Abstract
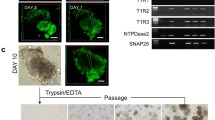
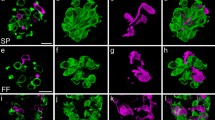
Establishment of primary and immortalized cultures of many cell types has facilitated efforts to understand the signals involved in proliferation and differentiation and yielded tools to rapidly assay new molecules targeting specific receptor pathways. Taste cells are specialized sensory epithelial cells which reside within taste buds on the lingual epithelium. Only recently have successful culturing protocols been developed which maintain essential molecular and functional characteristics. These protocols provide a tractable tool to examine the molecular, regenerative, and functional properties of these unique sensory cells within a controlled environment. The method involves an enzymatic isolation procedure and standardized culture conditions, and may be applied to either dissected rodent tissue or human fungiform papillae obtained by biopsy. Human fungiform cells can be maintained in culture for more than seven passages, without loss of viability and with retention of the molecular and biochemical properties of acutely isolated taste cells. Cultured primary human fungiform papillae cells also exhibit functional responses to taste stimuli indicating the presence of taste receptors and at least some relevant signaling pathways. While the loss of the three-dimensional structure of the intact taste bud must be taken into consideration in interpreting results obtained with these cells, this culture protocol provides a useful model for molecular studies of the proliferation, differentiation, and physiological function of mammalian taste receptor cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Arvidson K (1979) Location and variation in number of taste buds in human fungiform papillae. Scand J Dent Res 87:435–442
Barlow LA, Northcutt RG (1994) Analysis of the embryonic lineage of vertebrate taste buds. Chem Senses 19:715–724
Beidler LM, Smallman RL (1965) Renewal of cells within taste buds. J Cell Biol 27:263–272
Bezencon C, Le Coutre J, Damak S (2007) Taste-signaling proteins are coexpressed in solitary intestinal epithelial cells. Chem Senses 32:41–49
Caicedo A, Jafri MS, Roper SD (2000) In situ Ca2+ imaging reveals neurotransmitter receptors for glutamate in taste receptor cells. J Neurosci 20:7978–7985
Chandrashekar J, Hoon MA, Ryba NJ, Zuker CS (2006) The receptors and cells for mammalian taste. Nature 444:288–294
Finger TE (2005) Cell types and lineages in taste buds. Chem Senses 30(Suppl 1):i54–i55
Gilbertson TA, Damak S, Margolskee RF (2000) The molecular physiology of taste transduction. Curr Opin Neurobiol 10:519–527
Huang YA, Maruyama Y, Stimac R, Roper SD (2008) Presynaptic (Type III) cells in mouse taste buds sense sour (acid) taste. J Physiol 586:2903–2912
Kishi M, Emori Y, Tsukamoto Y, Abe K (2001) Primary culture of rat taste bud cells that retain molecular markers for taste buds and permit functional expression of foreign genes. Neuroscience 106:217–225
Mbiene JP, Maccallum DK, Mistretta CM (1997) Organ cultures of embryonic rat tongue support tongue and gustatory papilla morphogenesis in vitro without intact sensory ganglia. J Comp Neurol 377:324–340
Ozdener H, Yee KK, Cao J, Brand JG, Teeter JH, Rawson NE (2006) Characterization and long-term maintenance of rat taste cells in culture. Chem Senses 31:279–290
Paran N, Mattern CF, Henkin RI (1975) Ultrastructure of the taste bud of the human fungiform papilla. Cell Tissue Res 161:1–10
Perez CA, Huang L, Rong M, Kozak JA, Preuss AK, Zhang H, Max M, Margolskee RF (2002) A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci 5:1169–1176
Ruiz CJ, Stone LM, McPheeters M, Ogura T, Bottger B, Lasher RS, Finger TE, Kinnamon SC (2001) Maintenance of rat taste buds in primary culture. Chem Senses 26:861–873
Spielman AI, Mody I, Brand JG, Whitney G, MacDonald JF, Salter MW (1989) A method for isolating and patch-clamping single mammalian taste receptor cells. Brain Res 503:326–329
Stone LM, Wilcox CL, Kinnamon SC (2002) Virus-mediated transfer of foreign DNA into taste receptor cells. Chem Senses 27:779–787
Stone LM, Tan SS, Tam PP, Finger TE (2002) Analysis of cell lineage relationships in taste buds. J Neurosci 22:4522–4529
Zhao GQ, Zhang Y, Hoon MA, Chandrashekar J, Erlenbach I, Ryba NJ, Zuker CS (2003) The receptors for mammalian sweet and umami taste. Cell 115:255–266
Travers SP, Nicklas K (1990) Taste bud distribution in the rat pharynx and larynx. Anat Rec 227:373–379
Acknowledgement
We acknowledge the technical assistance of Esi Quayson, Aimee Myers, Linda Wysocki, and Valerie Audige. This work was supported in part by P50-DC006760, National Science Foundation (NSF) 0216310, and Givaudan Inc. grant.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Ozdener, M.H., Rawson, N.E. (2012). Primary Culture of Mammalian Taste Epithelium. In: Randell, S., Fulcher, M. (eds) Epithelial Cell Culture Protocols. Methods in Molecular Biology, vol 945. Humana Press, Totowa, NJ. https://doi.org/10.1007/978-1-62703-125-7_7
Download citation
DOI: https://doi.org/10.1007/978-1-62703-125-7_7
Published:
Publisher Name: Humana Press, Totowa, NJ
Print ISBN: 978-1-62703-124-0
Online ISBN: 978-1-62703-125-7
eBook Packages: Springer Protocols