Abstract
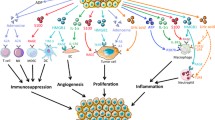
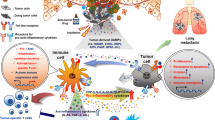
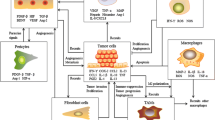
Damage-associated molecular pattern molecules (DAMPs) are proteins released from cells under stress due to nutrient deprivation, hypoxia, trauma, or treatment with chemotherapy, among a variety of other causes. When released, DAMPs activate innate immunity, providing a pathway to a systemic inflammatory response in the absence of infection. By regulating inflammation in the tumor microenvironment, promoting angiogenesis, and increasing autophagy with evasion of apoptosis, DAMPs facilitate cancer growth. DAMPs and DAMP receptors have a key role in melanoma pathogenesis. Due to their crucial role in the development of melanoma and chemoresistance, DAMPs represent intriguing targets at a time when novel treatments are desperately needed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Matzinger P (2002) The danger model: a renewed sense of self. Science 296:301–305
Lotze MT, Zeh HJ, Rubartelli A et al (2007) The grateful dead: damage-associated molecular pattern molecules and reduction/oxidation regulate immunity. Immunol Rev 220:60–81
Marenholz I, Heizmann CW, Fritz G (2004) S100 proteins in mouse and man: from evolution to function and pathology (including an update of the nomenclature). Biochem Biophys Res Commun 322:1111–1122
Leclerc E, Fritz G, Vetter SW, Heizmann CW (2009) Binding of S100 proteins to RAGE: an update. Biochim Biophys Acta 1793:993–1007
Ilg EC, Schafer BW, Heizmann CW (1996) Expression pattern of S100 calcium-binding proteins in human tumors. Int J Cancer 68:325–332
Maelandsmo GM, Florenes VA, Mellingsaeter T et al (1997) Differential expression patterns of S100A2, S100A4 and S100A6 during progression of human malignant melanoma. Int J Cancer 74:464–469
Helfman DM, Kim EJ, Lukanidin E, Grigorian M (2005) The metastasis associated protein S100A4: role in tumour progression and metastasis. Br J Cancer 92:1955–1958
Baudier J, Delphin C, Grunwald D et al (1992) Characterization of the tumor suppressor protein p53 as a protein kinase C substrate and a S100b-binding protein. Proc Natl Acad Sci U S A 89:11627–11631
Lin J, Yang Q, Wilder PT et al (2010) The calcium-binding protein S100B down-regulates p53 and apoptosis in malignant melanoma. J Biol Chem 285:27487–27498
Lin J, Yang Q, Yan Z et al (2004) Inhibiting S100B restores p53 levels in primary malignant melanoma cancer cells. J Biol Chem 279:34071–34077
Markowitz J, Chen I, Gitti R et al (2004) Identification and characterization of small molecule inhibitors of the calcium-dependent S100B-p53 tumor suppressor interaction. J Med Chem 47:5085–5093
Smith J, Stewart BJ, Glaysher S et al (2010) The effect of pentamidine on melanoma ex vivo. Anticancer Drugs 21:181–185
deBlacam C, Byrne C, Hughes E et al (2011) HOXC11-SRC-1 regulation of S100beta in cutaneous melanoma: new targets for the kinase inhibitor dasatinib. Br J Cancer 105:118–123
Eustace AJ, Crown J, Clynes M, O’Donovan N (2008) Preclinical evaluation of dasatinib, a potent Src kinase inhibitor, in melanoma cell lines. J Transl Med 6:53
Kluger HM, Dudek AZ, McCann C et al (2011) A phase 2 trial of dasatinib in advanced melanoma. Cancer 117:2202–2208
Hofmeister-Mueller V, Vetter-Kauczok CS, Ullrich R et al (2009) Immunogenicity of HLA-A1-restricted peptides derived from S100A4 (metastasin 1) in melanoma patients. Cancer Immunol Immunother 58:1265–1273
Hegyi Z, Zwicker S, Bureik D et al (2012) Vitamin D analog calcipotriol suppresses the Th17 cytokine-induced proinflammatory S100 “alarmins” psoriasin (S100A7) and koebnerisin (S100A15) in psoriasis. J Invest Dermatol 132:1416–1424
Godar DE, Landry RJ, Lucas AD (2009) Increased UVA exposures and decreased cutaneous vitamin D(3) levels may be responsible for the increasing incidence of melanoma. Med Hypotheses 72:434–443
Goodwin GH, Johns EW (1977) The isolation and purification of the high mobility group (HMG) nonhistone chromosomal proteins. Methods Cell Biol 16:257–267
Tang D, Kang R, Zeh HJ 3rd, Lotze MT (2011) High-mobility group box 1, oxidative stress, and disease. Antioxid Redox Signal 14:1315–1335
Tang D, Kang R, Zeh HJ 3rd, Lotze MT (2010) High-mobility group box 1 and cancer. Biochim Biophys Acta 1799:131–140
Ito N, DeMarco RA, Mailliard RB et al (2007) Cytolytic cells induce HMGB1 release from melanoma cell lines. J Leukoc Biol 81:75–83
Perez RP, Zhang P, Bosserhoff AK et al (2000) Expression of melanoma inhibitory activity in melanoma and nonmelanoma tissue specimens. Hum Pathol 31:1381–1388
Poser I, Golob M, Buettner R, Bosserhoff AK (2003) Upregulation of HMG1 leads to melanoma inhibitory activity expression in malignant melanoma cells and contributes to their malignancy phenotype. Mol Cell Biol 23:2991–2998
Dong Xda E, Ito N, Lotze MT et al (2007) High mobility group box I (HMGB1) release from tumor cells after treatment: implications for development of targeted chemoimmunotherapy. J Immunother 30:596–606
Pasheva EA, Ugrinova I, Spassovska NC, Pashev IG (2002) The binding affinity of HMG1 protein to DNA modified by cis-platin and its analogs correlates with their antitumor activity. Int J Biochem Cell Biol 34:87–92
Ulloa L, Ochani M, Yang H et al (2002) Ethyl pyruvate prevents lethality in mice with established lethal sepsis and systemic inflammation. Proc Natl Acad Sci U S A 99:12351–12356
Muller AJ, DuHadaway JB, Jaller D et al (2010) Immunotherapeutic suppression of indoleamine 2,3-dioxygenase and tumor growth with ethyl pyruvate. Cancer Res 70:1845–1853
Tang D, Kang R, Xiao W et al (2009) Quercetin prevents LPS-induced high-mobility group box 1 release and proinflammatory function. Am J Respir Cell Mol Biol 41:651–660
Rosner K, Ropke C, Pless V, Skovgaard GL (2006) Late type apoptosis and apoptosis free lethal effect of quercetin in human melanoma. Biosci Biotechnol Biochem 70:2169–2177
Camby I, Le Mercier M, Lefranc F, Kiss R (2006) Galectin-1: a small protein with major functions. Glycobiology 16:137R–157R
Vereecken P, Debray C, Petein M et al (2005) Expression of galectin-3 in primary and metastatic melanoma: immunohistochemical studies on human lesions and nude mice xenograft tumors. Arch Dermatol Res 296:353–358
Le QT, Shi G, Cao H et al (2005) Galectin-1: a link between tumor hypoxia and tumor immune privilege. J Clin Oncol 23:8932–8941
Lefranc F, Mathieu V, Kiss R (2011) Galectin-1-mediated biochemical controls of melanoma and glioma aggressive behavior. World J Biol Chem 2:193–201
Mourad-Zeidan AA, Melnikova VO, Wang H et al (2008) Expression profiling of Galectin-3-depleted melanoma cells reveals its major role in melanoma cell plasticity and vasculogenic mimicry. Am J Pathol 173:1839–1852
Inohara H, Raz A (1994) Effects of natural complex carbohydrate (citrus pectin) on murine melanoma cell properties related to galectin-3 functions. Glycoconj J 11:527–532
Platt D, Raz A (1992) Modulation of the lung colonization of B16-F1 melanoma cells by citrus pectin. J Natl Cancer Inst 84:438–442
Nangia-Makker P, Hogan V, Honjo Y et al (2002) Inhibition of human cancer cell growth and metastasis in nude mice by oral intake of modified citrus pectin. J Natl Cancer Inst 94:1854–1862
Calderwood SK, Khaleque MA, Sawyer DB, Ciocca DR (2006) Heat shock proteins in cancer: chaperones of tumorigenesis. Trends Biochem Sci 31:164–172
Shipp C, Weide B, Derhovanessian E, Pawelec G (2012) Hsps are up-regulated in melanoma tissue and correlate with patient clinical parameters. Cell Stress Chaperones 18(2):145–154
Ciocca DR, Cayado-Gutierrez N, Maccioni M, Cuello-Carrion FD (2012) Heat shock proteins (HSPs) based anti-cancer vaccines. Curr Mol Med 12(9):1183–1197
Pilla L, Patuzzo R, Rivoltini L et al (2006) A phase II trial of vaccination with autologous, tumor-derived heat-shock protein peptide complexes Gp96, in combination with GM-CSF and interferon-alpha in metastatic melanoma patients. Cancer Immunol Immunother 55:958–968
Testori A, Richards J, Whitman E et al (2008) Phase III comparison of vitespen, an autologous tumor-derived heat shock protein gp96 peptide complex vaccine, with physician’s choice of treatment for stage IV melanoma: the C-100-21 Study Group. J Clin Oncol 26:955–962
Liu KS, Ding WC, Wang SX et al (2012) The heat shock protein 90 inhibitor SNX-2112 inhibits B16 melanoma cell growth in vitro and in vivo. Oncol Rep 27:1904–1910
Ohshima Y, Tsukimoto M, Takenouchi T et al (2010) gamma-Irradiation induces P2X(7) receptor-dependent ATP release from B16 melanoma cells. Biochim Biophys Acta 1800:40–46
Zanetti M (2004) Cathelicidins, multifunctional peptides of the innate immunity. J Leukoc Biol 75:39–48
Kim JE, Kim HJ, Choi JM et al (2010) The antimicrobial peptide human cationic antimicrobial protein-18/cathelicidin LL-37 as a putative growth factor for malignant melanoma. Br J Dermatol 163:959–967
Territo MC, Ganz T, Selsted ME, Lehrer R (1989) Monocyte-chemotactic activity of defensins from human neutrophils. J Clin Invest 84:2017–2020
Fernandez LP, Milne RL, Pita G et al (2009) Human beta-defensins (HBD1 and HBD3) and malignant melanoma susceptibility. Melanoma Res 19:340–341
Mei HF, Jin XB, Zhu JY et al (2012) Beta-defensin 2 as an adjuvant promotes anti-melanoma immune responses and inhibits the growth of implanted murine melanoma in vivo. PLoS One 7:e31328
Abdelmohsen K, Gorospe M (2012) RNA-binding protein nucleolin in disease. RNA Biol 9(6):799–808
Mourmouras V, Cevenini G, Cosci E et al (2009) Nucleolin protein expression in cutaneous melanocytic lesions. J Cutan Pathol 36:637–646
Sparvero LJ, Asafu-Adjei D, Kang R et al (2009) RAGE (receptor for advanced glycation endproducts), RAGE ligands, and their role in cancer and inflammation. J Transl Med 7:17
Eichmuller S, Usener D, Jochim A, Schadendorf D (2002) mRNA expression of tumor-associated antigens in melanoma tissues and cell lines. Exp Dermatol 11:292–301
Leclerc E, Heizmann CW, Vetter SW (2009) RAGE and S100 protein transcription levels are highly variable in human melanoma tumors and cells. Gen Physiol Biophys 28 Spec No Focus:F65–F75
Masaki H, Okano Y, Sakurai H (1999) Generation of active oxygen species from advanced glycation end-products (AGEs) during ultraviolet light A (UVA) irradiation and a possible mechanism for cell damaging. Biochim Biophys Acta 1428:45–56
Abe R, Shimizu T, Sugawara H et al (2004) Regulation of human melanoma growth and metastasis by AGE-AGE receptor interactions. J Invest Dermatol 122:461–467
Raucci A, Cugusi S, Antonelli A et al (2008) A soluble form of the receptor for advanced glycation endproducts (RAGE) is produced by proteolytic cleavage of the membrane-bound form by the sheddase a disintegrin and metalloprotease 10 (ADAM10). FASEB J 22:3716–3727
Taguchi A, Blood DC, del Toro G et al (2000) Blockade of RAGE-amphoterin signalling suppresses tumour growth and metastases. Nature 405:354–360
Arumugam T, Ramachandran V, Gomez SB et al (2012) S100P-derived RAGE antagonistic peptide reduces tumor growth and metastasis. Clin Cancer Res 18:4356–4364
Zhu C, Anderson AC, Schubart A et al (2005) The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat Immunol 6:1245–1252
Chiba S, Baghdadi M, Akiba H et al (2012) Tumor-infiltrating DCs suppress nucleic acid-mediated innate immune responses through interactions between the receptor TIM-3 and the alarmin HMGB1. Nat Immunol 13:832–842
Wiener Z, Kohalmi B, Pocza P et al (2007) TIM-3 is expressed in melanoma cells and is upregulated in TGF-beta stimulated mast cells. J Invest Dermatol 127:906–914
Wu FH, Yuan Y, Li D et al (2010) Endothelial cell-expressed Tim-3 facilitates metastasis of melanoma cells by activating the NF-kappaB pathway. Oncol Rep 24:693–699
Fourcade J, Sun Z, Benallaoua M et al (2010) Upregulation of Tim-3 and PD-1 expression is associated with tumor antigen-specific CD8+ T cell dysfunction in melanoma patients. J Exp Med 207:2175–2186
Yu M, Wang H, Ding A et al (2006) HMGB1 signals through toll-like receptor (TLR) 4 and TLR2. Shock 26:174–179
Park JS, Svetkauskaite D, He Q et al (2004) Involvement of toll-like receptors 2 and 4 in cellular activation by high mobility group box 1 protein. J Biol Chem 279:7370–7377
Saint-Jean M, Knol AC, Nguyen JM et al (2011) TLR expression in human melanoma cells. Eur J Dermatol 21:899–905
Yu N, Zhang S, Zuo F et al (2009) Cultured human melanocytes express functional toll-like receptors 2-4, 7 and 9. J Dermatol Sci 56:113–120
Mittal D, Saccheri F, Venereau E et al (2010) TLR4-mediated skin carcinogenesis is dependent on immune and radioresistant cells. EMBO J 29:2242–2252
Matijevic T, Pavelic J (2010) Toll-like receptors: cost or benefit for cancer? Curr Pharm Des 16:1081–1090
Huang B, Zhao J, Unkeless JC et al (2008) TLR signaling by tumor and immune cells: a double-edged sword. Oncogene 27:218–224
Nunez NG, Andreani V, Crespo MI et al (2012) IFNbeta produced by TLR4-activated tumor cells is involved in improving the antitumoral immune response. Cancer Res 72:592–603
Tittarelli A, Gonzalez FE, Pereda C et al (2012) Toll-like receptor 4 gene polymorphism influences dendritic cell in vitro function and clinical outcomes in vaccinated melanoma patients. Cancer Immunol Immunother 61(11):2067–2077
Zhu XM, Yao YM, Liang HP et al (2011) High mobility group box-1 protein regulate immunosuppression of regulatory T cells through toll-like receptor 4. Cytokine 54:296–304
Pashenkov M, Goess G, Wagner C et al (2006) Phase II trial of a toll-like receptor 9-activating oligonucleotide in patients with metastatic melanoma. J Clin Oncol 24:5716–5724
Yang HZ, Cui B, Liu HZ et al (2009) Blocking TLR2 activity attenuates pulmonary metastases of tumor. PLoS One 4:e6520
Yan J, Hua F, Liu HZ et al (2012) Simultaneous TLR2 inhibition and TLR9 activation synergistically suppress tumor metastasis in mice. Acta Pharmacol Sin 33:503–512
Sims GP, Rowe DC, Rietdijk ST et al (2010) HMGB1 and RAGE in inflammation and cancer. Annu Rev Immunol 28:367–388
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media, New York
About this protocol
Cite this protocol
Boone, B.A., Lotze, M.T. (2014). Targeting Damage-Associated Molecular Pattern Molecules (DAMPs) and DAMP Receptors in Melanoma. In: Thurin, M., Marincola, F. (eds) Molecular Diagnostics for Melanoma. Methods in Molecular Biology, vol 1102. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-727-3_29
Download citation
DOI: https://doi.org/10.1007/978-1-62703-727-3_29
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-726-6
Online ISBN: 978-1-62703-727-3
eBook Packages: Springer Protocols