Abstract
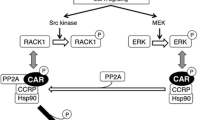
As the closest relative of the aforementioned PXR, the constitutive androstane/activator receptor (CAR, NR1I3) also governs the transcription of numerous hepatic drug-metabolizing enzymes and transporters in response to various xenobiotic exposures. Unlike most prototypical nuclear receptors, however, CAR can be activated via both direct ligand-binding and ligand-independent indirect mechanisms. Moreover, whereas CAR predominantly resides in the cytoplasm of primary hepatocytes in the absence of chemical stimulation, it simultaneously localizes in the nucleus of nearly all immortalized cell lines and is constitutively activated without chemical activation, making in vitro identification of CAR activators extremely challenging. In this chapter, we provide detailed step-by-step instructions for the application of two recently developed in vitro human (h) CAR activation assays: the hCAR1 + A-based luciferase assay in HepG2 cells and the adenovirus-based hCAR translocation assay in human primary hepatocytes. In combination, these assays are efficient in the identification of both direct and indirect activators of hCAR in vitro.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Honkakoski P et al (1998) The nuclear orphan receptor CAR-retinoid X receptor heterodimer activates the phenobarbital-responsive enhancer module of the CYP2B gene. Mol Cell Biol 18(10):5652–5658
Moore LB et al (2000) Orphan nuclear receptors constitutive androstane receptor and pregnane X receptor share xenobiotic and steroid ligands. J Biol Chem 275(20):15122–15127
Wang HB, LeCluyse EL (2003) Role of orphan nuclear receptors in the regulation of drug-metabolising enzymes. Clin Pharmacokinet 42(15):1331–1357
Qatanani M, Moore DD (2005) CAR, the continuously advancing receptor, in drug metabolism and disease. Curr Drug Metab 6(4):329–339
Yap KY, Chui WK, Chan A (2008) Drug interactions between chemotherapeutic regimens and antiepileptics. Clin Ther 30(8):1385–1407
Ma Q, Lu AY (2008) The challenges of dealing with promiscuous drug-metabolizing enzymes, receptors and transporters. Curr Drug Metab 9(5):374–383
Chakraborty S, Kanakasabai S, Bright JJ (2011) Constitutive androstane receptor agonist CITCO inhibits growth and expansion of brain tumour stem cells. Br J Cancer 104(3):448–459
Wada T, Gao J, Xie W (2009) PXR and CAR in energy metabolism. Trends Endocrinol Metab 20(6):273–279
Yamamoto Y et al (2004) The orphan nuclear receptor constitutive active/androstane receptor is essential for liver tumor promotion by phenobarbital in mice. Cancer Res 64(20):7197–7200
Huang W et al (2005) Xenobiotic stress induces hepatomegaly and liver tumors via the nuclear receptor constitutive androstane receptor. Mol Endocrinol 19(6):1646–1653
Gao J et al (2009) The constitutive androstane receptor is an anti-obesity nuclear receptor that improves insulin sensitivity. J Biol Chem 284(38):25984–25992
Dong B et al (2009) Activation of nuclear receptor CAR ameliorates diabetes and fatty liver disease. Proc Natl Acad Sci USA 106(44):18831–18836
Maglich JM et al (2004) The nuclear receptor CAR is a regulator of thyroid hormone metabolism during caloric restriction. J Biol Chem 279(19):19832–19838
Kawamoto T et al (1999) Phenobarbital-responsive nuclear translocation of the receptor CAR in induction of the CYP2B gene. Mol Cell Biol 19(9):6318–6322
Li H et al (2009) Nuclear translocation of adenoviral-enhanced yellow fluorescent protein-tagged-human constitutive androstane receptor (hCAR): a novel tool for screening hCAR activators in human primary hepatocytes. Drug Metab Dispos 37(5):1098–1106
Chen T et al (2010) A single amino acid controls the functional switch of human constitutive androstane receptor (CAR) 1 to the xenobiotic-sensitive splicing variant CAR3. J Pharmacol Exp Ther 332(1):106–115
Haché RJG et al (1999) Nucleocytoplasmic trafficking of steroid-free glucocorticoid receptor. J Biol Chem 274(3):1432–1439
Rowlands JC, Gustafsson J-Å (1997) Aryl hydrocarbon receptor-mediated signal transduction. Crit Rev Toxicol 27(2):109–134
Zelko I et al (2001) The peptide near the C terminus regulates receptor CAR nuclear translocation induced by xenochemicals in mouse liver. Mol Cell Biol 21(8):2838–2846
Maglich JM et al (2003) Identification of a novel human constitutive androstane receptor (CAR) agonist and its use in the identification of CAR target genes. J Biol Chem 278(19):17277–17283
Auerbach SS et al (2005) Retinoid X receptor-alpha-dependent transactivation by a naturally occurring structural variant of human constitutive androstane receptor (NR1I3). Mol Pharmacol 68(5):1239–1253
Auerbach SS et al (2007) CAR2 displays unique ligand binding and RXRalpha heterodimerization characteristics. Drug Metab Dispos 35(3):428–439
Arnold KA, Eichelbaum M, Burk O (2004) Alternative splicing affects the function and tissue-specific expression of the human constitutive androstane receptor. Nucl Recept 2(1):1
Lynch C et al (2013) Identification of novel activators of constitutive androstane receptor from FDA-approved drugs by integrated computational and biological approaches. Pharm Res 30(2):489–501
Wang H et al (2003) A novel distal enhancer module regulated by pregnane X receptor/constitutive androstane receptor is essential for the maximal induction of CYP2B6 gene expression. J Biol Chem 278(16):14146–14152
LeCluyse EL et al (2005) Isolation and culture of primary human hepatocytes. Methods Mol Biol 290:207–229
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Lynch, C., Li, H., Wang, H. (2014). Characterization of Constitutive Androstane Receptor (CAR) Activation. In: Caldwell, G., Yan, Z. (eds) Optimization in Drug Discovery. Methods in Pharmacology and Toxicology. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-742-6_11
Download citation
DOI: https://doi.org/10.1007/978-1-62703-742-6_11
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-741-9
Online ISBN: 978-1-62703-742-6
eBook Packages: Springer Protocols