Summary
Transgenesis is an important methodology for studying the function of genes and genomes in model plants and animals. For the model vertebrate zebrafish, methods using the Tol2 transposable element have been developed for this purpose. With these methods, the function of the transgene can be analyzed in both transient and stable transgenic fish. Recently, cis-sequences necessary for transposition of the Tol2 element were revealed. This enabled development of transposon vectors containing minimal DNA sequences that are easily manipulated. More recently, several transposon vectors containing the Gateway sequence were created and reported. These are useful because any foreign sequences can be cloned into a transposon vector fairly easily and rapidly. This chapter describes the features of these transposon vectors, and protocols to perform transgenesis in zebrafish.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Stuart, G. W., McMurray, J. V., and Westerfield, M. (1988). Replication, integration and stable germ-line transmission of foreign sequences injected into early zebrafish embryos. Development 103, 403–12.
Amsterdam, A., Lin, S., and Hopkins, N. (1995). The Aequorea victoria green fluorescent protein can be used as a reporter in live zebrafish embryos. Dev Biol 171, 123–9.
Long, Q., Meng, A., Wang, H., Jessen, J. R., Farrell, M. J., and Lin, S. (1997). GATA-1 expression pattern can be recapitulated in living transgenic zebrafish using GFP reporter gene. Development 124, 4105–11.
Higashijima, S., Okamoto, H., Ueno, N., Hotta, Y., and Eguchi, G. (1997). High-frequency generation of transgenic zebrafish which reliably express GFP in whole muscles or the whole body by using promoters of zebrafish origin. Dev Biol 192, 289–99.
Raz, E., van Luenen, H. G., Schaerringer, B., Plasterk, R. H., and Driever, W. (1998). Transposition of the nematode Caenorhabditis elegans Tc3 element in the zebrafish Danio rerio. Curr Biol 8, 82–8.
Fadool, J. M., Hartl, D. L., and Dowling, J. E. (1998). Transposition of the mariner element from Drosophila mauritiana in zebrafish. Proc Natl Acad Sci U S A 95, 5182–6.
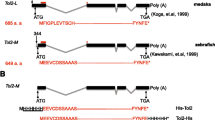
Kawakami, K., Koga, A., Hori, H., and Shima, A. (1998). Excision of the Tol2 transposable element of the medaka fish, Oryzias latipes, in zebrafish, Danio rerio. Gene 225, 17–22.
Davidson, A. E., Balciunas, D., Mohn, D., Shaffer, J., Hermanson, S., Sivasubbu, S., Cliff, M. P., Hackett, P. B., and Ekker, S. C. (2003). Efficient gene delivery and gene expression in zebrafish using the Sleeping Beauty transposon. Dev Biol 263, 191–202.
Kawakami, K., Takeda, H., Kawakami, N., Kobayashi, M., Matsuda, N., and Mishina, M. (2004). A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev Cell 7, 133–44.
Kawakami, K. (2005). Transposon tools and methods in zebrafish. Dev Dyn 234, 244–54.
Urasaki, A., Morvan, G., and Kawakami, K. (2006). Functional dissection of the Tol2 transposable element identified the minimal cis-sequence and a highly repetitive sequence in the subterminal region essential for transposition. Genetics 174, 639–49.
Balciunas, D., Wangensteen, K. J., Wilber, A., Bell, J., Geurts, A., Sivasubbu, S., Wang, X., Hackett, P. B., Largaespada, D. A., McIvor, R. S., and Ekker, S. C. (2006). Harnessing a high cargo-capacity transposon for genetic applications in vertebrates. PLoS Genet 2, e169.
Kwan, K. M., Fujimoto, E., Grabher, C., Mangum, B. D., Hardy, M. E., Campbell, D. S., Parant, J. M., Yost, H. J., Kanki, J. P., and Chien, C. B. (2007). The Tol2kit: a multisite gateway-based construction kit for Tol2 transposon transgenesis constructs. Dev Dyn 236, 3088–99.
Villefranc, J. A., Amigo, J., and Lawson, N. D. (2007). Gateway compatible vectors for analysis of gene function in the zebrafish. Dev Dyn 236, 3077–87.
Fisher, S., Grice, E. A., Vinton, R. M., Bessling, S. L., and McCallion, A. S. (2006). Conservation of RET regulatory function from human to zebrafish without sequence similarity. Science 312, 276–9.
Fisher, S., Grice, E. A., Vinton, R. M., Bessling, S. L., Urasaki, A., Kawakami, K., and McCallion, A. S. (2006). Evaluating the biological relevance of putative enhancers using Tol2 transposon-mediated transgenesis in zebrafish. Nat Protoc 1, 1297–305.
Zou, J., Beermann, F., Wang, J., Kawakami, K., and Wei, X. (2006). The Fugu tyrp1 promoter directs specific GFP expression in zebrafish: tools to study the RPE and the neural crest-derived melanophores. Pigment Cell Res 19, 615–27.
Blaser, H., Reichman-Fried, M., Castanon, I., Dumstrei, K., Marlow, F. L., Kawakami, K., Solnica-Krezel, L., Heisenberg, C. P., and Raz, E. (2006). Migration of zebrafish primordial germ cells: a role for myosin contraction and cytoplasmic flow. Dev Cell 11, 613–27.
Acknowledgments
We would like to thank Dr. Chi-Bin Chien for helpful comments on the manuscript; a postdoctoral fellowship from National Institute of Genetics (to HK); and the National BioResource Project and grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Humana Press, a part of Springer Science+Business Media, LLC
About this protocol
Cite this protocol
Kikuta, H., Kawakami, K. (2009). Transient and Stable Transgenesis Using Tol2 Transposon Vectors. In: Lieschke, G., Oates, A., Kawakami, K. (eds) Zebrafish. Methods in Molecular Biology, vol 546. Humana Press. https://doi.org/10.1007/978-1-60327-977-2_5
Download citation
DOI: https://doi.org/10.1007/978-1-60327-977-2_5
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-60327-976-5
Online ISBN: 978-1-60327-977-2
eBook Packages: Springer Protocols