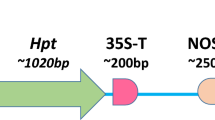
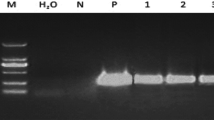
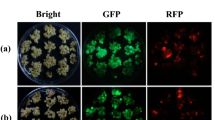
Abstract
Fluorescent proteins such as green fluorescent protein (GFP) allow direct visualization of transformed cells without the need for exogenous substrates. Furthermore, visual selection using GFP is a powerful tool that can be used to isolate transformed cells without antibiotic or herbicide pressure and can be applied to transformation systems in plants hypersensitive to these agents. Moreover, we propose that visual selection enables isolation of calli in which the gene of interest is expressed to a high level, by selecting calli in which a strong GFP signal is observed. However, until now, the efficiency of clonal propagation using visual selection has been lower than that in antibiotic selection because of the technical difficulties involved in the isolation and clonal propagation of transformed calli with conventional transformation frequencies. We have succeeded in improving the efficiency of clonal propagation by the use of a rice cultivar that exhibits high competency for Agrobacterium-mediated transformation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hare, P. D., and Chua, N. H. (2002) Excision of selectable marker genes from transgenic plants. Nat. Biotechnol. 20, 575–580.
Richards, H. A., Halfhill, M. D., Millwood, R. J., and Stewart, C. N. (2003) Quantitative GFP fluorescence as an indicator of recombinant protein synthesis in transgenic plants. Plant Cell Rep. 22, 117–121.
Stewart, C. N. (2005) Monitoring the presence and expression of transgenes in living plants. Trends Plant Sci. 10, 390–396.
Niwa, Y., Hirano, T., Yoshimoto, K., Shimizu, M., and Kobayashi, H. (1999) Non-invasive quantitative detection and applications of non-toxic, S65T-type green fluorescent protein in living plants. Plant J. 18, 455–463.
Saika, H., and Toki, S. (2009) Visual selection allows immediate identification of transgenic rice calli efficiently accumulating transgene products. Plant Cell Rep. 28, 619–626.
Saika, H., Sakamoto, W., Maekawa, M., and Toki, S. (2011) Highly efficient visual selection of transgenic rice plants using green fluorescent protein or anthocyanin synthetic genes. Plant Biotechnol. 28, 107–110.
Hood, E. E., Gelvin, S. B., Melchers, L. S., and Hoekema, A. (1993) New Agrobacterium helper plasmids for gene-transfer to plants. Transgenic Res. 2, 208–218.
Hiei, Y., and Komari, T. (2008) Agrobacterium-mediated transformation of rice using immature embryos or calli induced from mature seed. Nature Protocols 3, 824–834.
Toki, S., Hara, N., Ono, K., Onodera, H., Tagiri, A., Oka, S., and Tanaka, H. (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J. 47, 969–976.
Vain, P., Worland, B., Kohli, A., Snape, J. W., and Christou, P. (1998) The green fluorescent protein (GFP) as a vital screenable marker in rice transformation. Theor. Appl. Genet. 96, 164–169.
Elliott, A. R., Campbell, J. A., Dugdale, B., Brettell, R. I. S., and Grof, C. P. L. (1999) Green fluorescent protein facilitates rapid in vivo detection of genetically transformed plant cells. Plant Cell Rep. 18, 707–714.
Jordan, M. C. (2000) Green fluorescent protein as a visual marker for wheat transformation. Plant Cell Rep. 19, 1069–1075.
Hraska, M., Rakousky, S., and Curn, V. (2006) Green fluorescent protein as a vital marker for non-destructive detection of transformation events in transgenic plants. Plant Cell Tiss. Org. Cult. 86, 303–318.
Saika, H., and Toki, S. Mature seed-derived callus of the model indica rice variety Kasalath is highly competent in Agrobacterium-mediated transformation. Plant Cell Rep. 29, 1351–1364.
Wenck, A., Pugieux, C., Turner, M., Dunn, M., Stacy, C., Tiozzo, A., Dunder, E., van Grinsven, E., Khan, R., Sigareva, M., Wang, W. C., Reed, J., Drayton, P., Oliver, D., Trafford, H., Legris, G., Rushton, H., Tayab, S., Launis, K., Chang, Y. F., Chen, D. F., and Melchers, L. (2003) Reef-coral proteins as visual, non-destructive reporters for plant transformation. Plant Cell Rep. 22, 244–251.
Ogawa, Y., and Mii, M. (2007) Meropenem and moxalactam: Novel b-lactam antibiotics for efficient Agrobacterium-mediated transformation. Plant Sci. 172, 564–572.
Hirochika, H., Sugimoto, K., Otsuki, Y., Tsugawa, H., and Kanda, M. (1996) Retrotransposons of rice involved in mutations induced by tissue culture. Proc. Natl. Acad. Sci. USA 93, 7783–7788.
Matsuo, N., Gilmartin, P. M., and Hiratsuka, K. (2004) Characterization of the EMCV-IRES mediated bicistronic translation in plant cells. Plant Biotechnol. 21, 119–126.
de Felipe, P., Luke, G. A., Hughes, L. E., Gani, D., Halpin, C., and Ryan, M. D. (2006) E unum pluribus: multiple proteins from a self-processing polyprotein. Trends Biotechnol. 24, 68–75.
Urwin, P., Yi, L., Martin, H., Atkinson, H., and Gilmartin, P. M. (2000) Functional characterization of the EMCV IRES in plants. Plant J. 24, 583–589.
Ha, S. H., Liang, Y. S., Jung, H., Ahn, M. J., Suh, S. C., Kweon, S. J., Kim, D. H., Kim, Y. M., and Kim, J. K. (2010) Application of two bicistronic systems involving 2A and IRES sequences to the biosynthesis of carotenoids in rice endosperm. Plant Biotechnol. J. 8, 928–938.
Acknowledgments
We thank Y. Niwa for providing sGFP construct. We also thank K. Osakabe, K. Abe, M. Endo, S. Nonaka, A. Yokoi, and N. Ohtsuki for discussion, and K. Amagai, R. Aoto, C. Furusawa, A. Nagashii, E. Ozawa, and F. Suzuki for technical assistance. This work was supported financially by a grant from the Ministry of Agriculture, Forestry and Fisheries of Japan to H.S. and S.T., and grants-in-aid from the Ministry of Education, Culture, Sports, Science and Technology of Japan to H.S. This work was also supported by a Program for Promotion of Basic Research Activities for Innovative Biosciences (PROBRAIN) grant to S.T. and the Budget for Nuclear Research of the Ministry of Education, Culture, Sports, Science and Technology, based on screening and counseling by the Atomic Energy Commission to S.T.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Saika, H., Onodera, H., Toki, S. (2012). Visual Selection in Rice: A Strategy for the Efficient Identification of Transgenic Calli Accumulating Transgene Products. In: Dunwell, J., Wetten, A. (eds) Transgenic Plants. Methods in Molecular Biology, vol 847. Humana Press. https://doi.org/10.1007/978-1-61779-558-9_7
Download citation
DOI: https://doi.org/10.1007/978-1-61779-558-9_7
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-557-2
Online ISBN: 978-1-61779-558-9
eBook Packages: Springer Protocols