Abstract
Silicon is known as an anti-stress agent and its protecting role against a wide range of environmental stresses such as drought, frost, salinity, high temperature, and heavy metal toxicity is well recognized. Silicon increases crop productivity and improves crop quality while the lack of this element reduces plant’s biological ability to withstand the adverse environmental conditions. Silicon is non-corrossive, non-pollutive, and not detrimental to plants even when applied in excess. Silicon fertilizer can provide economic as well as ecological benefits to plant growers. Hence, concerted efforts in the area of silicon research can lead to its accelerated and improved application in the form of fertilizer for sustainable agriculture.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
The world’s population is approximately seven billion, and it has been predicted to enhance by ten billion in the next 50 years (Glick 2014). Food security is considered as one of the most serious challenges. Recent trends of crop production indicate that fertility of soil is declining due to intensive use of natural resources, indiscriminate use of pesticides, and lack of proper soil management practices (Cakmak 2001). There is a need to use new technologies which could help us to curb the problem of food insecurity. Abiotic stress is known as one of the most important constraints to agricultural production in the world (Meena et al. 2017). Abiotic stresses have become more common and challenging due to the unpredictable climate conditions. The change in climatic conditions has exacerbated the frequency and severity of many abiotic stresses with significant reduction in crop yield (Carmen and Roberto 2011). More than 70% decrease in crop yield has been reported due to land degradation, undesirable effects on agriculture, loss of biodiversity, and abiotic stresses (Veatch-Blohm 2007). The main constraints to agricultural production are change in environmental conditions causing various abiotic stresses such as drought, flood, high and low temperature, salinity, UV-B radiation, inadequate mineral supply, and heavy metal toxicity (Jewell et al. 2010; Shrivastava and Kumar 2015). Hence, there is a need to find the reliable eco-friendly methods to alleviate the impact of abiotic stresses on agricultural system to sustainably meet growing global food demands. The management of the soil with mineral nutrients offers both environmental and economic benefits (Marschner 1995). The positive effects of silicon on the crop yield and quality have been well documented (Liang et al. 2015). The beneficial effects of silicon in stressed plants are more visible than non-stressed plants (Cooke and Leishman 2016). The role of silicon in enhancing the resistance of plants to various abiotic stresses has been widely recognized by researchers (Guo et al. 2005; Gunes et al. 2007; Etesami and Beattie 2017; Etesami and Jeong 2018).
Application of silicon in agriculture is a sustainable strategy for the alleviation of abiotic stresses in plants (Cooke and Leishman 2011; Guntzer et al. 2012; Van Bockhaven et al. 2013; Hernandez-Apaolaza 2014; Adrees et al. 2015; Coskun et al. 2016; Imtiaz et al. 2016; Luyckx et al. 2017; Wang et al. 2017; Etesami and Jeong 2018). Silicon fertilizers are safe, eco-friendly, and cost-effective in comparison to other chemical/synthetic fertilizers even for small and marginal plant growers. Hence, silicon can be used as a growth regulator to improve plant growth and resistance under stress conditions.
Occurrence of Silicon in the Soil
Silicon is the second most abundant element in earth’s crust in terms of quantity (Mcginnity 2015), and it comprises up to 70% of soil mass (Ma and Yamaji 2008; Ahmed et al. 2014). Silicon forms a major portion of soil as silicate but its availability to plants is low (Zhu and Gong 2014). Balakhnina and Borkowska (2013) reported that most of the sources of silicon are insoluble and in a plant-unavailable form. Silicon exists in the form of silicic acid (H4SiO4), a non-charged molecule in soil solution at the concentration 0.1–0.6 mM and pH less than 9 (Epstein 2009). The concentration of silicon in soil solution is equivalent to some of the macro-elements such as potassium, calcium, and phosphorus (Epstein 1994). The soil contains 50–400 g silicon/kg of soil but the silicon content in the soil depends on the type of soil as sandy soil contains more silicon in comparison to clay soil (Matichenkov and Calvert 2002). Lovering and Engel (1959) reported that forest in the 1 hectare land can extract about 5000 tonnes of silicon in 5000 years. According to the reports of Food and Agricultural Organization (FAO 1998), approximately 210–224 million tonnes of silicon is removed from the arable soils all around the world and soils become less fertile due to low availability of silicon content. The incorporation of silicon in soil improves the fertility of soil through physical and chemical properties, improved water absorption capacity, and maintenance of nutrients in a soil in the plant available form.
Sources of Silicon
The surface of earth is covered with 27.7% of silicon next to oxygen but the existence of silicon in its pure form is rare (Mitra 2015). Silicon is deposited in the form of quartz (SiO2), sand and sandstone in the earth crust (Rédei 2008). Agricultural wastes such as silicate slag, steel slag, electric furnace slag, baggase furnace ash, lignite fly ash, and rice straw are rich in silicon and can be used in crop fields to increase the crop yield (Kalra et al. 2003). Calcium silicate obtained as a by-product of phosphorus production is one of the most widely used silicon fertilizer. Potassium silicate is highly soluble and can be used in hydroponic culture for laboratory experiments but it is expensive. The other sources of silicon have been used commercially are silica gel, calcium silicate hydrate, etc.
Silicon: Essential Element for Plants
Earlier researchers reported that silicon is not an essential element for plants but difficulties in growing plants in silicon-free environment made this assumption incorrect and silicon was designated as an essential element for the growth and development of plants. Justus Von Leibig proposed the use of sodium silicate as a silicon fertilizer as early as in the year 1840. Silicon is a beneficial element to higher plants as its effects are frequently linked to morphological, physiological, and molecular aspects of plants (Ma 2004; Lobato et al. 2009). Silicon acts as a quasi-essential element for plants because its deficiency can cause various abnormalities in growth, reproduction, and overall development of plants (Epstein and Bloom 2005). The sand is applied to rice fields at 2–3 tonnes/acre once in 2 or 3 years in southern part of India is considered as a good source of silicon. The farmers in Texas and Florida use certified green sand containing silicates to get higher crop yield. Silicon has been recognized as an agronomically essential element for rice cultivation in Japan because silicon promotes the growth and yield of rice. Silicon nutrition to the plants improves plant protective mechanism against diseases, insect attack, and unfavorable environmental conditions (Guntzer et al. 2011; Dallagnol et al. 2011; Liu et al. 2014).
Silicon Content in Plants
The content of silicon in plants shows a large variability ranging from 0.1 to 10% dry weight (Hodson et al. 2005). According to Marschner (1995), there are three classes of silicon absorbers: (1) silicon accumulator plants as they accumulate large amount of silicon—wheat, rice, millet, and sugarcane; (2) silicon non-accumulator plants—snapdragon plant; (3) silicon-excluder plants—soybean. Silicon accumulators have silicon concentration more than 1% and silicon/calcium ratio > 1 and excluders have silicon concentration below 0.5% and silicon/calcium ratio < 0.5. The plants which do not meet these criteria are considered as intermediate plants. Monocotyledons such as rice, sugarcane, and maize absorb silicon in large quantities due to the presence of silicon transporters in comparison to dicotyledons (Ma et al. 2016). The aerial plant parts accumulate more silicon in comparison to roots. Silicon content in plants increased with the age of crop plants and due to this older leaves are rich in silicon concentration than younger leaves (Henriet et al. 2006).
Silicon Mitigates Abiotic Stresses in Plants
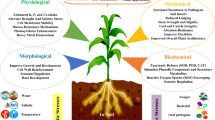
Silicon is known as the only element that is able to increase plant resistance to environmental stresses. Silicon-mediated alleviation of abiotic stresses such as salinity, drought, UV-B radiation, flooding, freezing, lodging, high temperature, and heavy metal toxicity has been reported (Reynolds et al. 2016; Debona et al. 2017; Kim et al. 2017; Etesami and Jeong 2018). The benefits of silicon accumulation in wetland environment have been studied in terms of increased ability to resist water current and allow roots to better penetrate the mud (Struyf and Conley 2008). The most important function of silicon lies in its potential to confer tolerance in plants to multiple stresses (Fig. 1).
Salinity Stress
Salinity is one of the most significant abiotic stresses for crop plants (Chinnusamy et al. 2005). Approximately 20% of the total arable area has been degraded by salinity (Shrivastava and Kumar 2015). Salt interferes with plant growth by ion cytotoxicity (Greenway and Munns 1980), nutrient imbalance (Khan et al. 2000), and oxidative damage (Hernandez et al. 2000). Silicon has been reported to improve salt tolerance in different crops (Liang et al. 2007; Reezi et al. 2009). Silicon can reduce Na+ uptake by plants under salt stress by (1) reducing membrane permeability of root cells, (2) improving structure and stability of root cells (Exley 2015; Luyckx et al. 2017), (3) mediating stimulation of the root plasma membrane H+-ATPase, and (4) facilitating Na+ export from the cell. Exogenous application of silicon improved gas exchange characteristics in many plant species under salinity condition (Etesami and Jeong 2018). The water status of leaf and water use efficiency of crops was increased by silicon application in many salt-stressed plants (Coşkun et al. 2016). Silicon deposited as silica gel in the cell wall of the roots provides binding sites for the salts and reduces their translocation to shoots which consequently improved plant growth and development (Lux et al. 2003).
Silicon deposition and polymerization in leaves limits transpiration and salt accumulation and improved water storage within plant tissues, which allowed higher growth rate, salt dilution within the plant cells, and mitigating salt toxicity effects (Bradbury and Ahmad 1990). Increase in salinity rises Na+, Cl−, and SO42− contents and decrease in K+, Ca2+, and Mg2+contents in a number of crop species (John et al. 2003). The addition of silicon to saline environment significantly decreased the Na+content but increased K+ concentration (Ashraf et al. 2010). Salinity-induced accumulation of ROS including superoxide radical, hydroxyl radical, and hydrogen peroxide are detrimental to cells at higher concentration because they cause oxidative damage to membrane lipids, proteins, chlorophyll, and nucleic acids (Mittler 2002; Keles et al. 2004; Gunes et al. 2007). The application of silicon under salt stress significantly increased superoxide dismutase, peroxidase, catalase, and glutathione reductase activity in barley roots (Liang et al. 2003) and cucumber (Zhu et al. 2004). The higher efficiency of antioxidant defense enzymes in salt-stressed plants with silicon application coincided with a decrease in their electrolyte leakage and lipid peroxidation level and H2O2 content, suggesting that oxidative damage induced by salinity stress can be alleviated by silicon supplementation (Moussa 2006; Wang et al. 2010).
Drought Stress
Drought is one of the major abiotic stresses that adversely affects growth and productivity of agricultural crops (Bodner et al. 2015). Drought or water deficit leads to stomatal closure, reduced transpiration rate, decrease in water potential of plant tissues, decrease in photosynthesis, and ultimately plant growth is inhibited (Yordanov et al. 2003). The ameliorative effect of silicon on drought stress has been related to the reduction in loss of water by transpiration (Romero-Aranda et al. 2006), osmotic adjustment (Trenholm et al. 2004), improved nutrient uptake (Gunes et al. 2008a), and activation of antioxidant defense system (Gunes et al. 2008b). Agarie et al. (1998) reported that deposition of silicon in the cell wall reduced transpiration and increased internal storage of water under drought stress. The mitigating effect of silicon on drought stress could be related to the hydrophilic nature of SiO2.nH2O which maintains the water in plant tissues and protects plant tissues from drought (Gong et al. 2005). An important mechanism of drought tolerance is the osmotic adjustment as it helps to retain water in plant tissues despite low water potential (Romero-Aranda et al. 2006). Kaya et al. (2006) found that silicon supply increased Ca concentration which played a vital role in maintaining membrane permeability and stability. Liang (1999) reported that under water stress condition, K+ concentration increased in the presence of silicon possibly because of the activation of H+-ATPase in the cell membrane. Potassium is one of the primary osmotic substances which contribute to osmotic adjustment (Ashraf et al. 2001). Silicon-mediated maintenance of higher level of K+ is essential in achieving better survival with improved growth under water stress (Umar 2002). Drought-induced reduction in chlorophyll content can be alleviated by silicon and hence improved photosynthesis under drought stress (Kaya et al. 2006). The effect of silicon stimulation on root growth may be due to increased root elongation caused by an increase in cell wall extensibility in the growth region as observed in sorghum (Hattori et al. 2003).
Wang et al. (2015) reported significant increase in the root/shoot ratio in silicon-treated plants, and it was due to the silicon-mediated modification in root morphology which increased water uptake ability of silicon-treated plants. The augmented water uptake during the addition of silicon in water deficit condition is the result of improved root hydraulic conductance (Hattori et al. 2008) and root activity (Chen et al. 2011). It has been reported that improved structural stability due to the binding of silicon with cell wall hemicellulose can be beneficial in water shortage condition for the plants (Ma et al. 2015).
Thermal Stress
Temperature is one of the most important environmental factors governing plant growth and development (Monjardino et al. 2005). Thermal stress (cold and heat) may disturb the balance between ROS and antioxidant activity in plants. Application of silicon has been reported to alleviate heat stress in different plant species (Epstein 1999). Heat stress is detrimental to plant growth and dry matter accumulation because it inhibits protein synthesis, enzyme activity, chlorophyll formation, photosynthesis, and increases transpiration (Gibson and Paulsen 1999). Commuri and Jones (2001) reported that extended periods of drought and high temperature lead to poor vigor and shallow rooting. According to Takahashi and Kurata (2007), silicon deposition in the cell wall of xylem vessels prevents the compression of vessels under the conditions of high transpiration caused by heat stress (Hattori et al. 2005; Liang et al. 2007). However, high level of silicon in cell wall reduced transpiration loss caused by higher temperature thus allowing continued metabolic functions under high temperature. According to Epstein (1999), plants wilt less, resist sunburn and are generally more tolerant to heat stress when silicon is applied to plant growth medium. The supply of silicon has been found to produce higher concentrations of RUBP-carboxylase enzyme in leaf tissues which help in CO2 metabolism in stress environment (Gunes et al. 2008b). Soundararajan et al. (2014) treated Salvia splendens with silicon under high temperature and found that the activities of SOD, APX, and GPX were increased.
The chilling and frost stresses can cause irreversible damage to plant cells due to mechanical forces generated by the formation of extracellular ice crystals, cellular dehydration, and increased concentration of intracellular salts (Steponkus 1984). It has been reported that freezing increases the level of ROS in plants and increased lipid peroxidation arising from the accumulation of ROS is the major cause of membrane damage (McKersie et al. 1993). Studies have shown that supply of silicon under low temperature stress regulated the activity of antioxidant defense system and alleviated oxidative damage caused by chilling or frost (Ma et al. 2001). It has been reported that silicon improves leaf and stem strength through deposition in the cuticle and by maintaining cell wall polysaccharide and lignin polymers, thus protected plants from low temperature and frost (Ohyama 1985; Hull 2004). The photosynthetic electron transport, CO2 fixation, Rubisco activity, and stomatal conductance are the major targets impaired by low temperature stress in plants. Application of silicon under frost conditions resulted in higher concentration of chlorophyll (Schmidt et al. 1999) and RUBP-carboxylase enzyme in leaf tissue. This enzyme regulated the metabolism of CO2 and enables the plant to make more efficient use of available levels of CO2 under low temperature stress. The leaves of silicon-treated plants (cucumber, rice, maize, and sunflower) grown hydroponically at low temperature (0–4 °C) were more resistant to cold-induced wilting and root ability to absorb nutrients was higher. Silicon-mediated alleviation of freezing injury may be attributed to enhanced antioxidant defense activities and consequent reduced membrane oxidative damage through better water retention in leaf tissues (Liang et al. 2003; Zhu et al. 2004; Gong et al. 2005).
Radiation
Exogenous application of silicon showed alleviating effects of UV-B stress on soybean, wheat, and maize (Yao et al. 2011; Shen et al. 2014). Many studies have revealed that silicon application can induce resistance to UV-B stress by the modification of physiological and biochemical processes in plants (Schaller et al. 2013; Tripathi et al. 2017). According to Tripathi et al. (2017), UV-B stress was significantly improved with the use of silicon nanoparticles on wheat seedlings.
Wounding Stress
Wounding stress is caused by physical injury in plants, and it may be due to wind or herbivore attack. Such injuries can cause the death of plant tissues and make it vulnerable to pathogenic attack. Wounds initiate oxidative stress thereby damaging the cell membranes. The antioxidant activities of catalase and peroxidase are significantly increased in silicon-treated wounded rice plants compared to control plants and improved mechanical strength to overcome losses from wounding stress (Kim et al. 2014). A similar trend of increased antioxidant enzyme activity was also observed in wheat (Gong et al. 2005), maize, and barley (Liang et al. 2005). The accumulation of antioxidative enzymes in silicon-treated plants functions as a strategy for coping with wounding stress.
Heavy Metal Toxicity
Heavy metal pollution is rapidly increasing, and it originates from natural sources as well as anthropogenic activities including mining, industrial waste, sewage sludge, and excessive use of fertilizer and pesticides (Nagajyoti et al. 2010). The relationship between silicon supplementation and metal tolerance in plants has been widely studied (Shi et al. 2005; da Cunha and do Nascimento 2009; Kaya et al. 2010). Silicon supplementation decreased metal toxicity such as toxicity of aluminum (Wang et al. 2004), boron (Gunes et al. 2007), cadmium (Liang et al. 2005), chromium (Tripathi et al. 2012), copper (Li et al. 2008), and zinc (Neumann and Zur Nieden 2001). The possible mechanism behind the silicon inhibition of metal transport in plants may be due to the thickening of the casparian strips in the endodermis and cell wall of the xylem due to the deposition of silicon in the cell wall (Chen et al. 2000; da Cunha and do Nascimento 2009).
Excessive metal exposure induces leaf chlorosis and necrosis by affecting chlorophyll synthesis and breakdown of the cell membrane. It was reported that silicon reduced the toxic effects of metals in plants by stimulating the production of root exudates (Adrees et al. 2015; Imtiaz et al. 2016). Silicon can reduce metal toxicity in plants by increasing the content and accumulation of macronutrients (Ca, Mg, P, and K) and micronutrients (Zn, Fe, and Mn) (Etesami and Jeong 2018).
The key mechanism of silicon-mediated metal toxicity alleviation in higher plants include: (1) complexation or co-precipitation of toxic metal ions with silicon; (2) immobilization of toxic metal ions in the growth media; (3) stimulation of antioxidant defense system; (4) uptake processes; (5) compartmentation or homogenous distribution of metal ions within plants (Liang et al. 2005); (6) accelerating suberin lamellae, casparian bands, root vascular tissues development, and lignification; (7) enhancing the activity of gas exchange characteristics (net photosynthetic rate, stomatal conductance, transpiration rate, and water use efficiency); (8) change in plant morphological traits (increase in length of leaves, leaf area, number of leaves per plant, root volume, root length, and total root surface area); (9) regulating plant phytohormones; (10) helping the formation of the tertiary endodermal cell wall; (11) localizing in root endodermis, which serve as a barrier to block heavy metal entrance into cells (Adrees et al. 2015; Imtiaz et al. 2016). Silicon has been shown to alleviate the adverse effects of nutritional imbalance stress in plants (Hernandez-Apaolaza 2014; Etesami and Beattie 2017; Etesami and Jeong 2018).
Macronutrients
The excessive use of nitrogen fertilizer causes lodging, mutual shading, and susceptibility of plants to pests and diseases (Ma 2004). The adverse effects due to excessive nitrogen fertilizers can be minimized by the use of silicon as the presence of silicate crystals in plant tissues provide mechanical barrier which hinders feeding of insects and inhibits fungal diseases by inhibiting fungal germ tube penetration of epidermis (Savant et al. 1999). Silicon can alleviate nitrogen deficiency-induced stress in plants by (1) increasing the uptake of nitrogen, (2) improving nodulation and better N2 fixation in legume plants, (3) increasing N use efficiency, and (4) altering primary metabolism due to stimulating amino acid remobilization.
Phosphorus is essential mineral element but most of phosphorus in soils is in biologically unavailable form. Soundararajan et al. (2016) reported that silicon fertilization increased P availability in various graminaceous species. Silicon can alleviate P shortage-induced stress in plants by (1) improving utilization of P by increasing phosphorylation; (2) decreasing excess Fe, Mn, and Al uptake and (3) increasing water-soluble phosphorus concentration. Owino-Gerroh and Gascho (2005) reported that application of soluble silicon in acid soils can decrease adsorption of phosphorus in soils and increase the amount of bioavailable phosphorus.
Potassium is one of the major macronutrients which has an important role in the growth and development of plants. Silicon can alleviate K-deficiency-induced stress by influencing the availability of K in the soil and plants and modulating antioxidant enzymes to alleviate K-deficiency-induced membrane lipid peroxidation and oxidative stress (Miao et al. 2010; Pei et al. 2010). Silicon can increase calcium and magnesium level in soil and plants. The improved absorption of calcium may be due to an increase in the movement of plasma membrane H+-ATPase due to the addition of silicon (Etesami and Jeong 2018).
Silicon application influences the nutrient content of sunflower by increasing the accumulation of both macro- and micronutrients (Savić and Marjanović-Jeromela 2013). Silicon can alleviate the effect of micronutrient deficiency stress on the plants grown under nutrient-poor conditions (Pavlovic et al. 2013; Bityutskii et al. 2014; Hernandez-Apaolaza 2014). It has been reported that silicon alleviates chlorosis in plants due to Fe deficiency by (1) forming Fe plaque on root; (2) increasing Fe transport from root to shoot; (3) maintaining the balance of other micronutrients such as Fe/Mn ratio; (4) accumulating Fe-mobilizing compounds such as citrate (in xylem sap and root and shoot tissues) or catechins (in roots); (5) the activation of Fe-deficiency-associated genes and (6) Fe chelate reductase (Etesami and Jeong 2018). Under Fe toxicity conditions, silicon can protect plants from excess of Fe stress by (1) releasing OH− by roots; (2) increasing the oxidizing capacity of roots, which changes ferrous iron (Fe2+) into ferric iron (Fe3+) and (3) increasing the iron precipitation in the growth media or at root surfaces (iron plaque) as Fe (III)-silicates (You-Qiang et al. 2012).
Silicon is able to mitigate manganese deficiency in plants by enhancing the roots oxidizing capacity, which gives a higher Mn-oxidation rate in the rhizosphere (Li et al. 1999). Silicon can also mitigate the excess stress of Mn by affecting Mn-oxidation state, which promotes its precipitation, regulates the Fenton reaction, and mediates the Mn adsorption on cell walls.
Silicon influences zinc plant nutrition under deficiency stress conditions (Pascual et al. 2016) by (1) remobilizing the Zn pools in the roots, (2) changing Zn distribution in plants, (3) increasing citrate in plants, and (4) activating the Zn-deficiency mechanism. Silicon can also mitigate the excess stress of Zn by avoiding the Zn transport to more sensitive organs of plant by enhancing Zn2+ adsorption on the deposits of the silicate.
Si also influences copper plant nutrition in the plants grown under Cu stress conditions by affecting the distribution of Cu within leaves (Frantz et al. 2011). Silicon can mitigate the excess stress of Cu in the plants by the formation of silicon deposits on the cell wall, which increases the Cu-binding sites.
Silicon-Induced Growth Regulation and Abiotic Stress Tolerance in Plants
Plant Growth Improvement
An increase in plant growth and development due to silicon application has been reported in rice (Ma and Yamaji 2008; Kim et al. 2012), cotton (Li et al. 1989), soybean (Hamayun et al. 2010), wheat (Gong et al. 2005), sorghum (Hattori et al. 2005), cucumber (Feng et al. 2009), barley (Savant et al. 1999), bean (Zuccarini 2008), tomato (Al-aghabary et al. 2005; Liang et al. 2007), spinach (Eraslan et al. 2008), and maize (Liang et al. 2005).
Silicon foliar fertilizer is advantageous for sugar beet production and a significant increase has been reported in sugar yield (Artyszak et al. 2014). Silicon has increased yield by 22% in potato (Luz et al. 2008), 30% in rice, and 45% in sugarcane (Kingston 2008). The application of silicon in canola improved yield and oil content and reduced pesticide and fungicide usage (Lynch 2008). Silicon augmentation in soilless cultivation of corn salad improved the yield, quality, and shelf life of corn by the regulation of nutrient acquisition, uptake of nitrate/iron, phenoloxidase gene expression, and protection of chlorophyll pigment from degradation (Gottardi et al. 2012). Silicon inclusion in tissue culture medium resulted in the enhancement of axillary shoot induction, alleviation of hyperhydricity (Soundararajan et al. 2017), callus induction (Islam et al. 2005), and root morphogenesis (Asmar et al. 2013).
Aquaporins are essential transmembrane proteins that maintain the uptake and movement of water molecules across the cell membranes particularly under abiotic stress condition (Boursiac et al. 2005). According to Boursiac et al. (2008), aquaporin activity is susceptible to change in the ROS level as H2O2 stimulated by salt stress resulted in the prevention of aquaporin function.
The higher expression of genes related to aquaporin results in the rapid water uptake which also dilutes the excess concentration of Na+ ions lethal for the plants (Gao et al. 2010). The findings of Sutka et al. (2011) illustrated that the abundance of aquaporin genes in the roots balance the water uptake by the plants even under water deficit conditions. The enhancement of aquaporin-related genes by silicon might improve the water status in plants under salinity and drought stress. The improvement of water status and ion balance aid in the reclamation of plants from abiotic stress. It is known that silicon can increase the uptake of nutrients by increasing root activity (Chen et al. 2011), enhancing water uptake (Sonobe et al. 2010), and improving hydraulic conductance of roots (Hattori et al. 2008).
Increase in Root System of Plants
Abiotic stresses adversely affect the growth and yield of plants by limiting the uptake and translocation of water and essential nutritional ions (Hu and Schmidhalter 2005). Nutrient absorption is linked to root surface region and root length. An increase in root surface area provides more exposed areas for the absorption of dispersed ions (Barber 1995). Silicon can improve root growth and subsequently enhance nutrient uptake, improve nutrient balance, and plant shoot biomass under salinity (Kim et al. 2016; Li et al. 2015) and drought (Chen et al. 2011). It has been reported that silicon facilitates root growth through increasing cell wall extensibility in the growth zone of roots (Vaculík et al. 2009). Silicon has also contributed to stimulation of nutrient uptake by increasing water uptake (Sonobe et al. 2010). The higher root growth increases water acquisition (increased water use efficiency) and nutrient uptake, it subsequently alleviates the adverse stress effects in silicon-treated plants. Water deficit also limits nutrient uptake through roots and subsequent transport to shoots, thereby reducing nutrient availability and metabolism (Farooq et al. 2009). Silicon-mediated selective transport capacity for K+ over Na+ and thereby increases in K+/Na+, which may be one of the main mechanisms improving plant growth and yield under abiotic stress.
Improvement of Water Relations
Romero-Aranda et al. (2006) stated that silicon helps in water restoration in salt-stressed plants due to its hydrophilic nature. Silicon can decrease salt toxicity for plants and improve plant growth under salinity stress by different mechanism including: (1) increasing water storage in plants which contributes to salt dilution (Tuna et al. 2008); (2) increasing thickness of leaves which results in leaf water content and water potential (Gong et al. 2003) and (3) deposition of silicon in leaves which reduces transpiration from leaf surface and decreases the escape of water molecules from the leaf surface (Keller et al. 2015). It has been recognized that drought stress substantially reduces the leaf water potential and water content in drought-stressed plants. Silicon also influences water relation and improves significantly the photosynthesis and water status in non-irrigated crops (Rizwan et al. 2015). Gao et al. (2005) observed that silicon application enhanced water use efficiency in drought-stressed maize by reducing leaf transpiration rate. According to Rizwan et al. (2015), silicon can influence stomatal movement in plants.
Some of the mechanisms by which silicon can result in higher efficient use of water, enhance root water uptake by roots, and increase drought tolerance in plants under water deficit stress (Zhu and Gong 2014) include: (1) altering transpiration (Farooq et al. 2009); (2) forming a cuticle-silica double layer on leaf epidermal tissue which reduces leaf transpiration and water flow rate in xylem vessels (Zhu and Gong 2014); (3) accumulation of soluble sugars and amino acids (Sonobe et al. 2010); (4) deposition of silicon on root cell wall, which affects the wetting properties of xylem vessels and water/solute transport (Gao et al. 2005); (5) maintaining photosynthetic pigments content (Lobato et al. 2009); (6) improving chloroplast ultrastructure; (7) increasing activities of antioxidant enzymes (Gong et al. 2005); (8) enhancing the activity of phosphoenolpyruvate carboxylase and the concentration of inorganic phosphate in plant leaves (Gong and Chen 2012).
Regulation of Biosynthesis of Compatible Solutes
Under stressful conditions such as salinity and drought, concentration of compatible solutes or osmolytes such as proline (Pei et al. 2010), glycine betaine, and polyols (Parida and Das 2005) increased in plants. Silicon application also increased plant tolerance to salinity and drought stress by modifying the levels of solutes such as proline (Yin et al. 2013), glycine betaine (Torabi et al. 2015), carbohydrates (Ming et al. 2012), polyols, antioxidant compounds (Hashemi et al. 2010), and soluble sugars and free amino acids (Hajiboland et al. 2016). These compatible solutes may increase plant resistance to abiotic stresses by (1) maintaining higher leaf water potential during stress; (2) scavenging free radicals and buffering cellular redox potential, which keep plants protected against oxidative stress and (3) stabilizing subcellular structures (membranes and proteins) (Fahad et al. 2015). It has been found that the compatible solutes may also act as oxygen radical scavengers (An and Liang 2013; Abbas et al. 2015). Silicon alleviated drought stress in the plants by osmotic adjustment inducing the production of soluble sugars and amino acids such as alanine and glutamic acid (Zhu and Gong 2014).
Photosynthesis
It has been reported that silicon delays chlorophyll degradation in plant species under abiotic stresses (Al-aghabary et al. 2004; Feng et al. 2010; Gottardi et al. 2012). Silicon has also been shown to result in higher concentration of chlorophyll per unit area of leaf tissue. There are two possible mechanisms have been reported: The first one was related with the structural protection of the chloroplast membranes by silicon (Al-aghabary et al. 2004; Feng et al. 2010) and second was the effect of silicon on strengthened cell walls which might contribute to a more favorable position of leaves to intercept light and increased the photosynthesis (Ma and Takahashi 2002). Silicon contributes to maintain micronutrients balance such as Fe/Mn ratio (Pich et al. 1994), which is also beneficial in enhancing chlorophyll synthesis and provides a possible explanation for the stimulation in growth of Fe-deficient plants supplied with silicon (Pavlovic et al. 2013; Bityutskii et al. 2014). The increase in citrate concentration in plants has been reported with silicon supply. Citrate is one of the molecules that joined iron on its transport through the xylem (Rellán-Álvarez et al. 2010).
Polyamines Synthesis and Metabolism
Plants with high level of polyamines such as putrescine, spermidine, and spermine reported to possess more resistance against environmental stresses (Quinet et al. 2010; Pottosin and Shabala 2014). The elevated levels of genes responsible for the synthesis of polyamines mitigate the negative effects of oxidative stress (Roy and Wu 2001; Tang et al. 2007). The augmentation of silicon elevated the expression level of S-adenosyl-l-methionine decarboxylase (SAMDC) gene which encodes a vital enzyme involved in the biosynthesis of polyamines. Polyamines are involved in various vital processes such as replication, transcription, translation, stabilization of membranes, and modulation of enzyme activities in addition to stress tolerance. Hence, the regulation of genes involved in polyamine biosynthesis by silicon could not only help in the stress alleviation but also improve the fundamental processes in cells and increased the growth and development of plants.
Antioxidative Defense System
Plants continuously produce several active oxygen species (AOS) or reactive oxygen species (ROS) such as superoxide anion (O2−), hydrogen peroxide (H2O2), and hydroxyl radical (OH−) during photosynthesis and respiration processes in different cell organelles such as mitochondria, chloroplast, and peroxisomes. ROS can cause serious oxidative damage to the biomolecules such as protein, membrane lipids, and nucleic acids in the cells (Apel and Hirt 2004; Tripathi et al. 2017). The scavenging of ROS is most important defense mechanism to cope with stress conditions in plants (Baxter et al. 2014). Silicon nutrition in plants enhances the production of antioxidants and enzymes involved in detoxification of the free radicals (Zhu et al. 2004). Plants can maintain homeostasis by two different detoxification mechanisms involving enzymatic and non-enzymatic antioxidants (Mittler 2002; Sytar et al. 2013; Wu et al. 2017). Application of silicon can improve ROS scavenging ability in plants by development of ROS scavenging enzymes such as superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), and ascorbate peroxidase (APX) and non-enzymatic antioxidants such as glutathione, ascorbate, and carotenoids (Crusciol 2009; Shen et al. 2010; Torabi et al. 2015).
Etesami and Jeong (2018) reported that silicon maintained cell membrane permeability and stability of the plants grown under abiotic stresses. Silicon application can also increase the glutathione concentration and suppress the malondialdehyde concentration (Liang et al. 2006). Zhu et al. (2004) reported that silicon is responsible for inhibiting the membrane damage caused by the formation of malondialdehyde, which causes the lipid peroxidation of membranes. By regulating antioxidant defense system, silicon can decrease lipid peroxidation in plants (Kim et al. 2017).
Silicon enhanced abiotic stress tolerance capacity which is linked to accumulation of photorespiratory enzymes (Nwugo and Huerta 2011). Silicon has been reported to prevent the damage of membrane caused by the formation of malondialdehyde (MDA) (Zhu et al. 2004) by regulating antioxidant defense system in plants (Zhu and Gong 2014). Silicon has also been found to decrease the concentration of MDA, the end-product of lipid peroxidation, in salt-stressed barley (Liang et al. 2003), maize (Moussa 2006), and grapevine (Soylemezoglu et al. 2009), thus may help to maintain membrane integrity and decrease membrane permeability.
Ma et al. (2016) found that silicon reduced H2O2 accumulation and increased expression of antioxidant enzyme genes in wheat under drought stress. Farooq et al. (2016) observed that silicon treatment increased the antioxidant capacity of rice plants under cadmium stress. Many experiments have assessed the impact of silicon on plants subjected to abiotic stress at the level of gene expression (Liu et al. 2014; Yin et al. 2016), and it is now becoming apparent that silicon may effect primary metabolism in higher plants (Sanglard et al. 2014).
Phytohormone Regulation
Phytohormones are known to play vital role in the ability of plants to acclimatize to different environment by different mechanism (Fahad et al. 2015). It has been reported that silicon application may enhance the plant tolerance to abiotic stresses by adjusting the level of phytohormones (Kim et al. 2014). Gibberellin regulates all aspects of plant life from seed germination to vegetative growth and fruiting (Colebrook et al. 2014). Endogenous bioactive GA1 and GA4 content increased when higher doses of silicon were applied to cucumber plants under salinity and drought stress (Hamayun et al. 2010). This clearly suggests that GAs play significant role in salt and drought stress alleviation. Silicon is known to increase plant growth, which can also be associated with the effect of exogenous gibberellin application to crop plants. It shows that silicon application activates gibberellin biosynthesis in order to maintain growth and impart stress tolerance.
Silicon is essential for life processes such as DNA replication (Okita and Volcani 1978; Martin-Jézéquel et al. 2000). Silicon was found to delay leaf senescence by activation of the cytokinin pathway in both silicon accumulating and non-accumulating plant species (Markovich et al. 2017). Silicon can increase plant tolerance to diseases by increasing the expression of genes involved in plant resistance to pests and diseases, enhancing the production of phenolic compounds, lignin and enzymes of phenylpropanoid pathway and by enhancing the concentration of polyphenol, antimicrobial flavonoids, and anthocyanin (Etesami and Jeong 2018).
Future Perspectives
Based on current knowledge, reviewed here, it can be stated that silicon is non-corrosive, non-pollutive element, and its excessive amount in soil does not pose any harm to plants. Silicon is a modest and major element of soil with enormous benefits to plants especially in the mitigation of abiotic stresses.
We suggest several future avenues of research:
-
1.
Researches are needed to investigate the effect of silicon on plant biochemistry and gene regulation under abiotic stresses.
-
2.
Nanotechnology-related applications can be used to explore possibilities for the formulation of silicon nanoparticles to elevate stress tolerance in plants.
-
3.
The resistance of plants to various environmental stresses can be enhanced by genetic modification of root ability to take up silicon and its accumulation in different plant parts. It will reduce the silicon expenditure in crop fields.
-
4.
Silicon possess multifaceted role in the regulation of genes involved in various physiological mechanism such as photosynthesis, secondary metabolism, polyamine biosynthesis, and transcription. Hence, there is a need of deep investigation related to the molecular level modulations triggered by silicon supplementation for physiological improvement of plant growth under stress conditions.
-
5.
The meta-analysis-related studies on the alleviation of abiotic stresses by silicon highlighted that most studies have focused on single species and single-stress models but there is a lack of studies which looked to compare effects between species or stress types.
-
6.
Farmers and researchers should be aware about the potential application of silicon in agriculture. Farmers should know the correct dose of silicon for particular crop and mode and time of silicon application on the crop plants to increase crop productivity.
Silicon is useful and sustainable fertilizer for crops facing a spectrum of environmental stresses. The application of silicon in the agriculture sector can improve food security by raising crop tolerance to adverse environmental conditions by enhancing adaptability of crops to different stresses. Silicon may be complementary option in silicon-deficient areas as silicon fertilizer can provide economic as well as ecological benefits.
References
Abbas T, Balal RM, Shahid MA, Pervez MA, Ayyub CM, Aqueel MA, Javaid MM (2015) Silicon-induced alleviation of NaCl toxicity in okra (Abelmoschus esculentus) is associated with enhanced photosynthesis, osmoprotectants and antioxidant metabolism. Acta Physiol Plant 37:1–15
Adrees M, Ali S, Rizwan M, Zia-ur-Rehman M, Ibrahim M, Abbas F, Farid M, Qayyum MF, Irshad MK (2015) Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: a review. Ecotoxicol Environ Saf 119:186–197
Agarie S, Uchida H, Agata W, Kubota F, Kaufman PB (1998) Effects of silicon on transpiration and leaf conductance in rice plants (Oryza sativa L.). Plant Prod Sci 1:89–95
Ahmed M, Asif M, Hassan FU (2014) Augmenting drought tolerance in sorghum by silicon nutrition. Acta Physiol Plant 36:473–483
Al-Aghabary K, Zhu Z, Shi QH (2004) Influence of silicon supply on chloro- phyll content, chlorophyll II fluorescence and antioxidant enzyme activities in tomato plants under salt stress. J Plant Nutr 27:2101–2115
Al-aghabary K, Zhu Z, Shi Q (2005) Influence of silicon supply on chlorophyll content, chlorophyll fluorescence, and antioxidative enzyme activities in tomato plants under salt stress. J Plant Nutr 27:2101–2115
An Y, Liang Z (2013) Drought tolerance of Periploca sepium during seed germination: antioxidant defense and compatible solutes accumulation. Acta Physiol Plant 35:959–967
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Artyszak A, Gozdowski D, Kucińska K (2014) The effect of foliar fertilization with marine calcite in sugar beet. Plant Soil Environ 60:413–417
Ashraf M, Ahmad A, MeNeilly T (2001) Growth and photosynthetic charac-teristics in the pearl millet under water stress and the different potassium supply. Photosynthetica 39:389–394
Ashraf M, Rahmatullah AM, Ahmad R, Mujeeb F, Sarwar A, Ali L (2010) Alleviation of detrimental effects of NaCl by silicon nutrition in salt-sensitive and salt-tolerant genotypes of sugarcane (Saccharum officinarum L). Plant and Soil 326:381–391
Asmar SA, Castro EM, Pasqual M, Pereira FJ, Soares JDR (2013) Changes in leaf anatomy and photosynthesis of micropropagated banana plantlets under different silicon sources. Sci Hortic 161:328–332
Balakhnina T, Borkowska A (2013) Effects of silicon on plant resistanceto environmental stresses: review. Int Agrophys 27:225–232
Barber SA (1995) Soil nutrient bioavailability: a mechanistic approach. Wiley, New York
Baxter A, Mittler R, Suzuki N (2014) ROS as key players in plant stress signalling. J Exp Bot 65:1229–1240
Bityutskii N, Pavlovic J, Yakkonen K, Maksimović V, Nikolic M (2014) Contrasting effect of silicon on iron, zinc and manganese status and accumulation of metal-mobilizing compounds in micronutrient-deficient cucumber. Plant Physiol Biochem 74:205–211
Bodner G, Nakhforoosh A, Kaul HP (2015) Management of crop water under drought: a review. Agron Sustain Dev 35:401–442
Boursiac Y, Chen S, Luu DT, Sorieul M, van den Dries N, Maurel C (2005) Early effects of salinity on water transport in Arabidopsis roots. Molecular and cellular features of aquaporin expression. Plant Physiol 139:790–805
Boursiac Y, Prak S, Boudet J, Postaire O, Luu DT, Tournaire-Roux C (2008) The response of Arabidopsis root water transport to a challenging environment implicates reactive oxygen species-and phosphorylation-dependent internalization of aquaporins. Plant Signal Behav 3:1096–1098
Bradbury M, Ahmad R (1990) The effect of silicon on the growth of Prosopis juliflora growing in saline soil. Plant and Soil 125:71–74
Cakmak I (2001) Plant nutrition research: priorities to meet human needs for food in sustainable ways. Plant and Soil 247:3–24
Carmen B, Roberto D (2011) Soil bacteria support and protect plants against abiotic stresses. In: Shan A (ed) Abiotic stress in plants mechanisms and adaptations. InTech, Croatia, pp 143–170
Chen J, Caldwell RD, Robinson CA, Steinkamp R (2000) Silicon: the estranged medium element. Bulletin 341, Institute of Food and Agricultural Sciences, University of Florida
Chen W, Yao X, Cai K, Chen J (2011) Silicon alleviates drought stress of rice plants by improving plant water status, photosynthesis and mineral nutrient absorption. Biol Trace Elem Res 142:67–76
Chinnusamy V, Jagendorf A, Zhu JK (2005) Understanding and improving salt tolerance in plants. Crop Sci 45:437–448
Colebrook EH, Thomas SG, Phillips AL (2014) The role of gibberellin signalling in plant responses to abiotic stress. J Exp Biol 217:67–75
Commuri PD, Jones RJ (2001) High temperatures during endosperm cell division in maize. A genotypic comparison under in vitro and field conditions. Crop Sci 41:1122–1130
Cooke J, Leishman MR (2011) Silicon concentration and leaf longevity: is silicon a player in the leaf dry mass spectrum? Funct Ecol 25:1181–1188
Cooke J, Leishman MR (2016) Consistent alleviation of abiotic stress with silicon addition: a meta-analysis. Funct Ecol 30:1340–1357
Coskun D, Britto DT, Huynh WQ, Kronzucker HJ (2016) The role of silicon in higher plants under salinity and drought stress. Front Plant Sci 7:1072. https://doi.org/10.3389/fpls.2016.01072
Crusciol CAC (2009) Effects of silicon and drought stress on tuber yield and leaf biochemical characteristics in potato. Crop Sci 49:949–954
da Cunha KPV, do Nascimento CWA (2009). Silicon effects on metal tolerance and structural changes in maize (Zea mays L.) grown on a cadmium and zinc enriched soil. Water Air and Soil Pollution. 197: 323–330. https://doi.org/10.1007/s11270-008-9814-9
Dallagnol LJ, Rodrigues FA, DaMatta FM, Mielli MVB, Pereira SC (2011) Deficiency in silicon uptake affects cytological, physiological and biochemical events in the rice–Bipolaris oryzae interaction. Phytopathology 101:92–104
Debona D, Rodrigues FA, Datnoff LE (2017) Silicon's role in abiotic and biotic plant stresses. Annu Rev Phytopathol 55:85–107
Epstein E (1994) The anomaly of silicon in plant biology. Proc Natl Acad Sci U S A 91:11–17
Epstein E (1999) Silicon. Annu Rev Plant Physiol Plant Mol Biol 50:641–664
Epstein E (2009) Silicon: its manifold roles in plants. Ann Appl Biol 155:155–160
Epstein E, Bloom AJ (2005) Mineral nutrition of plants: principles and perspectives, 2nd edn. Sinuar Associates, Sunderland, MA, pp 201–240
Eraslan F, Ali I, David J, Gunes A (2008) Interactive effects of salicylic acid and silicon on oxidative damage and antioxidant activity in spinach (Spinacia oleracea L cv Matador) grown under boron toxicity and salinity. Plant Growth Regul 55:207–219
Etesami H, Beattie GA (2017) Plant-microbe interactions in adaptation of agricultural crops to abiotic stress conditions. In: Probiotics and plant health. Springer, Berlin, pp 163–200
Etesami H, Jeong BR (2018) Silicon (Si): review and future prospects on the action mechanisms in alleviating biotic and abiotic stresses in plants. Ecotoxicol Environ Saf 147:881–896
Exley C (2015) A possible mechanism of biological silicification in plants. Front Plant Sci 6:853. https://doi.org/10.3389/fpls.2015.00853
Fahad S, Hussain S, Matloob A, Khan FA, Khaliq A, Saud S, Huang J (2015) Phytohormones and plant responses to salinity stress: a review. Plant Growth Regul 75:391–404
FAO (1998) World Agricultural Center, FAOSTAT agricultural statistic data—base gateway
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. In: Sustainable agriculture. Springer, Berlin, pp 153–188
Farooq MA, Detterbeck A, Clemens S, Dietz K-J (2016) Silicon-induced reversibility of cadmium toxicity in rice. J Exp Bot 67:3573–3585
Feng JP, Shi QH, Wang XF (2009) Effects of exogenous silicon on photosynthetic capacity and antioxidant enzyme activities in chloroplast of cucumber seedlings under excess manganese. Agric Sci China 8:40–50
Feng J, Shi Q, Wang X, Wei M, Yang F, Xu H (2010) Silicon supplementation ameliorated the inhibition of photosynthesis and nitrate metabolism by cadmium (Cd) toxicity in Cucumis sativus L. Sci Hortic 123:521–530
Frantz JM, Khandekar S, Leisner S (2011) Silicon differentially influences copper toxicity response in silicon-accumulator and non-accumulator species. J Am Soc Hort Sci 136:329–338
Gao X, Zou C, Wang L, Zhang F (2005) Silicon improves water use efficiency in maize plants. J Plant Nutr 27:1457–1470
Gao Z, He X, Zhao B, Zhou C, Liang Y, Ge R (2010) Overexpressing a putative aquaporin gene from wheat, TaNIP, enhances salt tolerance in transgenic Arabidopsis. Plant Cell Physiol 51:767–775
Gibson LR, Paulsen GM (1999) Yield components of wheat grown under high temperature stress during reproductive growth. Crop Sci 39:1841–1184
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169:30–39
Gong H, Chen K (2012) The regulatory role of silicon on water relations, photosynthetic gas exchange and carboxylation activities of wheat leaves in field drought conditions. Acta Physiol Plant 34:1589–1594
Gong HJ, Chen KM, Chen GC, Wang SM, Zhang CI (2003) Effects of silicon on growth of wheat under drought. J Plant Nutr 26:1055–1063
Gong H, Zhu X, Chen K, Wang S, Zhang C (2005) Silicon alleviates oxidative damage of wheat plants in pots under drought. Plant Sci 169:313–321
Gottardi S, Iacuzzo F, Tomasi N, Cortella G, Manzocco L, Pinton R (2012) Beneficial effects of silicon on hydroponically grown corn salad (Valerianella locusta L.) plants. Plant Physiol Biochem 56:14–23
Greenway H, Munns R (1980) Mechanism of salt tolerance in nonhalophytes. Annu Rev Plant Physiol 31:149–190
Gunes AA, Bagci IEG, Pilbeam DJ (2007) Silicon-mediated changes of some physiological and enzymatic parameters symptomatic for oxidative stress in spinach and tomato grown in sodic-B toxic soil. Plant and Soil 290:103–114
Gunes A, Kadioglu YK, Pilbeam DJ, Inal A, Coban S, Aksu A (2008a) Influence of silicon on sunflower cultivars under drought stress, II: essential and nonessential element uptake determined by polarized energy dispersive X-ray fluorescence. Commun Soil Sci Plant Anal 39:1904–1927
Gunes A, Pilbeam DJ, Inal A, Coban S (2008b) Influence of silicon on sunflower cultivars under drought stress, I: growth, antioxidant mechanisms, and lipid peroxidation. Commun Soil Sci Plant Anal 39:1885–1903
Guntzer F, Keller C, Meunier JD (2011) Benefits of plant silicon for crops: a review. Agron Sustain Dev 32:201–213
Guntzer F, Keller C, Poulton PR, McGrath SP, Meunier JD (2012) Long-term removal of wheat straw decreases soil amorphous silica at Broadbalk, Rothamsted. Plant and Soil 352:173–184
Guo W, Hou YL, Wang SG, Zhu YG (2005) Effect of silicate on the growth and arsenate uptake by rice (Oryza sativa L.) seedlings in solution culture. Plant and Soil 272:173–181
Hajiboland R, Cherghvareh L, Dashtebani F (2016) Effect of silicon supplementation on wheat plants under salt stress. J Plant Proc Funct 5:1–12
Hamayun M, Sohn EY, Khan SA, Shinwari ZK, Khan AL, Lee IJ (2010) Silicon alleviates the adverse effects of salinity and drought stress on growth and endogenous plant growth hormones of soybean (Glycine max L.). Pak J Bot 42:1713–1722
Hashemi A, Abdolzadeh A, Sadeghipour HR (2010) Beneficial effects of silicon nutrition in alleviating salinity stress in hydroponically grown canola, Brassica napus L. plants. J Soil Sci Plant Nutr 56:244–253
Hattori T, Inanaga S, Tanimoto E, Lux A, Luxová M, Sugimoto Y (2003) Silicon induced changes in viscoelastic properties of sorghum root cell walls. Plant Cell Physiol 44:743–749
Hattori T, Inanaga H, Araki H, An P, Morita S, Luxova M, Lux A (2005) Application of silicon enhanced drought tolerance in Sorghum bicolor. Physiol Plant 123:459–466
Hattori T, Sonobe K, Araki H, Inanaga S, An P, Morita S (2008) Silicon application by sorghum through the alleviation of stress-induced increase in hydraulic resistance. J Plant Nutr 31:1482–1495
Henriet C, Draye X, Oppitz I, Swennen R, Delvaux B (2006) Effects, distribution and uptake of silicon in banana (Musa spp.) under controlled conditions. Plant and Soil 287:359–374
Hernandez JA, Jim-enez A, Mullineaux P, Sevilla F (2000) Tolerance of pea (Pisum sativum L.) to long-term salt stress is associated with induction of antioxidant defenses. Plant Cell Environ 23:853–862
Hernandez-Apaolaza L (2014) Can silicon partially alleviate micronutrient deficiency in plants? A review. Planta 240:447–458
Hodson MJ, White PJ, Mead A, Broadley MR (2005) Phylogenetic variation in the silicon composition of plants. Ann Bot 96:1027–1046
Hu Y, Schmidhalter U (2005) Drought and salinity: a comparison of their effects on mineral nutrition of plants. J Plant Nutr Soil Sci 168:541–549
Hull RJ (2004) Scientists start to recognize silicon’s beneficial effects. Turf Grass Trends 8:69–73
Imtiaz M, Rizwan MS, Mushtaq MA, Ashraf M, Shahzad SM, Yousaf B, Saeed DA, Rizwan M, Nawaz MA, Mehmood S, Tu S (2016) Silicon occurrence, uptake, transport and mechanisms of heavy metals, minerals and salinity enhanced tolerance in plants with future prospects: a review. J Environ Manage 183:521–529
Islam MM, Ahmed M, Mahaldar D (2005) In vitro callus induction andplant regeneration in seed explants of rice (Oryza sativa L.). Res J Agric Biol Sci 1:72–75
Jewell MC, Campbell BC, Godwin ID (2010) Transgenic plants for abiotic stress resistance. In: Transgenic crop plants. Springer, Berlin, pp 67–132
John VS, Cabot C, Poschenrieder C, Barcelo J (2003) Efficient leaf ion partitioning, an overriding condition for abscisic acid-controlled stomatal and leaf growth responses to NaCl salinization in two legumes. J Exp Bot 54:2111–2119
Kalra N, Jain MC, Joshi HC, Chaudhary R, Sushilkumar PH, Sharma SK, Kumar V, Kumar R, Harit RC, Khan SA, Hussain MJ (2003) Soil properties and crop productivity as influenced by fly ash incorporation in soil. Environ Monit Assess 87:93–109
Kaya C, Tuna L, Higgs D (2006) Effect of silicon on plant growth and mineral nutrition of maize grown under water-stress conditions. J Plant Nutr 29:1469–1480
Kaya C, Tuna AL, Sonmez O, Ince F, Higgs D (2010) Mitigation effects of silicon on maize plants grown at high zinc. J Plant Nutr 32:1788–1798
Keles Y, Oncel I, Yenice N (2004) Relationship between boron content and antioxidant compounds in Citrus leaves taken from fields with different water source. Plant and Soil 265:343–353
Keller C, Rizwan M, Davidian JC, Pokrovsky OS, Bovet N, Chaurand P, Meunier JD (2015) Effect of silicon on wheat seedlings (Triticum turgidum L.) grown in hydroponics and exposed to 0 to 30 μM Cu. Planta 241:847–860
Khan MA, Ungar IA, Showalter AM (2000) Effects of sodium chloride treatments on growth and ion accumulation of the halophyte Haloxylon recurvum. Commun Soil Sci Plant Anal 31:2763–2774
Kim YH, Khan AL, Shinwari ZK, Kim DH, Waqas M, Kamran M (2012) Silicon treatment to rice (Oryza sativa L. cv. ‘Gopumbyeo’) plants during different growth periods and its effects on growth and grain yield. Pak J Bot 44:891–897
Kim YH, Khan AL, Waqas M, Shim JK, Kim DH, Lee KY, Lee IJ (2014) Silicon application to rice root zone influenced the phytohormonal and antioxidant responses under salinity stress. J Plant Growth Regul 33:137–149
Kim YH, Khan AL, Waqas M, Shahzad R, Lee IJ (2016). Silicon mediated mitigation of wounding stress acts by up-regulating the rice antioxidant system. Cereal Research Communications. 44: 111–121.
Kim YH, Khan AL, Waqas M, Lee IJ (2017) Silicon regulates antioxidant activities of crop plants under abiotic-induced oxidative stress: a review. Front Plant Sci 8:510. https://doi.org/10.3389/fpls.2017.00510
Kingston G (2008) Silicon fertilisers—requirements and field experiences. In: Silicon in Agriculture 4th International Conference, 31 October, Port Edward, South Africa, p 52
Li YC, Alva AK, Summer ME (1989) Response of cotton cultivars to aluminium in solutions with varying silicon concentrations. J Plant Nutr 12:881–892
Li CH, Chu TD, Liu XB, Yang Q (1999) Silicon nutrition effects and its study and application development in China. In: Proceedings of Symposium of Plant Nutrition, Shaanxi Science and Technology Press, China, pp 329–333
Li J, Leisner SM, Frantz J (2008) Alleviation of copper toxicity in Arabidopsis thaliana by silicon addition to hydroponic solutions. J Am Soc Hort Sci 133:670–677
Li H, Zhu Y, Hu Y, Han W, Gong H (2015) Beneficial effects of silicon in alleviating salinity stress of tomato seedlings grown under sand culture. Acta Physiol Plant 37:1–9
Liang YC (1999) Effects of silicon on enzyme activity and sodium, potassium and calcium concentration in barley under salt stress. Plant and Soil 29:217–224
Liang YC, Chen Q, Liu Q, Zhang WH, Ding RX (2003) Exogenous silicon (Si) increases antioxidant enzyme activity and reduces lipid peroxidation in roots of salt-stressed barley (Hordeum vulgare L.). J Plant Physiol 160:1157–1164
Liang Y, Si J, Romheld V (2005) Silicon uptake and transport is an active process in Cucumis sativus. New Phytol 167:797–804
Liang YC, Hua H, Zhu YG, Zhang J, Cheng C, Romheld V (2006) Importance of plant species and external silicon concentration to active silicon uptake and transport. New Phytol 172:63–67
Liang YC, Sun WC, Zhu YG, Christie P (2007) Mechanisms of silicon-mediated alleviation of abiotic stresses in higher plants: a review. Environ Pollut 147:422–428
Liang Y, Nikolic M, Bélanger R, Gong H, Song A (2015) Effect of silicon on crop growth, yield and quality. In: Liang Y, Nikolic M, Bélanger R, Gong H, Song A (eds) Silicon in agriculture: from theory to practice. Springer, Dordrecht, pp 209–223
Liu P, Yin L, Deng X, Wang S, Tanaka K, Zhang S (2014) Aquaporin mediated increase in root hydraulic conductance is involved in silicon induced improved root water uptake under osmotic stress in Sorghum bicolor L. J Exp Bot 65:4747–4756
Lobato AKS, Coimbra GK, Neto MAM, Costa RCL, Santos Filho BG, Oliveira Neto CF, Luz LM, Barreto AGT, Pereira BWF, Alves GAR, Monteiro BS, Marochio CA (2009) Protective action of silicon on relations and photosynthetic pigments in pepper plants induced to water deficit. Res J Agric Biol Sci 4:617–623
Lovering TS, Engel C (1959) Significance of accumulator plants in rock weathering. Bull Geol Soc Am 70:781–800
Lux A, Luxová M, Abe J, Tanimoto E, Hattori T, Inanaga S (2003) The dynamics of silicon deposition in the sorghum root endodermis. New Phytol 158:437–441
Luyckx M, Hausman JF, Lutts S, Guerriero G (2017) Silicon and plants: current knowledge and technological perspectives. Front Plant Sci 8:411. https://doi.org/10.3389/fpls.2017.00411
Luz JMQ, Rodrigues CR, Goncalves MV, Coelho L (2008) The effect of silicate on potatoes in Minas Gerais, Brazil. IV Silicon in Agriculture Conference, 31 October, Universidade Federal de Uberlandia, Amazonas 4C 127 Uberlandia, Brazil, p 67
Lynch M (2008) Silicates in contemporary Australian farming: a 20 year review. IV Silicon in Agriculture Conference, 31 October, South Africa, North Coast Testing Services, Bellingen NSW 2454, Australia, p 49
Ma JF (2004) Role of silicon in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci Plant Nutr 50:11–18
Ma JF, Takahashi E (2002) Soil, fertilizer and plant silicon research in Japan. Elsevier, Amsterdam
Ma JF, Yamaji N (2008) Functions and transport of silicon in plants. Cell Mol Life Sci 65:3049–3057
Ma JF, Goto S, Tamai K, Ichii M (2001) Role of root hairs and lateral roots in silicon uptake by rice. Plant Physiol 127:1773–1780
Ma J, Cai H, He C, Zhang W, Wang L (2015) Ahemicellulose-bound form of silicon inhibits cadmium ion uptake in rice (Oryza sativa) cells. New Phytol 206:1063–1074
Ma D, Sun D, Wang C, Qin H, Ding H, Li Y (2016) Silicon application alleviates drought stress in wheat through transcriptional regulation of multiple antioxidant defense pathways. J Plant Growth Regul 35:1–10
Markovich O, Steiner E, Kouøil S, Tarkowski P, Aharoni A, Elbaum R (2017) Silicon promotes cytokinin biosynthesis and delays senescence in Arabidopsis and Sorghum. Plant Cell Environ 40:1189–1196
Marschner H (1995) Mineral nutrition of higher plants. Academic Press, London, p 16
Martin-Jézéquel V, Hildebrand M, Brzezinski MA (2000) Silicon metabolism in diatoms: implications for growth. J Phycol 36:821–840
Matichenkov VV, Calvert DV (2002) Silicon as a beneficial element for sugarcane. J Am Soc Sugar Cane Technol 22:21–30
Mcginnity P (2015) Silicon and its role in crop production. PhD thesis. http://planttuff.com/wpcontentwpcontent/uploads/2015/12/silicon-agricultureiiterature-rvw-1.pdf
McKersie BD, Chen Y, De Beus M, Bowley SR, Bowler C (1993) Superoxide dismutase enhances tolerance of freezing stress in transgenic alfalfa (Medicago sativa L). Plant Physiol 103:1155–1163
Meena KK, Sorty AM, Bitla UM, Choudhary K, Gupta P, Pareek A, Singh DP, Prabha R, Sahu PK, Gupta VK, Singh HB, Krishanani KK, Minhas PS (2017) Abiotic stress responses and microbe-mediated mitigation in plants: the omics strategies. Front Plant Sci 8:172. https://doi.org/10.3389/fpls.2017.00172
Miao BH, Han XG, Zhang WH (2010) The ameliorative effect of silicon on soybean seedlings grown in potassium-deficient medium. Ann Bot 105:967–973
Ming DF, Pei ZF, Naeem MS, Gong HJ, Zhou WJ (2012) Silicon alleviates PEG-induced water-deficit stress in upland rice seedlings by enhancing osmotic adjustment. J Agron Crop Sci 198:14–26
Mitra GN (2015) Regulation of nutrient uptake by plants. Springer, New Delhi
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Monjardino P, Smith AG, Jones RJ (2005) Heat stress effects on protein ac-cumulation of maize endosperm. Crop Sci 45:1203–1210
Moussa HR (2006) Influence of exogenous application of silicon on physiological response of salt-stressed maize (Zea mays L.). Int J Agric Biol 8:293–297
Nagajyoti PC, Lee KD, Sreekanth TVM (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:199–216
Neumann D, Zur Nieden U (2001) Silicon and heavy metal tolerance of higher plants. Phytochemistry 56:685–692
Nwugo CC, Huerta AJ (2011) The effect of silicon on the leaf proteome of rice (Oryza sativa L.) plants under cadmium-stress. J Proteome Res 10:518–528
Ohyama N (1985) Amelioration of cold weather damage of rice by silicate fertilizer application. Agric Hortic J 60:1385–1389
Okita TH, Volcani BE (1978) Role of silicon in diatom metabolism IX. Differential synthesis of DNA polymerases and DNA-binding proteins during silicate starvation and recovery in Cylindrotheca fusiformis. Biochim Biophys Acta 519:76–86
Owino-Gerroh C, Gascho G (2005) Effect of silicon on low pH soil phosphorus sorption and on uptake and growth of maize. Commun Soil Sci Plant Anal 35:2369–2378
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349
Pascual MB, Echevarria V, Gonzalo MJ, Hernández-Apaolaza L (2016) Silicon addition to soybean (Glycine max L.) plants alleviates zinc deficiency. Plant Physiol Biochem 108:132–138
Pavlovic J, Samardzic J, Maksimovic V, Timotijevic G, Stevic N, Laursen KH, Hansen TH, Husted S, Schjoerring JK, Liang Y, Nikolic M (2013) Silicon alleviates iron deficiency in cucumber by promoting mobilization of iron in the root apoplast. New Phytol 198:1096–1107
Pei ZF, Ming DF, Liu D, Wan GL, Geng XX, Gong HJ, Zhou WJ (2010) Silicon improves the tolerance of water-deficit stress induced by polyethylene glycol in wheat (Triticum aestivum L.) seedlings. J Plant Growth Regul 29:106–115
Pich A, Scholz G, Stephan UW (1994) Iron-dependent changes of heavy metals, nicotianamine, and citrate in different plant organs and in the xylem exudate of two tomato genotypes. Nicotianamine as possible copper translocator. Plant and Soil 165:189–196
Pottosin I, Shabala S (2014) Polyamines control of cation transport across plant membranes: implications for ion homeostasis and abiotic stress signaling. Front Plant Sci 5:154. https://doi.org/10.3389/fpls.2014.00154
Quinet M, Ndayiragije A, Lefèvre I, Lambillotte B, Dupont-Gillain CC, Lutts S (2010) Putrescine differently influences the effect of salt stress on polyamine metabolism and ethylene synthesis in rice cultivars differing in salt resistance. J Exp Bot 61:2719–2733
Rédei GP (2008) Silicon (Si). In: Encyclopedia of genetics, genomics, proteomics, and informatics. Springer, Berlin, p 1817
Reezi S, Babalar M, Kalantari S (2009) Silicon alleviates salt stress, decreases malondialdehyde content and affects petal color of salt stressed cut rose (Rosa xhybrida L.) Hot Lady. Afr J Biotechnol 8:1502–1508
Rellán-Álvarez R, Giner-Martínez-Sierra J, Orduna J, Orera I, Rodríguez-Castrillón JÁ, García-Alonso JI, Abadía J, Álvarez-Fernández A (2010) Identification of atri-iron (III), tri-citrate complex in the xylem sap of iron-deficient tomato resupplied with iron: new insights into plant iron long-distance transport. Plant Cell Physiol 51:91–102
Reynolds OL, Padula MP, Zeng R, Gurr GM (2016) Silicon: potential to promote direct and indirect effects on plant defense against arthropod pests in agriculture. Front Plant Sci 7:744. https://doi.org/10.3389/fpls.2016.00744
Rizwan M, Ali S, Ibrahim M, Farid M, Adrees M, Bharwana SA, Zia-ur-Rehman M, Qayyum MF, Abbas F (2015) Mechanisms of silicon-mediated alleviation of drought and salt stress in plants: a review. Environ Sci Pollut Res 22:15416–15431
Romero-Aranda MR, Jurado O, Cuartero J (2006) Silicon alleviates the deleterious salt effect on tomato plant growth by improving plant water status. J Plant Physiol 163:847–855
Roy M, Wu R (2001) Arginine decarboxylase transgene expression and analysis of environmental stress tolerance in transgenic rice. Plant Sci 160:869–875
Sanglard LMVP, Martins SCV, Detmann KC (2014) Silicon nutrition alleviates the negative impacts of arsenic on the photosynthetic apparatus of rice leaves: an analysis of the key limitations of photosynthesis. Physiol Plant 152:355–366
Savant NK, Korndorfer GH, Datnoff LE, Snyder GH (1999) Silicon nutrition and sugarcane production: a review. J Plant Nutr 22:1853–1190
Savić J, Marjanović-Jeromela A (2013) Effect of silicon on sunflower growth and nutrient accumulation under low boron supply. Helia 36:61–68
Schaller J, Brackhage C, B€aucker E, Dudel EG (2013) UV-screening of grasses by plant silica layer? J Biosci 38:413–416
Schmidt RE, Zhang X, Chalmers DR (1999) Response of photosynthesis and superoxide dismutase to silica applied to creeping bent-grass grown under two fertility levels. J Plant Nutr 22:1763–1773
Shen X, ZhouY DL, Li Z, Eneji AE, Li J (2010) Silicon effects on photosynthesis and antioxidant parameters of soybean seedlings under drought and ultraviolet-B radiation. J Plant Physiol 167:1248–1252
Shen X, Li Z, Duan L, Eneji AE, Li J (2014) Silicon effects on the partitioning of mineral elements in soybean seedlings under drought and ultraviolet-B radiation. J Plant Nutr 37:828–836
Shi X, Zhang C, Wang H, Zhang F (2005) Effect of Si on the distribution of cd in rice seedlings. Plant and Soil 272:53–60
Shrivastava P, Kumar R (2015) Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22:123–131
Sonobe K, Hattori T, An P, Tsuji W, Eneji AE, Kobayashi S, Kawamura Y, Tanaka K, Inanaga S (2010) Effect of silicon application on sorghum root responses to water stress. J Plant Nutr 34:71–82
Soundararajan P, Sivanesan I, Jana S, Jeong BR (2014) Influence of silicon supplementation on the growth and tolerance to high temperature in Salvia splendens. Hortic Environ Biotechnol 55:271–279
Soundararajan P, Manivannan A, Jeong BR (2016) Regulatory mechanisms by silicon to overcome the salinity-induced imbalance of essential nutrient elements, silicon in plants: advances and future prospects. CRC, Boca Raton, FL, pp 47–66
Soundararajan P, Manivannan A, Cho YS, Jeong BR (2017) Exogenous supplementation of silicon improved the recovery of hyperhydric shoots in Dianthus caryophyllus L. by stabilizing the physiology and protein expression. Front Plant Sci 8:738. https://doi.org/10.3389/fpls.2017.00738
Soylemezoglu G, Demir K, Inal A, Gunes A (2009) Effect of silicon on antioxidant and stomatal response of two grapevine (Vitis vinifera L.) rootstocks grown in boron toxic, saline and boron toxic-saline soil. Sci Hortic 123:240–246
Steponkus PL (1984) Role of the plasma membrane in freezing injury and cold acclimation. Annu Rev Plant Physiol 35:543–584
Struyf E, Conley DJ (2008) Silica: an essential nutrient in wetland biogeochemistry. Front Ecol Environ 7:88–94
Sutka M, Li G, Boudet J, Boursiac Y, Doumas P, Maurel C (2011) Natural variation of root hydraulics in Arabidopsis grown in normal and salt-stressed conditions. Plant Physiol 155:1264–1267
Sytar O, Kumar A, Latowski D, Kuczynska P, Strzałka K, Prasad MNV (2013) Heavy metal-induced oxidative damage, defense reactions and detoxification mechanisms in plants. Acta Physiol Plant 35:985–999
Takahashi N, Kurata K (2007) Relationship between transpiration and silica content of the rice panicle under elevated atmospheric carbon dioxide concentration. J Agric Meteorol 63:89–94
Tang W, Newton RJ, Li C, Charles TM (2007) Enhanced stress tolerance in transgenic pine expressing the pepper CaPF1 gene is associated with the polyamine biosynthesis. Plant Cell Rep 26:115–124
Torabi F, Majd A, Enteshari S (2015) The effect of silicon on alleviation of salt stress in borage (Borago officinalis L.). Soil Sci Plant Nutr 61:788–798
Trenholm LE, Datnoff LE, Nagara RT (2004) Influence of silicon on drought and shade tolerance of St. Augustine grass. Hort Technol 14:487–490
Tripathi DK, Singh VP, Kumar D, Chauhan DK (2012) Impact of exogenous silicon addition on chromium uptake, growth, mineral elements, oxidative stress, antioxidant capacity, and leaf and root structures in rice seedlings exposed to hexavalent chromium. Acta Physiol Plant 34:279–289
Tripathi DK, Singh S, Singh VP, Prasad SM, Dubey NK, Chauhan DK (2017) Silicon nanoparticles more effectively alleviated UV-B stress than silicon in wheat (Triticum aestivum) seedlings. Plant Physiol Biochem 110:70–81
Tuna AL, Kaya C, Higgs D, Murillo-Amador B, Aydemir S, Girgin AR (2008) Silicon improves salinity tolerance in wheat plants. Environ Exp Bot 62:10–16
Umar SM (2002) Genotypic differences in yield and quality of groundnut as affected by potassium nutrition under erratic rainfall conditions. J Plant Nutr 25:1549–1562
Vaculík M, Lux A, Luxová M, Tanimoto E, Lichtscheidl I (2009) Silicon mitigates cadmium inhibitory effects in young maize plants. Environ Exp Bot 67:52–58
Van Bockhaven J, De Vleesschauwer D, Höfte M (2013) Towards establishing broad-spectrum disease resistance in plants: silicon leads the way. J Exp Bot 64:1281–1129
Veatch-Blohm ME (2007) Principles of plant genetics and breeding. Crop Sci 47:1763
Wang YX, Stass A, Horst WJ (2004) Apoplastic binding of aluminum is involved in silicon-induced amelioration of aluminum toxicity in maize. Plant Physiol 136:3762–3770
Wang XD, Ou-yang C, Fan ZR, Gao S, Chen F, Tang L (2010) Effects of exogenous silicon on seed germination and antioxidant enzyme activities of Momordica charantia under salt stress. J Anim Plant Sci 6:700–708
Wang S, Liu P, Chen D, Yin L, Li H, Deng X (2015) Silicon enhanced salt tolerance by improving the root water uptake and decreasing the ion toxicity in cucumber. Front Plant Sci 6:759. https://doi.org/10.3389/fpls.2015.00759
Wang M, Gao L, Dong S, Sun Y, Shen Q, Guo S (2017) Role of silicon on plant pathogen interactions. Front Plant Sci 8:701. https://doi.org/10.3389/fpls.2017.00701
Wu X, Yu Y, Baerson SR, Song Y, Liang G, Ding C, Niu J, Pan Z, Zeng R (2017) Interactions between nitrogen and silicon in rice and their effects on resistance toward the brown plant hopper Nilaparvata lugens. Front Plant Sci 8:28. https://doi.org/10.3389/fpls.2017.00028
Yao D, Zhang X, Zhao X, Liu C, Wang C, Zhang Z (2011) Transcriptome analysis reveals salt-stressregulated biological processes and key pathways in roots of cotton (Gossypium hirsutum L.). Genomics 98:47–55
Yin L, Wang S, Li J, Tanaka K, Oka M (2013) Application of silicon improves salt tolerance through ameliorating osmotic and ionic stresses in the seedling of Sorghum bicolor. Acta Physiol Plant 35:3099–3107
Yin L, Wang S, Tanaka K, Fujihara S, Itai A, Den X (2016) Silicon mediated changes in polyamines participate in silicon-induced salt tolerance in Sorghum bicolor L. Plant Cell Environ 39:245–258
Yordanov I, Velikova V, Tsonev T (2003) Plant responses to drought and stress tolerance. Bulg J Plant Physiol (Special Issue) 187–206
You-Qiang FU, Hong S, Dao-Ming WU, Kun-Zheng CAI (2012) Silicon-mediated amelioration of Fe2+ toxicity in rice (Oryza sativa L.) roots. Pedosphere 22:795–802
Zhu Y, Gong H (2014) Beneficial effects of silicon on salt and drought tolerance in plants. Agron Sustain Dev 34:455–472
Zhu ZJ, Wei GQ, Li J, Qian QQ, Yu JQ (2004) Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci 167:527–533
Zuccarini P (2008) Effects of silicon on photosynthesis, water relations and nutrient uptake of Phaseolus vulgaris under NaCl stress. Biol Plant 52:157–160
Acknowledgements
Authors are thankful to Dr. Chanderdeep Tandon, Director, Amity Institute of Biotechnology, Amity University, Noida, for his persistent encouragement and valuable suggestions.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Malhotra, C., Kapoor, R.T. (2019). Silicon: A Sustainable Tool in Abiotic Stress Tolerance in Plants. In: Hasanuzzaman, M., Hakeem, K., Nahar, K., Alharby, H. (eds) Plant Abiotic Stress Tolerance. Springer, Cham. https://doi.org/10.1007/978-3-030-06118-0_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-06118-0_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-06117-3
Online ISBN: 978-3-030-06118-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)