Abstract
The possibility that a combination of neurotrophins induces long-lasting neuroprotection of the cord following spinal cord injury (SCI) was examined in a rat model. The SCI was performed by making a unilateral incision into the right dorsal horn of the T10-11 segments and the animals were allowed to survive 5 h after trauma. Different combination of neurotrophins, i.e., BDNF in combination with GDNF, NT-3, or NGF was applied topically over the traumatized spinal cord and motor dysfunction, blood-spinal cord barrier (BSCB) breakdown, edema formation, and cell injury were examined. Topical application of BDNF in combination with GDNF and NGF 30 min (but not 60 or 90 min) at high concentrations (0.5 µg each) after SCI significantly improved motor function and reduced BSCB breakdown, edema formation, and cell injury at 5 h. However, concurrent application of BDNF, IGF-1, and GDNF (but not with NT-3 or NGF) even 60 or 90 min after trauma induced a significant reduction in motor dysfunction and spinal cord pathology. These observations suggest that a combination of neurotrophins may have added therapeutic value in the treatment of SCI, not reorted earlier.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Spinal cord injury (SCI) is a serious clinical problem that induces lifelong disabilities for the victims and places a huge burden on society (see (5,6,11)). Although our knowledge on the basic mechanisms of the pathophysiology of SCI has expanded tremendously in the last decades (2,3,5,6), suitable and effective therapeutic approaches to enhance neuroprotection and/or neurorepair are still not worked out. Thus, further studies in SCI are needed to enhance neurorepair mechanisms in order to improve the quality of life of trauma victims.
Experimental studies in SCI suggest that neurotrophins could play important roles in the repair mechanisms of the spinal cord following injury (2,3,5–10). Thus, dysregulation of neurotrophin receptors and/or alerted expression of neurotrophins in spinal cord motoneurons following injury are quite common (see (12) for review). This indicates that therapeutic intervention using neurotrophic factors may influence recovery following SCI by enhancing neuroregeneration or neurorepair processes.
One of the important goals for the treatment of SCI with neurotrophins is to thwart the propagation of spinal cord pathology to the uninjured portion of the cord and to rescue damaged nerve cells and axons in order to improve functional outcome (10–15). Another therapeutic aim of SCI is to find a suitable combination of neurotrophins that can help to achieve neuroprotection and neurorepair even when applied at longer time intervals after the initial injury (7–10,12–15).
Keeping this in mind, our laboratory is engaged in exploring the possibility of enhancing functional recovery and enhancing neuroprotection using several neurotrophic factors, either alone or in combination, in experimental models of SCI (7–15). Considering modern day patient care, the time delay from the accident to the initiation of treatment can easily be reduced from 180 to 90 min (see (15)). Thus, any suitable combination of neurotrophins that can delay the propagation of cord pathology within 90 min after trauma will certainly be an added value to the current therapeutic strategies used in SCI.
This investigation is aimed at exploring the possibility of using a select combination of neurotrophins to improve functional outcome and reduce spinal cord edema formation and cell injury when applied 60 to 90 min after SCI in our rat model.
2 Materials and Methods
2.1 Animals
Experiments were carried out on Male Sprague Dawley rats (250 to 300 g) housed at controlled room temperature (21 ± 1°C) on a 12 h light and 12 h dark schedule. Food and tap water were supplied ad libitum before the experiments.
2.2 Spinal Cord Injury
Under equithesin anesthesia (0.3 mL/100 g, i.p.) a one segment laminectomy was made at the T10-11 segments. The SCI was inflicted using a longitudinal incision (4 mm long and 1.5 mm deep) on the right dorsal horn using a sterile scalpel blade (see Fig. 1). The deepest part of the incision was limited to Rexed’s laminae VII or VIII (16). All experiments were carried out according to the National Institute of Health Guidelines for care of experimental animals and approved by Local Institutional Ethics Committee. Sham operated animals without SCI served as controls.
The new model of rat spinal cord injury (SCI) and tissue sampling (see (11)). A focal incision to the right dorsal horn of the T10-11 segments was used to inflict SCI. (a) Longitudinal view of the lesion within the spinal cord. The focal incision is limited to the right dorsal horn of the T10-11 segment. Tissue pieces for morphological or biochemical studies are taken from the adjacent T9 (rostral) and T12 (caudal) segments. (b) Cross section of the spinal cord showing the extent of the lesion within the dorsal horn. The deepest part of the lesion is mainly present around Rexed’s laminae VII or VIII. The white matter is largely intact. For high resolution light microscopy, tissue pieces were embedded in Epon, and approximately 1 µm thick whole spinal cord sections were cut and stained with toluidine blue. These sections were examined with light microscopy and photographed (see b). For immunohistochemistry or morphology, small areas from either the contralateral dorsal horn (1) or ventral horn (2) were selected to examine neuroprotection (modified after (11)). Bar: a ≈ 5 mm, b ≈ 2 mm (modified after 17)
2.3 Neurotrophin Treatment
Separate groups of animals were treated with various combination of neurotrophins, e.g., BDNF with GDNF, NGF, NT-3 or IGF-1, or GDNF. Neurotrophins were applied 30 to 90 min after SCI and the animals were allowed to survive 5 h after injury. The total dose of the neurotrophins was adjusted (0.5 µg each) in 30 µL (0.5 µg of BDNF + 0.5 µg of GDNF, or IGF-1, NGF or NT-3) and applied topically over the injured spinal cord with in 10 s (see (7–11)). The control group received 30 µL of 0.9% saline instead of neurotrophins (11–15).
2.4 Motor Functions
Tarlov scale and capacity angle were monitored in spinal cord injured animals in order to assess the functional outcome in neurotrophin- or saline-treated animals according to standard procedures (see (12–14)).
2.5 Blood–Spinal Cord Barrier Permeability
The blood–spinal cord barrier (BSCB) permeability to Evans blue albumin and radioiodine was examined in perifocal spinal cord segments (T9 and T12) according to standard protocol (see Fig. 1, Ref. 4,17.
2.6 Spinal Cord Edema
Spinal cord edema in T9 and T12 segments was assessed by measuring water content (1,16,18).
2.7 Spinal Cord Pathology
The spinal cord pathology in T9 and T12 segments (Fig. 1) was examined using Nissl or Hematoxylin and Eosin staining using standard histological techniques as described previously (17,18).
2.8 Statistical Evaluation of Data
ANOVA followed by Dunnet’s test for multiple group comparison from one control was used to evaluate statistical significance between control, injured, and neurotrophin treated groups. A p-value <0.05 was considered significant.
3 Results
3.1 Effects of Neurotrophins on Functional Outcome after SCI
Functional outcome was markedly improved when BDNF in combination with NT-3 or NGF (0.5 µg each) was applied 30 min after SCI, as measured by the Tarlov scale or capacity angle test (Table 1). This effect was markedly absent when these neurotrophins were applied either 60 or 90 min after SCI. Interestingly, even a triple combination of these neurotrophins (BDNF+NT-3+NGF, 0.5 µg each) did not induce significant improvement in motor function beyond 60 min SCI (results not shown). On the other hand, when BDNF was combined with IGF-1 and GDNF (0.5 µg each), this triple combination was able to improve motor function even after 90 min of SCI (Table 1).
3.2 Effects of Neurotrophins on BSCB Permeability in SCI
Marked reduction in BSCB permeability to Evans blue and radioiodine was noted in traumatized animals that received BDNF in combination with NT-3 or NGF 30 min after SCI (Table 1). No apparent reduction in BSCB permeability was seen even when these neurotrophins were applied in a combination (BDNF+NT-3+NGF, 0.5 µg each) beyond 60 min SCI (results not shown). However, a triple combination of BDNF with IGF-1 and GDNF (0.5 µg each) significantly reduced BSCB leakage even after 90 min of SCI (Table 1).
3.3 Effects of Neurotrophins on Edema Formation in SCI
A combination of BDNF with NT-3 and/or NGF effectively reduced edema formation if applied at 30 min but not after 60 or 90 min SCI (Table 1). In contrast, a combination of BDNF with IGF-1 and GDNF remarkably reduced spinal cord edema formation when applied even 60 to 90 min after injury (Table 1).
3.4 Effects of Neurotrophins on Spinal Cord Pathology in SCI
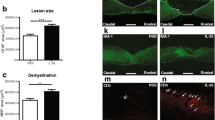
The cell changes in the spinal cord were markedly reduced by the triple combination of BDNF, IGF-1, and GDNF treatment when these neurotrophins were administered 60 to 90 min after SCI (Table 1, Fig. 2). On the other hand, a combination of BDNF with NT-3 and/or NGF did not reduce cell changes in the spinal cord if applied 60 min after SCI (Table 1).
Neuroprotective effects of combinations of neurotrophins 5 h after SCI. A combination of three neurotrophins, BDNF, IGF-1, and GDNF, almost completely attenuated nerve cell damage in the spinal cord when administered topically over the injured cord even 60 min after trauma (c). In contrast, other combinations of neurotrophins, i.e., BDNF, NT-3, and NGF, were not as effective in reducing nerve cell damage when applied either 60 (b) or 90 min (a) after injury as compared to the untreated injured rat (d). Several damaged nerve cells (arrows) are seen in the spinal cord following trauma in the untreated rat (d). Bar: 25 µm
4 Discussion
The results presented here are the first to demonstrate that a select combination of triple neurotrophins enhance function outcome and spinal cord pathology if applied topically 30 to 90 min after SCI. This effect appears to be very specific to the specific combination of neurotrophins used and the time of their application at different post-trauma periods. Thus, a combination of BDNF with NT-3 and NGF remarkably reduced functional deficits and spinal cord pathology if administered 30 min after SCI. On the other hand, this combination of neurotrophins remained ineffective if applied 60 to 90 min after injury. This indicates that SCI induces a time-related deficit of specific neurotrophins that are normally required to maintain spinal cord functions.
Our observations further show that a triple combination of neurotrophins (BDNF, NT-3, and NGF) 30 min after SCI is far superior in inducing neuroprotection than a combination of the two of the neurotrophins (BDNF and NT-3 or BDNF and NGF). This suggests that exogenous supplements with the right kind of growth factors at the right time are needed to attenuate spinal cord pathology and to improve functional outcome following trauma (see 5,6,19).
We applied a dose of 0.5 µg of each neurotrophin to the traumatized spinal cord in this investigation. However, there are reasons to believe that a specific combination of neurotrophins rather than the total amount of growth factors is responsible for the observed neuroprotection following the multiple combination of neurotrophins in SCI (2,6). This idea is further supported by the fact that no suitable neuroprotective effects on functional recovery or spinal cord pathology is seen in our experiments when the same amount of growth factors, i.e., BDNF, NT-3 and NGF (0.5 µg each) were applied 60 or 90 min after injury. Thus, exogenous application of right kind of neurotrophins at different time intervals after SCI is needed to achieve neuroprotection and to improve functional outcome.
In SCI, all of the components of the spinal cord, i.e., neurons, glial, and endothelial cells are damaged progressively with time (11,12). Thus, maintenance of neuronal, glial, and endothelial cell functions after SCI is needed to achieve neuroprotection and improve functional outcome (see (14,15)). It appears that during the early phase of SCI, treatment with neurotrophins, (BDNF, NT-3 and NGF) is sufficient to thwart spinal cord pathology. However, later damage to glial cell function along with neuronal damage worsens the outcome of SCI induced cord pathology. Thus, a combination of neurotrophins acting on neuronal and glial cells is needed to improve spinal cord function during the later phases of SCI.
Keeping these views in consideration, we used a combination of BDNF with IGF-1 and GDNF and applied then at 60 to 90 min after SCI. Our results show that a combination of neurotrophins acting on nerve cells (BDNF) and glial cells (GDNF), together with IGF-1 is effective in attenuating spinal cord pathology and improving functional outcome during later phases of SCI. These observations are in line with the idea that exogenous supplementation of neurotrophins supporting neural, glial, and endothelial cell functions after SCI could effectively induce neuroprotection and enhance functional recovery.
The possible mechanisms of neurotrophins-induced neuroprotection are not well understood. However, there are reasons to believe that neurotrophins may neutralize several neurotoxins released form neurons, glia, and endothelial cells following injury, which could adversely affect spinal cord functions (2,5,6,11,19). Thus, a suitable combination of neurotrophins that improves neural, glial, and endothelial cell functions will certainly improve the spinal cord cell and tissue structures and thus attenuate functional deficits. Obviously, a reduction in the breakdown of the BSCB by neurotrophins is in line with this idea (4). A reduction in BSCB permeability to proteins will attenuate vasogenic edema formation and thus reduce cell injury (16–18).
Further studies in our laboratory are in progress to identify the expression of various neurotrophin receptors in spinal cord injury at different time intervals after trauma that could shed additional light on the possible mechanisms of neurotrophins-induced neuroprotection in SCI.
Conflict of interest statement HSS has no conflict of interest with any organizations mentioned below.
References
Elliott KAC, Jasper H (1949) Measurement of experimentally induced brain swelling and shrinkage. Am J Physiol 157:122–129
Gibbons A, Wreford N, Pankhurst J, Bailey K (2005) Continuous supply of the neurotrophins BDNF and NT-3 improve chick motor neuron survival in vivo. Int J Dev Neurosci 23(4):389–396
Kaplan DR, Miller FD (2000) Neurotrophin signal transduction in the nervous system. Curr Opin Neurobiol 10:381–391
Rapoport SI (1976) Blood–brain barrier in physiology and medicine. Raven, New York, NY, pp 1–316
Schwab ME, Bartholdi D (1996) Degeneration and regeneration of axons in the lesioned spinal cord. Physiol Rev 76(2):319–370
Schwab JM, Brechtel K, Mueller CA, Failli V, Kaps HP, Tuli SK, Schluesener HJ (2006) Experimental strategies to promote spinal cord regeneration – an integrative perspective. Prog Neurobiol Feb 78(2):91–116. Review
Sharma HS (2005) Neuroprotective effects of neurotrophins and melanocortins in spinal cord injury: an experimental study in the rat using pharmacological and morphological approaches. Ann N Y Acad Sci Aug 1053:407–421
Sharma HS (2003) Neurotrophic factors attenuate microvascular permeability disturbances and axonal injury following trauma to the rat spinal cord. Acta Neurochir Suppl 86:383–388
Sharma HS (2005) Pathophysiology of blood–spinal cord barrier in traumatic injury and repair. Curr Pharm Des 11(11):1353–1389. Review
Sharma HS (2005) Neuroprotective effects of neurotrophins and melanocortins in spinal cord injury: an experimental study in the rat using pharmacological and morphological approaches. Ann N Y Acad Sci Aug 1053:407–421
Sharma HS (2004) Pathophysiology of the blood–spinal cord barrier in traumatic injury. In: Sharma HS, Westman J (eds) The blood–spinal cord and brain barriers in health and disease. Elsevier Academic, San Diego, pp 437–518
Sharma HS (2007) Neurotrophic factors in combination: a possible new therapeutic strategy to influence pathophysiology of spinal cord injury and repair mechanisms. Curr Pharm Des 13(18):1841–1874
Sharma HS (2007) A select combination of neurotrophins enhances neuroprotection and functional recovery following spinal cord injury. Ann NY Acad Sci Dec 1122:95–111
Sharma HS (2006) Post-traumatic application of brain-derived neurotrophic factor and glia-derived neurotrophic factor on the rat spinal cord enhances neuroprotection and improves motor function. Acta Neurochir Suppl 96:329–334
Sharma HS, Olsson Y (1990) Edema formation and cellular alterations following spinal cord injury in the rat and their modification with p-chlorophenylalanine. Acta Neuropathol (Berl) 79(6):604–610
Sharma HS, Olsson Y, Pearsson S, Nyberg F (1995) Trauma induced opening of the blood–spinal cord barrier is reduced by indomethacin, an inhibitor of prostaglandin synthesis. Experimental observations in the rat using 131I-sodium, Evans blue and lanthanum as tracers. Restor Neurol Neurosci 7:207–215
Sharma HS, Olsson Y, Cervós-Navarro J (1993) Early perifocal cell changes and edema in traumatic injury of the spinal cord are reduced by indomethacin, an inhibitor of prostaglandin synthesis. Experimental study in the rat. Acta Neuropathol 85(2):145–153
Sharma HS (2008) New perspectives for the treatment options in spinal cord injury. Expert Opin Pharmacother (16):2773–800. Review.
Tobias CA, Han SS, Shumsky JS, Kim D, Tumolo M, Dhoot NO, Wheatley MA, Fischer I, Tessler A, Murray M (2005) Alginate encapsulated BDNF-producing fibroblast grafts permit recovery of function after spinal cord injury in the absence of immune suppression. J Neurotrauma Jan 22(1):138–156
Acknowledgements
This investigation was supported by grants from Swedish Medical Research Council (2710 HSS), Stockholm, Sweden; Göran Gustafsson Foundation, Stockholm, Sweden; National Brain Research Centre, Sweden; Alexander von Humboldt Foundation, Bonn, Germany; Astra-Zeneca, Mölndal, Sweden; Acure Pharma, Sweden; IPSEN-Medical, Paris, France; The University Grants Commission, New Delhi, India; The Indian Medical Research Council, New Delhi, India; Ministry of Science & Technology, Govt. of India, New Delhi. The Secretarial help of Aruna Sharma and the Graphic assistance of Suraj Sharma are highly appreciated with thanks. The author (HSS) has no conflict of interest with any Granting authorities mentioned above.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2010 Springer-Verlag/Wien
About this paper
Cite this paper
Sharma, H.S. (2010). Selected Combination of Neurotrophins Potentiate Neuroprotection and Functional Recovery Following Spinal Cord Injury in the Rat. In: Czernicki, Z., Baethmann, A., Ito, U., Katayama, Y., Kuroiwa, T., Mendelow, D. (eds) Brain Edema XIV. Acta Neurochirurgica Supplementum, vol 106. Springer, Vienna. https://doi.org/10.1007/978-3-211-98811-4_55
Download citation
DOI: https://doi.org/10.1007/978-3-211-98811-4_55
Published:
Publisher Name: Springer, Vienna
Print ISBN: 978-3-211-98758-2
Online ISBN: 978-3-211-98811-4
eBook Packages: MedicineMedicine (R0)