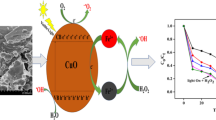
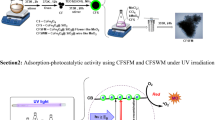
Abstract
Dye degradation by photocatalysis method has been attracting much research interest because of its wide applications in renewable energy and environmental remediation. Recently, many researchers were published about the degradation of dyes using magnetic nanoparticles as photocatalysts. This chapter aims to highlight the application of magnetic nanoparticles as photocatalysts in the degradation of organic dyes. The synthetic routes for magnetic nanoparticles such as Solvothermal, Co-precipitation, Hydrothermal, Sonochemical method, and Sol-gel methods have been summarized. Degradation of several organic dyes using magnetic nanoparticles is studied. Most of the articles focus on the degradation of methylene blue (MB), methyl orange (MO), and Rhodamine-B (RhB) dyes. There are many materials that are found to show good photocatalytic activity in the presence of ultraviolet (UV), visible light, and sunlight irradiation. Surface modifications of Iron oxide by incorporating impurity doping and composite semiconductors to improve their efficiency are reviewed. The efficiency can be controlled by optimizing various parameters such as pH, catalyst dosage, concentration of the dyes, time of irradiation, and light intensity during the reaction. The experimental conditions significantly affect the performance of photocatalyst in the photodegradation of dyes. Optimization of operating parameters and their effect on photocatalytic degradation of dyes are discussed here.
Similar content being viewed by others
References
Akbarzadeh A, Samiei M, Davaran S (2012) Magnetic nanoparticles: preparation, physical properties, and applications in biomedicine. Nanoscale Res Lett 7(1):144
Alonso J, Barandiarán JM, Fernández Barquín L, García-Arribas A (2018) Magnetic Nanoparticles, Synthesis, Properties, and Applications. Magn Nanostruct Mater:1–40
Atrak K, Ramazani A, Taghavi Fardood S (2018) A novel sol–gel synthesis and characterization of MgFe2O4@γ–Al2O3 magnetic nanoparticles using tragacanth gel and its application as a magnetically separable photocatalyst for degradation of organic dyes under visible light. J Mater Sci Mater Electron 29:6702–6710
Atrak K, Ramazani A, Fardood ST (2019a) Eco-friendly synthesis of Mg0.5Ni0.5AlxFe2-xO4 magnetic nanoparticles and study of their photocatalytic activity for degradation of direct blue 129 dye. J Photochem Photobiol A 382:111942
Atrak K, Ramazani A, Fardood ST (2019b) Green synthesis of Zn0.5Ni0.5AlFeO4 magnetic nanoparticles and investigation of their photocatalytic activity for degradation of reactive blue 21 dye. Environ Technol 41:2760–2770
Bayat S, Ghanbari D, Salavati-Niasari M (2016) Pechini synthesis of Co2SiO4 magnetic nanoparticles and its application in photo-degradation of azo dyes. J Mol Liq 220:223–231
Bishnoi S, Kumar A, Selvaraj R (2017) Facile synthesis of magnetic iron oxide nanoparticles using inedible Cynometraramiflora fruit extract waste and their photocatalytic degradation of methylene blue dye. J Mater Res Bull S:0025-5408 (17)31181-9
Boutra B, Güy N, Özacar M, Trari M (2019) Magnetically Separable MnFe2O4/TA/ZnO Nanocomposites for Photocatalytic Degradation of Congo Red Under Visible Light. J Magnetism Magn Mater:S0304-8853(19)31805-0
Cruz JC, Nascimento MA, Amaral AVH, Lima SDD, Teixeira APC, Lopes PR (2019) Synthesis and characterization of cobalt nanoparticles for application in the removal of textile dye. J Environ Manag:220–228
de Toledo LDAS, Rosseto HC, Bruschi ML (2018) Iron oxide magnetic nanoparticles as antimicrobials for therapeutics. Pharm Dev Technol 23:316–323
Fardood ST, Atrak K, Ramazani A (2017) Green synthesis using tragacanth gum and characterization of Ni–Cu–Zn ferrite nanoparticles as a magnetically separable photocatalyst for organic dyes degradation from aqueous solution under visible light. J Mater Sci Mater Electron 28:10739–10746
Feng K, Song B, Li X, Liao F, Gong J (2019) Enhanced photocatalytic performance of magnetic multi-walled carbon nanotubes/cerium dioxide nanocomposite. Ecotoxicol Environ Saf 171:587–593
Guo Y, Wang Z, Shao H, Jiang X (2013) Hydrothermal synthesis of highly fluorescent carbon nanoparticles from sodium citrate and their use for the detection of mercury ions. Carbon 52:583–589
Gupta AK, Gupta M (2005) Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 26(18):3995–4021
Hangxun X, Zeiger BW, Suslick KS (2013) Sonochemical synthesis of nanomaterials. Chem Soc Rev 42(7):2555–2567
Hasany SF, Rehman A, Jose R, Ahmed I (2012) Iron oxide magnetic nanoparticles: a short review. AIP Conf Proc 1502:298
Hyeon T, Lee SS, Park J, Chung Y, Na HB (2001) Synthesis of highly crystalline and monodisperse Maghemite Nanocrystallites without a size-selection process. J Am Chem Soc 123(51):12798–12801
Kalam A, Al-Sehemi AG, Assiri M, Du G, Ahmad T, Ahmad I, Pannipara M (2018) Modified Solvothermal synthesis of cobalt ferrite (CoFe2O4) magnetic nanoparticles photocatalysts for degradation of methylene blue with H2O2/visible light. J Res Phys:S2211-3797 (18)30042-1
Khan H, Khali AK, Khan A (2019) Photocatalytic degradation of alizarin yellow in aqueous medium and real samples using chiston conjugated tin magnetic nanocomposites. Mater Electron:21332–21342
Kim DK, Zhang Y, Voit W, Rao KV, Kehr J, Bjelke B, Muhammed M (2001) Superparamagneticiron oxide nanoparticles for bio-medical applications. Scr Mater 44:1713–1717
Kozakova Z, Kuritka I, Kazantseva NE, Babayan V, Pastorek M, Machovsky M, Saha P (2015) The formation mechanism of iron oxide nanoparticles within the microwave-assisted solvothermal synthesis and its correlation with the structural and magnetic properties. Dalton Trans 44(48):21099–21108
Liang H, Jiang X, Chen W, Wang S, Xu B, Wang Z (2014) α-Fe2O3/Pt hybrid nanorings and their enhanced photocatalytic activities. Ceram Int 40(4):5653–5658
Lu A-H, Salabas EL, Schüth F (2007) Magnetic nanoparticles: synthesis, protection, functionalization, and application. Angew Chem Int Ed 46(8):1222–1244
Lu D, Zhang Y, Lin S, Wang L (2013) Synthesis of magnetic ZnFe2O4/graphene composite and its application in photocatalytic degradation of dyes. J Alloys Compd 579:336–342
Lucas MS, Tavares PB, Peres JA, Faria JL, Rocha M, Pereira C, Freire C (2013) Photocatalytic degradation of reactive black 5 with TiO2-coated magnetic nanoparticles. Catal Today 209:116–121
Majidi S, Sehrig FZ, Farkhani MS, Goloujeh SM, Akbarzadeh A (2016) Current methods for synthesis of magnetic nanoparticles. Artif Cells Nanomed Biotechnol 44(2):722–734
Masoumi S, Nabiyouni G, Ghanbari D (2016) Photo-degradation of azo dyes: photo catalyst and magnetic investigation of CuFe2O4–TiO2 nanoparticles and nanocomposites. J Mater Sci Mater Electron 27:9962–9975
Mishra D, Senapati KK, Borgohain C, Perumal A (2012) CoFe2O4-Fe3O4 magnetic nanocomposites as photocatalyst for the degradation of methylene Orange. J Nanotechnol:52–58
Morrison SA, Cahill CL, Carpenter EE, Calvin S, Swaminathan R, McHenry ME, Harris VG (2004) Magnetic and structural properties of nickel zinc ferrite nanoparticles synthesized at room temperature. J Appl Phys 95(11):6392–6395
Mortazavi-Derazkola S, Salavati-Niasari M, Amiri O, Abbasi A (2017) Fabrication and characterization of Fe3O4@SiO2@TiO2@Ho nanostructures as a novel and highly efficient photocatalyst for degradation of organic pollution. J Energy Chem 26(1):17–23
Pang YL, Lim S, Ong HC, Chong WT (2016) Research progress on iron oxide-based magnetic materials: synthesis techniques and photocatalytic applications. Ceram Int 42(1):9–34
Qu L, Peng ZA, Peng X (2001) Alternative routes toward high quality CdSe nanocrystals. Nano Letters 1(6):333–337
Rahimi R, Tadjarodi A, Rabbani M, Kerdari H, Imani M (2012) Preparation, characterization and photocatalytic properties of Ba-cd-Sr-Ti doped Fe3O4 Nanohollow spheres on removal of Congo red under visible-light irradiation. J Supercond Nov Magn 26(1):219–228
Saffari J, Mir N, Ghanbari D, Khandan-Barani K, Hassanabadi A, Hosseini-Tabatabaei MR (2015) Sonochemical synthesis of Fe3O4/ZnO magnetic nanocomposites and their application in photo-catalytic degradation of various organic dyes. J Mater Sci Mater Electron 26:9591–9599
Sun S (2000) Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal Superlattices. Science 287(5460):1989–1992
Tang B, Wang G, Zhuo L, Ge J, Cui L (2006) Facile route to α-FeOOH and α-Fe2O3 Nanorods and magnetic property of α-Fe2O3 Nanorods. Chem Inform 37(38)
Tavakoli A, Sohrabi M, Kargari A (2007) A review of methods for synthesis of nanostructured metals with emphasis on iron compounds. Chem Pap 61:151–170
Wahab A, Imran M, Ikram M, Naz M, Aqeel M, Rafiq A, Ali S (2019) Dye degradation property of cobalt and manganese doped iron oxide nanoparticles. Appl Nanosci 9:1823–1832
Wang, Xuanze. Preparation, synthesis and application of Sol-gel method, 2020
Weissleder R, Mahmood U (2001) Molecular Imaging. Radiology 219(2):316–333
Xiao Z, Li J, Zhong J, Hu W, Zeng J, Huang S, Li M (2014) Enhanced photocatalytic decolorization of methyl orange by gallium-doped α-Fe2O3. Mater Sci Semicond Process 24:104–109
Xu H, Zeiger BW, Suslick KS (2013) Sonochemical synthesis of nanomaterials. Chem Soc Rev 42(7):2555–2567
Zazouli MA, Ghanbari F, Yousefi M, Madihi-Bidgoli S (2017) Photocatalytic degradation of food dye by Fe3O4–TiO 2 nanoparticles in presence of peroxymonosulfate: the effect of UV sources. J Environ Chem Eng 5(3):2459–2468
Zhang Z, Hossain MF, Takahashi T (2010) Self-assembled hematite (α-Fe2O3) nanotube arrays for photoelectrocatalytic degradation of azo dye under simulated solar light irradiation. Appl Catal B Environ 95(3–4):423–429
Author information
Authors and Affiliations
Corresponding author
Section Editor information
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Manikandan, A. et al. (2022). Photocatalytic Applications of Magnetic Hybrid Nanoalloys and Their Nanocomposites. In: Handbook of Magnetic Hybrid Nanoalloys and their Nanocomposites. Springer, Cham. https://doi.org/10.1007/978-3-030-34007-0_59-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-34007-0_59-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-34007-0
Online ISBN: 978-3-030-34007-0
eBook Packages: Springer Reference Chemistry and Mat. ScienceReference Module Physical and Materials ScienceReference Module Chemistry, Materials and Physics