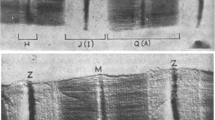
Abstract
In this chapter, first I will briefly describe the molecular properties of titin and nebulin — two extremely large, myofibrillar proteins -- and discuss their distribution and organization in the sarcomere. Although these novel proteins are major myofibrillar components of a wide range of striated muscles, they have escaped the attention of muscle biochemists until very recently. As I shall point out below, biochemical studies of these proteins have been unexpectedly challenging: many standard techniques had to be modified before they became capable of handling such giant proteins. In addition, our structural studies of these proteins have encountered a challange of a different nature: how to explain their distribution in the sarcomere according to the currently accepted two filament sarcomere model, because these proteins do not appear to be thick or thin filament-associated regulatory or anchoring proteins. These studies have led us to reexamine the question of whether continuous, longitudinal filaments exist within the sarcomere of striated muscle. I will attempt to integrate our results, as well as available literature data, within the framework of a hypothetical sarcomere model which incorporates an elastic filamentous matrix consisting of titin and nebulin as additional sarcomere constituents. Finally, I will very briefly mention our recent findings that an extensive three dimensional network of intermediate (10 nm) filaments, distinct from titin and nebulin, is intimately associated with the sarcomere of adult striated muscle.
I believe that the recognition of the existence of two sets of sarcomereassociated cytoskeletal filaments within adult striated muscle fibers may be a significant step toward resolving some of the unsettled questions in muscle mechanics such as those that have been discussed in this meeting
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Ashby, B., Frieden, C., and Bischoff, R. (1979). Immunofluorescent and histochemical localization of AMP deaminase in skeletal muscle J. Cell Biol. 81: 361–373.
Auber, J., and Couteaux, R. (1963). Ultrastructure de la strie Z dans des muscules de dipteres. J. Microscopy 2: 309.
Carlesen, F., Knappeis, G.G., and Buchtal, F. (1961). Ultrastructure of the resting and contracted striated muscle fiber at different degrees of stretch. J. Biophys. Biochem. Cyto. 11: 91–117.
dos Remedios, C., and Gilmour, D. (1978). Is there a third type of filament in striated muscle ? J. Biochem. 84: 235–238.
Ernst, E. and Straub, F.B. (1968). Symposium on Muscle. Budapest, Akademiai Kiado.
Etlinger, J.D., Zak, R, and Fischmann, D.A. (1976). Compositional studies of myofibrils from rabbit striated muscle. J. Cell Biol. 68: 123–141.
Franzini-Armstrong, C. (1970). Details of the I-band structure as revealed by the localization of ferritin. Tissue and Cell 2: 327–338.
Garamvolgyi, N., and Kerner, J. (1966). The ultrastructure of the insect flight muscle fibril ghost. Acta Biochim. et Biophys. Acad. Sol.. Hung. 1: 81–88.
Guba, F., Harsanyi, V., and Vajda, E. (1968). The muscle protein fibrillin. Acta Biochim. et Biophys. Acad. Sci. Hung. 3: 433–440.
Haseigrove, J.C., and Rodger, C.D. (1981). The interpretation of X-ray diffraction patterns from vertebrate striated muscle. J. Musc. Res. Cell Motil. 1: 371–390.
Hoyle, G. (1968). In: Symposium on Muscle ed. Ernst, E. and Straub, F.B. pp. 34 Budapest; Akademiai Kiado.
Huxley, A.F., and Peachey, L.D. (1981). The maximum length for contraction in vertebrate striated muscle. J. Physiol. 156: 150–165.
Huxley, H.E., and Hanson, J. (1954). Changes in the cross striations of muscle during contraction and stretch and their structural interpretation. Nature 173: 973–976.
Huxley, H.E. (1968). In: Symposium on Muscle ed. Ernst, E. and Straub, F.B. p. 247. Budapest; Akademiai Kiado.
Huxley, H.E. (1972). Molecular basis of contraction in cross-striated muscles. In: The Structure and Function of Muscle, Bourne, G.H. Ed. 2nd edition, Volume 1 pp. 301–387, Academic Press, London.
King, N.L. and Kurth, L. (1980). SDS gel electrophoresis studies of connectin. In: Fibrous Proteins: Scientific, Industrial and Medical aspects. Eds. Parry, D.A.D., and Creamer, L.K. Vol. 2, pp. 57–68, Academic Press, London.
Lazarides, E. (1980). Intermediate filaments as mechanical integrators of cellular space. Nature 283: 249–256.
Locker, R.H., and Leet, N.G. (1976). Histology of highly stretched beef muscle. H. Further evidence on the location and nature of gap filaments. J. Ultrastruc. Res. 55: 157–172.
Locker, R.H., and Dianes, G.J. (1980). Gap filaments–the third set in the myofibril. In: Fibrous Proteins: Scientific, Industrial and Medical Aspects. eds. Parry, D.A.D., and Creamer, L.K. Vol. 2, pp 43–55. Academic Press, London.
Maruyama, K., Matsubara, S., Natori, S.R., Nonomura, Y., Kimura, S., Ohashi, K., Murakami, F., Banda, S. and Eguchi, G. (1977). Connectin, and elastic protein in muscle. Characterization and function. J. Biochem. 82: 317–337.
Maruyama, K., Kimura, S., Ohashi, K., and Juwano, Y. (1980). Connectin, an elastic protein of muscle. In: Fibrous Proteins: Scientific, Industrial and Medical Aspects. eds. Parry, D.A.D., and Creamer, L.K., Volume 2, pp. 30–36. Academic Press, London.
McNeil, P.A., and Hoyle, G. (1967). Evidence for superthin filaments. Am. Zoologist 7: 483–498.
Organization of the Cytoplasm, Cold Spring Harbor Symp. Quant. Biol. vol. 46. Cold Spring Harbor Laboratory, New York.
Porzio, M.A., and Pearson, A.M. (1977). Improved resolution of myofibrillar proteins with sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Biochim. Biophys. Acta 490: 21–34.
Pringle, J.W.S. (1978). Stretch activation of muscle: function and mechanism. Proc. Roy. Soc. London B. 201: 107–130.
Sjostrand, F.S. (1962). The connection between A- and I-band filaments in striated frog muscle. J. Ultrastruct. Res. 7: 225–246.
Trinick, J. (1981). End filaments, a new structural element of vertebrate skeletal muscle thick filaments. J. Mol. Biol. 151: 309–314.
Walcott, B., and Ridgeway, E.B. (1967). The ultrastructure of myosin-extracted striated muscle fibers. Am. Zoologist 7: 499–503.
Wang, K., Ash, J.G., and Singer, S.J. (1975). Filamin, a new high molecular weight protein of smooth muscle and non-muscle cells. Proc. Natl. Acad. Sci. USA 72: 4483–4486.
Wang, K. and McClure, J. (1978). Extremely large proteins of vertebrate striated muscle myofibrils. J. Cell Biol. 79: 334a
Wang, K., McClure, J., and Tu, A. (1979). Titin: major myofibrillar components of striated muscle. Proc. Natl. Acad. Sci. USA 76: 3698–3702.
Wang, K., and Ramirez-Mitchell, R. (1979). Titin: possible candidate as components of putative longitudinal filaments in striated muscle. J. Cell Biol. 83: 389a.
Wang, K. and Williamson, C.L. (1980). Identification of an N2-line protein of striated muscle. Proc. Natl. Acad. Sci. USA 77: 3254–3258.
Wang, K., and Ramirez-Mitchell, R. (1981a). Myofibrillar connections. The role of titin, N2-line protein and intermediate filaments. Biophys. J. 33: 21a.
Wang, K., and Ramirez-Mitchell, R. (1981). Myofibrillar connections: The role of titin, nebulin, and intermediate filaments. J. Cell Biol. 83: 389a.
Wang, K. (1981). Nebulin, a giant protein component of N2-line striated muscle. J. Cell Biol. 91: 355a.
Wang, K., Feramìsco, J.R., and Ash, J.F. (1982). Fluorescent localization of contractile proteins. Methods in Enzymol. 85: 514–562.
Wang, K., and Ramirez-Mitchell, R. (1983). A network of transverse and longitudinal intermediate filaments is associated with the sarcomeres of adult vertebrate skeletal muscle. J. Cell Biol. 96: 562–570.
Yarom, R. and Meiri, U. (1971). N lines in striated muscle: a site of intracellular Cat*. Nature New Biology 234: 254–255.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1984 Plenum Press, New York
About this chapter
Cite this chapter
Wang, K. (1984). Cytoskeletal Matrix in Striated Muscle: The Role of Titin, Nebulin and Intermediate Filaments. In: Pollack, G.H., Sugi, H. (eds) Contractile Mechanisms in Muscle. Advances in Experimental Medicine and Biology, vol 37. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-4703-3_25
Download citation
DOI: https://doi.org/10.1007/978-1-4684-4703-3_25
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-4705-7
Online ISBN: 978-1-4684-4703-3
eBook Packages: Springer Book Archive