Abstract
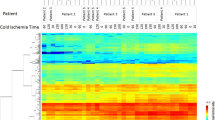
Phosphorylated proteins reflect the activity of specific cell signaling nodes in biological kinase protein networks. Cell signaling pathways can be either activated or deactivated depending on the phosphorylation state of the constituent proteins. The state of these kinase pathways reflects the in vivo activity of the cells and tissue at any given point in time. As such, cell signaling pathway information can be extrapolated to infer which phosphorylated proteins/pathways are driving an individual tumor’s growth. Reverse phase protein microarrays (RPMAs) are a sensitive and precise platform that can be applied to the quantitative measurement of hundreds of phosphorylated signal proteins from a small sample of tissue. Pre-analytical variability originating from tissue procurement and preservation may cause significant variability and bias in downstream molecular analysis. Depending on the ex vivo delay time in tissue processing, and the manner of tissue handling, protein biomarkers such as signal pathway phosphoproteins will be elevated or suppressed in a manner that does not represent the biomarker levels at the time of excision. Consequently, assessment of the state of these kinase networks requires stabilization, or preservation, of the phosphoproteins immediately post-tissue procurement. We have employed RPMA analysis of phosphoproteins to study the factors influencing stability of phosphoproteins in tissue following procurement. Based on this analysis we have established tissue procurement guidelines for clinical research with an emphasis on quantifying phosphoproteins by RPMA.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Espina, V., Edmiston, K. H., Heiby, M., Pierobon, M., Sciro, M., et al. (2008) A portrait of tissue phosphoprotein stability in the clinical tissue procurement process. Mol Cell Proteomics 7, 1998–2018.
Li, X., Friedman, A. B., Roh, M. S., and Jope, R. S. (2005) Anesthesia and post-mortem interval profoundly influence the regulatory serine phosphorylation of glycogen synthase kinase-3 in mouse brain. J Neurochem 92, 701–4.
Li, J., Gould, T. D., Yuan, P., Manji, H. K., and Chen, G. (2003) Post-mortem interval effects on the phosphorylation of signaling proteins. Neuropsychopharmacology 28, 1017–25.
Becker, K. F., Schott, C., Hipp, S., Metzger, V., Porschewski, P., et al. (2007) Quantitative protein analysis from formalin-fixed tissues: implications for translational clinical research and nanoscale molecular diagnosis. J Pathol 211, 370–8.
Fox, C. H., Johnson, F. B., Whiting, J., and Roller, P. P. (1985) Formaldehyde fixation. J Histochem Cytochem 33, 845–53.
Helander, K. G. (1994) Kinetic studies of formaldehyde binding in tissue. Biotech Histochem 69, 177–9.
Srinivasan, M., Sedmak, D., and Jewell, S. (2002) Effect of fixatives and tissue processing on the content and integrity of nucleic acids. Am J Pathol 161, 1961–71.
Nassiri, M., Ramos, S., Zohourian, H., Vincek, V., Morales, A. R., et al. (2008) Preservation of biomolecules in breast cancer tissue by a formalin-free histology system. BMC Clin Pathol 8, 1.
Devireddy, R. V. (2005) Predicted permeability parameters of human ovarian tissue cells to various cryoprotectants and water. Mol Reprod Dev 70, 333–43.
He, Y., and Devireddy, R. V. (2005) An inverse approach to determine solute and solvent permeability parameters in artificial tissues. Ann Biomed Eng 33, 709–18.
Goldstein, B. J. (2002) Protein-tyrosine phosphatases: emerging targets for therapeutic intervention in type 2 diabetes and related states of insulin resistance. J Clin Endocrinol Metab 87, 2474–80.
Neel, B. G., and Tonks, N. K. (1997) Protein tyrosine phosphatases in signal transduction. Curr Opin Cell Biol 9, 193–204.
Grellner, W., Vieler, S., and Madea, B. (2005) Transforming growth factors (TGF-alpha and TGF-beta1) in the determination of vitality and wound age: immunohistochemical study on human skin wounds. Forensic Sci Int 153, 174–80.
Grellner, W. (2002) Time-dependent immunohistochemical detection of proinflammatory cytokines (IL-1beta, IL-6, TNF-alpha) in human skin wounds. Forensic Sci Int 130, 90–6.
Grellner, W., and Madea, B. (2007) Demands on scientific studies: vitality of wounds and wound age estimation. Forensic Sci Int 165, 150–4.
Ohshima, T. (2000) Forensic wound examination. Forensic Sci Int 113, 153–64.
Oehmichen, M. (2004) Vitality and time course of wounds. Forensic Sci Int 144, 221–31.
Paweletz, C. P., Charboneau, L., Bichsel, V. E., Simone, N. L., Chen, T., et al. (2001) Reverse phase protein microarrays which capture disease progression show activation of pro-survival pathways at the cancer invasion front. Oncogene 20, 1981–9.
Petricoin, E. F., 3rd, Espina, V., Araujo, R. P., Midura, B., Yeung, C., et al. (2007) Phosphoprotein pathway mapping: Akt/mammalian target of rapamycin activation is negatively associated with childhood rhabdomyosarcoma survival. Cancer Res 67, 3431–40.
VanMeter, A., Signore, M., Pierobon, M., Espina, V., Liotta, L. A., et al. (2007) Reverse-phase protein microarrays: application to biomarker discovery and translational medicine. Expert Rev Mol Diagn 7, 625–33.
Wulfkuhle, J. D., Speer, R., Pierobon, M., Laird, J., Espina, V., et al. (2008) Multiplexed cell signaling analysis of human breast cancer applications for personalized therapy. J Proteome Res 7, 1508–17.
Espina, V., Mehta, A. I., Winters, M. E., Calvert, V., Wulfkuhle, J., et al. (2003) Protein microarrays: molecular profiling technologies for clinical specimens. Proteomics 3, 2091–100.
Belluco, C., Mammano, E., Petricoin, E., Prevedello, L., Calvert, V., et al. (2005) Kinase substrate protein microarray analysis of human colon cancer and hepatic metastasis. Clin Chim Acta 357, 180–3.
Berggren, K., Steinberg, T. H., Lauber, W. M., Carroll, J. A., Lopez, M. F., et al. (1999) A luminescent ruthenium complex for ultrasensitive detection of proteins immobilized on membrane supports. Anal Biochem 276, 129–43.
Berggren, K. N., Schulenberg, B., Lopez, M. F., Steinberg, T. H., Bogdanova, A., et al. (2002) An improved formulation of SYPRO Ruby protein gel stain: comparison with the original formulation and with a ruthenium II tris (bathophenanthroline disulfonate) formulation. Proteomics 2, 486–98.
Espina, V., Wulfkuhle, J. D., Calvert, V. S., VanMeter, A., Zhou, W., et al. (2006) Laser-capture microdissection. Nat Protoc 1, 586–603.
Stillman, B. A., and Tonkinson, J. L. (2000) FAST slides: a novel surface for microarrays. Biotechniques 29, 630–5.
Tonkinson, J. L., and Stillman, B. A. (2002) Nitrocellulose: a tried and true polymer finds utility as a post-genomic substrate. Front Biosci 7, c1–12.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Espina, V., Mueller, C., Liotta, L.A. (2011). Phosphoprotein Stability in Clinical Tissue and Its Relevance for Reverse Phase Protein Microarray Technology. In: Korf, U. (eds) Protein Microarrays. Methods in Molecular Biology, vol 785. Humana Press. https://doi.org/10.1007/978-1-61779-286-1_3
Download citation
DOI: https://doi.org/10.1007/978-1-61779-286-1_3
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-61779-285-4
Online ISBN: 978-1-61779-286-1
eBook Packages: Springer Protocols