Abstract
Introduction
The reversible nature of deep brain stimulation (DBS) brought renewed interest on surgery to medically intractable mental illnesses. The explosion of anatomical and functional imaging has allowed the development of new potential targets and the understanding of historical targets.
Methods
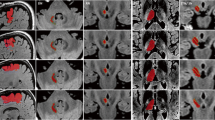
Fifteen patients undergoing stereotactic surgery for movement disorders, at UCLAs interventional MRI operating-room, were studied with fiber tracking. Stereotactic targets and fiber tracking were determined on MRIs using the Schaltenbrand-Wahren atlas for definition in the iPlan software. Cingulate, subcaudate, BA25/CgWM, amygdala, posterior hypothalamus, orbitofrontal cortex, nucleus accumbens, anterior limb of the internal capsule and dorsomedial thalamus were studied. DTI parameters used ranged from 10 to 20 mm for voxel size in the x/y/z planes, fiber length was kept constant at 36 mm, and fractional anisotropy (FA) threshold varied from 0.20 to 0.25.
Results
Reliable interconnectivity of targets were determined with DTI and related to PET imaging. Mental illness targets were observed with functional and fiber tract maps. This confirmation yields reliability to DTI imaging in order to determine novel targets and enhance the understanding of areas not well understood.
Conclusions
Currently available imaging techniques, the reversibility of DBS to modulate targets promises to bring a brighter future for surgery of mental illness.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Brody AL, Saxena S, Silverman DHS, Alborzian S, Fairbanks LA, Phelps ME, Huang SC, Wu HM, Maidment K, Baxter LR (1999) Brain metabolic changes in major depressive disorder from pre-to post-treatment with paroxetine. Psychiat Res Neuroim 91: 127–139
Buchsbaum MS, Joseph W, Siegel BV, Hackett E, Trenary M, Abel L, Reynolds C (1997) Effect of sertraline on regional metabolic rate in patients with affective disorder. Biol Psychiat 41: 15–22
Cummings JL (December 1995) Anatomic and behavioral aspects of frontal-subcortical circuits. Ann New York Acad Sci 769: 1–14
DaSilva AFM, Tuch DS, Wiegell MR, Hadjikhani N (2003) A primer on diffusion tensor imaging of anatomical substructures. Neurosurg Focus 15: 4
DeSalles AAF, Frighetto L, Behnke E, Sinha S, Tseng L, Torres R, Lee M, Cabatan-Awang C, Frysinger R (2004) Functional neuro-surgery in the MRI environment. Minim Invasive Neurosurg 47: 284–289
Driessen M, Herrmann J, Stahl K, Zwaan M, Meier S, Hill A, Osterheider M, Petersen D (2000) Magnetic resonance imaging volumes of the hippocampus and the amygdala in women with borderline personality disorder and early traumatization. Am Med Assoc 57: 1115–1122
Emery NJ, Capitanio JP, Mason WA, Machado CJ, Mendoza SP, Amaral DG (2001) The effects of bilateral lesions of the amygdala on dyadic social interactions in rhesus monkeys (Macaca mulatta). Behav Neurosci 115: 515–544
Gregg TR, Siegel A (2001) Brain structures and neurotransmitters regulating aggression in cats: implications for human aggression. Prog Neuropsychopharmacol Biol Psychiatry 25: 91–140
Groenewegen HJ, Mulder AB, Beijer AVJ, Wright CI, Lopes Da Silva FH, Pennartz CM A (1999) Hippocampal and amygdaloid interactions in the nucleus accumbens. Psychobiology (Austin, TX) 27: 149–164
Kier EL, Staib LH, Davis LM, Bronen RA (2004) MR Imaging of the temporal stem: anatomic dissection tractography of the uncinate fasciculus, inferior occipitofrontal fasciculus, and meyer’s loop of the optic radiation. Am J Neuroradiol 25: 677–691
Kumakura Y, Cumming P, Vernaleken I, Buchholz HG, Siessmeier T, Heinz A, Kienast T, Bartenstein P, Grunder G (2007) Elevated [18F] fluorodopamine turnover in brain of patients with schizo-phrenia: an [18F] Fluorodopa/positron emission tomography study. J Neurosci 27: 8080
Lee MWY, DeSalles AAF, Frighetto L, Torres R, Behnke E, Bronstein JM (2005) Deep brain stimulation in intraoperative MRI Environment-comparison of imaging techniques and electrode fixation methods. Minim Invasive Neurosurg 48: 1–6
Lindström LH, Gefvert O, Hagberg G, Lundberg T, Bergström M, Hartvig P, Langström B (1999) Increased dopamine synthesis rate in medial prefrontal cortex and striatum in schizophrenia indicated by L-G0-11C) DOPA and PET. Biol Psychiat 46: 681–688
Mayberg HS, Lozano AM, Voon V, McNeely HE, Seminowicz D, Hamani C, Schwalb JM, Kennedy SH (2005) Deep brain stimulation for treatment-resistant depression. Neuron 45: 651–660
McGowan S, Lawrence AD, Sales T, Quested D, Grasby P (2004) Presynaptic dopaminergic dysfunction in schizophrenia a positron emission tomographic [18F] Fluorodopa study. Am Med Assoc 61: 134–142
Meyer-Lindenberg A, Miletich RS, Kohn PD, Esposito G, Carson RE, Quarantelli M, Weinberger DR, Berman KF (2002) Reduced prefrontal activity predicts exaggerated striatal dopaminergic function in schizophrenia. Nature Neurosci 5: 267–271
New AS, Hazlett EA, Buchsbaum MS, Goodman M, Mitelman SA, Newmark R, Trisdorfer R, Haznedar MM, Koenigsberg HW, Flory J (2007) Amygdala-prefrontal disconnection in borderline personality disorder. Neuropsychopharmacology 32: 1629–1640
Ögren K, Sandlund M (2005) Psychosurgery in Sweden 1944-1964. J Hist Neurosci 14: 353–367
Rusch N, vanElst LT, Ludaescher P, Wilke M, Huppertz HJ, Thiel T, Schmahl C, Bohus M, Lieb K, Hesslinger B (2003) A voxel-based morphometric MRI study in female patients with borderline personality disorder. Neuroimage 20: 385–392
Soloff PH, Meltzer CC, Becker C, Greer PJ, Kelly TM, Constantine D (2003) Impulsivity and prefrontal hypometabolism in borderline personality disorder. Psychiat Res Neuroim 123: 153–163
Szabo K, Klein J, Voets N, Gass A, Hennerici MG, Johansen-Berg H, Matthews PM (2006) Probabilistic tractography of cortical connectivity of the human hippocampus and the anatomy of hippocampal-thalamic connections. Akt Neurol: 33
Wang Z, Hulihan TJ, Insel TR (1997) Sexual and social experience is associated with different patterns of behavior and neural activation in male prairie voles. Brain Res 767: 321–332
Wichmann T, DeLong MR (2006) Deep brain stimulation for neurologic and neuropsychiatric disorders. Neuron 52: 197–204
Wohlfahrt S (1950) III. Introductory lecture (2) anatamy, operative technic, pathological research. Acta Psychiatrica Scandinavica 25: 68–80
Yucel M, Harrison BJ, Wood SJ, Fornito A, Wellard RM, Pujol J, Clarke K, Phillips ML, Kyrios M, Velakoulis D (2007) Functional and biochemical alterations of the medial frontal cortex in obsessive-compulsive disorder. Arch Gen Psychiat 64: 946–955
Zetzsche T, Frodl T, Preuss UW, Schmitt G, Seifert D, Leinsinger G, Born C, Reiser M, Möller HJ, Meisenzahl EM (2006) Amygdala volume and depressive symptoms in patients with borderline personality disorder. Biol Psychiat 60: 302–310
Zipursky RB, Meyer JH, Verhoeff NP (2007) PET and SPECT imaging in psychiatric disorders. Can J Psychiatry 52: 146–157
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Springer-Verlag
About this paper
Cite this paper
Sedrak, M. et al. (2008). The role of modern imaging modalities on deep brain stimulation targeting for mental illness. In: Chiu, WT., et al. Reconstructive Neurosurgery. Acta Neurochirurgica Supplementum, vol 101. Springer, Vienna. https://doi.org/10.1007/978-3-211-78205-7_1
Download citation
DOI: https://doi.org/10.1007/978-3-211-78205-7_1
Publisher Name: Springer, Vienna
Print ISBN: 978-3-211-78204-0
Online ISBN: 978-3-211-78205-7
eBook Packages: MedicineMedicine (R0)