Abstract
The human endometrium contains a substantial population of leucocytes which vary in distribution during the menstrual cycle and pregnancy. An unusual population of natural killer (NK) cells, termed uterine NK (uNK) cells, are the most abundant of these cells in early pregnancy. The increase in number of uNK cells in the mid-secretory phase of the cycle with further increases in early pregnancy has focused attention on the role of uNK cells in early pregnancy. Despite many studies, the in vivo role of these cells is uncertain. This chapter reviews current information regarding the role of uNK cells in healthy human pregnancy and evidence indicating their importance in various reproductive and pregnancy problems. Studies in humans are limited by the availability of suitable tissues and the limitations of extrapolation from animal models.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Endometrial leucocyte
- Uterine NK cells
- T lymphocytes
- CD56
- Cytokines
- Chemokines
- Extravillous trophoblast
- Angiogenic growth factors
- Miscarriage
- Preeclampsia
5.1 Introduction
The human uterus fulfils a unique role, allowing implantation of the semi-allogeneic fetoplacental unit and, in normal human pregnancy, survival in safety for 40 weeks of gestation. Outside of pregnancy, the endometrium lining the uterine cavity is exposed to spermatozoa and seminal fluid on a regular basis and has to be able to resist infection potentially ascending through the cervix. The endometrium undergoes profound morphological changes during each menstrual cycle under the control of ovarian steroid hormones. After epithelial and stromal proliferation in the oestrogen-dependent follicular phase, after ovulation, influenced by progesterone, the glands become secretory and the stroma shows predecidual change, initially around spiral arterioles. In addition to epithelial and stromal cells, the endometrium contains a substantial leucocyte population (Bulmer et al. 1991), mainly within the stroma but with a small population of intraepithelial leucocytes (Pace et al. 1991). The endometrial stromal leucocyte population varies dramatically during the normal menstrual cycle and in pregnancy, and alterations in endometrial leucocyte populations have been described in various pathological situations, including recurrent miscarriage, recurrent implantation failure and pre-eclampsia (reviewed in Bulmer and Lash 2005; Lash and Bulmer 2011). In the 20–30 years since monoclonal antibodies allowed phenotypic identification of leucocyte populations within the endometrium, these cells have been the subject of numerous investigations, but despite considerable progress, the in vivo role of these cells is not yet fully understood.
5.2 Endometrial Leucocyte Populations
Leucocytes are present in both stratum basalis and the hormonally responsive stratum functionalis, and the populations differ between these two sites. Leucocyte populations in the stratum basalis have been less well characterised than those in the stratum functionalis.
Lymphoid aggregates are a common feature in the stratum basalis, sometimes with germinal centres. These follicles include T cells, predominantly CD8+, macrophages and B cells (Morris et al. 1985; Marshall and Jones 1988; Bulmer et al. 1988a); the aggregates have been reported to increase in size in the secretory compared with proliferative phase and to be absent in postmenopausal endometrium, suggesting possible hormonal regulation (Yeaman et al. 1997; Wira et al. 2008). The presence of CD56+ uterine natural killer (uNK) cells in the stratum basalis has not been documented in detail.
The proportion of endometrial stroma accounted for by leucocytes in the stratum functionalis varies during the menstrual cycle. Whereas leucocytes account for fewer than 10 % of stromal cells in the proliferative and early secretory phases, their numbers increase dramatically from the mid-secretory phase, increasing further in the late secretory phase and early pregnancy (Bulmer et al. 1991). Three main cell types account for the majority of endometrial stromal leucocytes in the secretory phase and early pregnancy: macrophages, T lymphocytes, including regulatory T cells, and uNK cells. Unlike other mucosal sites, including vagina, cervix and fallopian tube, B lymphocytes are uncommon in the endometrial stratum functionalis and plasma cells are not a normal feature of endometrium; their presence in non-pathological endometrium is generally considered to indicate chronic endometritis (Disep et al. 2004). Other leucocytes such as dendritic cells (Schulke et al. 2008), NK T cells (Shimada et al. 2003), eosinophils and mast cells are also present in endometrial stroma, in smaller numbers (Evans and Salamonsen 2012). Endometrial leucocyte populations also alter with the onset of menstruation, with neutrophil polymorphs becoming prominent components (Evans and Salamonsen 2012; Berbic and Fraser 2013). The mechanisms underlying the regulation of the various endometrial leucocyte populations during the menstrual cycle are not fully understood and are likely to involve complex interactions between steroid hormones, chemokine and cytokine products of endometrial stromal and epithelial cells, as well as placental trophoblast cells during pregnancy.
The relative proportion of endometrial leucocytes varies during the menstrual cycle. Whereas endometrial T lymphocytes remain relatively constant during the menstrual cycle, both macrophages and, to a greater extent, uNK cells increase in number in secretory endometrium and in early pregnancy (Bulmer et al. 1991). The prominence of uNK cells around the time of expected implantation in a fertile cycle and during early placentation has focused attention on these cells, with fewer studies of other leucocytes in human endometrium. Although uNK cells are the main focus of this chapter, it is useful to consider briefly other endometrial leucocyte populations.
5.2.1 T Lymphocytes
Although the numbers of CD3+ T cells remain relatively constant during the menstrual cycle, their proportion alters as other cells vary in number. T cells constitute 40–60 % of endometrial leucocytes in non-pregnant endometrium (Bulmer et al. 1991; Klentzeris et al. 1994) reducing to 10–20 % in early pregnancy as other cell populations become more abundant (Bulmer et al. 1991). Endometrial T lymphocyte subsets differ compared with those in peripheral blood; CD8+ cytotoxic T lymphocytes are the most abundant T cell in endometrium, with 30–45 % of CD4+ helper T cells. In vitro studies of CD8+ T cells purified from both non-pregnant endometrium and early pregnancy decidua have demonstrated that endometrial T cells are capable of cytotoxic activity (Yeaman et al. 1997; Scaife et al. 2006), although CD8+ T cells in normal pregnancy must maintain a complex balance between control of infection and tolerance towards the fetoplacental unit (Tilburgs et al. 2010; Tilburgs and Strominger 2013).
Besides CD8+ T cells, less abundant endometrial T lymphocyte populations include TCRγδ T cells, CD4-CD8-TCRαβ+ T cells and Vα24 + Vβ11+ NK T cells (Shimada et al. 2003), although the function of these cell types is largely unknown. Th17+ cells are reduced in decidua compared with peripheral blood (Mjosberg et al. 2010), although Th17 cell responses in decidua are abrogated by uNK cells (Fu et al. 2013). CD4+ CD25bright FOXP3+ regulatory T cells are the most studied T cell subset in pregnancy (Tilburgs et al. 2008, 2010). Although they represent a minor T cell population in human decidua, regulatory T cells may act as immune suppressors, contributing to immune homeostasis during pregnancy. They express immunosuppressive cytokines such as IL-10 and TGF-β, suppressing cytotoxic T cells (Aluvihare et al. 2004; Zenclussen et al. 2006). T regulatory cells may also play a role in vascular homeostasis at the maternal-fetal interface via production of IL-10 and TGF-β (Nevers et al. 2011).
T lymphocytes are a consistent and important component of the endometrial leucocyte population. Various subsets have been identified and even minor populations may play an important role in implantation and pregnancy, although additional studies are required to determine the role of specific T cells in both normal and pathological human pregnancy.
5.2.2 Macrophages
Macrophages are an important component of both non-pregnant and pregnant endometrium, although their in vivo role remains unclear. They express CD14, CD68 and class II MHC antigens as well as CD11c, CD86 and adhesion and activation markers depending on their activation state (Nagamatsu and Schust 2010; Houser 2012; Thiruchelvam et al. 2013). Macrophages have diverse functions, including remodelling of extracellular matrix, protease production, tissue regeneration and antigen presentation (Houser et al. 2011). Macrophages account for 20–25 % of the CD45+ leucocytes in non-pregnant endometrium and early pregnancy decidua, although the proportion of leucocytes accounted for by macrophages varies as uNK cell numbers increase during the menstrual cycle and pregnancy. Macrophages have been reported to increase in number in the late secretory and menstrual phases and in early pregnancy (Bulmer et al. 1991; Salamonsen et al. 2002; Thiruchelvam et al. 2013). Proteases and cytokines derived from macrophages may contribute to menstruation and clearance of menstrual debris, and macrophages may also play a role in remodelling of the stratum functionalis after menstruation (Maybin et al. 2012; Thiruchelvam et al. 2013).
In decidua, macrophages are detected in both decidua basalis and decidua parietalis, and in the former these cells are often closely associated with extravillous trophoblast (Bulmer et al. 1988b). Macrophages have been divided into two distinct subtypes, termed pro-inflammatory M1 and anti-inflammatory M2, but decidual macrophages do not conform to these subtypes; recent phenotypic studies reported two distinct populations of decidual macrophages based on CD11c expression as CD11chigh or CD11clow (Houser et al. 2011). CD11chigh macrophages exhibited increased antigen-presenting function with increased expression of CD1a, CD1c and CD1d compared with CD11clow cells. In addition, microarray studies suggest that decidual macrophages group closer to a M2 phenotype, but are a unique population of cells (Gustafsson et al. 2008). It has also been suggested that macrophages in decidua basalis show increased activation compared with decidua parietalis, evidenced by increased expression of HLA-DR (Repnik et al. 2008).
Despite the frequency of macrophages in endometrium and decidua, there have been relatively few studies of these cells, partly because of the difficulty in obtaining viable purified populations. Production of anti-inflammatory substances such as IL-10, IDO (Heikkinen et al. 2003) and prostaglandin E2 (Parhar et al. 1989) raises the possibility of an immunosuppressive function in pregnancy (Nagamatsu and Schust 2010). Our recent studies of macrophages enriched from human early pregnancy decidua have demonstrated production of both IL-6 and IL-8 by macrophages in first trimester decidua, with reduced production of both chemokines in association with first trimester miscarriage (Pitman et al. 2013).
5.2.3 Dendritic Cells
Mature dendritic cells (DCs) are numerically a small component of the human endometrial leucocyte population, but immature DCs are seen in greater numbers in early pregnancy, although studies are at a relatively early stage. In the cycling human endometrium, mature dendritic cells (DCs) are at their peak at the late secretory phase, in contrast with early pregnancy decidua, where the majority of the DCs express CD209 (DC-SIGN), a marker of immature or inactive DC (Gardner and Moffett 2003; Kammerer et al. 2003; Rieger et al. 2004). Although it has been suggested that DCs may play a role in remodelling the cycling endometrium following menstruation, current attention is focused on their potential role in the modulation of immune responses within the pregnant uterus (Dietl et al. 2006; Blois et al. 2011; Leno-Durán et al. 2014) with increasing interest into their potential interactions with uNK cells.
5.2.4 Uterine NK Cells
As the most abundant leucocyte population in endometrium at the time of implantation and early placentation, uNK cells have been the most investigated of the endometrial leucocyte populations, although their in vivo function is still not fully established. Although they were originally recognised by the presence of cytoplasmic granules (Hamperl and Hellweg 1958), uNK cells are now recognised by their unusual phenotype which is distinct from most peripheral blood NK cells (Bulmer et al. 1991; Trundley and Moffett 2004; Bulmer and Lash 2005). In contrast with most peripheral blood NK cells which are CD56dim CD16+, uNK cells express CD56 brightly but are CD16-. Around 5 % of peripheral blood NK cells are CD56bright CD16-, but, in contrast with the uNK cells which show prominent cytoplasmic granules, CD56bright CD16- NK cells in peripheral blood are mainly non-granulated (Cooper et al. 2001). There are other notable differences in the phenotype of uNK cells compared with peripheral blood NK cells, with expression of the tetraspanin family of proteins CD9 and CD151 by uterine but not by peripheral blood NK cells. uNK cells also show expression of NK surface receptors, with differing KIR repertoires noted between uNK cells and peripheral blood NK cells from the same individual (Verma et al. 1997).
5.2.4.1 Distribution of uNK Cells
Although relatively sparse in the proliferative and early secretory phases of the menstrual cycle, uNK cells increase in number dramatically in the mid-secretory phase of the cycle, increasing further as the cycle progresses and in early pregnancy (Bulmer et al. 1991). They reduce in number after the first half of pregnancy, but a substantial number of CD56+ cells remain in the decidua at term (Williams et al. 2009a). Early reports of a virtual absence of uNK cells at term may be explained by a reduction in the proportion of cells with perforin- and granzyme-containing cytoplasmic granules and therefore detectable using histochemical stains such as phloxine tartrazine to detect cytoplasmic granules (Bulmer et al. 2010). Electron microscope studies have suggested that the cells undergo some degranulation during pregnancy (Spornitz 1992), and this may reflect functional changes as pregnancy progresses.
uNK cells are often seen closely associated with extravillous trophoblast in the decidua basalis. Several studies have compared the distribution of uNK cells in decidua basalis and decidua parietalis with conflicting results. Williams et al. (2009a) noted no differences between uNK cell numbers in decidua basalis and decidua parietalis at various gestational ages, whereas others have suggested that uNK cell numbers are increased in decidua basalis associated with invasive EVT. Most recently, Helige et al. (2014) examined density of uNK cells in relation to EVT and noted an increased uNK cell density within 20 μm of trophoblast cells in decidua with density reducing in areas more distant from extravillous trophoblast.
uNK cells are also found adjacent to endometrial glands, as well as in aggregates adjacent to spiral arterioles and arteries (Bulmer et al. 1991). Although it was suggested that the perivascular distribution in the secretory phase may reflect influx of uNK cells from blood, the distribution of the uNK cells at this site may reflect their close association with predecidual change in the perivascular stromal cells. Both uNK cells and macrophages are closely associated with spiral arteries as pregnancy progresses, and this is likely to reflect their role in the transformation of uterine spiral arteries which is an essential feature of normal pregnancy.
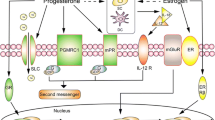
The mechanisms that control the altered numbers of uNK cells during the menstrual cycle and in pregnancy are not known. uNK cells are often associated with stromal cell decidualisation; in secretory phase endometrium, they accumulate in areas of stromal predecidual change. They are also associated with stromal decidualisation at ectopic sites such as ovarian serosal or cervical decidualisation in normal pregnancy or fallopian tube mucosa in ectopic tubal pregnancy. uNK cells are also particularly common in endometrium showing pseudodecidualisation due to high-dose progesterone treatment. This association has suggested regulation by progesterone, but uNK cells do not express progesterone receptor or oestrogen receptor (ER)α (Stewart et al. 1998; Henderson et al. 2003). Regulation of uNK cells by progesterone could occur indirectly via products of decidualised endometrial stromal cells; endometrial stromal cells produce interleukin (IL)-15 in the late secretory phase of the cycle and early pregnancy, and immunostaining for IL-15 has been shown to correlate with numbers of CD56+ cells in endometrium from women with recurrent miscarriage and implantation failure (Mariee et al. 2012). uNK cells do, however, express ERβ and glucocorticoid receptor (Henderson et al. 2003). Recent studies suggest that stromal decidualisation may be associated with secretion of oestrogen (Gibson et al. 2013), raising the possibility that oestrogen may play a role in stimulating uNK cell proliferation and/or differentiation in the secretory phase of the menstrual cycle.
5.2.4.2 Increased uNK Cells: Recruitment, Differentiation and/or Proliferation?
There is no consensus regarding the origin of uNK cells; there is evidence to support both recruitment and local differentiation (Koopman et al. 2003; Bulmer and Lash 2005; Bulmer et al. 2010; Zhang et al. 2012). In mice, uNK progenitor cells have been identified in primary and secondary lymphoid tissues, and CD127 is expressed at days 10.5 and 12.5 but not at day 6.5 suggesting local differentiation and regulation (Zhang et al. 2012). However, limited extrapolation is possible from mouse studies since uNK cells are only detected after blastocyst implantation in mouse and not in non-pregnant endometrium; and human and mouse placentation show important differences.
Early studies suggested recruitment of uNK cells into endometrium in response to chemokine and cytokine secretion. For example, production of CXCL-12 by extravillous trophoblast cells was suggested to attract uNK cells into decidua in pregnancy (Wu et al. 2005), and IL-15 which is produced by secretory endometrium and decidua has been shown to have a selective chemoattractant effect on peripheral blood CD16- NK cells (Kitaya et al. 2007). Increasing evidence, however, indicates that uNK cells differentiate in situ in endometrium from haematopoietic precursor cells (HPC) or immature NK cells recruited from peripheral blood into an environment rich in growth factors, cytokines and hormones. CD34+ CD45+ HPC have been reported in non-pregnant endometrium (Lynch et al. 2007) and early pregnancy decidua (Keskin et al. 2007; Vacca et al. 2011; Szereday et al. 2012), with a frequency of 0.1–4 %. CD34+ CD45+ HPC purified from decidua and cultured in decidual stromal cell-conditioned medium or combinations of c-kit ligand (KL), IL-15, Flt3L and IL-7 produced CD56bright CD16- CD9+ uNK-like cells (Keskin et al. 2007; Vacca et al. 2011). Transplantation of human proliferative endometrium into nonobese diabetic/severe combined immunodeficiency/γCnull immunodeficient mice provides additional evidence for local differentiation; increased CD56+ uNK cells were detected after hormone treatment mimicking the menstrual cycle (Matsuura-Sawada et al. 2005). In contrast, Male et al. (2010) detected stage 3 NK cell precursors (CD34- CD117+ CD94-) in uterine mucosa that were able to develop into stage 4 mature (CD34- CD117+/− CD94+) NK cells in vitro but did not detect CD34+ HPC in non-pregnant endometrial pipelle samples; other studies used curettage (Lynch et al. 2007) or hysterectomy samples (Matsuura-Sawada et al. 2005) which, in contrast with pipelle samples, would consistently include the stratum basalis which is preserved after menstruation and would be a likely source of CD34+ HPC.
Although mesenchymal stem cells have been reported in endometrium (Gargett and Masuda 2010), whether there are haematopoietic stem cells (HSCs) in non-pregnant endometrium is uncertain. It could be proposed that stem cells residing in the stratum basalis, which is retained after menstruation, could differentiate into uNK cells as the cycle progresses. We have demonstrated rare CD45+ CD34+ double-labelled cells in non-pregnant endometrium with most cells within the stratum basalis. It is interesting to note that if uNK cells do develop locally within endometrium during each menstrual cycle, abnormalities which have been detected in, for example, recurrent miscarriage (see below) would point to abnormal regulation within the endometrium as the cells develop and proliferate locally or an inherent defect in the stem cell population leading to abnormal development within each individual cycle.
Locally secreted chemokines/cytokines could attract distinct mature or immature NK cell subsets to endometrium and mediate further local differentiation. Male et al. (2010) proposed migration of stage 3 immature NK cells into endometrium and differentiation in situ within the endometrium into uNK cells. In contrast, Keskin et al. (2007) reported conversion of purified peripheral blood CD16+ CD9- NK cells into CD16- CD9+ uNK-like cells after culture with decidual stromal cell-conditioned medium or TGF-β1, suggesting conversion of peripheral blood NK cells to uNK cells locally within endometrium. Further evidence that uNK cells are an immature population comes from a report that 60 % of decidual uNK cells are CD11b- CD27- (Fu et al. 2011). There is also clear evidence that uNK cells are able to proliferate within endometrium. Stromal mitotic figures which are often seen in late secretory phase endometrium are due to proliferation in uNK cells (Pace et al. 1989), and several studies have demonstrated expression of the Ki67 proliferation marker by uNK cells (Pace et al. 1989; Kämmerer et al. 1999). In a study of CD56+ cells purified from non-pregnant endometrium by immunomagnetic selection, uNK cell expression of Ki67 was highest (>40 %) in the mid- and late secretory phase, reducing to ~12 % in early pregnancy decidua (Jones et al. 1998). Using a range of different techniques, Kämmerer et al. (1999) demonstrated proliferation in CD56+ cells in decidua from 5 to 11 weeks gestational age, with 7–23.5 % CD56+ cells co-expressing Ki67. CD56+ cells in pseudodecidualised endometrium after progesterone treatment also express Ki67.
Whether there is local development from HSCs within endometrium or local modification of peripheral blood NK cells recruited into endometrium, it is clear that there is local development of uNK cells within endometrium. In addition, the phenotype of uNK cells in early pregnancy varies with gestational age, suggesting ongoing differentiation: reduced expression of KIR specific for HLA-C (KIR2DL1/S1 and KIRDDL3/L2/S2) with increasing gestational age from 6 to 12 weeks (Sharkey et al. 2008; Marlin et al. 2012) has been reported, as well as the reduction of the CD85j + NKG2D- subset and increase of the CD85j- NKG2D+ with increasing gestation from 8 to 12 weeks (Marlin et al. 2012). In addition, in double immunohistochemical labelling studies, we noted increased expression of CD122 by CD56+ cells in LH+13 endometrium compared with LH+7 endometrium (Otun et al. 2009). It is possible that the origin of uNK cells differs between non-pregnant endometrium and also different stages of pregnancy, with varying populations being recruited as their required function changes with implantation and early placentation.
5.2.4.3 Regulation of uNK Cells by Growth Factors in Endometrium
Endometrial stromal cells undergo differentiation into decidual cells in the luteal phase, and human uNK cells localise to areas of stromal decidualisation. This association suggests a role for decidualised endometrial stromal cell products in uNK cell accumulation. A range of growth factors are produced within endometrium which could play a role in the development of uNK cells.
In peripheral blood, IL-15 and/or IL-2 promote differentiation and expansion of CD34+ HPCs to CD56+ NK cells (Mrózek et al. 1996). Although IL-2 is not present in normal endometrium, IL-15 has been detected in stromal cells in luteal phase endometrium and early pregnancy decidua; secretion is stimulated by progesterone, although the regulation of this appears complex (Okada et al. 2000), involving IL-1-β (Okada et al. 2004) and IFNγ (Dunn et al. 2002). Studies of luteal phase endometrium in women with recurrent reproductive failure have shown a correlation between uNK cell number and stromal cell IL-15 levels (Mariee et al. 2012). Although no data are available for human uNK cells, IL-11 is required for mouse uNK cell maturation (Ain et al. 2004). As well as epithelial expression, IL-11 is highly expressed in decidualised endometrial stromal cells in luteal phase endometrium (Dimitriadis et al. 2005). CD117 (c-kit) is expressed by the earliest IL-15-responsive HPC throughout human NK cell differentiation (Freud et al. 2006). c-kit ligand (KL, stem cell factor, SCF) triggers haematopoiesis on binding to CD117 (Broudy 1997) and enhances the proliferative action of both IL-2 and IL-15 on human CD56bright NK cells (Benson et al. 2009). Endometrial expression of KL/SCF increases in early pregnancy decidua (Kauma et al. 1996; Umekage et al. 1998). Reports on other NK cell differentiation factors in endometrium are limited: IL-7 and Flt3L have recently been reported in uterine fluid (Hannan et al. 2011); and IL-18 has been proposed as a marker of endometrial function and is related to uNK cell numbers and activation of NKp46 (Petitbarat et al. 2011). Transforming growth factor beta 1 (TGF-β1) inhibits peripheral blood NK cell cytotoxic activity, cytokine production and cell proliferation (Bellone et al. 1995) and suppresses expression of NK cell surface receptors, including NKp30 and NKG2D when cultured with human peripheral blood NK cells (Castriconi et al. 2003). TGF-β1 is present in human endometrium during the menstrual cycle and in early pregnancy (Jones et al. 2006; Omwandho et al. 2010; Lash et al. 2012).
5.2.4.4 Relationship of uNK Cells in Non-pregnant Endometrium and Decidua in Pregnancy
The relationship between uNK cells in non-pregnant endometrium and decidualised endometrium in pregnancy is uncertain, at least in part because the origin of uNK cells remains controversial. Although they were initially reported as absent in proliferative endometrium based on histochemical stains to detect cytoplasmic granules, CD56+ cells are detectable, albeit in relatively low numbers (Bulmer et al. 1991). The increase in numbers is seen from day 22 onwards (Russell et al. 2011, 2013), and at least some of the increase can be accounted for by local proliferation. The phenotype of uNK cells in non-pregnant endometrium has not been investigated to the same extent as those from early pregnancy decidua, where distinct gestational age differences in phenotype and function have been reported. In a study of CD56+ cells separated from non-pregnant endometrium (>98 % CD56+) at various menstrual cycle changes, expression of various cell surface markers was investigated. There was low to no expression of CD3, CD8, CD16, HML-1, l-selectin and CD25 (IL-2 receptor-α) on CD56+ cells isolated from non-pregnant and pregnant endometrium. Expression of CD2, CD49a and CD122 increased from the proliferative to the late secretory phase of the menstrual cycle, whereas CD11a, CD69 and CD49d expression was high and did not vary with menstrual cycle phase, although CD49d levels were significantly reduced in early pregnancy. There were also differential proliferative responses: in contrast with those from early pregnancy decidua, CD56+ cells from non-pregnant endometrium did not proliferate in response to phytohaemagglutinin (Searle et al. 1999).
Manaster et al. (2008) suggested that CD56+ cells in non-pregnant endometrium are immature, non-functional cells awaiting activation in pregnancy. This contrasts with other studies that have shown both proliferative and cytotoxic activity of uNK cells from non-pregnant endometrium (Jones et al. 1997; Searle et al. 1999). More recently, microarray studies (Kopcow et al. 2010) compared CD56+ cells from non-pregnant and early pregnancy endometrium: 450 genes were differentially expressed with >twofold difference, with ~70 % over-expressed in the non-pregnant uNK cell subset, suggesting that uNK cells in non-pregnant endometrium are far from inactive and likely to play an important role in implantation and early placentation.
Studies of uNK cells in non-pregnant endometrium are limited by availability of tissues. As nonsurgical and more conservative treatments for problems associated with heavy menstrual bleeding increase in popularity, it is increasingly difficult to obtain endometrial samples for functional studies of uNK cells in non-pregnant endometrium. This difficulty makes studies of the functional relevance of the increased luteal phase uNK cells that have been reported in some women with recurrent implantation failure (RIF) and recurrent miscarriage (RM) harder to achieve.
5.2.4.5 Function of uNK Cells
The in vivo roles of uNK cells are still not clear, but in vitro studies are providing clues to their function. Because of the difficulty in obtaining sufficient non-pregnant endometrium for cell purification, the vast majority of studies have been performed using uNK cells from early pregnancy decidua, with relatively few studies performed on uNK cells from non-pregnant endometrium.
5.2.4.5.1 Cytotoxicity
The recognition of uNK cells as a type of natural killer cell led to early studies of their cytotoxic activity and focus on this function as a means to control trophoblast invasion in early pregnancy. Although uNK cells isolated from early pregnancy decidua exhibit cytotoxic activity against the classical NK cell target K562, this cytotoxic activity is lower than that of peripheral blood NK cells (Ritson and Bulmer 1989; Kopcow et al. 2005). Early reports suggested that uNK cells were able to lyse choriocarcinoma and normal trophoblast cells after IL-2 activation (King and Loke 1990), but the current consensus is uNK cells do not lyse normal trophoblast, due to expression of HLA-G, HLA-E and HLA-C by extravillous trophoblast and expression by uNK cells of inhibitory receptors (Chumbly et al. 1994; Rouas-Freiss et al. 1997; Chen et al. 2010; Chazara et al. 2011). Vascular endothelial growth factor (VEGF)-C also upregulates expression of TAP-1 by extravillous trophoblast; TAP-1 plays a role in peptide loading for MHC class I assembly and antigen presentation in EVT cells, thereby also potentially protecting them from uNK cell cytotoxic activity (Kalkunte et al. 2009). The cytoplasmic granules which are a characteristic feature of uNK cells express perforin and granzyme (King et al. 1993) suggesting that they are capable of effector function, although the proportion expressing these molecules reduces with increasing gestational age (Bulmer et al. 2010). However, although they possess the machinery for effector function, uNK cells have been shown to form immature synapses with K562 cells, failing to polarise their microtubule-organising centres and perforin-containing granules to the synapse (Kopcow et al. 2005). Thus, although NK cells were originally defined by their cytolytic activity, it appears that cytolytic activity is unlikely to be a primary function of uNK cells in either normal or pathological human pregnancy.
5.2.4.5.2 Cytokine, Growth Factor and Protease Secretion
uNK cells purified from early pregnancy decidua are a rich source of a range of cytokines and growth factors, including tumour necrosis factor (TNF)-α, IL-10, granulocyte-macrophage colony-stimulating factor (GM-CSF), IL-1-β, TGF-β1, macrophage colony-stimulating factor (M-CSF), leukaemia inhibitor factor (LIF) and interferon (IFN)γ (Saito et al. 1993; Jokhi et al. 1994; Lash et al. 2010a). Interestingly, there are gestational age differences in cytokine secretion by uNK cells: secretion of IL-1-β, GM-CSF (Lash et al. 2010a), IL-6 (Champion et al. 2012), IL-8 (De Oliveira et al. 2010) and IFNγ (Lash et al. 2006a) has been shown to increase from 8–10 to 12–14 weeks gestational age.
uNK cells are also an important source of angiogenic growth factors within early pregnancy decidua. Production of angiopoietin (Ang) 1, Ang2, VEGF-C, placental growth factor (PlGF) and TGF-β1 by uNK cells from early pregnancy decidua has been reported (Lash et al. 2006b), and uNK cells in secretory phase endometrium also produce VEGF-C, Ang1, Ang2 and PlGF (Li et al. 2001). In contrast with cytokine production, uNK cell secretion of Ang2 and VEGF-C appears to reduce from 8–10 to 12–14 weeks gestational age (Lash et al. 2006b). The results of secretion studies suggest that uNK cells at 8–10 weeks gestational age are major local intrauterine producers of angiogenic growth factors, whereas at 12–14 weeks gestational age, they have switched their secretory profiles to become major cytokine producers. As the phenotype of uNK cells alters during the first trimester of pregnancy, the change from secretion of angiogenic growth factors to cytokines as pregnancy progresses may reflect these phenotypic alterations. Indeed, it is possible that the proportion of uNK cells developing locally or trafficking from the peripheral blood varies at different stages of the menstrual cycle and pregnancy in both non-pregnant endometrium and decidua. Detailed functional investigation of phenotypically distinct uNK cell populations in accurately dated samples would be required to resolve this question. An alternative explanation is that exposure to the changing intrauterine environment induces functional changes in uNK cells. There is evidence to suggest that exposure of peripheral blood NK cells to soluble HLA-G induces a senescent phenotype that is characterised by increased secretion of IL-6 and IL-8 (reviewed in Rajagopalan 2014). It could be proposed, therefore, that the exposure of uNK cells to soluble HLA-G within the pregnant uterus could lead to alterations in their secretory profile which could impact on their in vivo function.
During early pregnancy, cytotrophoblast proliferates from the tips of the chorionic villi to form cytotrophoblast columns which extend peripherally to form a cytotrophoblast shell. Extravillous trophoblast from the cytotrophoblast shell then invades into uterine decidua and superficial myometrium. There are two main pathways of trophoblast invasion: interstitial extravillous trophoblast invades through the decidua and superficial myometrium, whereas endovascular trophoblast extends within the lumen of the spiral arteries, transiently replacing the endothelium and ultimately remodelling the uterine spiral arteries from thick-walled musculoelastic vessels into dilated tubes whose wall comprises fibrinoid material containing intramural trophoblast cells (Pijnenborg et al. 2006). Both interstitial trophoblast invasion and spiral artery remodelling require breakdown of extracellular matrix (ECM) by proteolytic enzymes. Uterine NK cells secrete metalloproteinase (MMP)-1, MMP-2, MMP-7, MMP-9, MMP-10, tissue inhibitor of metalloproteinases (TIMP)-1, TIMP-2, TIMP-3, urokinase plasminogen activator (uPA) and uPA receptor (uPAR), although not plasminogen activator inhibitor (PAI)-1 and PAI-2 (Naruse et al. 2009a, b). In contrast with cytokines and angiogenic growth factors, no gestational age differences have been detected in secretion of these proteolytic enzymes. Immunoreactivity of MMP-7 and MMP-9 by leucocytes surrounding spiral arteries during early pregnancy has also been reported (Smith et al. 2009).
Both trophoblast invasion and spiral artery remodelling are tightly controlled processes, deficient trophoblast invasion and vascular transformation being associated with pregnancy pathology such as pre-eclampsia, fetal growth restriction, late miscarriage and preterm delivery (Pijnenborg et al. 2006). By their secretion of cytokines and angiogenic growth factors, uNK cells have been suggested to play a central role in these processes in early pregnancy.
5.2.4.5.3 Regulation of Trophoblast Invasion by uNK Cells
Hanna et al. (2006) demonstrated that IL-15-stimulated uNK cell supernatants stimulate invasion of isolated cytotrophoblast cells in vitro, this stimulatory effect being partially abrogated by neutralising antibodies to IL-8 and IP-10 (Hanna et al. 2006). This result is supported in part by studies demonstrating that uNK cell supernatants from 12 to 14 weeks gestational age stimulated invasion of extravillous trophoblast from placental explants (Lash et al. 2010b). This uNK-mediated stimulation of EVT invasion was partially abrogated in the presence of an IL-8 neutralising antibody (De Oliveira et al. 2010). In contrast, when both uNK cells and placenta were from 8 to 10 weeks gestational age, uNK cell supernatants had no effect on extravillous trophoblast invasion (Lash et al. 2010b; De Oliveira et al. 2010). Hu et al. (2006) demonstrated that IL-15-stimulated uNK cell supernatants inhibited migration of EVT in a two-dimensional migration assay by a mechanism dependent on IFNγ. Discrepancies between reports may, therefore, be due to variation in the gestational ages of the samples studied, as well as sources of trophoblast cells and the presence of prior activation of uNK cells by IL-15.
5.2.4.5.4 Role of uNK Cells in Spiral Artery Remodelling
Spiral artery remodelling is a key feature of successful human pregnancy (Pijnenborg et al. 2006). This process has often been attributed solely to the effect of extravillous trophoblast, but increasing evidence indicates that there is a ‘trophoblast-independent’ phase of spiral artery remodelling (Pijnenborg et al. 2006). Histological studies of the placental bed in early pregnancy demonstrated that initial stages of spiral artery remodelling, including dilatation, some fibrinoid deposition, endothelial swelling and vascular smooth muscle cell (VSMC) separation, occur in the absence of extravillous trophoblast (Craven et al. 1998; Kam et al. 1999), and more recent studies support this view.
uNK cells are often seen aggregated around the spiral arteries and arterioles in both the secretory phase of the menstrual cycle and in early human pregnancy; this distribution in pregnancy may reflect a role in mediating vascular changes in pregnancy. Increased numbers of both uNK cells and macrophages were detected within 25 μm of the vessel lumen in human decidual spiral arteries showing partial remodelling and an absence of EVT, compared with non-remodelled vessels and those with greater degrees of remodelling, including the presence of extravillous trophoblast (Smith et al. 2009). Using in vitro vessel models (either chorionic plate arteries from human term placenta or non-pregnant myometrial arteries), we demonstrated that supernatants produced by uNK cells from 8 to 10 weeks gestation were able to mediate separation of vascular smooth muscle cells within the vessels, while uNK cell supernatants from 12 to 14 weeks gestation had a greater effect on vascular smooth muscle cell dedifferentiation (Robson et al. 2012). The uNK cell-derived factors responsible for mediating the effects on uterine spiral arteries remain to be fully determined, but evidence from our in vitro studies implicates Ang1, Ang2 and VEGF-C in this process (Robson et al. 2012). It is possible that the stimulation of trophoblast invasion by uNK cells at 12–14 weeks gestational age may play a role in attracting interstitial trophoblast cells towards spiral arteries that have been partially remodelled by uNK cells for completion of the remodelling process.
5.2.4.6 Interaction of uNK Cells with Other Cell Types
uNK cells are intimately associated with a range of cell types in both non-pregnant and pregnant endometrium, including other endometrial leucocyte populations. Because of the intimate association of uNK cells with extravillous trophoblast in decidua basalis, several studies have focused on interactions between these two cell types.
5.2.4.6.1 Uterine NK Cell: Trophoblast Cell Interactions
Although early studies of uNK cells focused on the ability of uNK cells to lyse normal and pathological trophoblast cells, current studies are focused on their secretion of cytokines. Interactions could occur indirectly by secretion of growth factors and cytokines or directly via ligand-receptor interactions.
A key distinguishing feature of extravillous trophoblast, compared with villous cytotrophoblast and syncytiotrophoblast which do not express class I MHC antigens, is their expression of HLA-C, HLA-E and HLA-G, but not the classical, polymorphic MHC molecules HLA-A and HLA-B. HLA-C alleles are recognised by both inhibitory and activating KIRs, including KIR2DL1, KIR2DL2, KIR2DL3 and KIR2DS1. Genetic association studies have linked maternal KIR and fetal (paternal) HLA-C haplotypes and reproductive success. It has been suggested that a maternal KIR AA haplotype, with mainly inhibitory KIR in combination with a fetal HLA-C2 haplotype, is associated with pre-eclampsia and recurrent miscarriage (Hiby et al. 2004, 2010), while interaction between a fetal HLA-C2 haplotype and KIR BB, which is activating, may confer reproductive protection. However, it should be noted that the importance of this association has been disputed (Clark 2014), and it is not clear why only a proportion of pregnancies with the AA/C2 combination have compromised reproductive success.
As HLA-G expression appears to be restricted to extravillous trophoblast cells, initial assumptions were that it was important to protect these cells from lysis by uNK cells closely associated in decidua basalis. However, it is now clear that any protective effects can be attributed to interactions of HLA-G with T cells rather than NK cells (van der Meer et al. 2004, 2007), although interactions between HLA-G and uNK cells do appear to result in altered secretion of a range of soluble factors (van der Meer et al. 2004).
Several studies have investigated the effect of interactions between extravillous trophoblast and uNK cells on secretion of cytokines and angiogenic growth factors. Most studies have used either HLA-G-transfected cell lines or the choriocarcinoma cell lines JEG-3 (HLA-G positive) and JAR (HLA-G negative), and in addition, peripheral blood mononuclear cells and uterine mononuclear cells have often been used as a proxy for uNK cells. There have been few studies using primary isolates of extravillous trophoblast and uNK cells. Not surprisingly with the diverse approaches that have been used, the overall results of these studies have been variable.
We performed an extensive study of cytokine and angiogenic growth factor secretion after uNK cell coculture with either extravillous trophoblast or cytotrophoblast, with all cell types being isolated from the same patient. uNK cells and trophoblast were cultured both in direct contact or separated by a 0.4 μm pore filter (Lash et al. 2011). Secretion of both cytokines and angiogenic growth factors was reduced in cocultures, although whether this was due to uNK cells or trophoblast cells or both was not determined. In general, coculture of uNK cells with either extravillous trophoblast or cytotrophoblast produced similar results suggesting that any effects were not mediated by HLA molecules which differ between these two cell types, but by other shared molecules. Furthermore, secretion of angiogenic growth factors was altered after both direct and indirect coculture suggesting mediation by soluble factors, whereas alterations in cytokine secretion were only observed after direct coculture, suggesting involvement of a membrane-bound molecule. However, HLA-C and KIR haplotypes were not determined in this study, and recent studies indicate that secretion profiles may vary according to the HLA-C and KIR haplotypes (Xiong et al. 2013).
Xiong et al. (2013) studied the response of inhibitory KIR2DL1 or activating KIR2DS1 uNK cell subsets to coculture with 721.221 parent cells or those transfected with HLA-C2 (221-C2) by microarray analysis. uNK cell subsets were KIR2DS1+ KIR2DL1- (KIR2DS1 single positive (sp)), KIR2DS1- KIR2DL1+ (KIR2DL1sp), KIR2DS1+ KIR2DL1+ (double positive (dp)) or KIR2DS1- KIR2DL1- (double negative (dn)). Distinct differences in all four uNK cell subsets were demonstrated using cluster analysis; 45 transcripts were altered in the KIR2DL1sp group, 378 in the KIR2DS1sp group, 289 in the dp group and 3 in the dn group. There was little overlap between the 3 positive groups with only 24 altered transcripts common in the KIR2DS1sp and dp groups, despite both expressing KIR2DS1. Furthermore, there were only six common transcripts between the KIR2DL1sp and dp groups, as well as between the two different sp subsets. It is important to note that each individual has different subsets of uNK cells suggesting that differential local production of cytokines and growth factors may occur throughout the placental bed where uNK cells and EVT interact. In addition, the described study specifically concentrated on the effect of HLA-C2 on uNK cell transcription, whereas extravillous trophoblast cells express a range of ligands that would simultaneously interact with uNK cell-expressed receptors.
The functional consequence of interactions between uNK cells and trophoblast remains unclear. Although specific cytokines and growth factors have been implicated in these processes, it is likely that many different cytokines and chemokines are involved in the control of trophoblast invasion and spiral artery remodelling. Close association of extravillous trophoblast cells with uNK cells leads to reduced cytokine secretion, and it is possible that after recruitment of extravillous trophoblast or uNK cells to specific sites in the placental bed by higher cytokine levels, this serves to inhibit their onward passage. Extravillous trophoblasts are naturally highly invasive cells (Lash et al. 2006c), yet cease to invade in the inner third of the myometrium. Fusion of mononuclear trophoblast cells to form non-invasive multinucleate trophoblast giant cells may play a role in the control of trophoblast invasion. In women with focal placenta accreta, the numbers of myometrial multinucleate trophoblast giant cells are highly reduced in areas lacking decidua compared with areas with intact decidua, suggesting that decidual derived factors mediate or initiate this fusion process as the cells move towards the myometrium (Hannon et al. 2012). Furthermore, in placenta adhesiva, the mildest form of adherent placenta in which decidua basalis is present (van Beekhuizen et al. 2009), numbers of uNK cells were markedly reduced (van Beekhuizen et al. 2010). It is tempting to speculate that uNK cells may play a role in the fusion of mononuclear to multinucleate trophoblast.
5.2.4.6.2 Interaction of uNK Cells with Other Endometrial Cells
Many different cell types are present in human endometrium, and potentially uNK cells could interact with any of these cells. There is considerable interest in potential interactions between uNK cells and CD209+ (DC-SIGN) dendritic cells (DCs) in early pregnancy decidua. Although many studies have focused on mouse (Blois et al. 2011), there are a few studies that point to important interactions in human pregnancy. Using immunohistochemistry, Kammerer et al. (2003) noted intimate contact between DCs and uNK cells within human decidua, and this was confirmed in studies of decidual leucocyte suspensions in which it was suggested that a proportion of the DC-SIGN+ cells associated with uNK cells were apoptotic (Tirado-González et al. 2012). Investigation of the DC/uNK cell interaction in human pregnancy is at a very early stage, and technical issues in in vitro studies using a decidual source for both cell populations are challenging. However, studies in mouse highlight the potential importance, with evidence suggesting direct control of stromal cell proliferation, angiogenesis and homing and maturation of uNK cell precursors in the pregnant uterus (Blois et al. 2011).
5.2.4.7 uNK Cells in Pregnancy Pathology
As the predominant endometrial leucocyte population in the secretory phase and early pregnancy, when the seeds are sewn for pregnancy problems that may not manifest until much later in gestation, uNK cells have been a focus for studies of pathological pregnancy.
5.2.4.7.1 Miscarriage
Miscarriage is a common gynaecological problem; it has been estimated that 11–20 % of all clinically recognised pregnancies are lost before the 20th week of gestation (Everett 1997). Miscarriages are often separated into early (≤12 completed weeks from LMP) and late (≥13 weeks): early miscarriage accounts for the majority, with ~50 % being associated with aneuploidy, whereas late miscarriage affects only 1–2 % of pregnancies (Regan and Rai 2000) and is less likely to be associated with chromosomal abnormalities. Whereas some studies have focused on uNK cell populations in secretory phase endometrium, studies of decidua in miscarriage are hampered by the potential criticism that since clinical presentation may occur days after demise of the pregnancy, any alteration in leucocyte populations may simply reflect the result rather than the cause of the miscarriage.
Having been referred to by several names, such as Kornchenzellen, K cells and endometrial/decidual granulated/granular lymphocytes, the terminology of ‘uterine natural killer cell’ was adopted following the recognition that these cells are a type of NK cell, expressing NK receptors. Unfortunately, the term may suggest potential for these cells to ‘kill’ a pregnancy if activated, although there is no evidence for this and activation of uNK cells in pregnancy is considered beneficial. The focus on peripheral blood NK cells and uNK cells in recurrent miscarriage and implantation failure has led to patient demand for testing and treatment. At present, although some associations with NK cells have been reported, the functional consequences and importance of these are not known.
Sporadic Miscarriage
Increased uNK cells have been reported in immunohistochemical studies of decidua from women with sporadic miscarriage compared with controls (Zenclussen et al. 2001; Plaisier et al. 2009), although this was not reproduced in an immunohistochemical study of placental bed biopsies from 8 to 20 weeks gestation (Scaife et al. 2004). A recent study of chromosomally normal and abnormal miscarriage reported an increased incidence of NK cell aggregates in both euploid and aneuploid miscarriage compared with elective abortion (Lee et al. 2015). In contrast, Yamamoto et al. (1999) reported reduced CD56+ uNK cells in a flow cytometric study of decidua of women with sporadic miscarriage. Vassiliadou and Bulmer (1996) noted increased numbers of CD57+ NK cells in a subgroup of cases of sporadic miscarriage. Thus, whether numbers or phenotype of uNK cells are altered in sporadic miscarriage remains uncertain.
Altered NK cell function is likely to be more important than any change in NK cell numbers within decidua. Early suggestions that activation of NK cells in decidua could lead to pregnancy loss by cytotoxicity against fetoplacental tissues have not been supported by studies in human pregnancy. Compared with controls, Vassiliadou and Bulmer (1998) detected reduced cytotoxic activity against K562 cells in uNK cells isolated from decidua of women with early sporadic miscarriage. Yamada et al. (2005) reported an increased proportion of perforin-positive uNK cells in decidua associated with sporadic miscarriage compared with controls. In contrast, Nakashima et al. (2008) reported no difference in the proportion of uNK cells expressing perforin and granzyme B in sporadic miscarriage compared with controls, although the proportion of granulysin-positive uNK cells in decidua was increased and this was linked to an increase in EVT apoptosis.
Hence, there are several studies that suggest that uNK cells could play a role in sporadic early pregnancy loss. However, results are conflicting and studies are complicated by the potential of inflammation secondary to pregnancy demise. Furthermore, despite many studies of uNK cell function in the first trimester of pregnancy, understanding of their function around implantation and in very early pregnancy remains very limited.
Recurrent Miscarriage
Recurrent miscarriage (RM) is defined as three or more consecutive first trimester miscarriages and in 50 % of cases the cause remains unknown (Quenby and Farquharson 1993; Rai and Regan 2006). Several immunohistochemical studies from different groups have reported increased numbers of uNK cells in mid-secretory phase endometrium from women with a history of RM (Clifford et al. 1999; Quenby et al. 1999; Tuckerman et al. 2007). Quenby et al. (1999) noted significantly higher uNK cell numbers in women who went on to a further miscarriage compared with those who had a live birth, although a subsequent larger study failed to detect such an association (Tuckerman et al. 2007). In contrast, using flow cytometry, others have not detected altered numbers of endometrial uNK cell numbers in women with RM (Lachapelle et al. 1996; Shimada et al. 2004), although Lachapelle et al. (1996) did report reduced CD56bright CD16- and increased CD56dimCD16+ subsets. Michimata et al. (2002) also did not detect any differences in endometrial uNK cell numbers, although RM in this study was defined as two rather than three or more consecutive miscarriages. Although there are discrepancies in results, immunohistochemical studies have consistently shown increased uNK cell numbers in mid-secretory phase endometrium of women with a well-defined history of RM.
It is difficult to interpret studies of uNK cell numbers in the miscarried decidua of women with RM which have differing results; this may reflect the use of immunohistochemistry versus flow cytometry, but in addition whether any changes noted represent ‘cause’ or ‘effect’ is uncertain. Chao et al. (1995) reported similar numbers but increased NK activity in decidua associated with RM. Emmer et al. (2002) reported expression of CD16 by CD56+ cells in decidua from RM samples, while Kwak et al. (1995) noted increased expression of CD57+ cells in 29.6 % of RM cases. In contrast, Quack et al. (2001) reported reduced CD56+ cells in decidua from RM, although there were significantly increased numbers of CD25+ cells, suggesting increased leucocyte activation. Yamamoto et al. (1999) also reported reduced numbers of CD56+ cells in RM compared with controls, but an increased proportion expressed CD16.
In summary, reports of increased uNK cells in secretory phase endometrium from women with RM are consistent, but other approaches, including investigation of decidua, have not yielded consistent results. The functional importance of this is largely unknown. Quenby et al. (2009) linked increased uNK cells to altered blood flow in endometrium from women with both RM and RIF. However, little is known of uNK cell function in non-pregnant endometrium, and further studies of normal endometrium are required before the functional significance of these increased uNK cells is known.
5.2.4.7.2 Recurrent Implantation Failure and Infertility
There are several studies that have suggested that uNK cells may be implicated in both infertility and RIF, but results are variable. Although, using flow cytometry, Matteo et al. (2007) reported no difference in uNK cells in late secretory phase endometrium in women with RIF compared with normal fertile controls, others have reported increased uNK cells in pre-pregnancy endometrium in association with RIF. Tuckerman et al. (2010) reported a dramatic increase in uNK cells as a proportion of stromal cells in the mid-secretory phase in women with RIF after IVF compared with normal fertile controls, with uNK cell numbers correlating with stromal expression of IL-15 (Mariee et al. 2012). Similarly, Ledee-Bataille et al. (2004) demonstrated elevated uNK cell numbers in women with RIF in association with increased endometrial IL-12 or IL-18. This cohort also showed abnormal uterine artery Doppler compared with normal controls or RIF women without elevated uNK cells and cytokines. Quenby et al. (2009) also reported that elevated uNK cell numbers in women with RIF correlated with abnormal uterine artery Doppler.
Women with unexplained infertility have been less well studied. Klentzeris et al. (1994) reported reduced CD56+ cells in timed luteal phase biopsies from women with unexplained infertility compared with fertile controls. In contrast, using flow cytometry, Fukui et al. (1999) did not demonstrate alteration in overall CD56+ cell numbers but reported increased CD16+ CD56dim NK cells and reduced CD56bright CD16- NK cells in endometrium from women who subsequently went on to have failed IVF compared with those who had a successful pregnancy. McGrath et al. (2009) subsequently reported that while uNK cell numbers were not altered in infertility, uNK cell expression of CD94, CD158a and CD158a was increased, suggesting an altered phenotype in this cell type.
Infertility may be associated with uterine pathology, and although there are few studies, this may also be associated with altered uNK cells. Tremellen and Russell (2012) studied infertile women with adenomyosis and demonstrated increased uNK cells and macrophages in late secretory phase endometrium in women with diffuse or ‘adenomyoma’-type adenomyosis compared with focal adenomyosis or no disease. Leucocytes have also been investigated in the endometrium of women with leiomyomata (Kitaya and Yasuo 2010). Compared with controls, uNK cell numbers in proliferative and mid- to late secretory phase endometrium were lower in association with leiomyoma, with reduced uNK cells seen in ‘non-near nodule’ (i.e. distant from the leiomyoma) endometrium with a further reduction in ‘near nodule’ endometrium.
5.2.4.7.3 Clinical Applications of uNK Assessment in Recurrent Reproductive Failure
The association of altered uNK cells in endometrium has led to testing of uNK cells in endometrium from women suffering RM and RIF. Although increased uNK cell numbers have consistently been detected in a proportion of women suffering RM (Clifford et al. 1999; Quenby et al. 1999; Tuckerman et al. 2007), assessment differs between different laboratories and definitions of ‘abnormal’ uNK cell levels differs. Since there is variation in uNK cell numbers at different levels in endometrium (e.g. between zona spongiosum and zona compactum), an initial step should be to establish consistent methodology for quantification of uNK cell numbers in endometrium (Lash et al. 2014). Furthermore, the date of assessment may vary from LH+6 to LH+9 in different studies, reflecting a cycle stage when uNK cell numbers can vary dramatically (Russell et al. 2011, 2013). It is essential that sampling is on a specific day of the cycle or that ‘normal’ ranges for uNK cell numbers are established in large subject groups.
At present, the importance of increased uNK cells in recurrent reproductive failure remains unknown, and whether there is a correlation between high uNK cell levels and pregnancy outcome is uncertain and unclear. A recent meta-analysis did not demonstrate any difference in uNK cell numbers in RM subjects compared with controls, although percentages and numbers of peripheral blood NK cells were increased in women with RM (Seshadri and Sunkara 2014). Tang et al. (2011) conducted a systematic review and concluded that studies of uNK cells were not sufficiently large to determine whether high levels of uNK cells in the luteal phase predicted subsequent miscarriage.
5.2.4.7.4 Fetal Growth Restriction and Pre-eclampsia
Both fetal growth restriction (FGR) and pre-eclampsia (PE) are associated with failed spiral artery transformation and trophoblast invasion (Pijnenborg et al. 2006). Since uNK cells have been implicated in the control and stimulation of trophoblast invasion as well as in early stages of spiral artery transformation, it is feasible that altered uNK cell numbers or function may contribute to the aetiology of these pathological pregnancies. Furthermore, as already highlighted, there are genetic association studies that suggest that pre-eclampsia and recurrent miscarriage may be associated with specific maternal KIR haplotypes expressed by uNK cells interacting with paternal HLA-C haplotypes expressed by extravillous trophoblast (Moffett and Colucci 2014).
In common with RM and RIF, reports of uNK cells in FGR and PE are variable. Some groups have reported increased numbers of CD56+ uNK cells in decidua from women with pre-eclampsia compared with age-matched controls (Stallmach et al. 1999; Wilczynski et al. 2003; Bachmayer et al. 2006). However, in an immunohistochemical study of placental bed biopsies, we demonstrated reduced CD56+ uNK cells in both pre-eclampsia and FGR (Williams et al. 2009b). These results are similar to another report of reduced decidual CD56+ uNK cells in women with severe FGR with and without pre-eclampsia, although there were no differences in pre-eclampsia not associated with FGR (Eide et al. 2006). A flow cytometry study of decidual curettings reported no difference in the proportion of CD45+ cells that were CD56+ CD16- in pre-eclampsia compared with controls, although the proportion of CD45+ cells that were CD56+ CD16+ was reduced in pre-eclampsia (Rieger et al. 2009).
Studies of both pre-eclampsia and fetal growth restriction are limited by the fact that their clinical presentation occurs in the second half of pregnancy, whereas the pathogenetic lesions are likely to be established at a much earlier gestational age. Recent studies have investigated uNK cell function in early pregnancy decidua from pregnancies screened prior to pregnancy termination using uterine artery Doppler (Fraser et al. 2012; Wallace et al. 2013, 2014, 2015). Pregnancies were separated into those showing normal or high uterine artery Doppler resistance index as those at least risk (<1 %) and at most risk (21 %) of developing pre-eclampsia. Compared with uNK cells from low-risk pregnancies, those from pregnancies showing a high resistance index were less able to induce apoptosis of vascular smooth muscle cells and produce factors able to stimulate trophoblast invasion (Fraser et al. 2012). The cells were also less chemoattractant for trophoblast and less able to stimulate outgrowth from placental villous explants (Wallace et al. 2013), and uNK cell secretion of various angiogenic growth factors was increased (Wallace et al. 2014). Perhaps most interesting in the context of reports of the importance of paternal HLA-C/maternal KIR interactions in the pathogenesis of pre-eclampsia, a reduced proportion of uNK cells from the high-risk pregnancies expressed KIR2DL/S1, 3 and 5 and LILRB1 (Wallace et al. 2015), raising the possibility of an altered interaction with extravillous trophoblast via class I MHC antigens. Whether these results will be confirmed by others is not yet known, and it is notable that only a proportion of those pregnancies with a high uterine artery Doppler resistance index will go on to develop pre-eclampsia. Furthermore, the studies are based on vascular smooth muscle cell and extravillous trophoblast cell lines which may not be representative of the primary cells. However, these studies represent an exciting new approach to investigation of pre-eclampsia and potentially other pregnancy disorders that are associated with deficient trophoblast invasion.
5.2.4.7.5 Could uNK Cells Interact with Seminal Fluid Components?
Investigation of the role of uNK cells in pregnancy disorders has mainly focused on their potential role in mediating successful implantation and early placentation. However, uNK cells are potentially exposed to components within the seminal fluid, and this is an alternative mechanism whereby their function and/or differentiation and proliferation may be disturbed, thereby contributing to reproductive failure. Despite several studies, many in animals, that have indicated that seminal fluid contributes to reproductive success by signalling to elicit functional adaptations in the female (Robertson 2007; Schjenken and Robertson 2014), no studies have considered the potential effect of seminal fluid on uNK cell function.
Seminal fluid contains cytokines and prostaglandins that are synthesised in the male accessory glands which are transferred to the female at insemination. Early studies in humans demonstrated an influx of leucocytes, predominantly neutrophil polymorphs, into the cervix following insemination (Thompson et al. 1992), and more recent studies have demonstrated that the seminal fluid induces production of GM-CSF, IL-6, IL-8, MCP-1, MIP-3-α and IL-1-α (Sharkey et al. 2012). Evidence in mice suggests that seminal fluid induces T regulatory cells (Guerin et al. 2011), and there is some evidence for a similar effect in humans, although evidence to date is not definitive (Schjenken and Robertson 2014). Molecules within seminal fluid which may play a role in signalling are TGF-β, HLA-G5 and PGE2 (Kelly and Critchley 1997; Hutter and Dohr 1998; Robertson et al. 2002; Schjenken and Robertson 2014), and it is feasible that these could affect the function of uNK cells within endometrium. TGF-β affects the differentiation of NK cells, and incubation of peripheral blood NK cells in TGF-β1 has been shown to result in formation of cells with uNK-like phenotype (Keskin et al. 2007). TGF-β1 also affects uNK cell function by altering expression of cytokines (Eriksson et al. 2004). Soluble HLA-G has been detected in seminal fluid (Larsen et al. 2011); although effects on uNK cells have not been studied directly, it has been shown that soluble HLA-G induces a senescent phenotype in peripheral blood NK cells, altering their cytokine secretion profile (reviewed in Rajagopalan 2014).
Several pregnancy complications are commoner in the first pregnancy or when pregnancy occurs with a new partner in multiparous women, emphasising the importance of a paternal component in successful reproduction. Furthermore, women who have had recurrent miscarriages with one partner may go on to have successful pregnancy with a different partner. Although the focus has been on interactions of endometrial leucocytes with placental trophoblast cells as the pathogenetic mechanism in problems such as recurrent miscarriage and pre-eclampsia, the possibility that problems arise much earlier in the process should be considered. Given the prominence of uNK cells in endometrium around the time of implantation, as well as the known effects on NK cells in general, and in some cases on uNK cells, of the various components of seminal fluid, it is surprising that attention has not focused on the potential effect of seminal fluid on uNK cell function. This may be a promising area for future investigation.
5.3 Conclusions
Uterine NK cells have been the focus of many studies since it was appreciated that they are a major leucocytic component of the endometrial stroma at the time of implantation and in early pregnancy. There have been considerable advances in knowledge, but studies of very early pregnancy are limited by availability of tissues from humans and the limitations of extrapolating from animal (predominantly mouse) models. Functional studies of uNK cells from early pregnancy have highlighted their functions, with particular focus on secretion of cytokines and angiogenic growth factors pointing to roles in facilitation and control of trophoblast invasion and spiral artery remodelling in early pregnancy. Despite studies suggesting altered uNK cell numbers in recurrent reproductive failure, the functional consequences remain uncertain. It is important that the mechanisms that underlie the increased numbers of uNK cells as well as the functional consequences are determined before it will be possible to develop effective diagnostic and therapeutic approaches. Furthermore, the possible contribution of male factors such as components within seminal fluid should be considered in the context of uNK cell differentiation and function in recurrent reproductive failure.
References
Ain R, Trinh ML, Soares MJ (2004) Interleukin-11 signaling is required for the differentiation of natural killer cells at the maternal-fetal interface. Dev Dyn 231:700–708
Aluvihare VR, Kallikourdis M, Betz AG (2004) Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol 5:266–271
Bachmayer N, Rafik Hamad R, Liszka L, Bremme K, Sverremark-Ekstrom E (2006) Aberrant uterine natural killer (NK)-cell expression and altered placental and serum levels of the NK-cell promoting cytokine interleukin-12 in pre-eclampsia. Am J Reprod Immunol 56:292–301
Bellone G, Aste-Amezaga M, Trinchieri G, Rodeck U (1995) Regulation of NK cell functions by TGF-beta 1. J Immunol 155:1066–1073
Benson DM Jr, Yu J, Beckneil B, Wei M, Freud AG, Ferketich AK, Trotta R, Perrotti D, Briesewitz R, Caligiuri MA (2009) Stem cell factor and interleukin-2/15 combine to enhance MAPK-mediated proliferation of human natural killer cells. Blood 113:2706–2714
Berbic M, Fraser IS (2013) Immunology of normal and abnormal menstruation. Womens Health (Lond Engl) 9:387–395
Blois SM, Klapp BF, Barrientos G (2011) Decidualization and angiogenesis in early pregnancy: unravelling the functions of DC and NK cells. J Reprod Immunol 88:86–92
Broudy VC (1997) Stem cell factor and hematopoiesis. Blood 90:1345–1364
Bulmer JN, Lash GE (2005) Human uterine natural killer cells: a reappraisal. Mol Immunol 42:511–521
Bulmer JN, Lunny DP, Hagin SV (1988a) Immunohistochemical characterization of stromal leucocytes in nonpregnant human endometrium. Am J Reprod Immunol Microbiol 17:83–90
Bulmer JN, Smith J, Morrison L, Wells M (1988b) Maternal and fetal cellular relationships in the human placental basal plate. Placenta 9:237–246
Bulmer JN, Morrison L, Longfellow M, Ritson A, Pace D (1991) Granulated lymphocytes in human endometrium: histochemical and immunohistochemical studies. Hum Reprod 6:791–798
Bulmer JN, Williams PJ, Lash GE (2010) Immune cells in the placental bed. Int J Dev Biol 54:281–294
Castriconi R, Cantoni C, Della Chiesa M, Vitale M, Marcenaro E, Conte R, Biassoni R, Bottino C, Moretta L, Moretta A (2003) Transforming growth factor beta 1 inhibits expression of NKp30 and NKG2D receptors: consequences for the NK-mediated killing of dendritic cells. Proc Natl Acad Sci USA 100:4120–4125
Champion H, Innes BA, Robson SC, Lash GE, Bulmer JN (2012) Effects of interleukin-6 on extravillous trophoblast invasion in early human pregnancy. Mol Hum Reprod 18:391–400
Chao KH, Yang YS, Ho HN, Chen SU, Chen HF, Dai HJ, Huang SC, Gill TJ 3rd (1995) Decidual natural killer cytotoxicity decreased in normal pregnancy but not in anembryonic pregnancy and recurrent spontaneous abortion. Am J Reprod Immunol 34:274–280
Chazara O, Xiong S, Moffett A (2011) Maternal KIR and fetal HLA-C: a fine balance. J Leukoc Biol 90:703–716
Chen LJ, Han ZQ, Zhou H, Zou L, Zou P (2010) Inhibition of HLA-G expression via RNAi abolishes resistance of Extravillous trophoblast cell line TEV-1 to NK lysis. Placenta 31:519–527
Chumbly G, King A, Robertson K, Holmes N, Loke YW (1994) Resistance of HLA-G and HLA-A2 transfectants to lysis by decidual NK cells. Cell Immunol 155:312–322
Clark DA (2014) Popular myths in reproductive immunology. J Reprod Immunol 104–105:54–62
Clifford K, Flanagan AM, Regan L (1999) Endometrial CD56+ natural killer cells in women with recurrent miscarriage: a histomorphometric study. Hum Reprod 14:2727–2730
Cooper MA, Fehniger TA, Caligiuri MA (2001) The biology of human natural killer-cell subsets. Trends Immunol 22:633–640
Craven CM, Morgan T, Ward K (1998) Decidual spiral artery remodelling begins before cellular interaction with cytotrophoblasts. Placenta 19:241–252
De Oliveira LG, Lash GE, Murray-Dunning C, Bulmer JN, Innes BA, Searle RF, Sass N, Robson SC (2010) Role of interleukin 8 in uterine natural killer cell regulation of extravillous trophoblast cell invasion. Placenta 31:595–601
Dietl J, Hönig A, Kämmerer U, Rieger L (2006) Natural killer cells and dendritic cells at the human feto-maternal interface: an effective cooperation? Placenta 27:341–347
Dimitriadis E, White CA, Jones RL, Salamonsen LA (2005) Cytokines, chemokines and growth factors in endometrium related to implantation. Hum Reprod Update 11:613–630
Disep B, Innes BA, Cochrane HR, Tijani S, Bulmer JN (2004) Immunohistochemical characterization of endometrial leucocytes in endometritis. Histopathology 45:625–632
Dunn CL, Critchley HO, Kelly RW (2002) IL-15 regulation in human endometrial stromal cells. J Clin Endocrinol Metab 87:1898–1901
Eide IP, Rolfseng T, Isaksen CV, Mecsei R, Roald B, Lydersen S, Salvesen KA, Harsem NK, Austgulen R (2006) Serious foetal growth restriction is associated with reduced proportions of natural killer cells in decidua basalis. Virchows Arch 448:269–276
Emmer PM, Steegers EA, Kerstens HM, Bulten J, Nelen WL, Boer K, Joosten I (2002) Altered phenotype of HLA-G expressing trophoblast and decidual natural killer cells in pathological pregnancies. Hum Reprod 17:1072–1080
Eriksson M, Meadows SK, Wira CR, Sentman CL (2004) Unique phenotype of human uterine NK cells and their regulation by endogenous TGF-beta. J Leukoc Biol 76:667–675
Evans J, Salamonsen LA (2012) Inflammation, leukocytes and menstruation. Rev Endocr Metab Disord 13:277–288
Everett C (1997) Incidence and outcome of bleeding before the 20th week of pregnancy: prospective study from general practice. BMJ 315:32–34
Fraser R, Whitley GS, Johnstone AP et al (2012) Impaired decidual natural killer cell regulation of vascular remodelling in early human pregnancies with high uterine artery resistance. J Pathol 228:322–332
Freud AG, Yokohama A, Becknell B, Lee MT, Mao HC, Ferketich AK, Caligiuri MA (2006) Evidence for discrete stages of human natural killer cell differentiation in vivo. J Exp Med 203:1033–1043
Fu B, Wang F, Sun R, Ling B, Tian Z, Wei H (2011) CD11b and CD27 reflect distinct population and functional specialization in human natural killer cells. Immunology 133:350–359
Fu B, Li X, Sun R, Tong X, Ling B, Tian Z, Wei H (2013) Natural killer cells promote immune tolerance by regulating inflammatory TH17 cells at the human maternal-fetal interface. Proc Natl Acad Sci USA 110:E231–240
Fukui A, Fujii S, Yamaguchi E, Kimura H, Sato S, Saito Y (1999) Natural killer cell subpopulations and cytotoxicity for infertile patients undergoing in vitro fertilization. Am J Reprod Immunol 41:413–422
Gardner L, Moffett A (2003) Dendritic cells in the human decidua. Biol Reprod 69:1438–1446
Gargett CE, Masuda H (2010) Adult stem cells in the endometrium. Mol Hum Reprod 16:818–834
Gibson DA, McInnes KJ, Critchley HO, Saunders PT (2013) Endometrial Intracrinology—generation of an estrogen-dominated microenvironment in the secretory phase of women. J Clin Endocrinol Metab 98:E1802–1806
Guerin LR, Moldenhauer LM, Prins JR, Bromfield JJ, Hayball JD, Robertson SA (2011) Seminal fluid regulates accumulation of FOXP3+ regulatory T cells in the preimplantation mouse uterus through expanding the FOXP3+ cell pool and CCL19-mediated recruitment. Biol Reprod 85:397–408
Gustafsson C, Mjösberg J, Matussek A, Geffers R, Matthiesen L, Berg G, Sharma S, Buer J, Ernerudh J (2008) Gene expression profiling of human decidual macrophages: evidence for immunosuppressive phenotype. PLoS One 3, e2078
Hamperl H, Hellweg G (1958) Granular endometrial stroma cells. Obstet Gynecol 11:379–387
Hanna J, Goldman-Wohl D, Hamani Y, Avraham I, Greenfield C, Natanson-Yaron S, Prus D, Cohen-Daniel L, Arnon TI, Manaster I, Gazit R, Yutkin V, Benharroch D, Porgador A, Keshet E, Yagel S, Mandelboim O (2006) Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat Med 12:1065–1074
Hannan NJ, Paiva P, Meehan KL, Rombauts LJ, Gardner DK, Salamonsen LA (2011) Analysis of fertility-related soluble mediators in human uterine fluid identifies VEGF as a key regulator of embryo implantation. Endocrinology 152:4948–4956
Hannon T, Innes BA, Lash GE, Bulmer JN, Robson SC (2012) Effects of local decidua on trophoblast invasion and spiral artery remodeling in focal placenta creta—an immunohistochemical study. Placenta 33:998–1004
Heikkinen J, Mottonen M, Komi J, Alanen A, Lassila O (2003) Phenotypic characterization of human decidual macrophages. Clin Exp Immunol 131:498–505
Helige C, Ahammer H, Moser G, Hammer A, Dohr G, Huppertz B, Sedlmayr P (2014) Distribution of decidual natural killer cells and macrophages in the neighbourhood of the trophoblast invasion front: a quantitative evaluation. Hum Reprod 29:8–17
Henderson TA, Saunders PTK, Moffett-King A, Groome NP, Critchley HOD (2003) Steroid receptor expression in uterine natural killer cells. J Clin Endocrinol Metab 88:440–449
Hiby SE, Walker JJ, O’shaughnessy KM, Redman CW, Carrington M, Trowsdale J, Moffett A (2004) Combinations of maternal KIR and fetal HLA-C genes influence the risk of preeclampsia and reproductive success. J Exp Med 200:957–965
Hiby SE, Apps R, Sharkey AM, Farrell LE, Gardner L, Mulder A, Claas FH, Walker JJ, Redman CC, Morgan L, Tower C, Regan L, Moore GE, Carrington M, Moffett A (2010) Maternal activating KIRs protect against human reproductive failure mediated by fetal HLA-C2. J Clin Invest 120:4102–4110
Houser BL (2012) Decidual macrophages and their roles at the maternal-fetal interface. Yale J Biol Med 85:105–118
Houser BL, Tilburgs T, Hill J, Nicotra ML, Strominger JL (2011) Two unique human decidual macrophage populations. J Immunol 186:2633–2642
Hu Y, Dutz JP, Maccalman CD, Yong P, Tan R, Von Dadelszen P (2006) Decidual NK cells alter in vitro first trimester extravillous cytotrophoblast migration: a role for IFN-gamma. J Immunol 177:8522–8530
Hutter H, Dohr G (1998) HLA expression on immature and mature human germ cells. J Reprod Immunol 38:101–122
Jokhi PP, King A, Loke YW (1994) Production of granulocyte-macrophage colony-stimulating factor by human trophoblast cells and by decidual large granular lymphocytes. Hum Reprod 9:1660–1669
Jones RK, Bulmer JN, Searle RF (1997) Cytotoxic activity of endometrial granulated lymphocytes during the menstrual cycle in humans. Biol Reprod 57:1217–1222
Jones RK, Searle RF, Stewart JA, Turner S, Bulmer JN (1998) Apoptosis, bcl-2 expression, and proliferative activity in human endometrial stroma and endometrial granulated lymphocytes. Biol Reprod 58:995–1002
Jones RL, Stoikos C, Findlay JK, Salamonsen LA (2006) TGF-beta superfamily expression and actions in the endometrium and placenta. Reproduction 132:217–232
Kalkunte SS, Mselle TF, Norris WE, Wira CR, Sentman CL, Sharma S (2009) Vascular endothelial growth factor C facilitates immune tolerance and endovascular activity of human uterine NK cells at the maternal-fetal interface. J Immunol 182:4085–4092
Kam EP, Gardner L, Loke YW, King A (1999) The role of trophoblast in the physiological change in decidual spiral arteries. Hum Reprod 14:2131–2138
Kämmerer U, Marzusch K, Kröber S, Ruck P, Handgretinger R, Dietl J (1999) A subset of CD56+ large granular lymphocytes in first-trimester human decidua are proliferating cells. Fertil Steril 71:74–79
Kammerer U, Eggert AO, Kapp M, McLellan AD, Geijtenbeek TB, Dietl J, van Kooyk Y, Kampgen E (2003) Unique appearance of proliferating antigen-presenting cells expressing DC-SIGN (CD209) in the decidua of early human pregnancy. Am J Pathol 162:887–896
Kauma S, Huff T, Krystal G, Ryan J, Takacs P, Turner T (1996) The expression of stem cell factor and its receptor, c-kit in human endometrium and placental tissues during pregnancy. J Clin Endocrinol Metab 81:1261–1266
Kelly RW, Critchley HO (1997) Immunomodulation by human seminal plasma: a benefit for spermatozoon and pathogen? Hum Reprod 12:2200–2207
Keskin DB, Allan DSJ, Rybalov B, Andzelm MM, Stern JNH, Kopcow HD, Koopman LA, Strominger JL (2007) TGFβ promotes conversion of CD16+ peripheral blood NK cells into CD16- NK cells with similarities to decidual NK cells. Proc Natl Acad Sci USA 104:3378–3383
King A, Loke YW (1990) Human trophoblast and JEG choriocarcinoma cells are sensitive to lysis by IL-2-stimulated decidual NK cells. Cell Immunol 129:435–448
King A, Wooding P, Gardner L, Loke YW (1993) Expression of perforin, granzyme A and TIA-1 by human uterine CD56+ NK cells implies they are activated and capable of effector functions. Hum Reprod 8:2061–2067
Kitaya K, Yasuo T (2010) Leukocyte density and composition in human cycling endometrium with uterine fibroids. Hum Immunol 71:158–163
Kitaya K, Yamaguchi T, Yasuo T, Okubo T, Honjo H (2007) Post-ovulatory rise of endometrial CD16(−) natural killer cells: in situ proliferation of residual cells or selective recruitment from circulating peripheral blood? J Reprod Immunol 76:45–53
Klentzeris LD, Bulmer JN, Warren MA, Morrison L, Li TC, Cooke ID (1994) Lymphoid tissue in the endometrium of women with unexplained infertility: morphometric and immunohistochemical aspects. Hum Reprod 9:646–652
Koopman LA, Kopcow HD, Rybalov B, Boyson JE, Orange JS, Schatz F, Masch R, Lockwood CJ, Schachter AD, Park PJ, Strominger JL (2003) Human decidual natural killer cells are a unique NK cell subset with immunomodulatory potential. J Exp Med 198:1201–1212
Kopcow HD, Allan DS, Chen X, Rybalov B, Andzelm MM, Ge B, Strominger JL (2005) Human decidual NK cells form immature activating synapses and are not cytotoxic. Proc Natl Acad Sci USA 102:15563–15568
Kopcow HD, Eriksson M, Mselle TF, Damrauer SM, Wira CR, Sentman CL, Strominger JL (2010) Human decidual uNK cells from gravid uteri and NK cells from cycling endometrium are distinct NK cell subsets. Placenta 31:334–338
Kwak JY, Beaman KD, Gilman-Sachs A, Ruiz JE, Schewitz D, Beer AE (1995) Up-regulated expression of CD56+, CD56+/CD16+, and CD19+ cells in peripheral blood lymphocytes in pregnant women with recurrent pregnancy losses. Am J Reprod Immunol 34:93–99
Lachapelle MH, Miron P, Hemmings R, Roy DC (1996) Endometrial T, B, and NK cells in patients with recurrent spontaneous abortion. Altered profile and pregnancy outcome. J Immunol 156:4027–4034
Larsen MH, Bzorek M, Pass MB, Larsen LG, Nielsen MW, Svendsen SG, Lindhard A, Hviid TV (2011) Human leukocyte antigen-G in the male reproductive system and in seminal plasma. Mol Hum Reprod 17:727–738
Lash GE, Bulmer JN (2011) Do uterine natural killer (uNK) cells contribute to female reproductive disorders? J Reprod Immunol 88:156–164
Lash GE, Otun HA, Innes BA, Kirkley M, De Oliveira L, Searle RF, Robson SC, Bulmer JN (2006a) Interferon-gamma inhibits extravillous trophoblast cell invasion by a mechanism that involves both changes in apoptosis and protease levels. FASEB J 20:2512–2518
Lash GE, Schiessl B, Kirkley M, Innes BA, Cooper A, Searle RF, Robson SC, Bulmer JN (2006b) Expression of angiogenic growth factors by uterine natural killer cells during early pregnancy. J Leukoc Biol 80:572–580
Lash GE, Otun HA, Innes BA, Bulmer JN, Searle RF, Robson SC (2006c) Low oxygen concentrations inhibit trophoblast cell invasion from early gestation placental explants via alterations in levels of the urokinase plasminogen activator system. Biol Reprod 74:403–409
Lash GE, Robson SC, Bulmer JN (2010a) Review: functional role of uterine natural killer (uNK) cells in human early pregnancy decidua. Placenta 31:S87–S92
Lash GE, Otun HA, Innes BA, Percival K, Searle RF, Robson SC, Bulmer JN (2010b) Regulation of extravillous trophoblast invasion by uterine natural killer cells is dependent on gestational age. Hum Reprod 25:1137–1145
Lash GE, Naruse K, Robson A, Innes BA, Searle RF, Robson SC, Bulmer JN (2011) Interaction between uterine natural killer cells and extravillous trophoblast cells: effect on cytokine and angiogenic growth factor production. Hum Reprod 26:2289–2295
Lash GE, Innes BA, Drury JA, Robson SC, Quenby S, Bulmer JN (2012) Localization of angiogenic growth factors and their receptors in the human endometrium throughout the menstrual cycle and in recurrent miscarriage. Hum Reprod 27:183–195
Lash GE, Laird SM, Li TC, Innes BA, Mariee N, Patel N, Quenby S, Bulmer JN (2014) Standardisation of uterine natural killer (uNK) cell measurements in endometrium of women with recurrent miscarriage. ESHRE Annual Conference Abstract
Ledee-Bataille N, Dubanchet S, Coulomb-L'hermine A, Durand-Gasselin I, Frydman R, Chaouat G (2004) A new role for natural killer cells, interleukin (IL)-12, and IL-18 in repeated implantation failure after in vitro fertilization. Fertil Steril 81:59–65
Lee JY, Kim EN, Hong JS, Shim JY, Hwang D, Kim KC, Lee J, Kim CJ (2015) Diffuse decidual leucocytoclastic necrosis and NK cell aggregates: two distinct features of miscarriage. J Clin Pathol 68:119–124
Leno-Durán E, Muñoz-Fernández R, Olivares EG, Tirado-González I (2014) Liaison between natural killer cells and dendritic cells in human gestation. Cell Mol Immunol 11:449–455
Li XF, Charnock-Jones DS, Zhang E, Hiby S, Malik S, Day K, Licence D, Bowen JM, Gardner L, King A, Loke YW, Smith SK (2001) Angiogenic growth factor messenger ribonucleic acids in uterine natural killer cells. J Clin Endocrinol Metab 86:1823–1834
Lynch L, Golden-Mason L, Eogan M, O’Herlihy C, O’Farrelly C (2007) Cells with haematopoietic stem cell phenotype in adult human endometrium: relevance to infertility? Hum Reprod 22:919–926
Male V, Hughes T, McClory S, Colucci F, Caligiuri MA, Moffett A (2010) Immature NK cells, capable of producing IL-22, are present in human uterine mucosa. J Immunol 185:3913–3918
Manaster I, Mizrahi S, Goldman-Wohl D, Sela HY, Stern-Ginossar N, Lankry D, Gruda R, Hurwitz A, Bdolah Y, Haimov-Kochman R, Yagel S, Mandelboim O (2008) Endometrial NK cells are special immature cells that await pregnancy. J Immunol 181:1869–1876
Mariee N, Li TC, Laird SM (2012) Expression of leukaemia inhibitory factor and interleukin 15 in endometrium of women with recurrent implantation failure after IVF; correlation with the number of endometrial natural killer cells. Hum Reprod 27:1946–1954
Marlin R, Duriez M, Berkane N, de Truchis C, Madec Y, Rey-Cuille MA, Cummings JS, Cannou C, Quillay H, Barré-Sinoussi F, Nugeyre MT, Menu E (2012) Dynamic shift from CD85j/ILT-2 to NKG2D NK receptor expression pattern on human decidual NK during the first trimester of pregnancy. PLoS One 7, e30017
Marshall RJ, Jones DB (1988) An immunohistochemical study of lymphoid tissue in human endometrium. Int J Gynecol Pathol 3:225–235
Matsuura-Sawada R, Murakami T, Ozawa Y, Nabeshima H, Akahira J, Sato Y, Koyanagi Y, Ito M, Terada Y, Okamura K (2005) Reproduction of menstrual changes in transplanted human endometrial tissue in immunodeficient mice. Hum Reprod 20:1477–1484
Matteo MG, Greco P, Rosenberg P, Mestice A, Baldini D, Falagario T, Martino V, Santodirocco M, Massenzio F, Castellana L, Specchia G, Liso A (2007) Normal percentage of CD56bright natural killer cells in young patients with a history of repeated unexplained implantation failure after in vitro fertilization cycles. Fertil Steril 88:990–993
Maybin JA, Barcroft J, Thiruchelvam U, Hirani N, Jabbour HN, Critchley HO (2012) The presence and regulation of connective tissue growth factor in the human endometrium. Hum Reprod 27:1112–1121
McGrath E, Ryan EJ, Lynch L, Golden-Mason L, Mooney E, Eogan M, O’Herlihy C, O’Farrelly C (2009) Changes in endometrial natural killer cell expression of CD94, CD158a and CD158b are associated with infertility. Am J Reprod Immunol 61:265–276
Michimata T, Ogasawara MS, Tsuda H, Suzumori K, Aoki K, Sakai M, Fujimura M, Nagata K, Nakamura M, Saito S (2002) Distributions of endometrial NK cells, B cells, T cells, and Th2/Tc2 cells fail to predict pregnancy outcome following recurrent abortion. Am J Reprod Immunol 47:196–202
Mjosberg J, Berg G, Jenmalm MC, Ernerudh J (2010) FOXP3+ regulatory T cells and T helper 1, T helper 2, and T helper 17 cells in human early pregnancy decidua. Biol Reprod 82:698–705
Moffett A, Colucci F (2014) Uterine NK cells: active regulators at the maternal-fetal interface. J Clin Invest 124:1872–1879
Morris H, Edwards J, Tiltman A, Emms M (1985) Endometrial lymphoid tissue: an immunohistological study. J Clin Pathol 38:644–652
Mrózek E, Anderson P, Caligiuri MA (1996) Role of interleukin-15 in the development of human CD56+ natural killer cells from CD34+ hematopoietic progenitor cells. Blood 87:2632–2640
Nagamatsu T, Schust DJ (2010) The contribution of macrophages to normal and pathological pregnancies. Am J Reprod Immunol 63:460–471
Nakashima A, Shiozaki A, Myojo S, Ito M, Tatematsu M, Sakai M, Takamori Y, Ogawa K, Nagata K, Saito S (2008) Granulysin produced by uterine natural killer cells induces apoptosis of extravillous trophoblasts in spontaneous abortion. Am J Pathol 173:653–664
Naruse K, Lash GE, Innes BA, Otun HA, Searle RF, Robson SC, Bulmer JN (2009a) Localization of matrix metalloproteinase (MMP)-2, MMP-9 and tissue inhibitors for MMPs (TIMPs) in uterine natural killer cells in early human pregnancy. Hum Reprod 24:553–561
Naruse K, Lash GE, Bulmer JN, Innes BA, Otun HA, Searle RF, Robson SC (2009b) The urokinase plasminogen activator (uPA) system in uterine natural killer cells in the placental bed during early pregnancy. Placenta 30:398–404
Nevers T, Kalkunte S, Sharma S (2011) Uterine Regulatory T cells, IL-10 and hypertension. Am J Reprod Immunol 66:88–92
Okada S, Okada H, Sanezumi M, Nakajima T, Yasuda K, Kanzaki H (2000) Expression of interleukin-15 in human endometrium and decidua. Mol Hum Reprod 6:75–80
Okada H, Nakajima T, Yasuda K, Kanzaki H (2004) Interleukin-1 inhibits interleukin-15 production by progesterone during in vitro decidualization in human. J Reprod Immunol 61:3–12
Omwandho CO, Konrad L, Halis G, Oehmke F, Tinneberg HR (2010) Role of TGF-betas in normal human endometrium and endometriosis. Hum Reprod 25:101–109
Otun HA, Innes BA, Lash GE, Robson SC, Bulmer JN (2009) Immunohistochemical characterization of uterine NK cell differentiation markers in endometrium and decidua. Reprod Sci 16(Suppl):182A
Pace D, Morrison L, Bulmer JN (1989) Proliferative activity in endometrial stromal granulocytes throughout menstrual cycle and early pregnancy. J Clin Pathol 42:35–39
Pace D, Longfellow M, Bulmer JN (1991) Characterization of intraepithelial lymphocytes in human endometrium. J Reprod Fertil 91:165–174
Parhar RS, Yagel S, Lala PK (1989) PGE2-mediated immunosuppression by first trimester human decidual cells blocks activation of maternal leukocytes in the decidua with potential anti-trophoblast activity. Cell Immunol 120:61–74
Petitbarat M, Rahmati M, Sérazin V, Dubanchet S, Morvan C, Wainer R, de Mazancourt P, Chaouat G, Foidart JM, Munaut C, Lédée N (2011) TWEAK appears as a modulator of endometrial IL-18 related cytotoxic activity of uterine natural killers. PLoS One 6, e14497
Pijnenborg R, Vercruysse L, Hanssens M (2006) The uterine spiral arteries in human pregnancy: facts and controversies. Placenta 27:939–958
Pitman H, Innes BA, Robson SC, Bulmer JN, Lash GE (2013) Altered expression of interleukin-6, interleukin-8 and their receptors in decidua of women with sporadic miscarriage. Hum Reprod 28:2075–2086
Plaisier M, Dennert I, Rost E, Koolwijk P, van Hinsbergh VW, Helmerhorst FM (2009) Decidual vascularization and the expression of angiogenic growth factors and proteases in first trimester spontaneous abortions. Hum Reprod 24:185–197
Quack KC, Vassiliadou N, Pudney J, Anderson DJ, Hill JA (2001) Leukocyte activation in the decidua of chromosomally normal and abnormal fetuses from women with recurrent abortion. Hum Reprod 16:949–955
Quenby SM, Farquharson RG (1993) Predicting recurring miscarriage: what is important? Obstet Gynecol 82:132–138
Quenby S, Bates M, Doig T, Brewster J, Lewis-Jones DI, Johnson PM, Vince G (1999) Pre-implantation endometrial leukocytes in women with recurrent miscarriage. Hum Reprod 14:2386–2391
Quenby S, Nik H, Innes B, Lash G, Turner M, Drury J, Bulmer J (2009) Uterine natural killer cells and angiogenesis in recurrent reproductive failure. Hum Reprod 24:45–54
Rai R, Regan L (2006) Recurrent miscarriage. Lancet 368:601–611
Rajagopalan S (2014) HLA-G-mediated NK cell senescence promotes vascular remodeling: implications for reproduction. Cell Mol Immunol 11:460–466
Regan L, Rai R (2000) Epidemiology and the medical causes of miscarriage. Baillieres Best Pract Res Clin Obstet Gynaecol 14:839–854
Repnik U, Tilburgs T, Roelen DL, van der Mast BJ, Kanhai HH, Scherjon S, Claas FH (2008) Comparison of macrophage phenotype between decidua basalis and decidua parietalis by flow cytometry. Placenta 29:405–412
Rieger L, Honig A, Sutterlin M, Kapp M, Dietl J, Ruck P, Kammerer U (2004) Antigen-presenting cells in human endometrium during the menstrual cycle compared to early pregnancy. J Soc Gynecol Investig 11:488–493
Rieger L, Segerer S, Bernar T, Kapp M, Majic M, Morr AK, Dietl J, Kammerer U (2009) Specific subsets of immune cells in human decidua differ between normal pregnancy and preeclampsia—a prospective observational study. Reprod Biol Endocrinol 7:132
Ritson A, Bulmer JN (1989) Isolation and functional studies of granulated lymphocytes in first trimester human decidua. Clin Exp Immunol 77:263–268
Robertson SA (2007) Seminal fluid signaling in the female reproductive tract: lessons from rodents and pigs. J Anim Sci 85(Suppl):E36–44
Robertson SA, Ingman WV, O’Leary S, Sharkey DJ, Tremellen KP (2002) Transforming growth factor beta–a mediator of immune deviation in seminal plasma. J Reprod Immunol 57:109–128
Robson A, Harris LK, Innes BA, Lash GE, Aljunaidy MM, Aplin JD, Baker PN, Robson SC, Bulmer JN (2012) Uterine natural killer cells initiate spiral artery remodeling in human pregnancy. FASEB J 26:4876–4885
Rouas-Freiss N, Goncalves RM, Menier C, Dausset J, Carosella ED (1997) Direct evidence to support the role of HLA-G in protecting the fetus from maternal uterine natural killer cytolysis. Proc Natl Acad Sci USA 94:11520–11525
Russell P, Anderson L, Lieberman D, Tremellen K, Yilmaz H, Cheerala B, Sacks G (2011) The distribution of immune cells and macrophages in the endometrium of women with recurrent reproductive failure I: Techniques. J Reprod Immunol 91:90–102
Russell P, Sacks G, Tremellen K, Gee A (2013) The distribution of immune cells and macrophages in the endometrium of women with recurrent reproductive failure. III: Further observations and reference ranges. Pathology 45:393–401
Saito S, Nishikawa K, Morii T, Enomoto M, Narita N, Motooshi K, Ichijo M (1993) Cytokine production by CD16−CD56bright natural killer cells in the human early pregnancy decidua. Int Immunol 5:559–563
Salamonsen LA, Zhang J, Brasted M (2002) Leukocyte networks and human endometrial remodelling. J Reprod Immunol 57:95–108
Scaife PJ, Searle RF, Robson SC, Innes BA, Bulmer JN (2004) Comparison of decidual leucocyte populations in spontaneous early pregnancy loss. J Soc Gynecol Investig 11:285A
Scaife PJ, Bulmer JN, Robson SC, Innes BA, Searle RF (2006) Effector activity of decidual CD8+ T lymphocytes in early human pregnancy. Biol Reprod 75:562–567
Schjenken JE, Robertson SA (2014) Seminal fluid and immune adaptation for pregnancy–comparative biology in mammalian species. Reprod Domest Anim 49(Suppl 3):27–36
Schulke L, Manconi F, Markham R, Fraser IS (2008) Endometrial dendritic cell populations during the normal menstrual cycle. Hum Reprod 23:1574–1580
Searle RF, Jones RK, Bulmer JN (1999) Phenotypic analysis and proliferative responses of human endometrial granulated lymphocytes during the menstrual cycle. Biol Reprod 60:871–878
Seshadri S, Sunkara SK (2014) Natural killer cells in female infertility and recurrent miscarriage: a systematic review and meta-analysis. Hum Reprod Update 20:429–438
Sharkey AM, Gardner L, Hiby S, Farrell L, Apps R, Masters L, Goodridge J, Lathbury L, Stewart CA, Verma S, Moffett A (2008) Killer Ig-like receptor expression in uterine NK cells is biased toward recognition of HLA-C and alters with gestational age. J Immunol 181:39–46
Sharkey DJ, Tremellen KP, Jasper MJ, Gemzell-Danielsson K, Robertson SA (2012) Seminal fluid induces leukocyte recruitment and cytokine and chemokine mRNA expression in the human cervix after coitus. J Immunol 188:2445–2454
Shimada S, Iwabuchi K, Kato EH, Morikawa M, Sakuragi N, Onoé K, Minakami H, Yamada H (2003) No difference in natural-killer-T cell population, but Th2/Tc2 predominance in peripheral blood of recurrent aborters. Am J Reprod Immunol 50:334–339
Shimada S, Kato EH, Morikawa M, Iwabuchi K, Nishida R, Kishi R, Onoe K, Minakami H, Yamada H (2004) No difference in natural killer or natural killer T-cell population, but aberrant T-helper cell population in the endometrium of women with repeated miscarriage. Hum Reprod 19:1018–1024
Smith SD, Dunk CE, Aplin JD, Harris LK, Jones RL (2009) Evidence for immune cell involvement in decidual spiral arteriole remodeling in early human pregnancy. Am J Pathol 174:1959–1971
Spornitz UM (1992) The functional morphology of the human endometrium and decidua. Adv Anat Embryol Cell Biol 124:1–99
Stallmach T, Hebisch G, Orban P, Lu X (1999) Aberrant positioning of trophoblast and lymphocytes in the feto-maternal interface with pre-eclampsia. Virchows Arch 434:207–211
Stewart JA, Bulmer JN, Murdoch AP (1998) Endometrial leucocytes: expression of steroid hormone receptors. J Clin Pathol 51:121–126
Szereday L, Miko E, Meggyes M, Barakonyi A, Farkas B, Varnagy A, Bodis J, Lynch L, O'Farrelly C, Szekeres-Bartho J (2012) Commitment of decidual haematopoietic progenitor cells in first trimester pregnancy. Am J Reprod Immunol 67:9–16
Tang AW, Alfirevic Z, Quenby S (2011) Natural killer cells and pregnancy outcomes in women with recurrent miscarriage and infertility: a systematic review. Hum Reprod 26:1971–1980
Thiruchelvam U, Dransfield I, Saunders PT, Critchley HO (2013) The importance of the macrophage within the human endometrium. J Leukoc Biol 93:217–225
Thompson LA, Barratt CL, Bolton AE, Cooke ID (1992) The leukocytic reaction of the human uterine cervix. Am J Reprod Immunol 28:85–89
Tilburgs T, Strominger JL (2013) CD8+ effector T cells at the fetal-maternal interface, balancing fetal tolerance and antiviral immunity. Am J Reprod Immunol 69:395–407
Tilburgs T, Roelen DL, van der Mast BJ, de Groot-Swings GM, Kleijburg C, Scherjon SA, Claas FH (2008) Evidence for a selective migration of fetus-specific CD4 + CD25bright regulatory T cells from the peripheral blood to the decidua in human pregnancy. J Immunol 180:5737–5745
Tilburgs T, Claas FH, Scherjon SA (2010) Elsevier Trophoblast Research Award Lecture: Unique properties of decidual T cells and their role in immune regulation during human pregnancy. Placenta 31:S82–S86
Tirado-González I, Muñoz-Fernández R, Prados A, Leno-Durán E, Martin F, Abadía-Molina AC, Olivares EG (2012) Apoptotic DC-SIGN+ cells in normal human decidua. Placenta 33:257–263
Tremellen KP, Russell P (2012) The distribution of immune cells and macrophages in the endometrium of women with recurrent reproductive failure. II: adenomyosis and macrophages. J Reprod Immunol 93:58–63
Trundley A, Moffett A (2004) Human uterine leukocytes and pregnancy. Tissue Antigens 63:1–12
Tuckerman E, Laird SM, Prakash A, Li TC (2007) Prognostic value of the measurement of uterine natural killer cells in the endometrium of women with recurrent miscarriage. Hum Reprod 22:2208–2213
Tuckerman E, Mariee N, Prakash A, Li TC, Laird S (2010) Uterine natural killer cells in peri-implantation endometrium from women with repeated implantation failure after IVF. J Reprod Immunol 87:60–66
Umekage H, Saito S, Morikawa H (1998) Enhancement by stem cell factor of interleukin-2 (IL-2)-induced DNA synthesis in human decidual CD16- CD56bright natural killer cells mediated by increased expression of the IL-2 receptor alpha chain. J Reprod Immunol 40:1–24
Vacca P, Vitale C, Montaldo E, Conte R, Cantoni C, Fulcheri E, Darretta V, Moretta L, Mingari MC (2011) CD34+ hematopoietic precursors are present in human decidua and differentiate into natural killer cells upon interaction with stromal cells. Proc Natl Acad Sci USA 108:2402–2407
van Beekhuizen HJ, Joosten I, de Groot AN, Lotgering FK, van der Laak J, Bulten J (2009) The number of multinucleated trophoblastic giant cells in the basal decidua is decreased in retained placenta. J Clin Pathol 62:794–797
van Beekhuizen HJ, Joosten I, Lotgering FK, Bulten J, van Kempen LC (2010) Natural killer cells and HLA-G expression in the basal decidua of human placenta adhesiva. Placenta 31:1078–1084
van der Meer A, Lukassen HGM, van Lierop MJC, Wijnands F, Mosselman S, Braat DDM, Joosten I (2004) Membrane-bound HLA-G activates proliferation and interferon-γ production by uterine natural killer cells. Mol Hum Reprod 10:189–195
van der Meer A, Lukassen HG, van Cranenbroek B, Weiss EH, Braat DD, van Lierop MJ, Joosten I (2007) Soluble HLA-G promotes Th1-type cytokine production by cytokine-activated uterine and peripheral natural killer cells. Mol Hum Reprod 13:123–133
Vassiliadou N, Bulmer JN (1996) Immunohistochemical evidence for increased numbers of ‘classic’ CD57+ natural killer cells in the endometrium of women suffering spontaneous early pregnancy loss. Hum Reprod 11:1569–1574
Vassiliadou N, Bulmer JN (1998) Functional studies of human decidua in spontaneous early pregnancy loss: effect of soluble factors and purified CD56+ lymphocytes on killing of natural killer- and lymphokine-activated killer-sensitive targets. Biol Reprod 58:982–987
Verma S, King A, Loke YW (1997) Expression of killer cell inhibitory receptors on human uterine natural killer cells. Eur J Immunol 27:979–983
Wallace AE, Host AJ, Whitley GS, Cartwright JE (2013) Decidual natural killer cell interactions with trophoblasts are impaired in pregnancies at increased risk of preeclampsia. Am J Pathol 183:1853–186
Wallace AE, Fraser R, Gurung S, Goulwara SS, Whitley GS, Johnstone AP, Cartwright JE (2014) Increased angiogenic factor secretion by decidual natural killer cells from pregnancies with high uterine artery resistance alters trophoblast function. Hum Reprod 29:652–60
Wallace AE, Whitley GS, Thilaganathan B, Cartwright JE (2015) Decidual natural killer cell receptor expression is altered in pregnancies with impaired vascular remodeling and a higher risk of pre-eclampsia. J Leukoc Biol 97:79–86
Wilczynski JR, Tchorzewski H, Banasik M, Glowacka E, Wieczorek A, Lewkowicz P, Malinowski A, Szpakowski M, Wilczynski J (2003) Lymphocyte subset distribution and cytokine secretion in third trimester decidua in normal pregnancy and preeclampsia. Eur J Obstet Gynecol Reprod Biol 109:8–15
Williams PJ, Searle RF, Robson SC, Innes BA, Bulmer JN (2009a) Decidual leucocyte populations in early to late gestation normal human pregnancy. J Reprod Immunol 82:24–31
Williams PJ, Bulmer JN, Searle RF, Innes BA, Robson SC (2009b) Altered decidual leucocyte populations in the placental bed in pre-eclampsia and foetal growth restriction: a comparison with late normal pregnancy. Reproduction 138:177–184
Wira CR, Fahey JV, Schaefer TM, Pioli PA, Sentman CL, Shen L (2008) Innate and adaptive immunity in the human female reproductive tract:influence of the menstrual cycle and menopause on the mucosal immune system in the uterus. The endometrium: molecular, cellular and clinical perspectives, 2nd edn. Informa Healthcare, New York
Wu X, Jin LP, Yuan MM, Zhu Y, Wang MY, Li DJ (2005) Human first-trimester trophoblast cells recruit CD56brightCD16- NK cells into decidua by way of expressing and secreting of CXCL12/stromal cell-derived factor 1. J Immunol 175:61–68
Xiong S, Sharkey AM, Kennedy PR, Gardner L, Farrell LE, Chazara O, Bauer J, Hiby SE, Colucci F, Moffett A (2013) Maternal uterine NK cell-activating receptor KIR2DS1 enhances placentation. J Clin Invest 123:4264–4272
Yamada H, Shimada S, Morikawa M, Iwabuchi K, Kishi R, Onoe K, Minakami H (2005) Divergence of natural killer cell receptor and related molecule in the decidua from sporadic miscarriage with normal chromosome karyotype. Mol Hum Reprod 11:451–457
Yamamoto T, Takahashi Y, Kase N, Mori H (1999) Decidual natural killer cells in recurrent spontaneous abortion with normal chromosomal content. Am J Reprod Immunol 41:337–342
Yeaman GR, Guyre PM, Fanger MW, Collins JE, White HD, Rathbun W, Orndorff KA, Gonzalez J, Stern JE, Wira CR (1997) Unique CD8+ T cell-rich lymphoid aggregates in human uterine endometrium. J Leukoc Biol 61:427–435
Zenclussen AC, Fest S, Sehmsdorf US, Hagen E, Klapp BF, Arck PC (2001) Upregulation of decidual P-selectin expression is associated with an increased number of Th1 cell populations in patients suffering from spontaneous abortions. Cell Immunol 213:94–103
Zenclussen AC, Gerlof K, Zenclussen ML, Ritschel S, Zambon Bertoja A, Fest S, Hontsu S, Ueha S, Matsushima K, Leber J, Volk HD (2006) Regulatory T cells induce a privileged tolerant microenvironment at the fetal-maternal interface. Eur J Immunol 36:82–94
Zhang J, Chen Z, Fritz JH, Rochman Y, Leonard WJ, Gommerman JL, Plumb AW, Abraham N, Croy BA (2012) Unusual timing of CD127 expression by mouse uterine natural killer cells. J Leukoc Biol 91:417–426
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Bulmer, J.N., Lash, G.E. (2015). The Role of Uterine NK Cells in Normal Reproduction and Reproductive Disorders. In: Bronson, R. (eds) The Male Role in Pregnancy Loss and Embryo Implantation Failure. Advances in Experimental Medicine and Biology, vol 868. Springer, Cham. https://doi.org/10.1007/978-3-319-18881-2_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-18881-2_5
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-18880-5
Online ISBN: 978-3-319-18881-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)