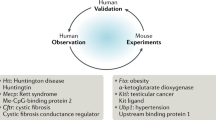
Abstract
The identification of causative genes underlying biomedically relevant phenotypes, particularly complex multigenic traits, is of vital interest to modern medicine. Using genome-wide association analysis, many studies have successfully identified thousands of loci (called quantitative trait loci or QTL), some of these associating with drug response phenotypes. However, the determination and validation of putative genes has been much more challenging. The actions of drugs, both efficacious and deleterious, are complex phenotypes that are controlled or influenced in part by genetic mechanisms.
Investigation for genetic correlates of complex traits and pharmacogenetic traits is often difficult to perform in human studies due to cost, availability of relevant sample population, and limited ability to control for environmental effects. These challenges can be circumvented with the use of mouse models for pharmacogenetic studies. In addition, the mouse can be treated at sub- and supratherapeutic doses and subjected to invasive procedures, which can facilitate measures of drug response phenotypes, making identification of pharmacogenetically relevant genes more feasible. The availability of multiple mouse genetic and phenotypic resources is an additional benefit to using the mouse for pharmacogenetic studies.
Here, we describe the contribution of animal models, specifically the mouse, towards the field of pharmacogenetics. In this chapter, we describe different mouse models, including the knockout mouse, recombinant mouse inbred strains, in vitro mouse cell-based assays, as well as novel experimental approaches like the Collaborative Cross recombinant mouse inbred panel, which can be applied to preclinical pharmacogenetics research. These approaches can be used to assess drug response phenotypes that are difficult to model in humans, thereby facilitating drug discovery, development, and application.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Zhang W, Dolan ME (2009) Use of cell lines in the investigation of pharmacogenetic loci. Curr Pharm Des 15(32):3782–3795
Bogaards JJ et al (2000) Determining the best animal model for human cytochrome P450 activities: a comparison of mouse, rat, rabbit, dog, micropig, monkey and man. Xenobiotica 30(12):1131–1152
Cheung C, Gonzalez FJ (2008) Humanized mouse lines and their application for prediction of human drug metabolism and toxicological risk assessment. J Pharmacol Exp Ther 327(2):288–299
Liggett SB (2004) Genetically modified mouse models for pharmacogenomic research. Nat Rev Genet 5(9):657–663
Hernandez D et al (2009) Deletion of the mouse Fmo1 gene results in enhanced pharmacological behavioural responses to imipramine. Pharmacogenet Genomics 19(4): 289–299
Henderson CJ et al (2000) Increased resistance to acetaminophen hepatotoxicity in mice lacking glutathione S-transferase Pi. Proc Natl Acad Sci USA 97(23):12741–12745
Eisener-Dorman AF et al (2009) Cautionary insights on knockout mouse studies: the gene or not the gene? Brain Behav Immun 23(3):318–324
Skarnes WC et al (2011) A conditional knockout resource for the genome-wide study of mouse gene function. Nature 474(7351):337–342
Darvasi A (1998) Experimental strategies for the genetic dissection of complex traits in animal models. Nat Genet 18(1):19–24
Zou F (2009) QTL mapping in intercross and backcross populations. Methods Mol Biol 573:157–173
Haston CK et al (2002) Bleomycin hydrolase and a genetic locus within the MHC affect risk for pulmonary fibrosis in mice. Hum Mol Genet 11(16):1855–1863
Drews E et al (2010) Quantitative trait loci contributing to physiological and behavioural ethanol responses after acute and chronic treatment. Int J Neuropsychopharmacol 13(2):155–169
Jones BC et al (1999) Quantitative-trait loci analysis of cocaine-related behaviours and neurochemistry. Pharmacogenetics 9(5):607–617
Boyle AE, Gill K (2001) Sensitivity of AXB/BXA recombinant inbred lines of mice to the locomotor activating effects of cocaine: a quantitative trait loci analysis. Pharmacogenetics 11(3):255–264
McClurg P et al (2006) Comparative analysis of haplotype association mapping algorithms. BMC Bioinformatics 7:61
Guo Y et al (2006) In silico pharmacogenetics: warfarin metabolism. Nat Biotechnol 24(5): 531–536
Harrill AH et al (2009) Mouse population-guided resequencing reveals that variants in CD44 contribute to acetaminophen-induced liver injury in humans. Genome Res 19(9):1507–1515
Zhang X et al (2011) In silico and in vitro pharmacogenetics: aldehyde oxidase rapidly metabolizes a p38 kinase inhibitor. Pharmacogenomics J 11(1):15–24
Kang HM et al (2008) Efficient control of population structure in model organism association mapping. Genetics 178(3):1709–1723
Pletcher MT et al (2004) Use of a dense single nucleotide polymorphism map for in silico mapping in the mouse. PLoS Biol 2(12):e393
McClurg P et al (2007) Genomewide association analysis in diverse inbred mice: power and population structure. Genetics 176(1):675–683
Grubb SC et al (2009) Mouse phenome database. Nucleic Acids Res 37(Database issue): D720–D730
Blake J.A., et al. (2009) The Mouse Genome Database genotypes::phenotypes. Nucleic Acids Res 37(Database issue):D712-9.
Olson H et al (2000) Concordance of the toxicity of pharmaceuticals in humans and in animals. Regul Toxicol Pharmacol 32(1):56–67
Jaeschke H et al (2002) Mechanisms of hepatotoxicity. Toxicol Sci 65(2):166–176
Rausch O (2006) High content cellular screening. Curr Opin Chem Biol 10(4):316–320
Bullen A (2008) Microscopic imaging techniques for drug discovery. Nat Rev Drug Discov 7(1):54–67
Abraham VC et al (2008) Application of a high-content multiparameter cytotoxicity assay to prioritize compounds based on toxicity potential in humans. J Biomol Screen 13(6): 527–537
Mayr LM, Bojanic D (2009) Novel trends in high-throughput screening. Curr Opin Pharmacol 9(5):580–588
Zock JM (2009) Applications of high content screening in life science research. Comb Chem High Throughput Screen 12(9):870–876
Zanella F et al (2010) High content screening: seeing is believing. Trends Biotechnol 28(5): 237–245
Taylor DL, Giuliano KA (2005) Multiplexed high content screening assays create a systems cell biology approach to drug discovery. Drug Discov Today Techn 2(2):149–154
Giuliano KA et al (2006) Systems cell biology based on high-content screening. Methods Enzymol 414:601–619
Abraham VC et al (2004) High content screening applied to large-scale cell biology. Trends Biotechnol 22(1):15–22
Shapiro HM (2003) Practical flow cytometry. Wiley, Hoboken
Huang RS et al (2007) Identification of genetic variants contributing to cisplatin-induced cytotoxicity by use of a genomewide approach. Am J Hum Genet 81(3):427–437
Shukla SJ et al (2009) Whole-genome approach implicates CD44 in cellular resistance to carboplatin. Hum Genomics 3(2): 128–142
Bleibel WK et al (2009) Identification of genomic regions contributing to etoposide-induced cytotoxicity. Hum Genet 125(2): 173–180
Marshak DR, Greenwalt DE (2007) Differentiating primary human cells in rapid-throughput discovery applications. Methods Mol Biol 356:121–128
Ward A, Tosh D (2010) Mouse cell culture: methods and protocols, 1st edn. Humana Press, New York
Freshney R (2005) Culture of animal cells—a manual of basic technique, 5th edn. Wiley, Hoboken, NJ
Kim TK, Eberwine JH (2010) Mammalian cell transfection: the present and the future. Anal Bioanal Chem 397(8):3173–3178
Churchill GA et al (2004) The Collaborative Cross, a community resource for the genetic analysis of complex traits. Nat Genet 36(11): 1133–1137
Aylor DL et al (2011) Genetic analysis of complex traits in the emerging collaborative cross. Genome Res 21:1213–1222
Krutzik PO, Nolan GP (2006) Fluorescent cell barcoding in flow cytometry allows high-throughput drug screening and signaling profiling. Nat Methods 3(5):361–368
Acknowledgments
The authors express their appreciation to Cristina Benton for valuable contributions and manuscript revision.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Frick, A., Suzuki, O., Butz, N., Chan, E., Wiltshire, T. (2013). In Vitro and In Vivo Mouse Models for Pharmacogenetic Studies. In: Innocenti, F., van Schaik, R. (eds) Pharmacogenomics. Methods in Molecular Biology, vol 1015. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-435-7_17
Download citation
DOI: https://doi.org/10.1007/978-1-62703-435-7_17
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-434-0
Online ISBN: 978-1-62703-435-7
eBook Packages: Springer Protocols