Abstract
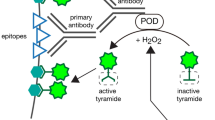
Enzyme-linked signal amplification is a key technique used to enhance the immunohistochemical detection of protein, mRNA, and other molecular species. Tyramide signal amplification (TSA) is based on a catalytic reporter deposit in close vicinity to the epitope of interest. The advantages of this technique are its simplicity, enhanced sensitivity, high specificity, and compatibility with modern multi-label fluorescent microscopy. Here, we describe the use of a TSA kit to increase the signal of enhanced green fluorescent protein (eGFP) expressed under the control of Slc17a6 regulatory elements in the brain of a transgenic mouse. The labeling procedure consists of 6 basic steps: (1) tissue preparation, (2) blocking of nonspecific epitopes, (3) binding with primary antibody, (4) binding with horseradish peroxidase-conjugated secondary antibody, (5) reacting with fluorescent tyramide substrate, and (6) imaging of the signal. The procedures described herein detail these steps and provide additional guidance and background to assist novice users.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bobrow MN, Moen PT, Jr (2001) Tyramide signal amplification (TSA) systems for the enhancement of ISH signals in cytogenetics. Curr Protoc Cytom Chapter 8: Unit 8.9
Macechko PT, Krueger L, Hirsch B et al (1997) Comparison of immunologic amplification vs enzymatic deposition of fluorochrome-conjugated tyramide as detection systems for FISH. J Histochem Cytochem 45:359–363
Ormanns W, Schaffer R (1985) An alkaline-phosphatase staining method in avidin-biotin immunohistochemistry. Histochemistry 82:421–424
Bobrow MN, Harris TD, Shaughnessy KJ et al (1989) Catalyzed reporter deposition, a novel method of signal amplification. Application to immunoassays. J Immunol Methods 125:279–285
Bobrow MN, Shaughnessy KJ, Litt GJ (1991) Catalyzed reporter deposition, a novel method of signal amplification. II. Application to membrane immunoassays. J Immunol Methods 137:103–112
Porstmann B, Porstmann T, Nugel E et al (1985) Which of the commonly used marker enzymes gives the best results in colorimetric and fluorimetric enzyme immunoassays: horseradish peroxidase, alkaline phosphatase or beta-galactosidase? J Immunol Methods 79:27–37
Zaidi AU, Enomoto H, Milbrandt J et al (2000) Dual fluorescent in situ hybridization and immunohistochemical detection with tyramide signal amplification. J Histochem Cytochem 48:1369–1375
Bobrow MN, Moen PT Jr (2001) Tyramide Signal Amplification (TSA) Systems for the Enhancement of ISH Signals in Cytogenetics. Current Protocols in Cytometry. Chapter 8:Unit 8.9
Mayer G, Bendayan M (1997) Biotinyl-tyramide: a novel approach for electron microscopic immunocytochemistry. J Histochem Cytochem 45:1449–1454
Van Heusden J, de Jong P, Ramaekers F et al (1997) Fluorescein-labeled tyramide strongly enhances the detection of low bromodeoxyuridine incorporation levels. J Histochem Cytochem 45:315–319
Raap AK, van de Corput MP, Vervenne RA et al (1995) Ultra-sensitive FISH using peroxidase-mediated deposition of biotin- or fluorochrome tyramides. Hum Mol Genet 4:529–534
van Gijlswijk RP, Wiegant J, Vervenne R et al (1996) Horseradish peroxidase-labeled oligonucleotides and fluorescent tyramides for rapid detection of chromosome-specific repeat sequences. Cytogenet Cell Genet 75:258–262
Sako W, Morigaki R, Kaji R et al (2011) Identification and localization of a neuron-specific isoform of TAF1 in rat brain: implications for neuropathology of DYT3 dystonia. Neuroscience 189:100–107
Okita S, Morigaki R, Koizumi H et al (2012) Cell type-specific localization of optineurin in the striatal neurons of mice: implications for neuronal vulnerability in Huntington’s disease. Neuroscience 202:363–370
Morigaki R, Sako W, Okita S et al (2011) Cyclin-dependent kinase 5 with phosphorylation of tyrosine 15 residue is enriched in striatal matrix compartment in adult mice. Neuroscience 189:25–31
Koizumi H, Morigaki R, Okita S et al (2013) Response of striosomal opioid signaling to dopamine depletion in 6-hydroxydopamine-lesioned rat model of Parkinson’s disease: a potential compensatory role. Front Cell Neurosci 7:74
Zhao C, Eisinger B, Gammie SC (2013) Characterization of GABAergic neurons in the mouse lateral septum: a double fluorescence in situ hybridization and immunohistochemical study using tyramide signal amplification. PLoS One 8:e73750
van Gijlswijk RP, Zijlmans HJ, Wiegant J et al (1997) Fluorochrome-labeled tyramides: use in immunocytochemistry and fluorescence in situ hybridization. J Histochem Cytochem 45:375–382
Gong H, Byers DM (2003) Glutamate-41 of Vibrio harveyi acyl carrier protein is essential for fatty acid synthase but not acyl-ACP synthetase activity. Biochem Biophys Res Commun 302:35–40
Hnasko TS, Chuhma N, Zhang H et al (2010) Vesicular glutamate transport promotes dopamine storage and glutamate corelease in vivo. Neuron 65:643–656
Hnasko TS, Hjelmstad GO, Fields HL et al (2012) Ventral tegmental area glutamate neurons: electrophysiological properties and projections. J Neurosci 32:15076–15085
Kapuscinski J (1990) Interactions of nucleic acids with fluorescent dyes: spectral properties of condensed complexes. J Histochem Cytochem 38:1323–1329
Horling L, Neuhuber WL, Raab M (2012) Pitfalls using tyramide signal amplification (TSA) in the mouse gastrointestinal tract: endogenous streptavidin-binding sites lead to false positive staining. J Neurosci Methods 204:124–132
Bussolati G, Leonardo E (2008) Technical pitfalls potentially affecting diagnoses in immunohistochemistry. J Clin Pathol 61:1184–1192
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Faget, L., Hnasko, T.S. (2015). Tyramide Signal Amplification for Immunofluorescent Enhancement. In: Hnasko, R. (eds) ELISA. Methods in Molecular Biology, vol 1318. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-2742-5_16
Download citation
DOI: https://doi.org/10.1007/978-1-4939-2742-5_16
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-2741-8
Online ISBN: 978-1-4939-2742-5
eBook Packages: Springer Protocols