Abstract
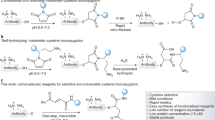
Interchain disulfide bonds of antibodies can be reduced by agents such as TCEP or DTT to form reactive cysteine residues. These endogenous cysteines are used for conjugation to biologically active drugs either directly or via linkers to prepare antibody drug conjugates (ADCs). The anti-notch 3 ADC described here is being evaluated in the early clinical development program as a potential treatment for a variety of cancers. The ADC is composed of an IgG1 mAb that is conjugated by endogenous cysteines to a cytotoxic microtubulin inhibitor via a maleimide-containing linker. The endogenous cysteine residues are produced by partial reduction of the mAb with TCEP reducing agent. The conjugation results in the formation of a mixture of 2, 4, 6, and 8 loaded ADC species. In addition to the desired product, several product-related impurities such as aggregates are generated during the conjugation reaction. The product- and process-related impurities are separated from the monomeric ADC by column chromatography and ultrafiltration-diafiltration techniques. The temperature of TCEP reduction step has an impact on the level of aggregates produced in the reaction. The temperature also impacts the isomeric composition of the 4 loaded ADC species.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Mallory RG, Mine C, Longyu L, Jiaming Z, Barbara O, Thayumanavan S (2015) Field guide to challenges and opportunities in antibody-drug conjugates for chemists. Bioconjug Chem 26(11):2198–2215
Zhou Q (2017) Site-specific antibody conjugation for ADC and beyond. Biomedicine 5:64
Hamann PR (2011) In: Barrish JC, Carter PH, Cheng PTW, Zahler R (eds) Accounts in drug discovery: case studies in medicinal chemistry. The genesis of the antibody conjugate gemtuzumab ozogamicin (Mylotarg®) for acute myeloid leukemia. Chapter 5: 103–119
Chen Y, Kim MT, Zheng L, Deperalta G, Jacobson F (2016) Structural characterization of cross-linked species in Trastuzumab Emtansine (Kadcyla). Bioconjug Chem 27:2037–2047
Lyon RP, Meyer DL, Setter JR, Senter PD (2012) Conjugation of anticancer drugs through endogenous monoclonal antibody cysteine residues. Methods Enzymol 502:123–138
Behrens CR, Liu B (2014) Methods for site-specific drug conjugation to antibodies. MAbs 6(1):46–53
Geles KG, Gao Y, Sapra P, Tchitstiakova LG, Zhou SB (2014) Anti-notch 3 antibodies and antibody drug conjugates. Pfizer Inc., New York, NY. Patent 0127211A1
Hamblett KJ, Senter PD, Chace DF, Sun MMC, Lenox J, Cerveny CG, Kissler KM, Bernhardt SX, Kopcha AK, Zabinski RF, Meyer DL, Francisco JA (2004) Effects of drug loading on the antitumor activity of a monoclonal antibody drug conjugate. Clin Cancer Res 10:7063–7070
Nadkarni DV, Jiang Q, Friese O, Bazhina N, Meng H, Guo J, Kutlik R, Borgmeyer J (2018) Process development and structural characterization of an anti-notch 3 antibody−drug conjugate. Org Process Res Dev 22:286–295
Hollander I, Kunz A, Hamann PR (2008) Selection of reaction additives used in the preparation of monomeric antibody−Calicheamicin conjugates. Bioconjug Chem 19:358–361
Li W, Prabakaran P, Chen W, Zhu Z, Feng Y, Dimitrov DS (2016) Antibody aggregation: insights from sequence and structure. Antibodies 5:19–23
Gagnon P, Beam K (2009) Antibody aggregate removal by hydroxyapatite chromatography. Curr Pharm Biotechnol 10:440–446
Nadkarni DV, Borgmeyer J, Meng H, Jiang Q (2017) Purification of antibody drug conjugates using a sodium phosphate gradient. Pfizer Inc., New York, NY. Patent WO 2017/109619 Al
Acknowledgement
The author thanks Qingping Jiang, He Meng, Jeffry Borgmeyer, Nataliya Bazhina, and Olga Friese for their contributions during the development of this work. The author also thanks Leo Letendre, Heyi Li, Bo Arve, and Aparna Deora for their support and encouragement.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Nadkarni, D.V. (2020). Conjugations to Endogenous Cysteine Residues. In: Tumey, L. (eds) Antibody-Drug Conjugates. Methods in Molecular Biology, vol 2078. Humana, New York, NY. https://doi.org/10.1007/978-1-4939-9929-3_3
Download citation
DOI: https://doi.org/10.1007/978-1-4939-9929-3_3
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-4939-9928-6
Online ISBN: 978-1-4939-9929-3
eBook Packages: Springer Protocols