Abstract
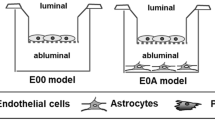
In vitro blood–brain barrier (BBB) models using primary cultured brain endothelial cells are important for establishing cellular and molecular mechanisms of BBB function. Co-culturing with BBB-associated cells especially astrocytes to mimic more closely the in vivo condition leads to upregulation of the BBB phenotype in the brain endothelial cells. Rat brain endothelial cells (RBECs) are a valuable tool allowing ready comparison with in vivo studies in rodents; however, it has been difficult to obtain pure brain endothelial cells, and few models achieve a transendothelial electrical resistance (TEER, measure of tight junction efficacy) of >200 Ω cm2, i.e. the models are still relatively leaky. Here, we describe methods for preparing high purity RBECs and neonatal rat astrocytes, and a co-culture method that generates a robust, stable BBB model that can achieve TEER >600 Ω cm2. The method is based on >20 years experience with RBEC culture, together with recent improvements to kill contaminating cells and encourage BBB differentiation.
Astrocytes are isolated by mechanical dissection and cell straining and are frozen for later co-culture. RBECs are isolated from 3-month-old rat cortices. The brains are cleaned of meninges and white matter and enzymatically and mechanically dissociated. Thereafter, the tissue homogenate is centrifuged in bovine serum albumin to separate vessel fragments from other cells that stick to the myelin plug. The vessel fragments undergo a second enzyme digestion to separate pericytes from vessels and break down vessels into shorter segments, after which a Percoll gradient is used to separate capillaries from venules, arterioles, and single cells. To kill remaining contaminating cells such as pericytes, the capillary fragments are plated in puromycin-containing medium and RBECs grown to 50–60% confluence. They are then passaged onto filters for co-culture with astrocytes grown in the bottom of the wells. The whole procedure takes ∼2 weeks, using pre-frozen astrocytes, from isolation of RBECs to generation of high-resistance/low-permeability RBEC monolayers.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abbott, N.J., Patabendige, A.A., Dolman, D.E., Yusof, S.R., and Begley, D.J. (2010) Structure and function of the blood–brain barrier. Neurobiol. Dis. 37, 13–25.
Abbott, N.J., Rönnbäck, L., and Hansson, E. (2006) Astrocyte-endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 7, 41–53.
Deli, M.A., Abrahám, C.S., Kataoka, Y., and Niwa, M. (2005) Permeability studies on in vitro blood–brain barrier models: physiology, pathology, and pharmacology. Cell Mol Neurobiol. 25, 59–127.
Nakagawa, S., Deli, M.A., Kawaguchi, H., Shimizudani, T., Shimono, T., Kittel, A. et al. (2009) A new blood–brain barrier model using primary rat brain endothelial cells, pericytes and astrocytes. Neurochem. Int. 54, 253–263.
Perrière, N. Demeuse, P., Garcia, E., Regina, A., Debray, M., Andreux, J.P. et al. (2005) Puromycin-based purification of rat brain capillary endothelial cell cultures. Effect on the expression of blood–brain barrier-specific properties. J. Neurochem. 93, 279–289.
Gaillard, P.J., Voorwinden, L.H., Nielsen, J.L., Ivanov, A., Atsumi, R., Engman, H. et al. (2001) Establishment and functional characterization of an in vitro model of the blood–brain barrier comprising a co-culture of brain capillary endothelial cells and astrocytes. Eur. J. Pharm. Sci. 12, 215–222.
Stanness, K.A., Guatteo, E., and Janigro, D. (1996) A dynamic model of the blood–brain barrier “in vitro”. Neurotoxicol. 17, 481–496.
Booher, J., and Sensenbrenner, M. (1972) Growth and cultivation of dissociated neurons and glial cells from embryonic chick, rat and human brain in flask cultures. Neurobiol. 2, 97–105.
Gómez-Lechón, M.J., Iborra, F.J., Azorín, I., Guerri, C., Renau-Piqueras, J. (1992) Cryopreservation of rat astrocytes from primary cultures. J. Tissue Cult. Methods 14, 73–82.
Bowman, P.D., Betz, A.L., Ar, D., Wolinsky, J.S., Penney, J.B. et al. (1981) Primary culture of capillary endothelium from rat brain. In Vitro 17, 353–362.
Bowman, P.D., Ennis, S.R., Rarey, K.E., Betz, A.L., and Goldstein, G.W. (1983) Brain microvessel endothelial cells in tissue culture: a model for study of blood–brain barrier permeability. Ann. Neurol. 14, 396–402.
Hughes, C.C., and Lantos, P.L. (1986) Brain capillary endothelial cells in vitro lack surface IgG Fc receptors. Neurosci. Lett. 68, 100–106.
Hughes, C.C., and Lantos, P.L. (1989) Uptake of leucine and alanine by cultured cerebral capillary endothelial cells. Brain Res. 480, 126–132.
Abbott, N.J., Hughes, C.C., Revest, P.A., and Greenwood, J. (1992) Development and characterization of a rat brain capillary endothelial culture: towards an in vitro blood–brain barrier. J. Cell Sci. 103, 23–37.
Dolman, D., Drndarski S., Abbott, N.J., and Rattray, M. (2005). Induction of aquaporin 1 but not aquaporin 4 messenger RNA in rat primary brain microvessel endothelial cells in culture. J. Neurochem. 93, 825–383.
Strom, S.C. and Michalopoulos, G. (1982) Collagen as a substrate for cell growth and differentiation. Methods Enzymol. 82, 544–555.
Fredriksson, S.M. (2009) Modulation of the blood–brain barrier by inflammatory mediators: relevance to multiple sclerosis. PhD Thesis, King’s College London, University of London.
Acknowledgements
The development and optimisation of this method has built on the earlier work of several previous members of the BBB Group King’s College, including Chris Hughes, Patricia Revest, Nacho Romero, Rob Rist, and Mike Dobbie. The work was supported by grants from the MRC, the Wellcome Trust, the Multiple Sclerosis Society U.K., and UCB.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Abbott, N.J., Dolman, D.E.M., Drndarski, S., Fredriksson, S.M. (2012). An Improved In Vitro Blood–Brain Barrier Model: Rat Brain Endothelial Cells Co-cultured with Astrocytes. In: Milner, R. (eds) Astrocytes. Methods in Molecular Biology, vol 814. Humana Press. https://doi.org/10.1007/978-1-61779-452-0_28
Download citation
DOI: https://doi.org/10.1007/978-1-61779-452-0_28
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-451-3
Online ISBN: 978-1-61779-452-0
eBook Packages: Springer Protocols