Abstract
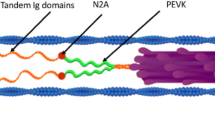
Titin (connectin) is a giant polypeptide that forms a single-molecule filamental structure extending from the M-line to the Z-line in the sarcomere of striated muscle. The primary structure of titin consists mainly of repeats of two types of ~100-amino acid modules (fibronectin type III and immunoglobulin-like motifs, respectively) and a Pro rich segment named the PEVK domain. The I-band region of titin shows an elasticity important to the passive properties of the myofibril. To investigate the biological function of titin, we cloned cDNA segments encoding single or linked structural modules of titin into expression vectors to produce non-fusion titin fragments in E. coli. High level expression of titin fragments was achieved and effective purification procedures were developed. We also developed specific monoclonal antibodies against the titin fragments and solid-phase protein-binding assays to investigate the interaction of the titin structural modules and other sarcomeric proteins. The results show that the immunoglobin-like module that are enriched in the I-band titin binds to F-actin. In contrast to the rigid association of A-band titin with the thick filament, the relatively weak titin-actin binding suggests that the I-band titin may reversibly interact with the thin filament during muscle contraction. This hypothesis is supported by an epitope similarity between the actin-binding site of caldesmon and the immunoglobulin-like module of titin, which suggests analogous functions of caldesmon and titin in organizing the contractile proteins. Together with data from mechanical studies demonstrating that the titin-actin interaction may contribute to the passive property of cardiac muscle in a Ca2+-dependent manner, we speculate that the Ca2+-mediated thin filament regulation may coordinate the function of titin during muscle contraction and relaxation.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Astier C, Raynaud F, Lebbart MC, Roustan C, Benyamin Y. Binding of native titin fragment to actin is regulated by PIP2. FEBS Lett 1998;429:95–98.
Ayme-Southgate A, Vigoreaux J, Benian G, Pardue ML. Drosophilahas a twitching/titin-related gene that appears to encode projectin. Proc NatlAcad Sci USA 1991;88:7973–7977.
Benian GM, Kiff JE, Neckelmann N, Moermann DG, Waterston RH. Sequence of an unusually large protein implicated in regulation of myosin activity in C. elegans. Nature 1989;342:45–50.
Eilertsen KJ, Kazmierski ST, Keller TC III. Cellular titin localization in stress fibers and interaction with myosin II filaments in vitro. J Cell Biol 1994;74:361–364.
Einheber S, Fischman DA. Isolation and characterization of a cDNA clone encoding avian skeletal muscle C-protein: an intracellular member of the immunoglobulin superfamily. Proc Natl Acad Sci USA 1990;87:2157–2161.
Granzier H, Irving TC. Passive tension in cardiac muscle: contribution of collagen, titin, microtubules, and intermediate filaments. Biophys J 1995;68:1027–1044.
Granzier H, Kellermayer M, Helmes M, Trombitás K. Titin elasticity and mechanism of passive force development in rat cardiac myocyte probed by thin-filament extraction. Biophys J 1997;73:2043–2053.
Helmes M, Trombitás K, Granzier H. Titin develops restoring force in rat cardiac myocytes. Circ Res 1996;79:619–626.
Jin J-P Molecular studies of cardiac troponin Tisoforms. Ph.D. thesis. The University of Iowa, Iowa City, Iowa, USA 1989.
Jin J-P. Cloned rat cardiac titin class I and class II motifs: Expression, purification, characterization and interaction with F-actin. J Biol Chem 1995;270:6908–6916.
Jin J-P, Wang K. Cloning, expression and protein interaction of human nebulin fragments composed of varying number of sequence modules. J Biol Chem 1991;266:21215–21223.
Jin J-P, Walsh MP, Resek ME, McMartin GA. Epitope structure and expression of calponin in different smooth muscles and during development. Biochem Cell Biol 1996;74:187–196.
Labeit S, Gautel M, Lakey A, Trinick J. Towards a molecular understanding of titin. EMBO J 1992;11:1711–1716.
Labeit S, Kolmerer B. Titin: giant protein in charge of muscle ultrastructure and elasticity. Science 1995;270:293–296.
Li Q, Jin J-P, Granzier H. The effect of genetically expressed cardiac titin fragments on in vitro actin motility. Biophys J 1995;69:1508–1518.
Lin JJ-C, Davis-Nanthakumar EJ, Jin J-P, Lourim D, Novy RE, Lin JL-C. Epitope mapping of monoclonal antibodies against caldesmon and their effects on the binding of caldesmon to Ca2+/calmodulin and to actin or actin-tropomyosin filaments. Cell Motil Cytoskel 1991;20:95–108.
Linke WA, Ivemeyer M, Labeit S, Hinssen H, Ruegg JC, Gautel M. Actin-titin interaction in cardiac myofibrils: Probing a physiological role. Biophys J 1997;73:905–919.
Linke WA, Stockmeier NR, Ivemeyer M, Hosser H, Mündel P. Characterizing titin’s I-b and Ig dom ain region as an entropie spring. J Cell Sci 1998;111:1567–1574.
Maruyama K. Connectin/titin, giant elastic protein of muscle. FASEB J 1997;11:341–345.
Ogut O, Jin J-P. Expression, zinc-affinity purification and characterization of a novel metal-binding cluster in troponin T: Metal-stabilized a-helical structure and effects of the NH2-terminal variable region on the conformation of intact troponin T and its association with tropomyosin. Biochemistry 1996;35:16581–16590.
Ogut O, Jin J-P. Developmental regulated, alternative RNA splicing-generated pectoral muscle-specific troponin T isoforms and role of the NH terrninal hypervariable region in the tolerance to acidosis. J Biol Chem.1998;273:27858–27866.
Olson NJ, Pearson RB, Needleman D, Hurwitz MY, Kemp BE, Means AR. Regulatory and structural motifs of chicken gizzard myosin light chain kinase. Proc Natl Acad Sci USA 1990;87:2284–2288.
Price MG, Gomer RH. Skelemin, A cytoskeletal M-disc periphery protein, contains motifs of adhesion/recognition and intermediate filament proteins. J Biol Chem 1993;268:21800–21810.
Raharjo WH, Lin JJ-C, Mak AS, Jin J-P. An epitope structure shared by titin and caldesmon. Biophys J 1996;70:A379.
Rief M, Gautel M, Schemmel A, Gaub HE. The mechanical stability of immunoglobulin and fibronectin III domains in the muscle protein titin measured by atomic force microscopy. Biophys J 1998;75:3008–3014.
Schagger H, von Jagow G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range of 1 ~ 100-kDa. Anal Biochem 1987;166:368–379.
Studier FW, Rosenberg AH, Dunn JJ, Dubendorff JW. Use of T7 RNA polymerase to direct expression of cloned genes. Method Enzymol 1990;185:60–89.
Stuyvers BD, Miura M, Jin J-P, ter Keurs HEDJ. Ca2+-dependence of diastolic properties of cardiac sarcomeres: involvement of titin. Prog Biophysics Mol Biol 1998a;vn69:425–443.
Stuyvers BD, Jin J-P, ter Keurs HEDJ. Involvement of titin in stiffness-[Ca2+] relationship of cardiac sarcomeres. Biophys J 1998b;74:A351.
Trinick J. Elastic filaments and giant proteins in muscle. Curr Opin Cell Biol 1991;3:112–118.
Trombitás K, Jin J-P, Granzier, H. The passive-tension domain of titin in cardiac muscle. Circ Res 1995;77:856–861.
Trombitás K, Greaser M, Labeit S, Jin J-P, Kellermayer M, Helmes M, Granzier H. Titin extensibility in situ: Entropicelasticity of both permanently folded and permanently unfolded molecular segments. J Cell Biol 1998;40:853–859.
Wang J, Jin J-P. Primary structure and developmental acidic to basic transition of 13 alternatively spliced mouse fast skeletal muscle troponin T isoforms. Gene 1997;193:105–114.
Wang J, Jin J-P. Conformationl modulation of troponin T by configuration of the NH2-terminal variable region and functional effects. Biochemistry 1998;37:14519–14528.
Wang K. “Sarcomere associated cytoskeletal lattice in striated muscle.” In Cell and Muscle Motility Vol. 6, JW Shay, ed. New York: Plenum Press, 1985;315–369.
Weikamp B, Jurk K, Beinbrech G. Projectin-thin filament interaction and modulation of the sensitivity of the actomyosin ATPase to calcium by projectin kinase. J Biol Chem 1998;273:19802–19808.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer Science+Business Media New York
About this chapter
Cite this chapter
Jin, JP. (2000). Titin-Thin Filament Interaction and Potential Role in Muscle Function. In: Granzier, H.L., Pollack, G.H. (eds) Elastic Filaments of the Cell. Advances in Experimental Medicine and Biology, vol 481. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-4267-4_19
Download citation
DOI: https://doi.org/10.1007/978-1-4615-4267-4_19
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6916-5
Online ISBN: 978-1-4615-4267-4
eBook Packages: Springer Book Archive