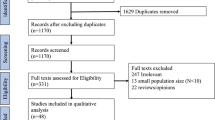
Abstract
Introduction: Delayed cerebral ischemia (DCI) is a major contributor to morbidity and mortality after subarachnoid hemorrhage (SAH). Data challenge vasospasm being the sole cause of ischemia and suggest other factors. We tested the hypothesis that early autoregulatory failure might predict DCI. Methods: This is a prospective observational study of cerebral autoregulation following SAH in which the primary end point was DCI at 21 days. Cox proportional hazards and multivariate models were used and the benefit of using multiple indices was analyzed. Results: Ninety-eight patients were included in the study. There was an increased risk of DCI with early dysautoregulation (odds ratio [OR]: 7.46, 95% confidence interval [CI]: 3.03–18.40 and OR: 4.52, 95 % CI: 1.84–11.07 for the transcranial Doppler index of autoregulation [Sxa] and near-infrared spectroscopy index of autoregulation [TOxa], respectively), but not vasospasm (OR: 1.36, 95 % CI: 0.56–3.33). Sxa and TOxa remained independent predictors of DCI in the multivariate model (OR: 12.66, 95 % CI: 2.97–54.07 and OR: 5.34, 95 % CI: 1.25–22.84 for Sxa and TOxa, respectively). There was good agreement between different indices. All 13 patients with impaired autoregulation in all three methods developed DCI. Conclusions: Disturbed autoregulation in the first 5 days after SAH is predictive of DCI. Although colinearities exist between the methods assessed, multimodal monitoring of cerebral autoregulation can aid the prediction of DCI.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cerebral autoregulation
- Delayed cerebral ischemia
- Near-infrared spectroscopy
- Prediction
- Subarachnoid hemorrhage
- Transcranial Doppler
Introduction
Delayed cerebral ischemia (DCI) is a major contributor to morbidity and mortality after subarachnoid hemorrhage (SAH) [5]. Data challenge the concept of vasospasm being the sole cause of ischemia following SAH [3, 9] and support disturbed cerebral autoregulation being a factor in the development of DCI [6, 8, 11].
We tested the hypothesis that early autoregulatory disturbances following SAH might be related to the development of DCI. We analysed the relationships between the various methods of testing autoregulation and their predictive value.
Materials and Methods
All patients with SAH admitted to the Department of Neurosurgery of this institution between June 2010 and January 2012 were screened. Inclusion criteria included: aneurysmal SAH within 5 days from ictus. The study was approved by the local research ethics committee. The primary end point was DCI within 21 days following SAH.
Calculation of Autoregulatory Indices
The following indices of autoregulation were calculated: transient hyperemic response ratio (THRR) [4, 12] (THRR ≤1.09 indicated impaired autoregulation [12]), transcranial Doppler index of autoregulation (Sxa) [2], and near-infrared spectroscopy index of autoregulation (TOxa) [1].
Statistical Analysis
Receiver operator characteristic (ROC) curve was used to predict DCI (data from days 0–5), Cox proportional hazards model (data from days 0–5) to assess the 21-day risk of DCI, and binary logistic regression to assess the predictive value of impaired autoregulation (data from days 0–5). Flow velocity (FV), THRR, Sxa, and TOxa were binned according to predefined arterial blood pressure (ABP) thresholds for identifying the lower and upper limits of autoregulation (LLA and ULA).
Results
Ninety-eight patients were included. Sixty-six had undergone all three methods of testing autoregulation. Cerebral vasospasm was diagnosed on a median of 6 days post-ictus (range 1–13 days), while DCI was diagnosed on day 8 (range 3–12 days).
Prediction of DCI
The ROC curve determined a good fit for predicting DCI for all indices (AUC: 0.80, 0.86, and 0.80 for THRR, Sxa, and TOxa, respectively; Fig. 1). There was a significantly higher risk of DCI when Sxa and TOxa demonstrated impaired autoregulation (odds ratio [OR]: 7.46, 95 % confidence interval [CI]: 3.03–18.40 and OR: 4.52, 95 %CI: 1.84–11.07, respectively; Table 1). Both indices remained independent predictors in a multivariate model (OR: 12.66, 95 % CI: 2.97–54.07 and OR: 5.34, 95 % CI: 1.25–22.84, respectively) along with a modified Fisher scale grade 3 (OR: 6.21, 95 % CI: 1.45–26.68).
Receiver operating characteristic (ROC) curves demonstrating the ability of the transient hyperemic response ratio (THRR), transcranial Doppler autoregulation (Sxa), and near-infrared spectroscopy-based autoregulation (TOxa). TOxa was used to predict delayed cerebral ischemia (DCI). In all cases, data from the first 5 days post-ictus were used. No significant differences between the area under the curve (AUC) were found for the different indices
Relationship Between Indices
Both Sxa and TOxa were able to accurately predict impaired autoregulation as demonstrated by THRR ≤1.09 (AUC: 0.788, 95 % CI: 0.723–0.854 and AUC: 0.827, 95 % CI: 0.769–0.885, respectively; Fig. 2). An autoregulatory curve could be obtained after binning FV from all recordings according to ABP, suggesting an LLA at 80 mmHg and a ULA at 120 mmHg. The static group autoregulation curves for each index were generated using the same ABP thresholds. All three indices showed impaired autoregulation below 80 mmHg. Sxa demonstrated impaired autoregulation above an ABP of 125 mmHg, while for TOxa it was above 105 mmHg. ULA was not seen using THRR. Table 2 summarizes the respective AUC and the sensitivity and specificity for all indices and combinations of indices for predicting DCI.
Discussion
The role of disturbed autoregulation in the pathophysiology of DCI has been previously demonstrated [6, 8, 11]. However, the small numbers, the inclusion of only poor-grade SAH patients, and the use of invasive monitoring techniques and univariate analysis did not allow for wide generalization. Nevertheless, it has been shown that early dysautoregulation is predictive of poor outcome [7, 11]. This study confirms these findings in a large cohort that comprises all clinical grades of SAH patients and uses noninvasive near-infrared spectroscopy and transcranial Doppler methodology. However, the temporal characteristics of the autoregulatory disturbances presented here differ from previous results, such as Jaeger et al. [6]. The inclusion of all WFNS grades in this study, as opposed to poor-grade patients only in the study by Jaeger et al. [6], provides a possible explanation for these differences.
Despite the good discriminatory value of both Sxa and TOxa shown using Kaplan–Meier analysis, 11 and 13 % of patients with intact autoregulation in the first 5 days post-SAH developed DCI, respectively. Experimental data suggest that DCI might be a multifactorial process, with a number of contributors likely to play a role.
There was good agreement between the methods in identifying the LLA at 80 mmHg, with some discrepancies in the ULA. Overall, the range obtained at which autoregulation was active was narrow compared with classical values of 50–150 mmHg. Because of the limited numbers, we were not able to identify the ULA and LLA separately for DCI and non-DCI groups.
Both Sxa and TOxa demonstrated good accuracy in predicting impaired autoregulation, as seen by a THRR ≤1.09. However, compared with the discrete assessment of autoregulation obtained using the THRR, Sxa and TOxa provide continuous autoregulation monitoring.
Simultaneous demonstration of autoregulatory failure using all three indices showed 100 % specificity for predicting DCI. Concomitant use of continuous Sxa and TOxa resulted in both high sensitivity and high specificity (close to 80 %). It seems that, while considerable colinearities undoubtedly exist between the methods, there is merit in using the multimodal monitoring of cerebral autoregulation following SAH.
References
Brady KM, Lee JK, Kibler KK, Smielewski P, Czosnyka M, Easley RB, Koehler RC, Shaffner DH (2007) Continuous time-domain analysis of cerebrovascular autoregulation using near-infrared spectroscopy. Stroke 38:2818–2825
Budohoski KP, Reinhard M, Aries MJ, Czosnyka Z, Smielewski P, Pickard JD, Kirkpatrick PJ, Czosnyka M (2012) Monitoring cerebral autoregulation after head injury. Which component of transcranial Doppler flow velocity is optimal? Neurocrit Care 17(2):211–218
Etminan N, Vergouwen MD, Ilodigwe D, Macdonald RL (2011) Effect of pharmaceutical treatment on vasospasm, delayed cerebral ischemia, and clinical outcome in patients with aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. J Cereb Blood Flow Metab 31:1443–1451
Giller CA (1991) A bedside test for cerebral autoregulation using transcranial Doppler ultrasound. Acta Neurochir (Wien) 108:7–14
Hop JW, Rinkel GJ, Algra A, van Gijn J (1997) Case-fatality rates and functional outcome after subarachnoid hemorrhage: a systematic review. Stroke 28:660–664
Jaeger M, Schuhmann MU, Soehle M, Nagel C, Meixensberger J (2007) Continuous monitoring of cerebrovascular autoregulation after subarachnoid hemorrhage by brain tissue oxygen pressure reactivity and its relation to delayed cerebral infarction. Stroke 38:981–986
Jaeger M, Soehle M, Schuhmann MU, Meixensberger J (2012) Clinical significance of impaired cerebrovascular autoregulation after severe aneurysmal subarachnoid hemorrhage. Stroke 43:2097–2101
Lam JM, Smielewski P, Czosnyka M, Pickard JD, Kirkpatrick PJ (2000) Predicting delayed ischemic deficits after aneurysmal subarachnoid hemorrhage using a transient hyperemic response test of cerebral autoregulation. Neurosurgery 47:819–825, discussions 825–826
Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, Vajkoczy P, Wanke I, Bach D, Frey A, Marr A, Roux S, Kassell N (2011) Clazosentan, an endothelin receptor antagonist, in patients with aneurysmal subarachnoid haemorrhage undergoing surgical clipping: a randomised, double-blind, placebo-controlled phase 3 trial (CONSCIOUS-2). Lancet Neurol 10:618–625
Macdonald RL (2014) Delayed neurological deterioration after subarachnoid haemorrhage. Nat Rev Neurol 10:44–58
Pickard JD, Matheson M, Patterson J, Wyper D (1980) Prediction of late ischemic complications after cerebral aneurysm surgery by the intraoperative measurement of cerebral blood flow. J Neurosurg 53:305–308
Smielewski P, Czosnyka M, Kirkpatrick P, McEroy H, Rutkowska H, Pickard JD (1996) Assessment of cerebral autoregulation using carotid artery compression. Stroke 27:2197–2203
Disclosure/Conflict of Interest
ICM+ Software is licensed by Cambridge Enterprise, Cambridge, UK, http://www.neurosurg.cam.ac.uk/icmplus/. MC and PS have a financial interest in part of the licensing fee.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Budohoski, K.P. et al. (2016). Monitoring Cerebral Autoregulation After Subarachnoid Hemorrhage. In: Ang, BT. (eds) Intracranial Pressure and Brain Monitoring XV. Acta Neurochirurgica Supplement, vol 122. Springer, Cham. https://doi.org/10.1007/978-3-319-22533-3_40
Download citation
DOI: https://doi.org/10.1007/978-3-319-22533-3_40
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-22532-6
Online ISBN: 978-3-319-22533-3
eBook Packages: MedicineMedicine (R0)