Abstract
Transcranial Doppler is a non-invasive ultrasound technique at the patient’s bedside, measuring cerebral blood flow velocities (CBFVs) of basal cerebral arteries and veins, based on pulsed-wave Doppler by low-frequency probe (1.5–2 MHz). TCD is a non-imaging method to assess intracranial blood flow velocity, which is the main difference from TCCS technique. The intracranial vessels are insonated through the skull acoustic windows: Transtemporal, transorbital, transoccipital, or transforaminal and submandibular.
The spectral Doppler (sonogram) obtained shows the blood flow velocity spectrum where the envelope wave defines the values of cerebral blood flow velocities (CBFVs) and derived hemodynamic indexes; peak systolic velocity (PSV), end-diastolic velocity (EDV), mean flow velocity (MFV), pulsatility index (PI), resistance index (RI), and Lindegaard ratio (LR). The flow toward the transducer is displayed as positive velocity wave and the flow away from it as negative velocity wave. The analysis of the different morphologies of the spectral Doppler wave allows estimating the patterns of cerebral hemodynamics (hyperemia, hypoperfusion, etc.), during different critical scenarios.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Transcranial Doppler
- pulsed Doppler
- M-Mode
- transcranial acoustic windows
- middle cerebral artery
- pulsatility index
- cerebral blood flow velocity
- anterior cerebral artery
- posterior cerebral artery
- vertebral arteries
- basilar artery
-
1.
TCD is a non-invasive ultrasound technique at the patient’s bedside, measuring cerebral blood flow velocities (CBFV) of basal cerebral arteries.
-
2.
It measures flow velocities and estimates changes in cerebral blood flow but does not measure cerebral blood flow in absolute values.
-
3.
It uses areas or natural orifices of the skull as acoustic windows to insonate intracerebral hemodynamic changes.
-
4.
The most used acoustic window to evaluate global cerebral hemodynamics is the transtemporal window through the middle cerebral artery (MCA) insonation.
-
5.
TCD, through an envelope wave, determines the three flow velocities and the derived hemodynamic indexes: PSV, EDV, MFV, PI, RI, and LR.
-
6.
In normal conditions, the intracranial blood flow circulates only in one direction, accelerating during systole and decelerating during diastole, without ever stopping or reversing its direction.
-
7.
The cerebral arteries’ blood flow is less pulsatile than in the rest of the arteries of the body, given the important collateral circulation of the system (circle of Willis) that maintains a low resistance pattern.
-
8.
It is essential to repeat the TCD examinations in order to measure the hemodynamic changes in real time and to assess the trends of the parameters evaluated and/or the response to an established therapeutic trial.
1 Introduction
Transcranial Doppler (TCD) is a non-invasive ultrasound method that measures cerebral blood flow velocities (CBFVs) in the basal cerebral arteries (circle of Willis), without transferring the patient out of the ICU.
It measures the circulatory velocity of the blood. It does not measure cerebral blood flow (C BF). However, under certain conditions, changes in CBFV are proportional to changes in CBF.
In intensive care unit (ICU), it can be used (a) uniquely, (b) repeatedly, observing the trend of changes in CBFVs and pulsatility, (c) continuously alone, or (d) combined with other variables, for example, arterial blood pressure and intracranial pressure (ICP), constituting multimodal neuromonitoring.
2 TCD: Spectral Wave
The screen of the TCD equipment shows the circulation velocity in time. The circulatory velocity of a complete arterial pulse cycle is called sonogram.
At each instant the blood particle velocities can be seen. Since the ultrasound is reflected by multiple moving blood particles in a certain segment of the artery (the depth and size of this segment are chosen by the operator), a “spectrum” of velocities is generated [1]. This spectrum is different if the flow is laminar or turbulent (Fig. 12.1). In arterial segments without stenosis or bifurcations, the flow is laminar.
Difference between laminar flow spectrum (upper) and turbulent flow spectrum (lower). In the first one most of the circulating particles are of high velocity and therefore form a thick line near the maximum velocity in each instant; the different colors translate more or less particles concentration. A lower triangle remains where there is a spectral window, indicated by a red circle. In turbulent flow, which can be seen in bifurcations, or in severe stenoses, the angles of intonation are formed between the direction of the particles rotating in turbulent form and the direction of the ultrasound emitted by the transducer are large, so they look as low velocities. The vascular murmur is marked with a circle, evidencing the turbulence, which has a special sound, like that of a ¨seagull's song¨, superimposed on the normal sound
By taking only the maximum velocities at each point in this spectrum, the TCD equipment outlines an “envelope” wave. The envelope wave, therefore, shows the values of the particles moving at the highest velocity within the arterial vessel. The values of the envelope wave thus generated are those analyzed and displayed by the ultrasound equipment. In the graph of the envelope wave of each arterial pulse, three variables are defined: peak systolic velocity (PSV), mean flow velocity (MFV), and end-diastolic velocity (EDV) (Fig. 12.2).
On the left you can see a Doppler spectrum velocities (sonogram) in the first four cycles. In the following cycles the spectrum has been erased and only the envelope wave is seen. The arrows show the point where the PSV, the EDV and the MFV. You can also see the graphical calculation of the average velocity which in this case coincides with the value calculated by the equipment since the envelope wave correctly follows the maximum values of the Doppler spectrum. The average velocity calculated graphically is the one that marks a horizontal line that divides the envelope wave in two parts of similar area as seen in the figure
Spectrum and envelope wave characteristics: Under normal conditions, cerebral blood flow moves in a single direction within the vessel, slows down in the diastole, and accelerates when a new blood pulse arrives. It does not stop or reverse its direction. The final diastolic rate is the lowest of each pulse, greater than zero.
By convention, the flow toward the transducer is given a positive value and the flow away from it is given a negative value. In the arterial bifurcations, a bidirectional flow is seen.
A relatively constant diameter of the studied vessel is assumed, and thus the changes of CBFV are directly proportional to the changes of CBF. For this, the transducer must remain fixed without changes in its angulation.
TCD measures CBFV and estimates changes in CBF but does not measure CBF in absolute values .
3 TCD: Identification of Vessels
Transcranial Doppler identifies the different arteries according to
-
1.
Characteristics of the Doppler signal.
-
2.
Topography of the Doppler signal (acoustic window, direction of the ultrasound beam, and depth).
-
3.
Hemodynamic response to maneuvers.
-
1.
Characteristics of the Doppler Signal:
By convention, if the flow is directed toward the transducer, it is shown as positive spectrum wave; if it is directed in the opposite direction, it is shown as negative spectrum wave. If a bifurcation is insonated, it is displayed as a bidirectional spectrum wave.
-
2.
Topography:
Different acoustic windows allow to insonate different arteries of the circle of Willis. From each acoustic window, the direction of the ultrasound beam from transducer allows the identification of the different basal cerebral arteries, as well as the depth at which each of them is located.
-
3.
M-Mode: Power Motion Mode Doppler
Transcranial Doppler emits pulsed ultrasound. This means that it obtains information only from a small volume of the intracranial space whose depth is permanently defined throughout the examination. This small volume is called the “sample volume.” The operator must, therefore, know which vessel he is going to look for, through which window, and at what depth he is going to look for it.
The operator must also know the insonation angle that he must give to the transducer. This generated the concept that the technique is operator dependent. The introduction of the M-mode (PMD) facilitates the examination, even for inexperienced operators, since it makes it possible to simultaneously see the intensity and direction of the flow along 6 cm or more of the intracranial space that the ultrasound beams cross.
When using the PMD, the screen of TCD machine is divided into two sectors:
-
1.
In the main sector (generally upper), you can see the sonogram of the vessel under examination (at a predetermined depth), that is, the full characteristics (spectrum, envelope wave, PSV, EDV, MFV, PI) of the flow velocity at the chosen point of the artery under study.
-
2.
In the M-mode sector screen (generally lower), the different flows that appear along the path (3–6 cm or more, configurable by the examiner) of the ultrasound beam in that direction are shown simultaneously (Fig. 12.3). This sector graphs the flows along the beam path, showing in the different depths, if there is any flow or not, and in case there is, in red or blue color if it is directed toward or moves away from the transducer, respectively.
Over time these segments are seen as red or blue horizontal bands crossing the screen. The display allows to choose any point of interest in this sector, so that the main screen shows the sonogram of the flow at the chosen point and the characteristics of both the spectrum and its envelope waves (PSV, EDV, MFV, PI, etc.).
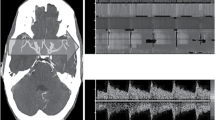
The M mode or "Power Mode". The two panels that appear on the computer screen when using this mode are shown. The insonation is through transtemporal acoustic window. In the upper panel you can see the Doppler spectrum, where equipment has delineated the spectral Doppler envelope. In this case it is a laminar blood flow. The lower panel shows the M-mode, which displays all the flows in the path of the ultrasonic beam, their depths in relation to the surface of the transducer and their directions (towards the transducer in red and away from it in blue). The scale on the left shows at what depth, in mm, these flows are seen. Between 40 and 60 mm there is a flow that approaches the transducer, which corresponds to the middle cerebral artery (MCA). In this red band there is a dotted line, which shows the depth at which the artery is being observed, in this case about 48 mm; at this depth corresponds the spectrum seen in the upper panel. Any point can be chosen, so that the volume of the sample will inspect and show on the upper display the spectrum and the corresponding envelope with its PSV, MFV, EDV and PI. Between 70 and 75 mm a flow is seen that moves away, corresponding to the anterior cerebral artery (ACA). The carotid bifurcation is not clearly seen in this image, but at about 62 mm a blue band can be seen at times, so it can be thought that this is the bifurcation and that by slightly angulating the transducer it could be clearly found
Normal values of CBFVs vary according to (a) insonated vessel and (b) physiological variables. CBFV in the cerebral arteries varies with age, where they are greatest in childhood and decrease with age. Normal values of CBFV in healthy adults for three age groups are shown in Table 12.1.
To have a global idea of the values in mind, the following scheme is useful: average velocity in cm/s (±10): ACM 60, ACA 50, ACP 40, and posterior circulation sector 35.
Other important physiological variables that influence the CBFV in the cerebral arteries are blood viscosity (one of whose main determinants is hematocrit), temperature, PaCO2, and heart rate.
The trends of values are more important than a single CBFV value.
4 TCD: Clinical Utility
TCD is known as the “Stethoscope of the brain.” The use of the TCD is directed to the detection and follow-up of cerebral hemodynamics alterations in the patients with neurological injury: acute ischemic stroke (AIS), subarachnoid hemorrhages (SAH), intracerebral hematoma, traumatic brain injury (TBI), and other pathologies such as central nervous system (CNS) infections, cerebral vasculitis, etc. It is especially useful in neurocritically ill patients whose state of consciousness is altered (secondary to different injuries and/or sedation/analgesia/neuromuscular block requirements), and complete neurological examination is not possible. Unlike the TCCS, it does not show anatomical images (B-mode) but only the blood flow velocities in the basal cerebral arteries.
The alterations of cerebral hemodynamics, monitored by TCD, are generally secondary to
-
1.
Increase of ICP.
-
2.
Vasospasm.
-
3.
Hyperemia.
-
4.
Alterations in cerebrovascular reactivity.
-
5.
Coexistence of two or more of the previously mentioned points.
When the interest is to assess whether there are global alterations of the cerebral hemodynamics, we must insonate the anterior circulation; middle cerebral artery (MCA), anterior cerebral artery (ACA), and the posterior circulation; and basilar artery (BA) and vertebral arteries (VA). When the interest of the study is to detect a segmental alteration (vasospasm), the assessment must be global initially and then detailed, along each vessel.
5 TCD: Frequent Uses in Intensive Care Unit
-
1.
Detect and estimate changes of intracranial pressure (ICP) [2,3,4,5,6,7,8,9,10,11].
-
2.
Diagnose cerebral circulatory arrest [12,13,14,15,16,17,18], collaborating in the diagnosis of brain death.
-
3.
Diagnosis and monitoring of cerebral artery vasospasm in the clinical evolution of SAH [19, 20], TBI [21, 22], and CNS infections [23,24,25,26,27].
-
4.
In AIS: Monitor arterial recanalization when thrombolytics are performed [28]. To increase the action of these drugs (sonothrombolysis) [29, 30]. To evaluate the presence of microembolism signals and the presence of right–left shunt in cryptogenic AIS.
-
5.
To evaluate the state of cerebral autoregulation (CA) [31], by means of integrated neuromonitoring through mean flow reactive index (Mx).
-
6.
Cerebral vasomotor reactivity to CO2. Time constant of cerebral circulation and critical closing pressure.
6 TCD: Technique
6.1 Position of the Patient and Examiner
Frequently , the neurocritical patient is in dorsal decubitus, with the head elevated about 30°, aligned with the trunk. It is not necessary to modify the patient’s position. The examiner can be placed at the patient’s bedside, but given the large number of catheters and devices that are frequently found around critical patients, it is usually placed at the side of the bed.
The different vessels are insonated through the different acoustic windows (Fig. 12.4). Acoustic windows are areas of the skull that are more permeable to ultrasound beams, because they are thinner or because they are natural orifices. The transducer should be placed by exerting moderate pressure (except when using transorbital window), with abundant gel to ensure proper coupling between the transducer and the skin. It must be known in which direction the ultrasound beam will be emitted, and at what depth each artery will be searched. In this way, the depth will be fixed in advance.
The circle of Willis and their relationships to the different acoustic windows. In (a) it is shown the whole circle of Willis, its main branches and the vessels that form it, the transducer in transtemporal acoustic window and the different intonation angles that must be given to it (in this plane) to find the lowest angle of insonation with the MCA and. In (b) the transoccipital acoustic window and its relationship with the blood flow in the posterior circulation can be observed. In (c) the anterior sector is seen in a coronal view and, in (d) the transorbital acoustic window and its relationship with the vessels of the carotid siphon and OA is observed in a sagittal section
The exploration will begin more superficially and will be deepened by a few millimeters, optimizing the angle of insonation at each point to find the spectrum whose blood flow velocity is maximum, which is the one that will coincide with the minimum angle of insonation (angle between the ultrasonic beam and the direction of blood flow at the point of insonation).
6.2 Transtemporal Acoustic Window
TCD assessment begins through the transtemporal acoustic window to insonate middle cerebral artery (MCA). The transtemporal acoustic window extends above the zygomatic arch, in front of the swallow, and behind the lateral corner of the homolateral eye. It has a projection that can be anterior, medial, and posterior. Each patient is different, so the best approach should always be sought in each case.
6.2.1 Anterior Circulation
6.2.1.1 Middle Cerebral Artery (MCA)
-
1.
Acoustic Window: Transtemporal
-
2.
Depth: (M1 Segment)
The depth should be between 45 and 60–65 mm and followed along its entire length.
-
3.
Acoustic Window: Transtemporal
-
4.
Depth: (M2 Segment)
In certain cases, it is important to insonate more superficially, from 45 mm to 30 mm (e.g., distal vasospasm).
In all cases, the transducer will be slightly rotated to apical (about 10°), caudal (about 10°), occipital, and frontal direction [7], independent of the sector of the transtemporal window that the operator is located to record Doppler signals.
As the flow of the MCA is toward the transducer, it will be seen as a positive sonogram.
At 65 mm depth of insonation is the carotid bifurcation, where the MCA and the anterior cerebral artery (ACA) originate. The carotid bifurcation is insonated as a bidirectional blood flow, with a simultaneous positive and negative sonogram.
At greater depth of insonation, the flow in the ACA (A1 segment) is seen as a sonogram with negative blood flow velocities that originates at 65 mm depth (in the carotid bifurcation) and extends about 5–10 mm inward and forward, with values always negative .
6.2.2 Posterior Circulation
6.2.2.1 Posterior Cerebral Artery (PCA)
-
1.
Acoustic Window: Transtemporal
-
2.
Depth:
The transducer should be rotated slightly to occipital direction, between 70 and 90 mm depths. The blood flow is directed toward the transducer, where it will be seen as a positive sonogram.
To differentiate whether a positive spectrum found in the transtemporal window is MCA or PCA, we must consider the following:
-
1.
Direction of the probe: PCA is found with transducer rotated to occipital direction.
-
2.
Depth: MCA is usually less than 65 mm, and PCA is deeper.
-
3.
Carotid compression maneuvers: Homolateral compression will generate decrease in sonogram blood flow velocity if it is MCA and increase if it is PCA, in which the blood flow in this artery originates in the posterior sector. This depends on the patient having a functioning posterior communicating artery. If the circle of Willis does not compensate, carotid compression will not generate changes if it is PCA .
6.3 Submandibular Acoustic Window
6.3.1 Internal Carotid Artery (ICA—Extracranial Portion)
-
1.
Transducer: 2 MHz
-
2.
Depth: 40–50 mm
The blood flow is away from the transducer (negative spectrum wave).
The velocity in this artery is approximately 30 cm/s.
As a complement to the insonation of MCA, mainly when the velocities are high, this velocity is compared with extracranial internal carotid artery (ICA) velocity, to study the Lindegaard ratio (vasospasm vs. hyperemia). The transducer is placed below the angle of the mandible, parallel to, and behind the upright branch.
6.4 Transoccipital Acoustic Window
6.4.1 Posterior Circulation
-
1.
Vessels: Basilar artery (BA) and vertebral artery (VA)
-
2.
Acoustic Window: Foramen magnum
The transducer is positioned under the external occipital protuberance and is directed toward the nasion.
-
3.
Depth: 80–100 mm (BA)
Follow the vertebral arteries to their central confluence with the BA.
-
4.
Depth: 60 mm (VA)
Therefore, with an initial depth of 60 mm, angulate the transducer to the right and left of the midline until the signals from the vertebral arteries are found. If no Doppler signal is found, the transducer can be moved slightly sideways to optimize the window through retromastoid position. The identification of right and left vertebral arteries will be based on the direction of the ultrasonic beam and landmarks between the observed vessels.
The patient can be positioned in a dorsal decubitus with a pillow under the occiput and the head flexed to the side opposite the operator, or (without absolute contraindications for cervical flexion) the patient can be lateralized very carefully to access the acoustic window .
6.5 Transorbital Acoustic Window
-
1.
Vessels: Ophthalmic artery (OA) and carotid siphon
-
2.
Depth: 40–60 mm (OA)
The OA flow is directed toward the transducer (positive spectral Doppler).
Unlike basal cerebral arteries, OA is an extracranial artery with high resistance flow pattern. Homolateral carotid compression results in a decrease of the Doppler signal. This artery can act as collateral in case of significant carotid disease. In this clinical context, a negative flow is visualized since the direction of blood flow away from the transducer.
-
3.
Depth: 60–80 mm (Carotid siphon)
The flow can be directed toward the transductor or away from it, according to the portion of the carotid siphon that is insonated.
The ultrasound intensity should be lowered to 10% to minimize eye exposure (prevent cavitation effects). The transducer, using abundant gel, is placed on the closed upper eyelid, a few millimeters inward from the middle of the eyelid. No pressure should be exerted on the eye, and the exposure should be for a short time.
Figure 12.5 shows a scheme of basal cerebral arteries studied by TCD and the depths at which they are found each .
Scheme of the basal cerebral arteries being studied with TCD, their approximate depths (mm), and CBF direction. Anterior circulation; ICA (terminal segment); Bif carotid bifurcation, MCA middle cerebral artery. (M1: M1 segment and M2 segment). ACA anterior cerebral artery. AComA anterior communicating artery, OA ophthalmic artery. In the posterior circulation; VA vertebral arteries, BA basilar artery. Connecting the anterior and posterior sector we see the PComA posterior communicating artery. Communicating arteries do not have flow in normal conditions, they do when they function as collaterals
6.6 Blood Flow Velocities and Hemodynamic Indexes
6.6.1 Interpretation of Doppler Spectrum Wave (Sonogram)
The PSV corresponds to the highest measuring of the Doppler spectrum wave (sonogram). It is related to the left ventricular contractility. The EDV corresponds to the lowest point of the sonogram, before starting a new cardiac cycle. It makes it possible to infer the blood flow velocity output, related to cerebrovascular resistances (CVR). In comparison with the circulatory flow velocities of the extracranial vessels, this CBFV output in the basal cerebral arteries is high. It evidenced a characteristic of the cerebral circulation: it is a system of low resistance. The MFV is calculated mathematically as the average on time of the CBFVs during each Doppler spectrum wave. It can also be calculated graphically in the Doppler sonogram .
6.6.2 Pulsatility Index
There are two hemodynamic indices: the pulsatility index (PI) or Gosling’s index and the resistance index (RI) or Pourcelot’s index.
The PI is the most widely used. It is calculated by the following formula (Eq. 12.1):
*Normal value: 0.6–1.2
The higher the differential velocity (high PSV and low EDV), the higher the PI value [7]. In general, it reflects a higher (high PI) or lower (low PI) resistance to the cerebral blood flow, and it can be modified by (1) conditions specific to the intracerebral arteries (small resistance vessels), (2) change of cerebral parenchyma compliance, and/or (3) changes in cerebral perfusion pressure. PI > 1.2 (integrating clinical evolution of the patient with PI absolute and trends values) in patients with acute neurological injury and risk of developing intracranial hypertension, should always alert the clinician. Repeated and bilateral neurological monitoring with TCD is crucial.
The other hemodynamic index that can be measured by TCD is RI (less used). The RI is calculated with the following formula (Eq. 12.2):
7 The Different Patterns of Cerebral Blood Flow
7.1 High-Velocity Pattern
It is seen mainly in vasospasm or hyperemia.
7.2 Low-Velocity Pattern
This pattern refers to cerebral hypoperfusion.
7.3 High Resistance Pattern
In general, it coexists with low CBFV. A PI > 1.2 should lead to suspicion of intracranial hypertension in critically ill patients with acute neurological injury (Fig. 12.6).
7.4 Cerebral Circulatory Arrest Pattern
Reverberant flow and systolic spikes are seen diffusely in the patient progressing to brain death.
All these patterns will be described in depth in the corresponding chapters.
8 TCD: Other Clinical Uses
Among the advantages that TCD has over other monitoring methods, it is the possibility of performing prolonged neurological monitoring, constituting a very useful tool in multimodal neuromonitoring (MMM).
8.1 Through the MMM You Can Study the Following
8.1.1 Cerebral Vascular Reactivity
Capacity of the cerebral arteriolar bed to respond to different stimuli with changes in CVR. Depending on the stimulus, cerebrovascular reactivity can be classified as follows:
-
1.
Reactivity to CO2.
-
2.
Metabolic reactivity.
-
3.
Reactivity to drugs.
-
4.
Cerebral autoregulation.
9 TCD: Limitations
The most important limitations of TCD are (a) operator dependent and (b) 10–15% of patients do not have a good acoustic window.
10 Conclusion
Transcranial Doppler is a useful monitoring tool for assessing cerebral hemodynamics in the critically ill patient. It allows suspecting or ruling out serious alterations that need urgent management, at the patient’s bedside, in a non-invasive way. Unlike imaging studies, which give an anatomical evaluation that provides little information about functionality, TCD can be done as many times as desired (repeated or continuous way), with excellent temporal resolution.
It is complementary to the anatomical evaluation. The TCD protocol in ICU is different from that performed in the neurological laboratory. The study can be done to evaluate hemodynamics globally and/or segmental changes (vasospasm, steno-occlusion, etc.). In this case, it should be remembered that there may be intracranial pressure gradients and, therefore, the arteries of the anterior and posterior circulations (bilaterally) should be assessed through the four acoustic windows in order to evaluate CBFV, hemodynamic indexes, and right/left CBF asymmetry.
References
Hakimi R, Alexandrov AV, Garami Z. Neuro-ultrasonography. Neurol Clin. 2020;38(1):215–29.
Nornes H, Aaslid R, Lindegaard KF. Intracranial pulse pressure dynamics in patients with intracranial hypertension. Acta Neurochir. 1977;38:177–86.
Hassler W, Steinmetz H, Gawlowski J. Transcranial Doppler ultrasonography in raised intracranial pressure and in intracranial circulatory arrest. J Neurosurg. 1988;68:745–51.
Melon E. Indications for monitoring intracranial pressure. Ann Fr Anesth Reanim. 1997;16:415–9.
Sidi A, Messinger G, Mahla ME. Transcranial Doppler monitoring compared with invasive monitoring of intracranial pressure during acute intracranial hypertension. J Clin Monit Comput. 1999;15:185–95.
Schmidt B, Czosnyka M, Klingelhöfer J. Clinical applications of a non-invasive ICP monitoring method. Eur J Ultrasound. 2002;16:37–45.
Reinhard M, Petrick M, Steinfurth G, Ziyeh S, Hetzel A. Acute increase in intracranial pressure revealed by transcranial Doppler sonography. J Clin Ultrasound. 2003;31:324–7.
Edouard AR, Vanhille E, Le Moigno S, Benhamou D, Mazoit JX. Non-invasive assessment of cerebral perfusion pressure in brain injured patients with moderate intracranial hypertension. Br J Anaesth. 2005;94:216–21.
Splavski B, Radanović B, Muzević D, Has B, Janculjak D, Kristek J, et al. Assessment of intra-cranial pressure after severe traumatic brain injury by transcranial Doppler ultrasonography. Brain Inj. 2006;20:1265–70.
Sharma VK, Chan BP. Comment on 'Assessment of intra-cranial pressure after severe traumatic brain injury by transcranial Doppler ultrasonography' by Splavski et al. Brain Inj. 2007;21:1093–4.
Aggarwal S, Brooks DM, Kang Y, Linden PK, Patzer JF 2nd. Noninvasive monitoring of cerebral perfusion pressure in patients with acute liver failure using transcranial doppler ultrasonography. Liver Transpl. 2008;14:1048–57.
Powers AD, Graeber MC, Smith RR. Transcranial Doppler ultrasonography in the determination of brain death. Neurosurgery. 1989;24:884–9.
Hassler W, Steinmetz H, Pirschel J. Transcranial Doppler study of intracranial circulatory arrest. J Neurosurg. 1989;71:95–201.
Werner C, Kochs E, Rau M, Schulte EJ. Transcranial Doppler sonography as a supplement in the detection of cerebral circulatory arrest. J Neurosurg Anesthesiol. 1990;2:159–65.
Ducrocq X, Hassler W, Moritake K, Newell DW, von Reutern GM, Shiogai T, et al. Consensus opinion on diagnosis of cerebral circulatory arrest using Doppler-sonography: Task Force Group on cerebral death of the Neurosonology Research Group of the World Federation of Neurology. J Neurol Sci. 1998;159:145–50.
Sloan MA, Alexandrov AV, Tegeler CH, Spencer MP, Caplan LR, Feldmann E, et al. Transcranial Doppler ultrasonography: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2004;62:1468–81.
Monteiro LM, Bollen CW, van Huffelen AC, Ackerstaff RG, Jansen NJ, van Vught AJ. Transcranial Doppler ultrasonography to confirm brain death: a meta-analysis. Intensive Care Med. 2006;32:1937–44.
Calleja S, Tembl JI, Segura T. Recommendations of the use of transcranial Doppler to determine the existence of cerebral circulatory arrest as diagnostic support of brain death. Neurologia. 2007;22:441–7.
Vora YY, Suarez-Almazor M, Steinke DE, Martin ML, Findlay JM. Role of transcranial Doppler monitoring in the diagnosis of cerebral vasospasm after subarachnoid hemorrhage. Neurosurgery. 1999;44:1237–47.
Soustiel JF, Shik V, Shreiber R, Tavor Y, Goldsher D. Basilar vasospasm diagnosis: investigation of a modified "Lindegaard Index" based on imaging studies and blood velocity measurements of the basilar artery. Stroke. 2002;33:72–7.
Martin NA, Doberstein C, Zane C, Caron MJ, Thomas K, Becker DP. Posttraumatic cerebral arterial spam: transcranial Doppler ultrasound, cerebral blood flow, and angiographic findings. J Neurosurg. 1992;77:575–583martin.
Lee JH, Martin NA, Alsina G, McArthur DL, Zaucha K, Hovda DA, et al. Hemodynamically significant cerebral vasospasm and outcome after head injury: a prospective study. J Neurosurg. 1997;87:221–33.
Müller M, Merkelbach S, Huss GP, Schimrigk K. Clinical relevance and frequency of transient stenoses of the middle and anterior cerebral arteries in bacterial meningitis. Stroke. 1995;26:1399–403.
Gupta R, Mahapatra AK, Bhatia R. Serial transcranial Doppler study in meningitis. Acta Neurochir. 1995;137:74–7.
Müller M, Merkelbach S, Hermes M, Schimrigk K. Transcranial Doppler sonography at the early stage of acute central nervous system infections in adults. Ultrasound Med Biol. 1996;22:173–8.
Müller M, Merkelbach S, Hermes M, König J, Schimrigk K. Relationship between short-term outcome and occurrence of cerebral artery stenosis in survivors of bacterial meningitis. J Neurol. 1998;245:87–92.
Kiliç T, Elmaci I, Ozek MM, Pamir MN. Utility of transcranial Doppler ultrasonography in the diagnosis and follow-up of tuberculous meningitis-related vasculopathy. Childs Nerv Syst. 2002;18:142–6.
Saqqur M, Tsivgoulis G, Molina CA, Demchuk AM, Shuaib A, Alexandrov AV, CLOTBUST Investigators. Residual flow at the site of intracranial occlusion on transcranial Doppler predicts response to intravenous thrombolysis: a multi-center study. Cerebrovasc Dis. 2009;27:5–12.
Alexandrov AV. Ultrasound enhancement of fibrinolysis. Stroke. 2009;40:S107–10.
Alexandrov AV, Köhrmann M, Soinne L, Tsivgoulis G, Barreto AD, Demchuk AM, et al. CLOTBUST-ER Trial Investigators. Safety and efficacy of sonothrombolysis for acute ischaemic stroke: a multicentre, double-blind, phase 3, randomised controlled trial. Lancet Neurol. 2019;18(4):338–47.
Martínez-Sánchez P, Tsívgoulis G, Lao A, Sharma V, Alexandrov A. Ultrasound in acute ischemic stroke. Neurologia. 2009;24:59–68.
Hennerici M, Rautenberg W, Sitzer G, Schwartz A. Transcranial Doppler ultrasound for the assessment of intracranial arterial flow velocity-Part 1. Examination technique and normal values. Surg Neurol. 1987;27:439–48.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Algorithm
Algorithm

ABCD Airway-breathing-circulation-disability, GCS Glasgow coma scale, ACA Anterior Cerebral Artery, PCA Posterior Cerebral Artery, BA Basilar Artery, VA Vertebral Artery, OA Ophthalmic artery, ICPn Noninvasive intracranial pressure, CBFVs Cerebral blood flow velocities, Mx Mean flow index, Sx systolic flow index, PRx Pressure reactivity index, CA Cerebral autoregulation, CrCP Critical closing pressure, CPP Cerebral perfusión pressure
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Puppo, C. (2022). Neurosonology in the ICU: Transcranial Doppler (TCD) Protocol. In: Rodríguez, C.N., et al. Neurosonology in Critical Care . Springer, Cham. https://doi.org/10.1007/978-3-030-81419-9_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-81419-9_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-81418-2
Online ISBN: 978-3-030-81419-9
eBook Packages: MedicineMedicine (R0)