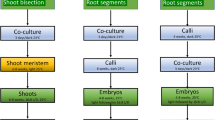
Abstract
Legumes have been for a long time recalcitrant to efficient Agrobacterium transformation. The choice and use of model legume plants (Medicago truncatula and Lotus japonicus) for molecular studies has triggered extensive studies devoted to the development of efficient Agrobacterium-mediated transformation protocols for these two plants. In M. truncatula, transformation protocols rely on the use of highly regenerable lines obtained by recurrent in vitro culture selection. These protocols are based on Agrobacterium-mediated transformation of M. truncatula followed by somatic embryogenesis-mediated plant regeneration. We describe here the protocol developed for M. truncatula R108-1 (c3).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Nolan KE, Rose RJ, Gorst JR (1989) Regeneration of Medicago truncatula from tissue culture: increased somatic embryogenesis using explants from regenerated plants. Plant Cell Rep 8:278–281
Chabaud M, Larsonneau C, Marmouget C, Huguet T (1996) Transformation of barrel medic (Medicago truncatula Gaertn.) by Agrobacterium tumefaciens and regeneration via somatic embryogenesis of transgenic plants with the MtENOD12 nodulin promoter fused to the gus reporter gene. Plant Cell Rep 15:305–310
Trieu AT, Harrison MJ (1996) Rapid transformation of Medicago truncatula: regeneration via shoot organogenesis. Plant Cell Rep 16:6–11
Hoffmann B, Trinh TH, Leung J, Kondorosi A, Kondorosi E (1997) A new Medicago truncatula line with superior in vitro regeneration, transformation, and symbiotic properties isolated through cell culture selection. Mol Plant Microbe Interact 10:307–315
Trinh TH, Ratet P, Kondorosi E, Durand P, Kamaté K, Bauer P, Kondorosi A (1998) Rapid and efficient transformation of diploid Medicago truncatula and Medicago sativa ssp. falcata in vitro lines improved in somatic embryogenesis. Plant Cell Rep 17:345–355
Kamaté K, Rodriguez-Llorente ID, Scholte M, Durand P, Ratet P, Kondorosi E, Kondorosi A, Trinh TH (2000) Transformation of floral organs with GFP in Medicago truncatula. Plant Cell Rep 19:647–653
Chabaud M, de Carvalho-Niebel F, Barker DG (2003) Efficient transformation of Medicago truncatula cv. Jemalong using the hypervirulent Agrobacterium tumefaciens strain AGL1. Plant Cell Rep 22:46–51
Zhou X, Chandrasekharan MB, Hall TC (2004) High rooting frequency and functional analysis of GUS and GFP expression in transgenic Medicago truncatula A17. New Phytol 162:813–822
Araújo SS, Duque AS, Santos D, Fevereiro P (2004) An efficient transformation method to regenerate a high number of transgenic plants using a new embryogenic line of Medicago truncatula cv. Jemalong. Plant Cell Tissue Organ Cult 78:123–131
Crane S, Wright E, Dixon RA, Wang Z-Y (2006) Transgenic Medicago truncatula plants obtained from Agrobacterium tumefaciens transformed roots and Agrobacterium rhizogenes transformed hairy roots. Planta 223:1344–1354
Iantcheva A, Chabaud M, Cosson V, Barascud M, Schutz B, Primard-Brisset C, Durand P, Barker DG, Atanasov A, Vlahova M, Ratet P (2009) Osmotic shock improves Tnt1 transposition frequency in Medicago truncatula cv Jemalong during in vitro regeneration. Plant Cell Rep 28(10):1563–1572
Scholte M, d’Erfurth I, Ripa S, Mondy S, Cosson V, Durand P, Breda C, Trinh T, Rodriguez-Llorente I, Kondorosi E, Schultze M, Kondorosi A, Ratet P (2002) T-DNA tagging in the model legume Medicago truncatula allows efficient gene discovery. Mol Breeding 10:203–215
d’Erfurth I, Cosson V, Eschstruth A, Lucas H, Kondorosi A, Ratet P (2003) Efficient transposition of the Tnt1 tobacco retrotransposon in the model legume Medicago truncatula. Plant J 34:95–106
Tadege M, Wen J, He J, Tu H, Kwak Y, Eschstruth A, Cayrel A, Endre G, Zhao PX, Chabaud M, Ratet P, Mysore KS (2008) Large scale insertional mutagenesis using Tnt1 retrotransposon in the model legume Medicago truncatula. Plant J 54(2):335–347
Chu CC (1978) The N6 medium and its applications to anther culture of cereals. In: Proceedings of symposium on plant tissue culture. Peking, Pitman Publishing Ltd. and The Science Press, pp 43–50
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204
Krall L, Wiedemann U, Unsin G, Weiss S, Domke N, Baron C (2002) Detergent extraction identifies different VirB protein subassemblies of the type IV secretion machinery in the membranes of Agrobacterium tumefaciens. Proc Natl Acad Sci U S A 99(17):11405–11410
Hellens R, Mullineaux P, Klee H (2000) A guide to Agrobacterium binary Ti vectors. Trends Plant Sci 5:446–451
Bevan M (1984) Binary Agrobacterium vectors for plant transformation. Nucleic Acids Res 12:8711–8721
Ratet P, Schell J, de Bruijn FJ (1988) Mini-Mulac transposons with broad-host-range origins of conjugal transfer and replication designed for gene regulation studies in Rhizobiaceae. Gene 63:41–52
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer Science+Business Media New York
About this protocol
Cite this protocol
Cosson, V., Eschstruth, A., Ratet, P. (2015). Medicago truncatula Transformation Using Leaf Explants. In: Wang, K. (eds) Agrobacterium Protocols. Methods in Molecular Biology, vol 1223. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-1695-5_4
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1695-5_4
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-1694-8
Online ISBN: 978-1-4939-1695-5
eBook Packages: Springer Protocols