Abstract
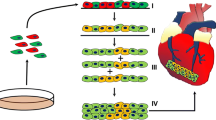
Considering the impaired regenerative capacity of adult mammalian heart tissue, cardiovascular tissue engineering aims to create functional substitutes that can restore the structure and function of the damaged cardiac tissue. The success of cardiac regenerative therapies has been limited mainly due to poor control on the structure and properties of the tissue substitute, lack of vascularization, and immunogenicity. In this study we introduce a new approach to rapidly engineer dense biomimetic scaffolds consisting of type I collagen, to protect the heart against severe ischemic injury. Scaffold biomechanical properties are adjusted to mimic embryonic epicardium which is shown to be optimal to support cardiomyocyte contractile work. Moreover, the designed patch can serve as a delivery device for targeted, controlled release of cells or therapeutic macromolecules into the lesion area.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abou Neel EA, Cheema U, Knowles JC et al (2006) Use of multiple unconfined compression for control of collagen gel scaffold density and mechanical properties. Soft Matter 2: 986–992
Akins RE, Boyce RA, Madonna ML et al (1999) Cardiac organogenesis in vitro: reestablishment of three-dimensional tissue architecture by dissociated neonatal rat ventricular cells. Tissue Eng 5:103–118
Badylak SF, Taylor D, Uygun K (2011) Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix scaffolds. Annu Rev Biomed Eng 13:27–53
Bitar M, Salih V, Brown RA, Nazhat SN (2007) Effect of multiple unconfined compression on cellular dense collagen scaffolds for bone tissue engineering. J Mater Sci Mater Med 18: 237–244
Brown RA, Wiseman M, Chuo CB et al (2005) Ultrarapid engineering of biomimetic materials and tissues: fabrication of nano- and microstructures by plastic compression. Adv Funct Mater 15:1762–1770
Chen Q-Z, Harding SE, Ali NN et al (2008) Biomaterials in cardiac tissue engineering: ten years of research survey. Mater Sci Eng R Rep 59:1–37
Chicatun F, Muja N, Serpooshan V et al (2013) Effect of chitosan incorporation on the consolidation process of highly hydrated collagen hydrogel scaffolds. Soft Matter. doi:10.1039/C3SM52176A
Dako (2010) Dako pathology educational guide; special stains and H & E
Dako (2009) Dako general instructions for immunohistochemical staining
Engler AJ, Carag-Krieger C, Johnson CP et al (2008) Embryonic cardiomyocytes beat best on a matrix with heart-like elasticity: scar-like rigidity inhibits beating. J Cell Sci 121: 3794–3802
Eschenhagen T, Fink C, Remmers U et al (1997) Three-dimensional reconstitution of embryonic cardiomyocytes in a collagen matrix: a new heart muscle model system. FASEB J 11:683–694
(2009) Stains file – paraffin processing. http://stainsfile.info/StainsFile/prepare/process/processing.htm
Jawad H, Ali NN, Lyon AR et al (2007) Myocardial tissue engineering: a review. J Tissue Eng Regen Med 1:327–342
Lee CH, Singla A, Lee Y (2001) Biomedical applications of collagen. Int J Pharm 221: 1–22
Lee MY, Cagavi Bozkulak E, Schliffke S et al (2011) High density cultures of embryoid bodies enhanced cardiac differentiation of murine embryonic stem cells. Biochem Biophys Res Commun 416:51–57
Lu T-Y, Lin B, Kim J et al (2013) Repopulation of decellularized mouse heart with human induced pluripotent stem cell-derived cardiovascular progenitor cells. Nat Commun. doi:10.1038/ncomms3307
Mercola M, Ruiz-Lozano P, Schneider MD (2011) Cardiac muscle regeneration: lessons from development. Genes Dev 25:299–309
Oberwallner B, Brodarac A, Choi Y-H et al (2013) Preparation of cardiac extracellular matrix scaffolds by decellularization of human myocardium. J Biomed Mater Res A. doi: 10.1002/jbma.35000
Pachence JM (1996) Collagen-based devices for soft tissue repair. J Biomed Mater Res 33: 35–40
Sachs HG, DeHaan RL (1973) Embryonic myocardial cell aggregates: volume and pulsation rate. Dev Biol 30:233–240
Serpooshan V, Julien M, Nguyen O et al (2010) Reduced hydraulic permeability of three-dimensional collagen scaffolds attenuates gel contraction and promotes the growth and differentiation of mesenchymal stem cells. Acta Biomater 6:3978–3987
Serpooshan V, Muja N, Marelli B, Nazhat SN (2011) Fibroblast contractility and growth in plastic compressed collagen gel scaffolds with microstructures correlated with hydraulic permeability. J Biomed Mater Res A 96: 609–620
Serpooshan V, Quinn TM, Muja N, Nazhat SN (2012) Hydraulic permeability of multilayered collagen gel scaffolds under plastic compression-induced unidirectional fluid flow. Acta Biomater. doi:10.1016/j.actbio.2012.08.031
Serpooshan V, Quinn TM, Muja N, Nazhat SN (2011) Characterization and modelling of a dense lamella formed during self-compression of fibrillar collagen gels: implications for biomimetic scaffolds. Soft Matter 7:2918–2926
Serpooshan V, Zhao M, Metzler SA et al (2013) The effect of bioengineered acellular collagen patch on cardiac remodeling and ventricular function post myocardial infarction. Biomaterials 34:9048–9055
Serpooshan V, Zhao M, Metzler SA, et al. (2014) Use of biomimetic 3D technology in therapeutics for heart disease. Bioengineered (in press)
Smart N, Bollini S, Dube KN et al (2011) De novo cardiomyocytes from within the activated adult heart after injury. Nature 474:640–U117
Venugopal JR, Prabhakaran MP, Mukherjee S et al (2012) Biomaterial strategies for alleviation of myocardial infarction. J R Soc Interface 9:1–19
Vunjak-Novakovic G, Tandon N, Godier A et al (2010) Challenges in cardiac tissue engineering. Tissue Eng Part B Rev 16: 169–187
Zhang D, Shadrin IY, Lam J et al (2013) Tissue-engineered cardiac patch for advanced functional maturation of human ESC-derived cardiomyocytes. Biomaterials 34:5813–5820
Zimmermann WH, Fink C, Kralisch D et al (2000) Three-dimensional engineered heart tissue from neonatal rat cardiac myocytes. Biotechnol Bioeng 68:106–114
Zimmermann W-H, Schneiderbanger K, Schubert P et al (2002) Tissue engineering of a differentiated cardiac muscle construct. Circ Res 90:223–230
Acknowledgements
This work was supported by NIH grants HL65484 and HL086879 (to P.R.L.). V.S. was an Oak Foundation postdoctoral fellow at Stanford.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this protocol
Cite this protocol
Serpooshan, V., Ruiz-Lozano, P. (2014). Ultra-rapid Manufacturing of Engineered Epicardial Substitute to Regenerate Cardiac Tissue Following Acute Ischemic Injury. In: Kioussi, C. (eds) Stem Cells and Tissue Repair. Methods in Molecular Biology, vol 1210. Humana Press, New York, NY. https://doi.org/10.1007/978-1-4939-1435-7_18
Download citation
DOI: https://doi.org/10.1007/978-1-4939-1435-7_18
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-1434-0
Online ISBN: 978-1-4939-1435-7
eBook Packages: Springer Protocols