eliminates-any-lingering-doubts-on-sustainability-of-krill. Accessed Feb 2015
Shahidi F, Wanasundara UN (1998) Omega-3 fatty acid concentrates: nutritional aspects and production technologies. Trends Food Sci Technol 9:230–240
Sidhu GS, Montgomery WA, Holloway GL, Johnson AR, Walker DM (1970) Biochemical composition and nutritive value of krill (Euphausia superba Dana). J Sci Food Agric 21:293–296
Siebert G, Kühl J, Hannover R (1980) Nutritional experiments with krill. In: Connell JJ (ed) Advances in fish science and technology. Blackwell Scientific Publications Ltd, London, pp 332–334
Siegel V, Watkins J (2016) Distribution, biomass and demography of Antarctic krill, Euphausia superba. In: Siegel V (ed) Biology and ecology of Antarctic krill. Springer, Cham, pp 21–100
Smith DM, Tabrett SJ, Barclay MC, Irvin SJ (2005) The efficacy of ingredients included in shrimp feeds to stimulate uptake. Aquac Nutr 11:263–272
Soevik T, Braekkan OR (1979) Fluoride in Antarctic Krill (Euphausia superba) and Atlantic Krill (Meganyctiphanes norvegica). J Fish Res Board Can 36:1414–1416
Suontama J, Kiessling A, Melle W, Waagbø R, Olsen RE (2007) Protein from Northern krill (Thysanoessa inermis), Antarctic krill (Euphausia superba) and the Arctic amphipod (Themisto libellula) can partially replace fish meal in diets to Atlantic salmon (Salmo salar) without affecting product quality. Aquac Nutr 13:50–58
Tandy S, Chung RWS, Wat E, Kamili A, Berge K, Griinari M, Cohn JS (2009) Dietary krill oil supplementation reduces hepatic steatosis, glycaemia, and hypercholesterolemia in high-fat-fed mice. J Agric Food Chem 57:9339–9345
Tenuto-Filho A (1993) Fluorine removal during production of krill paste and protein concentrates. Acta Alimaentaria 22:269–281
Tenuto-Filho A, Alvarenga RCC (1999) Reduction of the bioavailability of fluoride from Antarctic krill by calcium. Int J Food Sci Nutr 50:297–302
Tou JC, Jaczynski J, Chen-Chen Y (2007) Krill for human consumption: nutritional value and potential health benefits. Nutr Rev 65(2):63–77
Trathan PN, Hill S (2016) The importance of krill predation in the Southern Ocean. In: Siegel V (ed) Biology and ecology of Antarctic krill. Springer, Cham, pp 321–350
Voronia NM (1983) Biomass and production of Antarctic krill (Euphausia superba Dana). Oceanology 23(6):760–762
Wang Z, Yin X (2012) Comparison of the defluoridation efficiency of calcium phosphate and chitin in the exoskeleton of Antarctic krill. Adv Polar Sci 23(3):149–154
Wang L, Xue C, Wang Y, Yang B (2011) Extraction of proteins with low fluoride level from Antarctic krill (Euphausia superba) and their composition analysis. J Agric Food Chem 59(11):6108–6112
Washington S, Ababouch L (2011) Private standards and certification in fisheries and aquaculture: current practice and emerging issues. Food and Agriculture Organization of the United Nations, Rome
Xie CL, Kim HS, Shim KB, Kim YK, Yoon NY, Kim PH, Yoon HO (2012) Organic acid extraction of fluoride from Antarctic Krill Euphausia superba. Fish Aquat Sci 15(3):203–207
Yanase M (1981) Chemical composition of the exoskeleton of Antarctic krill. Bull Tokai Reg Fish Res Lab 83:1–6
Yoshitomi B, Aoki M, Oshima S (2007) Effect of total replacement of dietary fish meal by low fluoride krill (Euphausia superba) meal on growth performance of rainbow trout (Oncorhynchus mykiss) in fresh water. Aquaculture 266:219–225
Zhang L, Lu X, Wang Z, Qin L, Lin Z, Yuan L, Zhang W, Yin X (2014) Evaluation of the biological toxicity of fluorine in Antarctic krill. Adv Polar Sci 25(1):38–45
Acknowledgements
We would like to thank the CCAMLR Secretariat, in particular Drew Wright, Keith Reid, Lucy Robinson and David Ramm, for their assistance and patience.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Nicol, S., Foster, J. (2016). The Fishery for Antarctic Krill: Its Current Status and Management Regime. In: Siegel, V. (eds) Biology and Ecology of Antarctic Krill. Advances in Polar Ecology. Springer, Cham. https://doi.org/10.1007/978-3-319-29279-3_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-29279-3_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-29277-9
Online ISBN: 978-3-319-29279-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)