Abstract
Immobilized cells entrapped within a polymer matrix or attached onto the surface of a solid support have advantages over their free-cell counterpart, with easier harvesting of the biomass, enhanced wastewater treatment, and enriched bioproduct generation. Immobilized microalgae have been used for a diverse number of bioprocesses including gaining access to high-value products (biohydrogen, biodiesel, and photopigments), removal of nutrients (nitrate, phosphate, and ammonium ions), heavy metal ion removal, biosensors, and stock culture management. Wastewater treatment processes appear to be one of the most promising applications for immobilized microalgae, which mostly involve heavy metal and nutrient removal from liquid effluents. This chapter outlines the current applications of immobilized microalgae with an emphasis on alternative immobilization approaches. Advances in immobilization processes and possible research directions are also highlighted.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
2.1 Introduction
Algal bioprocesses are advantageous in integrating wastewater treatment processes with valuable biomass production. Algal biomass can be further exploited for various purposes such as biofuel generation in the form of biodiesel, biohydrogen, or biogas; food additives; slow-release fertilizers or soil conditioners; cosmetics; pharmaceuticals; and several other valuable chemicals (Johnson and An 1991; Mallick 2002; Mulbry et al. 2005). Microalgal cells have several other advantages in not requiring many resources to generate their biomass, providing an economical operation at lower costs, with the dissolved oxygen released by the algae being useful to elevate the oxygen levels of water effluents, and can be utilized for the reduction of carbon dioxide emissions using CO2 for their biomass and/or energy production.
Harvesting and dewatering of algal biomass from its liquid environment is one of the major challenges of algal bioprocesses. Several studies have focused on harvesting of microalgae using a wide range of technologies from sand filtration to high-speed centrifugation (Mallick 2002; Oswald 1988). Some of the most recent technologies for algal dewatering are further discussed in Chaps. 12, 13, and 14 of this book. Immobilization of algal cells has been proposed mainly to overcome the burdens of difficult harvesting and dewatering stages, in addition to providing the retention of the high-value-added algal biomass for further processes (de la Noue and de Pauw 1988; Mallick 2002).
Immobilization of various cells in either polymeric or biopolymeric matrices has several advantages over their free-cell counterparts, since immobilized cells occupy less space, are easier to handle, and can be used repeatedly for product generation (Mallick 2002). Immobilization of cells has also been proposed to increase the biosorption capacity and bioactivity of the biomass (Akhtar et al. 2004; de-Bashan and Bashan 2010). It allows bioprocesses with higher cell densities and also easy harvesting of biomass from its liquid environment (Mallick 2002). Immobilization processes have several other advantages as being resistant to harsh environments such as salinity, metal toxicity, and pH; protecting the aging cultures against the harmful effects of photoinhibition; yielding higher biomass concentrations; recovering the cells in a less-destructive way; and enhancing the cost-effectiveness of the process by reusing the regenerated biomass (Bailliez et al. 1986; Hall-Stoodley et al. 2004; Liu et al. 2009). Given the use of large-scale bioreactors represents a significant challenge associated with algal biomass recovery, immobilization systems are becoming attractive alternatives for scale-up processing (Christenson and Sims 2011; Hoffmann 1998).
Various immobilization processes are in use, such as adsorption, confinement in liquid–liquid emulsions, capturing with semipermeable membranes, covalent coupling, and entrapment within polymers (de-Bashan and Bashan 2010; Mallick 2002). Among others, the most common immobilization processes are the entrapment of the cells within polymeric matrices and self-adhesive attachment of cells onto the surfaces of solid-supports (Godlewska-Żyłkiewicz 2003). Both synthetic and natural polymers can be applied as the immobilization matrix (de-Bashan and Bashan 2010).
Important criteria for successful entrapment are to set the algal cells free within their partition, while pores inside the gel matrix allow the diffusion of substrates and the metabolic products toward and from the cells (Mallick 2002). Nevertheless, entrapment systems still hold some drawbacks in reducing the mass transfer kinetics of the uptake of metal ions (Aksu et al. 2002). However, these can be avoided by careful choice of the immobilization method and the nature of the matrix, which will be further discussed in detail.
Key applications suggested for immobilized algal cells include removal of nutrients from aqueous solutions (Chevalier and de la Noüe 1985), biodiesel production (Bailliez et al. 1985; Li et al. 2007), biosorption of heavy metals from wastewaters (de-Bashan and Bashan 2010), photoproduction of hydrogen and photopigments (Bailliez et al. 1986; Laurinavichene et al. 2008), providing an alternative technique to the common cryopreservation processes (Chen 2001; Faafeng et al. 1994; Hertzberg and Jensen 1989), and also toxicity testing (Bozeman et al. 1989). These processes will also be discussed in detail in the following sections of this review article.
2.2 Immobilization Techniques and Applied Matrices
Entrapment is one of the most common immobilization methods which consists of capturing the cells in a three-dimensional gel lattice, made of either natural (agar, cellulose, alginate, carrageenan) or synthetic (polyacrylamide, polyurethane, polyvinyl, polypropylene) polymers (de-Bashan and Bashan 2010; Hameed and Ebrahim 2007; Liu et al. 2009). Synthetic polymers are reported to be more stable in wastewater samples than the natural polymers, whereas natural polymers have higher nutrient/product diffusion rates and are more environmentally friendly (de-Bashan and Bashan 2010; Leenen et al. 1996).
Polysaccharide gel-immobilized algal cells have often been used for the removal of nitrate, phosphate, and heavy metal ions from their aqueous environment, in providing an alternative to the current physicochemical wastewater treatment technologies (Bayramoğlu et al. 2006). Microalgae cells entrapped within either alginate or carrageenan beads were shown to have sufficient immobilization and significant nutrient removal efficiencies from aqueous environments (Chevalier et al. 2000). Aguilar-May et al. (2007) reported that the immobilization of Synechococcus sp. cells in chitosan gels had a positive effect on protecting the cell walls from the toxic effect of high NaOH concentration, with immobilized cells displaying higher growth than their free-cell counterparts.
Alginate beads are one of the most common encapsulation matrices, being an anionic polysaccharide found mostly in the cell walls of brown algae (Andrade et al. 2004). Major advantages of alginate gel are it being nontoxic, easy to process, cost-effective, and transparent and permeable (de-Bashan and Bashan 2010). Despite these advantages, alginate beads have some drawbacks such as not retaining their polymeric structure in the presence of high phosphate concentrations or high content of some cations such as K+ or Mg2+ (Kuu and Polack 1983). Faafeng et al. (1994) observed the degradation of sodium alginate beads, used for the immobilization of Selenastrum capricornutum, after keeping them in polluted wastewater with high phosphorous (P) and nitrogen (N) content for longer than two weeks. This degradation problem can be minimized if the stability of the target gel is enhanced. In this context, Serp et al. (2000) found that the mechanical resistance of alginate beads was doubled after mixing them with chitosan. Japanese konjac flour was also used to increase the stability of chitosan gels during tertiary treatment of wastewaters with high phosphate concentrations (Kaya and Picard 1996). Kuu and Polack (1983) suggested that increasing the gel strength of carrageenan and agar gels by integrating them with polyacrylamide results in a more rigid support for microorganisms.
Most of the entrapment processes have a similar protocol, namely mixing the microalgal suspension with the monomers of the selected polymer, followed by solidification of the resulting algae/polymer mixture by some physical or chemical process such as cross-linking of the monomers of the polymer with di- or multivalent cations (Cohen 2001; de-Bashan and Bashan 2010). As an illustration, a general procedure for the entrapment of microalgae within alginate beads includes the following steps: (1) mixing of algal suspension with sodium alginate solution, (2) placing the homogenously distributed algae/alginate mixture in a vessel with a small orifice, such as a syringe, (3) gently dripping the mixture from the syringe as small droplets/beads into a cross-linking solution such as calcium chloride, (4) optimizing the time for algae/alginate beads inside the cross-linking solution to form cross-linked/hardened beads, (5) collecting the final algae/alginate beads, and rinsing them with deionized water several times (Smidsrød and Skjåk-Bræk 1990). Since a manual dripping process for bead production is not practical for larger scale processes, automated prototypes were also proposed for the mass production of gel beads (de-Bashan and Bashan 2010; Hunik and Tramper 1993).
There are some drawbacks of cellular entrapment due to limitations of the oxygen and/or carbon dioxide transfer from the liquid environment through the immobilization matrix, which would cause difficulties mainly for aerobic microorganisms (Toda and Sato 1985). Co-immobilization of the target microorganism with microalgal cells has been proposed as an interesting alternative to overcome any oxygen transfer limitations. Since microalgae are capable of generating oxygen from the photolysis of water, they function as ideal oxygen generators for their surrounding microenvironments (Adlercreutz et al. 1982; Chevalier and de la Noüe 1988). Selected microalgae–bacteria pairs have already been shown to benefit from each other, with microalgal cells generating oxygen and some organic compounds that are assimilated by bacteria. On the other hand, bacteria release some vitamins and phytohormones or provide an additional CO2 source that can enhance the algal growth (de-Bashan et al. 2005; Gonzalez and Bashan 2000; Mouget et al. 1995). Mouget et al. (1995) also found that Pseudomonas diminuta and Pseudomonas vesicularis bacterial cells isolated from the algal cultures of Chlorella sp. and Scenedesmus bicellularis stimulate the growth of those microalgal cells.
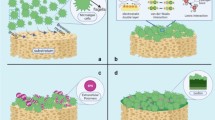
Previous attempts to immobilize viable algal cells inside gels faced other limitations, as the volume-to-surface ratios of spherical encapsulating materials are usually orders of magnitude larger than that of thin films. As a consequence, algal viability is a concern since the nutrients or reactants have to diffuse far into these materials to reach the algal cells. In order to overcome these problems, several other immobilization matrices have been proposed in the recent literature. Three different immobilization matrices with different geometries and chemical properties are given in Fig. 2.1.
a Alginate beads containing different amounts of immobilized Scenedesmus quadricauda: (i) ca. 2500; (ii) ca. 20,000; (iii) ca. 90,000 algal cells (modified from Chen 2001), b Chlorella sorokiniana cells covering the surface of a Luffa cylindrica sponge (modified from Akhtar et al. 2008), c Chlorella vulgaris cells attached on the surface of a chitosan nanofiber mat (3 × 2 cm) floating inside the algal growth media (modified from Eroglu et al. 2012)—reproduced by permission of The Royal Society of Chemistry
Algal biofilms are one of the alternatives to overcome the harvesting problems of algae in larger scale processes, where microalgal cells stick to each other on external surfaces (Chevalier et al. 2000; Wuertz et al. 2003). Microorganisms form a biofilm as a response to several factors, such as the cellular recognition of the specific functional groups on the targeted surfaces (Karatan and Watnick 2009). Microorganisms forming a biofilm on a surface secrete extracellular polymeric substance, which is mainly composed of phospholipids, proteins, polysaccharides, and extracellular DNA (Hall-Stoodley et al. 2004; Qureshi et al. 2005). Polystyrene disks (Przytocka-Jusiak et al. 1984), textured steel surfaces (Cao et al. 2009), aluminum disks (Torpey et al. 1971), and polystyrene surfaces (Johnson and Wen 2010) are some examples of biofilm surfaces used for algal growth for the primary application of nutrient removal from wastewaters.
The shape of algal cell composite material has two components, a global geometrical form and the surface detail which determines the texture of the surface, with nanomaterial processing techniques being the useful approaches for creating different shapes, from fibers to spheres and flat membranes (Crandall 1996). Various nanofabrication processes have featured in recent research from the authors’ laboratories, albeit in using more unconventional types of immobilization matrices for the immobilization of Chlorella vulgaris cells, such as electrospun nanofibers (Eroglu et al. 2012), laminar nanomaterials such as graphene and graphene oxide nanosheets (Wahid et al. 2013a, b), microfibers of ionic liquid-treated human hair (Boulos et al. 2013), and magnetic polymer matrix composed of magnetite nanoparticles embedded in polyvinylpyrrolidone (Eroglu et al. 2013). Electrospinning processes can create nanofiber mats with high porosities and surface-to-volume ratios and are generated by forcing a charged polymer solution through a very small-sized nozzle while applying an electrical field (Kelleher and Vacanti 2010). On the other hand, a recently developed vortex fluidic device has been successfully used for the exfoliation of laminar materials within the dynamic thin films formed on the walls of this microfluidic platform (Wahid et al. 2013a, b). Scanning electron microscopic images of different nanomaterial matrices, used for the immobilization of C. vulgaris microalgal cells, are given in Fig. 2.2.
Scanning electron microscopy images of a chitosan nanofibers (modified from Eroglu et al. (2012)—reproduced by permission of The Royal Society of Chemistry); b multilayer graphene oxide nanosheets (modified from Wahid et al. (2013a)—reproduced by permission of The Royal Society of Chemistry); c microfibers of ionic liquid-treated human hair (modified from Boulos et al. (2013)—reproduced by permission of The Royal Society of Chemistry), surrounding Chlorella vulgaris microalgal cells
2.3 Wastewater Treatment Using Immobilized Algae
Wastewater treatment consists of the removal of unwanted chemicals, or biological contaminants from impure water sources, such as from the liquid wastes released by houses, industrial operations, or agricultural processes. Conventional wastewater treatment methods include physical processes such as filtration and sedimentation; chemical processes such as flocculation and chlorination; and biological processes such as generation of activated sludge (Metcalf and Eddy 2003). However, these methods are mainly based on the separation of pollutants from the wastewater with a requirement for a further stage to eliminate these pollutants. This brings a need for an integrated wastewater treatment process that eliminates the undesired portion of the wastewater while converting them into valuable products, which can be successfully achieved by applying a selected immobilization process. Immobilized algal systems are particularly effective for the removal of nutrients (i.e., phosphate and nitrate) and various metals from wastewaters, which will be discussed in the following sections.
2.3.1 Nutrient Removal from Wastewater
Several studies have demonstrated the potential of microalgae for the removal of nitrogen and phosphorus elements from wastewater effluents, with cells taking them up as their nutrient sources. Some of those studies are listed in Table 2.1, while some are further described in the following text. It should be noted that the direct comparison of the nutrient removal efficiencies from various experiments is inherently difficult, because of variations in the initial nutrient concentration, duration of the experiment, pH of the working solution, selected algal species, and the type of immobilization matrix.
The most common algal species used for the removal of nitrates and phosphates are Chlorella, Scenedesmus, and Spirulina. Various open and closed bioreactors have been used for the removal of nutrients by algae, ranging from tubular photobioreactors to corrugated raceways and high-rate algae ponds (Borowitzka 1999; Cromar et al. 1996; Olguín et al. 2003). Increased nutrient removal efficiencies with immobilized algae are usually related, with the dual effect of the enhanced photosynthetic rate of the cells and the ionic exchange between the nutrient ions and the immobilization matrix. Gels which are anionic in nature, such as carrageenan, are usually associated with the adsorption of cations (such as ammonium (NH4 +)), while cationic gels such as chitosan yield adsorption of anions (phosphate (PO −34 ), nitrate (NO3 −), nitrite (NO2 −)) with higher efficiencies (Mallick and Rai 1994). Moreover, calcium ions of the alginate or chitosan gels are particularly efficient for the precipitation of PO −34 ions from wastewaters (Lau et al. 1997).
Immobilization of C. vulgaris cells within sodium alginate beads showed higher nutrient removal efficiencies from sewage wastewater compared to their externally immobilized counterparts on polyurethane foam (Travieso et al. 1996). de-Bashan et al. (2002b) obtained higher ammonium and phosphate removal efficiencies after co-immobilization of C. vulgaris microalgae with plant growth-promoting bacterium Azospirillum brasilense in alginate beads, relative to immobilized C. vulgaris cells alone. Tam and Wong (2000) obtained 78 % ammonium and 94 % phosphate removal efficiencies with immobilized C. vulgaris, entrapped in calcium alginate beads, compared to the 40 % ammonium and 59 % phosphate removal with free cells. Lau et al. (1997) also observed significantly higher ammonium (95 %) and phosphate (99 %) removal efficiencies for C. vulgaris cells immobilized in alginate beads relative to their free counterparts, resulting in only 50 % nitrogen and 50 % phosphate removal. In contrast, free cells of Nannochloropsis sp. cells yielded higher total phosphorus removal with respect to their immobilized cells within calcium alginate beads (Jimenez-Perez et al. 2004).
Pretreatment of the cells by starving them in a saline solution for three days was found to increase the cellular growth and phosphate removal efficiencies of the independently co-immobilized Chlorella sorokiniana & A. brasilense and C. vulgaris & A. brasilense pairs entrapped in alginate beads (Hernandez et al. 2006). Kaya et al. (1995) observed higher nutrient removal rates using S. bicellularis cells when they were immobilized on flat-surface alginate screens compared to their encapsulated form inside alginate beads.
Canizares et al. (1993) used immobilized Spirulina maxima cells in kappa-carrageenan gel beads for nutrient removal from swine waste. This immobilized system achieved around 90 % total phosphorus and ammonium-nitrogen removal, while it also allowed processing swine waste at higher concentrations. Chevalier and de la Noüe (1985) investigated Scenedesmus acutus and Scenedesmus obliquus cells individually immobilized in kappa-carrageenan beads for nutrient removal from a secondary effluent. Immobilized cells showed similar cellular growth and ammonium or phosphate uptake rates compared to their free-living cell counterparts. They observed around 90 % ammonium removal within the first 4 h, while all traces of phosphate were removed within 2 h (Chevalier and de la Noüe 1985).
C. vulgaris and Anabaena doliolum cells immobilized in chitosan have higher phosphate, nitrate, and nitrite removal efficiencies than when they were immobilized within agar, alginate, or carrageenan (Mallick and Rai 1994). In addition, the phosphate removal capacity of the immobilization process was increased when phosphate-deprived cells were initially entrapped within chitosan. Fierro et al. (2008) investigated the nitrate and phosphate removal efficiencies of individually entrapped Scenedesmus sp. cells within chitosan beads. Immobilized cells achieved approximately 94 % phosphate and 70 % nitrate removal within the first 12 h after incubation, whereas by themselves chitosan beads removed 60 % phosphate and 20 % nitrate by the end of the experiment. The reason for yielding a significant phosphate removal rate (60 %) by chitosan beads alone was explained by the increased pH values, which eventually triggered the release of some calcium ions from chitosan polymer, resulting in the precipitation of phosphate ions (Fierro et al. 2008; Tam and Wong 2000).
Other immobilization matrices have also been proposed as alternatives to the gel beads. Immobilized cells of Trentepohlia aurea microalgal cells on a filter paper formed a biofilm layer that reduced the concentration of ammonium, nitrate, and nitrite ions, for around 40 days (Abe et al. 2003). Shi et al. (2007) proposed a twin-layer system, where the microalgal cells are attached on an ultrathin and microporous “substrate layer” composed of a nitrocellulose membrane, which is surrounded by a “source layer” of macroporous glass fiber providing the growth medium (Shi et al. 2007). They observed phosphate, ammonium, and nitrate removal when C. vulgaris and Scenedesmus rubescens microalgal cells were entrapped in this twin-layer system.
In a recent study, C. vulgaris cells immobilized on electrospun chitosan nanofiber mats yielded an efficient nitrate removal rate (87 %) as a result of the dual action of nitrate removal by the microalgal cells and electrostatic binding of the nitrate ions on chitosan nanofibers (Eroglu et al. 2012). In other studies from the authors’ laboratories, the resulting microalgal composites with multilayer graphene (Wahid et al. 2013b) or graphene oxide sheets (Wahid et al. 2013a) also achieved significant nitrate uptake rates, without being toxic for the microalgal cells.
2.3.2 Metal Removal from Wastewater
Conventional methods used for the removal of heavy metal ions include chemical precipitation, adsorption, chemical oxidation/reduction, membrane filtration, ion exchange, and electrochemical processes. However, these techniques have some drawback, such as partial removal of metal ions, costly installation requirements, high energy demands, and the generation of toxic waste products which require additional elimination stages (Aksu et al. 2002).
Both live and dead cells can be successfully used for the biosorption of metal ions, while uptake of metal ions by living microorganisms, referred to as bioaccumulation, occurs when an active metabolic process is involved (Aksu et al. 2002; Brady and Duncan 1994; Moreno-Garrido et al. 1998). Biosorption is a reversible process, since it is possible to desorb the metal ions bound to the surfaces of cells by a simple acid treatment, whereas bioaccumulation processes are only partially reversible (de-Bashan and Bashan 2010; Dönmez and Aksu 2002; Velásquez and Dussan 2009).
Compared to the other organisms used for biosorption processes, namely fungi, cyanobacteria, and bacteria, algal cells have higher heavy metal biosorption capacities which relates to the different structure and composition of their cell wall (Bayramoğlu et al. 2006; Gekeler et al. 1988). Cell walls of different microorganisms have different functional groups which are involved in metal ion binding, such as amino, amide, carbonyl, carboxyl, hydroxyl, imidazole, phosphate, sulfate, sulfhydryl, and phenol moieties (Barkley 1991; Schiewer and Volesky 2000). Depending on the variations in the cell wall composition, there will also be differences in the metal ion binding mechanisms and affinities (Godlewska-Żyłkiewicz 2003; Leusch et al. 1995).
The chemical characteristics of the functional adsorbent (i.e., functional groups, polarity, and solubility) are responsible for determining the binding mechanism and the nature of the adsorption process. Different physicochemical forces, such as covalent bonding, van der Waals bonding, ion exchange, and dipole/dipole interactions can be responsible for the uptake of ions on the adsorbents (Aksu et al. 2002).
Free cells have some disadvantages when used for large-scale applications of metal ion biosorption studies, due to the otherwise risk of clogging problems on the filters and flow lines. Nevertheless, this problem was overcome by using immobilized cells in natural matrices such as carrageenan, alginate, chitosan, agarose; polymeric supports such as polyacrylamide, polypropylene, and polysulfone; cross-linked copolymers; or biomatrices such as sponges (Akhtar et al. 2003a, b; Robinson 1998). Some of those studies are highlighted in Table 2.2.
The presence of more than one type of metal ion within the wastewater might have a negative effect on the adsorption of one type of metal ion over another. Mehta and Gaur (2001) observed nearly complete removal of copper and nickel metals by alginate-entrapped C. vulgaris cells when they were in separate solutions. On the other hand, the presence of copper in the nickel solution inhibited the biosorption of both metals either by immobilized or free cells, due to the competition of different metal ions on the same active sites of microalgae. da Costa and Leite (1991) used alginate-immobilized Chlorella homosphaera for the removal of cadmium and zinc metals. They also observed that the biosorption of cadmium and zinc alone was much higher than the case when these two metal ions were combined.
Biological materials were also used as the immobilization matrices for microalgal cells. da Costa and de França (1996) attached the microalgae Tetraselmis chuii and cyanobacteria Spirulina maxima on the surface of two different seaweeds (Sargassum sp. and the Ulva sp.), which eventually increased the overall cadmium biosorption efficiencies. In series of studies by Akhtar et al., C. sorokiniana algal cells were immobilized on a biological matrix of Luffa cylindrica sponge for the removal of nickel (Akhtar et al. 2003a), cadmium (Akhtar et al. 2003b), chromium (Akhtar et al. 2008), and lead (Akhtar et al. 2004) ions from liquid effluents. L. cylindrica sponge was chosen as the immobilization matrix due its rigid structure, low cost, and high porosity, while its fibrous network provides an efficient contact between the immobilized cells with their surrounding aqueous environment (see Fig. 2.1b). They reported high maximum adsorption capacities in a continuous liquid flow column, as 192 mg cadmium and 71 mg nickel per gram of immobilized biomass. They also achieved successful desorption of cadmium and nickel metal ions with HCl solution, and the regenerated immobilized samples were reusable with a similar biosorption efficiency.
The biosorption of lead (Pb) ions by C. sorokiniana cells immobilized on L. cylindrica sponge was another efficient method, with 96 % adsorption efficiency of the metal ions within the first 5 min of the experiments (Akhtar et al. 2004). They also observed a maximum adsorption of lead ions at around pH 5.0. Higher removal rates were associated with the fibrous structure of the immobilization matrix, increased surface area, and easier access of the targeted metal ion to the sorption sites (Akhtar et al. 2003a, b).
Leusch et al. (1995) used two marine brown algae, Sargassum fluitans and Ascophyllum nodosum, for the biosorption of cadmium, copper, nickel, lead, and zinc heavy metal ions. They observed the highest metal uptakes when the cells were cross-linked with glutaraldehyde, followed by cross-linking with formaldehyde. Both species had the highest biosorption efficiencies for lead and the lowest for zinc. Introducing formaldehyde possibly involves cross-linking of the hydroxylic groups with the sugars of the cell wall, while glutaraldehyde cross-links mostly with the amino groups (Leusch et al. 1995).
Significant amounts of pollutants were removed using a mixed-immobilization of selected consortium of several microalgal species (Chlorella sp., S. obliquus, Stichococcus sp.) and several bacteria (Rhodococcus sp., Kibdelosporangium aridum) inside a highly contaminated pond, after the separate immobilization of microalgae and bacteria in solid carriers such as capron fibers and ceramics. They established 62 % copper, 62 % nickel, 90 % zinc, 70 % manganese, and 64 % iron removal efficiencies (Safonova et al. 2004).
Bayramoğlu et al. (2006) used immobilized Chlamydomonas reinhardtii cells in calcium alginate beads for the removal of mercury, cadmium, and lead ions from aqueous solutions. They observed the highest adsorption capacities for immobilized cells for a pH in the range 5.0–6.0, achieving mercury, cadmium, and lead ion adsorption capacities of 89.5, 66.5, and 253.6 mg g−1 dry adsorbent, respectively. On the other hand, control samples composed of only calcium alginate beads provided less metal-binding sites and yielded lower adsorption capacities of mercury, cadmium, and lead ions at 32.4, 27.9, and 173.9 mg g−1 dry adsorbent, respectively. Acidic pH conditions were not optimal due to the protonation of the cell wall components. In contrast, mildly acid conditions (pH range 5.0–6.0) allowed sufficient interaction of the heavy metal ions with the carboxylate and phosphate groups of the algal cell wall (Bayramoğlu et al. 2006). Neutral pH was found to be the optimal condition for an efficient chromium biosorption by immobilized C. vulgaris and freshwater cyanobacterium A. doliolum cells in alginate (Mallick and Rai 1993).
Barkley (1991) investigated the utilization of immobilized algae in a permeable polymeric matrix for the adsorption of mercury ions from groundwater in both laboratory and pilot-scale field tests. Their resulting immobilization product (AlgaSORB) was quite robust and can be packed within adsorption columns, having sufficient porosity to allow easy diffusion of the ions toward the cells. Field test results showed that AlgaSORB was a highly reasonable alternative to the conventional ion-exchange resins (Barkley 1991).
Nakajima et al. (1982) achieved the removal of uranium ions from both freshwater and seawater samples using the immobilized cells of Chlorella sp. in polyacrylamide gels. They also reported that this system can be used several times by applying consecutive adsorption and desorption stages.
Recovery of precious metals with immobilization methods can be a highly cost-effective process. da Costa and Leite (1991) used immobilized C. homosphaera cells within alginate beads for the adsorption of gold metal, which achieved a very high absorption yield of around 90 % of the initial quantity of gold present in solution.
Due to their exclusive catalytic properties, corrosion, and oxidation resistivity, palladium and platinum noble metal s have been widely used in various areas from metallurgical processes, chemical synthesis, petroleum processing, electronics to automotive industry (Dziwulska et al. 2004). As a result of the high emission risks of these metals into the environment, it has become important to monitor their concentration in environmental samples. Thus, several microorganisms have been investigated for the separation and preconcentration of some trace metals such as palladium, platinum, copper, cadmium, lead, and gold via biosorption processes, which then allows the use of analytical methods such as atomic absorption spectrometry and inductively coupled plasma optical emission spectrometry (Carrilho et al. 2003; Dziwulska et al. 2004; Elmahadi and Greenway 1991; Godlewska-Żyłkiewicz 2003).
Dziwulska et al. (2004) demonstrated the selective biosorption of palladium and platinum ions from strong acidic solutions (pH below 2), using immobilized C. vulgaris cells on anion-exchange resin Cellex-T. This technique was also used for the preconcentration and analysis of these noble metals for graphite furnace atomic absorption spectrometry in different environmental samples including wastewater , tap water, and grass. Elmahadi and Greenway (1991) used Chlamydomonus reinhartii and S. capricornutum algal cells immobilized on controlled-pore glass for the preconcentration of copper, silver, and chromium metals for atomic adsorption spectrophotometric detection. In their work, they also found that the presence of some compounds, such as sodium chloride, humic acid, and sodium bicarbonate, can interfere with metal biosorption process by competing for the metal ions. Silica gel was used as the immobilization matrix for Stichococcus bacillaris microalgae for lead preconcentration (Mahan and Holcombe 1992), while silica gel-entrapped Pilayella littoralis brown microalgae was used for the preconcentration of copper, iron, aluminum, and cobalt ions for their detection by inductively coupled plasma optical emission spectrometry (Carrilho et al. 2003).
2.4 Secondary Products Using Immobilized Algae
Photosynthesis is responsible for the conversion of light into chemical energy which can be used for biofuel production, including biohydrogen, biodiesel, bioethanol, and biomethane generation (Hankamer et al. 2007). Immobilized microalgal cultures have also been in use for the enhancement of these secondary product formations, as explained further below.
2.4.1 Biohydrogen Production
Several unicellular green algae are capable of generating hydrogen through their [FeFe]-hydrogenase enzyme by reducing water protons to molecular hydrogen. However, given the sensitivity of [FeFe]-hydrogenase to oxygen, which is generated by photosystem II (PSII), new approaches have been developed for increasing the practical application of microalgae for biohydrogen production (Laurinavichene et al. 2008). For instance, higher hydrogen production efficiencies were achieved by growing the microalgal cells under sulfur-deprived conditions (Melis et al. 2000). Sulfur deprivation causes partial inactivation of PSII, which is responsible for O2 generation, resulting an enhanced synthesis of [FeFe]-hydrogenase enzyme (Laurinavichene et al. 2008).
Immobilization processes have been proposed by several researchers for enhancing hydrogen production by sulfur-deprived microalgae and also allowing an easy exchange step between “sulfur-replete” and “sulfur-depleted” stages of the experiment (Laurinavichene et al. 2006, 2008). Several challenges require addressing for scaling up the current hydrogen production systems, while immobilization processes offer an alternative approach to the current technology. Immobilized cells were also reported to have higher light utilization efficiencies per area and higher cell densities (Kosourov and Seibert 2009).
In a study by Kosourov and Seibert (2009), C. reinhardtii cells were immobilized inside alginate films for the photoproduction of hydrogen. The cells were previously deprived of sulfur and phosphorus nutrients before being entrapped inside the alginate films. They observed higher cell densities and specific hydrogen production rates after the immobilization process. An immobilization strategy also provided easy protection of the hydrogenase enzyme from oxygen inhibition, yielding higher hydrogen production rates compared to the free cells.
Laurinavichene et al. (2006, 2008) used immobilized C. reinhardtii cells on a fiber glass matrix under sulfur-deprived conditions and observed a prolonged hydrogen production phase for the immobilized cells, while the specific hydrogen production rate was similar to the free-cell counterparts. In another study, algal cells were immobilized on fumed silica particles, which had similar hydrogen production rates with the suspended cultures (Hahn et al. 2007). Song et al. (2011) recently used agar-immobilized Chlorella sp. cells for a two-stage cyclic hydrogen production involving the oxygenic photosynthesis followed by anaerobic incubation under sulfur-deprived conditions.
2.4.2 Biodiesel Production
Biodiesel is a diesel fuel consisting of mono-alkyl esters of long-chain fatty acids that are generally made by the transesterification of lipids in animal fat or vegetable oils such as soybean, sunflower, rapeseed, and oil palm (Hankamer et al. 2007; Li et al. 2007; Ma and Hanna 1999). As an alternative, microalgae have become popular for the renewable generation of hydrocarbon-based biofuels with high biofuel yields relative to those from plants (Eroglu and Melis 2009; Li et al. 2007).
Immobilization of the hydrocarbon-rich microalgae, Botryococcus braunii and Botryococcus protuberans, in alginate beads yielded a significant increase in the chlorophyll, carotenoids, cellular growth, and lipid contents of the cells during their stationary growth phase (Singh 2003). Bailliez et al. (1985) also observed enhanced hydrocarbon production for the B. braunii cells immobilized in calcium alginate gel as a result of enhanced photosynthetic activity.
In a study by de-Bashan et al. (2002a), C. vulgaris and C. sorokiniana microalgal cells were individually co-immobilized with A. brasilense growth-promoting bacterium in alginate beads. They found that the presence of the growth-promoting bacterium within the immobilization matrix significantly enhanced the metabolism of Chlorella strains and yielded higher lipid and fatty acid production.
Li et al. (2007) used immobilization technology for the transesterification of algal oils, using immobilized lipase enzyme from Candidia sp. Initially, they grew Chlorella protothecoides cells in large-scale photobioreactors at three different sizes (5, 750, 11,000 L) yielding high lipid contents in the range 44–49 % per dry cell weight. Then, immobilized lipase enzyme from Candidia sp. was used to catalyze the transesterification of the lipids from C. protothecoides, yielding biodiesel production rates of 7.02, 6.12, and 6.24 g L−1 from 5, 750, and 11,000 L bioreactors, respectively. They also highlighted that the quality of this Chlorella biodiesel was comparable to that for conventional diesel fuels (Li et al. 2007).
2.4.3 Pigment Production
Bailliez et al. (1986) found that the immobilized B. braunii cultures in calcium alginate beads had higher chlorophyll and photosynthetic activities compared to their free cells. S. obliquus cells immobilized in alginate (Brouers et al. 1983), and C. vulgaris and Anacystis nidulans in agar (Kayano et al. 1981; Weetall and Krampitz 1980), also showed a significant increase in their chlorophyll content. Enhanced chlorophyll and photosynthetic activity was explained by the protection of immobilized cells from photoinhibition due to the self-shadowing effect, and a possible increase in the concentrations of particular ions in the microenvironment of cells which can improve photosynthesis (Bailliez et al. 1986; Tamponnet et al. 1985).
Individually co-immobilized cells of C. vulgaris and C. sorokiniana with A. brasilense growth-promoting bacterium also yielded higher chlorophyll a and b, violaxanthin, and lutein accumulation compared to the immobilized algal cells without any bacterium (de-Bashan et al. 2002a).
Lebeau et al. (2000) reported that the immobilization of the marine diatom Haslea ostrearia in agar had a positive effect on the continuous production of the marennin pigment, which is primarily used for the oyster-breeding industry.
Some potential limitations of the secondary product formation by immobilized cells are the commonly reported slower growth rates of the microorganisms compared to their free-cell suspension systems and slower diffusion rates of the target-products (i.e., hydrogen) from the cells into their environment. Resolving these issues with the combination of optimized immobilization matrices and innovative bioreactor designs (e.g., some attempts include membrane-based cell recycle bioreactor (Chang et al. 1994); dual-layer coaxial hollow fiber-type bioreactor (Yang et al. 2006); and a multimembrane bioreactor in a pressure cycling mode (Efthymiou and Shuler 1987), aiming to increase the nutrient transfer to the cells) can potentially bring other dimensions to the research areas of those aforementioned bioprocesses.
2.5 Biosensors
Biosensor research often focuses on the application of enzyme sensors for the detection of toxic chemicals (Dennison and Turner 1995; Shul’ga et al. 1994). Due to the drawbacks of this technology, such as enzyme stability, cost of the process, and difficulty to prepare multienzymatic biosensors, immobilized cells have been proposed as an alternative biosensor technology. Using the entire cells has the advantage of involving various enzymes at the same time, which allows establishing information about the toxicological effects of different pollutants directly on the selected organisms. Immobilized cells had more stable metabolic activities than free cells during the long testing periods (Lukavský et al. 1986) and also higher resistivity to turbid/colored effluents (Bozeman et al. 1989).
The generation or consumption of charged chemicals during bioreactions results in a significant change in the ionic composition of the test sample that can be detected by conductometric biosensors. For this reason, Chouteau et al. (2004) investigated the development of conductometric biosensors using immobilized C. vulgaris cells for alkaline phosphatase analysis and cadmium ion detection. C. vulgaris cells were immobilized inside bovine serum albumin membranes that were cross-linked with glutaraldehyde vapors.
Frense et al. (1998) used immobilized Scenesdesmus subspicatus algal cells as optical biosensors for the determination of the herbicide content in wastewater samples. The algal cells were initially immobilized on a filter paper, which was then covered by alginate and then cross-linked with CaCl2 solution. They used a fiber optics-based electronic device for measuring the chlorophyll fluorescence of algal cells as a response to the presence or absence of the toxic substances in the liquid sample.
C. vulgaris cells immobilized in a membrane of oxygen electrode has been used as a biosensor for the detection of perchloroethylene aerosols by monitoring the photosynthetic activity of the microalgae through oxygen production (Naessens and Tran-Minh 1999). Shitanda et al. (2005) also immobilized alginate-entrapped C. vulgaris cells on the surface of an indium tin oxide electrode, for the monitoring toxic compounds such as atrazine, toluene, benzene, and 3-(3,4-dichlorophenyl)-1,1-diethylurea (DCMU).
Immobilized algal cells of S. capricornutum in alginate beads were used for the toxicity testing of various chemicals, such as cadmium ions, copper ions, pentachlorophenol, sodium dodecyl sulfate, and herbicides (glyphosate, hydrothol, paraquat) (Bozeman et al. 1989). In subsequent studies, alginate-immobilized S. capricornutum cells were also successively used for the toxicity testing of various pesticides, herbicides, and fungicide (Abdel-Hamid 1996; Van Donk et al. 1992). The immobilization process reduced the toxic effect of these tested chemicals on the algal cells compared to their free-cell equivalents.
2.6 Stock Culture Management
Some researchers have applied immobilization technologies to the stock culture management as an alternative to the common cryopreservation processes, since entrapment processes are cheaper and easier (Chen 2001; Faafeng et al. 1994; Hertzberg and Jensen 1989). Immobilization can also provide protection of the cells toward being consumed by any zooplankton present in the same aquatic ecosystems.
Chen (2001) observed that the immobilized Scenedesmus quadricauda cells in alginate beads can preserve their physiological activities for a long time, even after three years of storage in darkness at 4 °C. This observation was explained by the entrapped cells self-consuming their own pyrenoid reserves. Transmission electron microscopic images of immobilized S. quadricauda cells showed that they lose their pyrenoids after extended storage, which is then rebuilt when the cultures are placed back into their nutrient media under light conditions.
Lebeau et al. (1998) also established the ability to store marine diatom H. ostrearia cells for nearly 2 months after their entrapment in calcium alginate beads and later used them as a substrate source for the greening of oysters. As a follow-up study, the same group achieved a longer term storage when H. ostrearia diatom cells were entrapped in alginate beads and kept at 4 °C (Gaudin et al. 2006). Chen (2003) stored Isochrysis galbana marine microalgal cells for more than a year after immobilizing them in alginate at 4 °C in dark conditions, and the cells were then used for feeding clam cultures.
2.7 Other Applications
In some studies, more than one culture was immobilized to achieve a multifunctional immobilization matrix. For example, Adlercreutz et al. (1982) co-immobilized mixed cultures of algae (Chlorella pyrenoidosa) and bacteria (Gluconobacter oxydans) inside calcium alginate beads for the continuous production of dihydroxyacetone. They did not observe any significant loss of activity within the first six days of this bioprocess. They used the algal cells as an in situ oxygen supplier, which was directly used by the bacteria during the conversion of glycerol to dihydroxyacetone (Adlercreutz et al. 1982). Co-immobilization of microalga S. obliquus with Bacillus subtilis bacteria in carrageenan beads was studied inside air-lift reactors, for enhancing the production of alpha-amylase enzyme (Chevalier and de la Noüe 1988). Microalgal cells were again used as an in situ oxygen generator for the bacterial cells, which were mainly responsible for the synthesis of alpha-amylase enzyme. Co-immobilization overcame the existing oxygen diffusion problems and yielded higher alpha-amylase activity by a factor of around 20 %. They also observed higher growth rates for the algal cells when co-immobilized with bacteria, compared to the immobilization with algal cells alone (Chevalier and de la Noüe 1988).
Immobilization of Dunaliella tertiolecta in alginate (Grizeau and Navarro 1986) and Dunaliella salina in agar-agar (Thakur and Kumar 1999) increased the amount of glycerol production. Immobilized algae were also used for the generation of keto acids from amino acids (Wikström et al. 1982).
Luan et al. (2006) achieved successful removal (90 %) of a highly toxic tributyltin using alginate-immobilized C. vulgaris cells. They observed that less than 10 % of the tributyltin was accumulated inside the cells, while the remainder was adsorbed by both the immobilization matrix and the cell walls.
He et al. (2014) recently constructed an algal fuel cell with immobilized C. vulgaris cells in sodium alginate placed inside a cathode chamber of the fuel cell. The aim was to achieve a complete process that combines biomass production, electricity generation, and wastewater treatment all at the same time. They observed a significant chemical oxygen demand (COD) removal efficiency of 92.1 %.
2.8 Conclusions and Future Directions
immobilization of cells brings several advantages over current suspension bioprocessing, such as (1) providing flexibility to the photobioreactor designs; (2) increasing reaction rates arising from higher cell density; (3) enhancing operational stability; (4) avoiding cell washouts; (5) facilitating cultivation and easy harvesting of microorganisms; (6) minimizing the volume of growth medium as the immobilized cellular matter occupies less space; (7) easier handling of the products; (8) permitting the easy replacement of the algae at any stage of the experiment; (9) protecting the cell cultures from the harsh environmental conditions such as salinity, metal toxicity, variations in pH, and any product inhibition; and (10) allowing continuous utilization of algae in a non-destructive way. Enhanced survival rates of immobilized cells in toxic environments provide a significant alternative to achieve sufficient bioremediation of chemically contaminated environments. It is also important to stress that continuous biomass production, opportunity for product recycling, and nearly spontaneous biomass harvesting will have the potential to outweigh the difficulties and added costs associated with applying the technology on a larger scale.
Conventional wastewater treatment methods are mostly focused on the separation of pollutants from the liquid effluents with a requirement for a further stage to eliminate them. Developing integrated wastewater treatment processes that eliminate the undesired portion of the wastewater while converting it into valuable products is important in developing sustainable processes for the future. immobilization of algal cells is important in the development of an integrated process while simplifying the harvesting of biomass and providing the retention of the high-value algal biomass for further processing.
There are, however, technical issues to address, such as the hybridization of different polymers for creating more efficient and stronger immobilization matrix for algal cells. Immobilization of viable algae inside three-dimensional gel lattices also faces several limitations given that the encapsulating materials can have high volume-to-surface ratios. As a consequence, algal viability decreases since the light, nutrients, or reactants have to diffuse far into these materials to reach the algal cells. One of the other restrictions for the gel-entrapped cultures is their lower growth rates compared to their free-living counterparts. Such drawbacks can be addressed by optimizing the immobilization processes, that is, by choosing different encapsulating materials with lower volume-to-surface ratios such as thin films. Overcoming the difficulties of the current technology will increase the applicability of immobilized algae systems for various industrial applications.
Current immobilization projects have been often confined to the laboratory in providing an effective proof-of-concept rather than quick-install industrial prototypes. For larger scale wastewater treatment and biofuel production bioprocesses, the cost of immobilization matrix becomes a significant parameter that needs to be improved by further innovative designs and additional profits through generating valuable by-products.
Discovering the optimal microalgae–bacteria combinations for co-immobilization processes can also be a good alternative for large-scale wastewater treatment practices, since algal cultures in nature are usually associated with bacteria.
Application of innovative composite materials for use as the algal immobilization matrices can have a significant contribution to the economic and environmental development by sustainable utilization and recovery of the local resources, while bringing valuable strategies for solving important environmental issues.
References
Abdel-Hamid MI (1996) Development and application of a simple procedure for toxicity testing using immobilized algae. Water Sci Technol 33(6):129–138
Abe K, Matsumura I, Imamaki A, Hirano M (2003) Removal of inorganic nitrogen sources from water by the algal biofilm of the aerial microalga Trentepohlia aurea. World J Microb Biot 19(3):325–328
Adlercreutz P, Holst O, Mattiasson B (1982) Oxygen supply to immobilized cells: 2. Studies on a co immobilized algae—bacteria preparation with in situ oxygen generation. Enzyme Microb Tech 4(6):395–400
Aguilar-May B, del Pilar Sánchez-Saavedra M, Lizardi J, Voltolina D (2007) Growth of Synechococcus sp. immobilized in chitosan with different times of contact with NaOH. J Appl Phycol 19(2):181–183
Akhtar N, Iqbal J, Iqbal M (2003a) Microalgal-luffa sponge immobilized disc: a new efficient biosorbent for the removal of Ni (II) from aqueous solution. Lett Appl Microbiol 37(2):149–153
Akhtar N, Saeed A, Iqbal M (2003b) Chlorella sorokiniana immobilized on the biomatrix of vegetable sponge of Luffa cylindrica: a new system to remove cadmium from contaminated aqueous medium. Bioresource Technol 88(2):163–165
Akhtar N, Iqbal J, Iqbal M (2004) Enhancement of lead(II) biosorption by microalgal biomass immobilized onto loofa (Luffa cylindrica) sponge. Eng Life Sci 4(2):171–178
Akhtar N, Iqbal M, Zafar SI, Iqbal J (2008) Biosorption characteristics of unicellular green alga Chlorella sorokiniana immobilized in loofa sponge for removal of Cr(III). J Environ Sci (China) 20(2):231–239
Aksu Z, Gönen F, Demircan Z (2002) Biosorption of chromium(VI) ions by Mowital® B30H resin immobilized activated sludge in a packed bed: comparison with granular activated carbon. Process Biochem 38(2):175–186
Andrade LR, Salgado LT, Farina M, Pereira MS, Mourão PAS, Amado Filho GM (2004) Ultrastructure of acidic polysaccharides from the cell walls of brown algae. J Struct Biol 145(3):216–225
Bailliez C, Largeau C, Casadevall E (1985) Growth and hydrocarbon production of Botryococcus braunii immobilized in calcium alginate gel. Appl Microbiol Biot 23(2):99–105
Bailliez C, Largeau C, Berkaloff C, Casadevall E (1986) Immobilization of Botryococcus braunii in alginate: influence on chlorophyll content, photosynthetic activity and degeneration during batch cultures. Appl Microbiol Biot 23(5):361–366
Barkley NP (1991) Extraction of mercury from groundwater using immobilized algae. J Air Waste Manage 41(10):1387–1393
Bayramoğlu G, Tuzun I, Celik G, Yilmaz M, Arica MY (2006) Biosorption of mercury(II), cadmium(II) and lead(II) ions from aqueous system by microalgae Chlamydomonas reinhardtii immobilized in alginate beads. Int J Miner Process 81(1):35–43
Borowitzka MA (1999) Commercial production of microalgae: ponds, tanks, and fermenters. Prog Ind M 35:313–321
Boulos RA, Eroglu E, Chen X, Scaffidi A, Edwards BR, Toster J, Raston CL (2013) Unravelling the structure and function of human hair. Green Chem 15(5):1268–1273
Bozeman J, Koopman B, Bitton G (1989) Toxicity testing using immobilized algae. Aquat Toxicol 14(4):345–352
Brady D, Duncan J (1994) Bioaccumulation of metal cations by Saccharomyces cerevisiae. Appl Microbiol Biot 41(1):149–154
Brouers M, Collard F, Jeanfils J, Loudeche R (1983) Long term stabilization of photobiological activities of immobilized algae. Photoproduction of hydrogen by immobilized “adapted” Scenedesmus cells. Sol Energy R & D Eur Community Ser D 2:171–178
Canizares R, Dominguez A, Rivas L, Montes M, Travieso L, Benitez F (1993) Free and immobilized cultures of Spirulina maxima for swine waste treatment. Biotechnol Lett 15(3):321–326
Cao J, Yuan W, Pei Z, Davis T, Cui Y, Beltran M (2009) A preliminary study of the effect of surface texture on algae cell attachment for a mechanical-biological energy manufacturing system. J Manuf Sci Eng ASME 131(6):064505
Carrilho ENV, Nóbrega JA, Gilbert TR (2003) The use of silica-immobilized brown alga (Pilayella littoralis) for metal preconcentration and determination by inductively coupled plasma optical emission spectrometry. Talanta 60(6):1131–1140
Chang HN, Yoo I-K, Kim BS (1994) High density cell culture by membrane-based cell recycle. Biotechnol Adv 12(3):467–487
Chen Y-C (2001) Immobilized microalga Scenedesmus quadricauda (Chlorophyta, Chlorococcales) for long-term storage and for application for water quality control in fish culture. Aquaculture 195(1–2):71–80
Chen Y-C (2003) Immobilized Isochrysis galbana (Haptophyta) for long-term storage and applications for feed and water quality control in clam (Meretrix lusoria) cultures. J Appl Phycol 15(5):439–444
Chevalier P, de la Noüe J (1985) Wastewater nutrient removal with microalgae immobilized in carrageenan. Enzyme Microb Tech 7(12):621–624
Chevalier P, de la Noüe J (1988) Behavior of algae and bacteria co-immobilized in carrageenan, in a fluidized bed. Enzyme Microb Tech 10(1):19–23
Chevalier P, Proulx D, Lessard P, Vincent WF, de la Noüe J (2000) Nitrogen and phosphorus removal by high latitude mat-forming cyanobacteria for potential use in tertiary wastewater treatment. J Appl Phycol 12(2):105–112
Chouteau C, Dzyadevych S, Chovelon J-M, Durrieu C (2004) Development of novel conductometric biosensors based on immobilised whole cell Chlorella vulgaris microalgae. Biosens Bioelectron 19(9):1089–1096
Christenson L, Sims R (2011) Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol Adv 29(6):686–702
Cohen Y (2001) Biofiltration–the treatment of fluids by microorganisms immobilized into the filter bedding material: a review. Bioresource Technol 77(3):257–274
Crandall B (1996) Nanotechnology: molecular speculations on global abundance. MIT Press, Cambridge
Cromar N, Fallowfield HJ, Martin N (1996) Influence of environmental parameters on biomass production and nutrient removal in a high rate algal pond operated by continuous culture. Water Sci Technol 34(11):133–140
da Costa ACA, de França FP (1996) The use of seaweeds as immobilization supports for microorganisms used in continuous cadmium biosorption. Biotechnol Tech 10(10):761–766
da Costa ACA, Leite SGF (1991) Metals biosorption by sodium alginate immobilized Chlorella homosphaera cells. Biotechnol Lett 13(8):559–562
de-Bashan LE, Bashan Y (2010) Immobilized microalgae for removing pollutants: review of practical aspects. Bioresource Technol 101(6):1611–1627
de-Bashan LE, Bashan Y, Moreno M, Lebsky VK, Bustillos JJ (2002a) Increased pigment and lipid content, lipid variety, and cell and population size of the microalgae Chlorella spp. when co-immobilized in alginate beads with the microalgae-growth-promoting bacterium Azospirillum brasilense. Can J Microbiol 48(6):514–521
de-Bashan LE, Moreno M, Hernandez J-P, Bashan Y (2002b) Removal of ammonium and phosphorus ions from synthetic wastewater by the microalgae Chlorella vulgaris coimmobilized in alginate beads with the microalgae growth-promoting bacterium Azospirillum brasilense. Water Res 36(12):2941–2948
de-Bashan LE, Antoun H, Bashan Y (2005) Cultivation factors and population size control the uptake of nitrogen by the microalgae Chlorella vulgaris when interacting with the microalgae growth-promoting bacterium Azospirillum brasilense. FEMS Microbiol Ecol 54(2):197–203
de la Noue J, de Pauw N (1988) The potential of microalgal biotechnology: a review of production and uses of microalgae. Biotechnol Adv 6(4):725–770
Dennison M, Turner AP (1995) Biosensors for environmental monitoring. Biotechnol Adv 13(1):1–12
Dönmez G, Aksu Z (2002) Removal of chromium (VI) from saline wastewaters by Dunaliella species. Process Biochem 38(5):751–762
Dziwulska U, Bajguz A, Godlewska-Żyłkiewicz B (2004) The use of algae Chlorella vulgaris immobilized on Cellex-T support for separation/preconcentration of trace amounts of platinum and palladium before GFAAS determination. Anal Lett 37(10):2189–2203
Efthymiou G, Shuler M (1987) Elimination of diffusional limitations in a membrane entrapped cell reactor by pressure cycling. Biotechnol Progr 3(4):259–264
Elmahadi HAM, Greenway GM (1991) Immobilized alga as a reagent for preconcentration in trace element atomic absorption spectrometry. J Anal Atom Spectrom 6(8):643–646
Eroglu E, Melis A (2009) “Density equilibrium” method for the quantitative and rapid in situ determination of lipid, hydrocarbon, or biopolymer content in microorganisms. Biotechnol Bioeng 102(5):1406–1415
Eroglu E, Agarwal V, Bradshaw M, Chen X, Smith SM, Raston CL, Swaminathan Iyer K (2012) Nitrate removal from liquid effluents using microalgae immobilized on chitosan nanofiber mats. Green Chem 14(10):2682–2685
Eroglu E, D’Alonzo NJ, Smith SM, Raston CL (2013) Vortex fluidic entrapment of functional microalgal cells in a magnetic polymer matrix. Nanoscale 5(7):2627–2631
Faafeng BA, van Donk E, Källqvist ST (1994) In situ measurement of algal growth potential in aquatic ecosystems by immobilized algae. J Appl Phycol 6(3):301–308
Fierro S, del Pilar Sanchez-Saavedra M, Copalcua C (2008) Nitrate and phosphate removal by chitosan immobilized Scenedesmus. Bioresource Technol 99(5):1274–1279
Frense D, Müller A, Beckmann D (1998) Detection of environmental pollutants using optical biosensor with immobilized algae cells. Sensor Actuat B-Chem 51(1):256–260
Gaudin P, Lebeau T, Robert J-M (2006) Microalgal cell immobilization for the long-term storage of the marine diatom Haslea ostrearia. J Appl Phycol 18(2):175–184
Gekeler W, Grill E, Winnacker E-L, Zenk MH (1988) Algae sequester heavy metals via synthesis of phytochelatin complexes. Arch Microbiol 150(2):197–202
Godlewska-Żyłkiewicz B (2003) Biosorption of platinum and palladium for their separation/preconcentration prior to graphite furnace atomic absorption spectrometric determination. Spectrochim Acta B 58(8):1531–1540
Gonzalez LE, Bashan Y (2000) Increased growth of the microalga Chlorella vulgaris when coimmobilized and cocultured in alginate beads with the plant-growth-promoting bacterium Azospirillum brasilense. Appl Environ Microb 66(4):1527–1531
Grizeau D, Navarro J (1986) Glycerol production by Dunaliella tertiolecta immobilized within Ca-alginate beads. Biotechnol Lett 8(4):261–264
Hahn JJ, Ghirardi ML, Jacoby WA (2007) Immobilized algal cells used for hydrogen production. Biochem Eng J 37(1):75–79
Hall-Stoodley L, Costerton JW, Stoodley P (2004) Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Micro 2(2):95–108
Hameed M, Ebrahim O (2007) Review: biotechnological potential uses of immobilized algae. Int J Agri Biol 9:183–192
Hankamer B, Lehr F, Rupprecht J, Mussgnug JH, Posten C, Kruse O (2007) Photosynthetic biomass and H2 production by green algae: from bioengineering to bioreactor scale-up. Physiol Plantarum 131(1):10–21
He H, Zhou M, Yang J, Hu Y, Zhao Y (2014) Simultaneous wastewater treatment, electricity generation and biomass production by an immobilized photosynthetic algal microbial fuel cell. Bioproc Biosyst Eng 37(5):873–880
Hernandez J-P, de-Bashan LE, Bashan Y (2006) Starvation enhances phosphorus removal from wastewater by the microalga Chlorella spp. co-immobilized with Azospirillum brasilense. Enzyme Microb Technol 38(1):190–198
Hertzberg S, Jensen A (1989) Studies of alginate immobilized marine microalgae. Bot Mar 32:267–273
Hoffmann JP (1998) Wastewater treatment with suspended and nonsuspended algae. J Phycol 34(5):757–763
Hunik JH, Tramper J (1993) Large-scale production of kappa-carrageenan droplets for gel-bead production: theoretical and practical limitations of size and production rate. Biotechnol Progr 9(2):186–192
Jimenez-Perez M, Sanchez-Castillo P, Romera O, Fernandez-Moreno D, Pérez-Martınez C (2004) Growth and nutrient removal in free and immobilized planktonic green algae isolated from pig manure. Enzyme Microb Tech 34(5):392–398
Johnson EA, An G-H (1991) Astaxanthin from microbial sources. Crit Rev Biotechnol 11(4):297–326
Johnson MB, Wen Z (2010) Development of an attached microalgal growth system for biofuel production. Appl Microbiol Biotechnol 85(3):525–534
Karatan E, Watnick P (2009) Signals, regulatory networks, and materials that build and break bacterial biofilms. Microbiol Mol Biol Rev 73(2):310–347
Kaya VM, Picard G (1996) Stability of chitosan gel as entrapment matrix of viable Scenedesmus bicellularis cells immobilized on screens for tertiary treatment of wastewater. Bioresource Technol 56(2–3):147–155
Kaya VM, de la Noüe J, Picard G (1995) A comparative study of four systems for tertiary wastewater treatment by Scenedesmus bicellularis: new technology for immobilization. J Appl Phycol 7(1):85–95
Kayano H, Matsunaga T, Karube I, Suzuki S (1981) Hydrogen evolution by co-immobilized Chlorella vulgaris and Clostridium butyricum cells. Biochim Biophys Acta 638(1):80–85
Kelleher CM, Vacanti JP (2010) Engineering extracellular matrix through nanotechnology. J R Soc Interface 7:S717–S729
Kosourov SN, Seibert M (2009) Hydrogen photoproduction by nutrient-deprived Chlamydomonas reinhardtii cells immobilized within thin alginate films under aerobic and anaerobic conditions. Biotechnol Bioeng 102(1):50–58
Kuu WY, Polack JA (1983) Improving immobilized biocatalysts by gel phase polymerization. Biotechnol Bioeng 25(8):1995–2006
Lau P, Tam N, Wong Y (1997) Wastewater nutrients (N and P) removal by carrageenan and alginate immobilized Chlorella vulgaris. Environ Technol 18(9):945–951
Laurinavichene TV, Fedorov AS, Ghirardi ML, Seibert M, Tsygankov AA (2006) Demonstration of sustained hydrogen photoproduction by immobilized, sulfur-deprived Chlamydomonas reinhardtii cells. Int J Hydrogen Energ 31(5):659–667
Laurinavichene TV, Kosourov SN, Ghirardi ML, Seibert M, Tsygankov AA (2008) Prolongation of H2 photoproduction by immobilized, sulfur-limited Chlamydomonas reinhardtii cultures. J Biotechnol 134(3–4):275–277
Lebeau T, Moan R, Turpin V, Robert J-M (1998) Alginate-entrapped Haslea ostrearia as inoculum for the greening of oysters. Biotechnol Tech 12(11):847–850
Lebeau T, Gaudin P, Junter G-A, Mignot L, Robert J-M (2000) Continuous marennin production by agar-entrapped Haslea ostrearia using a tubular photobioreactor with internal illumination. Appl Microbiol Biot 54(5):634–640
Leenen EJTM, Dos Santos VAP, Grolle KCF, Tramper J, Wijffels R (1996) Characteristics of and selection criteria for support materials for cell immobilization in wastewater treatment. Water Res 30(12):2985–2996
Leusch A, Holan ZR, Volesky B (1995) Biosorption of heavy metals (Cd, Cu, Ni, Pb, Zn) by chemically-reinforced biomass of marine algae. J Chem Technol Biot 62(3):279–288
Li X, Xu H, Wu Q (2007) Large-scale biodiesel production from microalga Chlorella protothecoides through heterotrophic cultivation in bioreactors. Biotechnol Bioeng 98(4):764–771
Liu Y, Rafailovich MH, Malal R, Cohn D, Chidambaram D (2009) Engineering of bio-hybrid materials by electrospinning polymer-microbe fibers. Proc Natl Acad Sci USA 106(34):14201–14206
Luan T, Jin J, Chan S, Wong Y, Tam NF (2006) Biosorption and biodegradation of tributyltin (TBT) by alginate immobilized Chlorella vulgaris beads in several treatment cycles. Process Biochem 41(7):1560–1565
Lukavský J, Komárek J, Lukavská A, Ludvík J, Pokorný J (1986) Metabolic activity and cell structure of immobilized algal cells (Chlorella, Scenedesmus). Arch Hydrobiol Suppl 73:261–279
Ma F, Hanna MA (1999) Biodiesel production: a review. Bioresource Technol 70(1):1–15
Mahan CA, Holcombe JA (1992) Immobilization of algae cells on silica gel and their characterization for trace metal preconcentration. Anal Chem 64(17):1933–1939
Mallick N (2002) Biotechnological potential of immobilized algae for wastewater N, P and metal removal: A review. Biometals 15(4):377–390
Mallick N, Rai L (1993) Influence of culture density, pH, organic acids and divalent cations on the removal of nutrients and metals by immobilized Anabaena doliolum and Chlorella vulgaris. World J Microbiol Biotechnol 9(2):196–201
Mallick N, Rai L (1994) Removal of inorganic ions from wastewaters by immobilized microalgae. World J Microbiol Biotechnol 10(4):439–443
Mehta SK, Gaur JP (2001) Removal of Ni and Cu from single and binary metal solutions by free and immobilized Chlorella vulgaris. Eur J Protistol 37(3):261–271
Melis A, Zhang L, Forestier M, Ghirardi ML, Seibert M (2000) Sustained photobiological hydrogen gas production upon reversible inactivation of oxygen evolution in the green alga Chlamydomonas reinhardtii. Plant Physiol 122(1):127–136
Metcalf INC, Eddy H (2003) Wastewater engineering: treatment and reuse. McGraw-Hill, New York
Moreno-Garrido I, Blasco J, Gonzalez-Delvalle M, Lubián L (1998) Differences in copper accumulation by the marine microalga Nannochloropsis gaditana, submitted to two different thermal treatments. Ecotoxicol Environ Restor 1:43–47
Moreno-Garrido I, Campana O, Lubián L, Blasco J (2005) Calcium alginate immobilized marine microalgae: experiments on growth and short-term heavy metal accumulation. Mar Pollut Bull 51(8):823–829
Mouget J-L, Dakhama A, Lavoie MC, de la Noüe J (1995) Algal growth enhancement by bacteria: Is consumption of photosynthetic oxygen involved? FEMS Microbiol Ecol 18(1):35–43
Mulbry W, Westhead EK, Pizarro C, Sikora L (2005) Recycling of manure nutrients: use of algal biomass from dairy manure treatment as a slow release fertilizer. Bioresource Technol 96(4):451–458
Naessens M, Tran-Minh C (1999) Biosensor using immobilized Chlorella microalgae for determination of volatile organic compounds. Sensor Actuat B Chem 59(2):100–102
Nakajima A, Horikoshi T, Sakaguchi T (1982) Recovery of uranium by immobilized microorganisms. Eur J Appl Microbiol 16(2–3):88–91
Olguín EJ, Galicia S, Mercado G, Pérez T (2003) Annual productivity of Spirulina (Arthrospira) and nutrient removal in a pig wastewater recycling process under tropical conditions. J Appl Phycol 15(2–3):249–257
Onyancha D, Mavura W, Ngila JC, Ongoma P, Chacha J (2008) Studies of chromium removal from tannery wastewaters by algae biosorbents, Spirogyra condensata and Rhizoclonium hieroglyphicum. J Hazard Mater 158(2):605–614
Oswald WJ (1988) Micro-algae and waste-water treatment. In: Borowitzka MA, Borowitzka LJ (eds) Micro-algal biotechnology. Cambridge University Press, New York, pp 305–328
Przytocka-Jusiak M, Ba̵szczyk Mw, Kosińska E, Bisz-Konarzewska A (1984) Removal of nitrogen from industrial wastewaters with the use of algal rotating disks and denitrification packed bed reactor. Water Res 18(9):1077–1082
Qureshi N, Annous BA, Ezeji TC, Karcher P, Maddox IS (2005) Biofilm reactors for industrial bioconversion processes: employing potential of enhanced reaction rates. Microb Cell Fact 4(1):24
Robinson PK (1998) Immobilized algal technology for wastewater treatment purposes. In: Wong YS, Tam FY (eds) Wastewater treatment with algae. Springer, Berlin, pp 1–16
Safonova E, Kvitko K, Iankevitch M, Surgko L, Afti I, Reisser W (2004) Biotreatment of industrial wastewater by selected algal-bacterial consortia. Eng Life Sci 4:347–353
Schiewer S, Volesky B (2000) Biosorption processes for heavy metal removal. In: Lovley DR (ed) Environmental microbe–metal interactions. ASM Press, Washington DC, pp 329–362
Serp D, Cantana E, Heinzen C, Von Stockar U, Marison IW(2000) Characterization of an encapsulation device for the production of monodisperse alginate beads for cell immobilization. Biotechnol Bioeng 70(1):41–53
Shi J, Podola B, Melkonian M (2007) Removal of nitrogen and phosphorus from wastewater using microalgae immobilized on twin layers: an experimental study. J Appl Phycol 19(5):417–423
Shitanda I, Takada K, Sakai Y, Tatsuma T (2005) Compact amperometric algal biosensors for the evaluation of water toxicity. Anal Chim Acta 530(2):191–197
Shul’ga A, Soldatkin A, El’skaya A, Dzyadevich S, Patskovsky S, Strikha V (1994) Thin-film conductometric biosensors for glucose and urea determination. Biosens Bioelectron 9(3):217–223
Singh Y (2003) Photosynthetic activity, and lipid and hydrocarbon production by alginate-immobilized cells of Botryococcus in relation to growth phase. J Microbiol Biotechnol 13(5):687–691
Smidsrød O, Skjåk-Bræk G (1990) Alginate as immobilization matrix for cells. Trends Biotechnol 8:71–78
Song W, Rashid N, Choi W, Lee K (2011) Biohydrogen production by immobilized Chlorella sp. using cycles of oxygenic photosynthesis and anaerobiosis. Bioresource Technol 102(18):8676–8681
Tam N, Wong Y (2000) Effect of immobilized microalgal bead concentrations on wastewater nutrient removal. Environ Pollut 107(1):145–151
Tamponnet C, Coslantino F, Barbotin JN, Calvayrac R (1985) Cytological and physiological behaviour of Euglena gracillis cells entrapped in a calcium alginate gel. Physiol Plant 63(3):277–283
Thakur A, Kumar H (1999) Use of natural polymers as immobilizing agents and effects on the growth of Dunaliella salina and its glycerol production. Acta Biotechnol 19(1):37–44
Toda K, Sato K (1985) Simulation study on oxygen uptake rate of immobilized growing microorganisms. J Ferment Technol 63(3):251–258
Torpey WN, Heukelekian H, Kaplovsky AJ, Epstein R (1971) Rotating disks with biological growths prepare wastewater for disposal or reuse. J Water Pollut Control Fed 43(11):2181–2188
Travieso L, Benitez F, Weiland P, Sanchez E, Dupeyron R, Dominguez A (1996) Experiments on immobilization of microalgae for nutrient removal in wastewater treatments. Bioresource Technol 55(3):181–186
Travieso L, Canizares R, Borja R, Benitez F, Dominguez A, Dupeyrón y R, Valiente V (1999) Heavy metal removal by microalgae. Bull Environ Contam Toxicol 62(2):144–151
Urrutia I, Serra JL, Llama MJ (1995) Nitrate removal from water by Scenedesmus obliquus immobilized in polymeric foams. Enzyme Microb Technol 17(3):200–205
Van Donk E, Abdel-Hamid M, Faafeng B, Källqvist T (1992) Effects of Dursban® 4E and its carrier on three algal species during exponential and P-limited growth. Aquat Toxicol 23(3):181–191
Velásquez L, Dussan J (2009) Biosorption and bioaccumulation of heavy metals on dead and living biomass of Bacillus sphaericus. J Hazard Mater 167(1):713–716
Wahid MH, Eroglu E, Chen X, Smith SM, Raston CL (2013a) Entrapment of Chlorella vulgaris cells within graphene oxide layers. RSC Adv 3(22):8180–8183
Wahid MH, Eroglu E, Chen X, Smith SM, Raston CL (2013b) Functional multi-layer graphene–algae hybrid material formed using vortex fluidics. Green Chem 15(3):650
Weetall HH, Krampitz LO (1980) Production of hydrogen from water using biophotolytic methods. J Solid-Phase Biochem 5(2):115–124
Wikström P, Szwajcer E, Brodelius P, Nilsson K, Mosbach K (1982) Formation of α-keto acids from amino acids using immobilized bacteria and algae. Biotechnol Lett 4(3):153–158
Wilkinson S, Goulding K, Robinson P (1990) Mercury removal by immobilized algae in batch culture systems. J Appl Phycol 2(3):223–230
Wuertz S, Bishop PL, Wilderer PA (2003) Biofilms in wastewater treatment: an interdisciplinary approach. IWA Publishing, London
Yang P, Teo W-K, Ting Y-P (2006) Design and performance study of a novel immobilized hollow fiber membrane bioreactor. Bioresource Technol 97(1):39–46
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Eroglu, E., Smith, S.M., Raston, C.L. (2015). Application of Various Immobilization Techniques for Algal Bioprocesses. In: Moheimani, N., McHenry, M., de Boer, K., Bahri, P. (eds) Biomass and Biofuels from Microalgae. Biofuel and Biorefinery Technologies, vol 2. Springer, Cham. https://doi.org/10.1007/978-3-319-16640-7_2
Download citation
DOI: https://doi.org/10.1007/978-3-319-16640-7_2
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-16639-1
Online ISBN: 978-3-319-16640-7
eBook Packages: EnergyEnergy (R0)