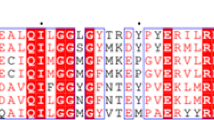
There have been several recent advances in the elucidation of the structure and mechanism of action of aldose reductase (ALR2; EC 1.1.1.21). The enzyme exhibits ordered sequential kinetics and during the kinetic mechanism a conformational change occurs upon coenzyme binding (Grimshaw et al., 1990; Kubiseski et al., 1992). Unlike most oxidoreductases the rate limiting step is not the release of NADP+ but rather, in the forward direction at least, the rate of isomerization of the binary enzyme-coenzyme complex, E*•NADP⇄E•NADP (Kubiseski et al., 1992). Chemical modification studies previously have suggested several amino acid residues essential for the function of the enzyme e.g. lysine (Morjana et al., 1989), arginine (Halder et al., 1985) and cysteine (Liu et al., 1989). The cloning and sequencing of ALR2 from several species has enabled the precise location of such residues in the primary structure (Carper et al., 1987; Garcia-Perez et al., 1989; Nishimura et al., 1990; Schade et al., 1990). Moreover, the development of expression systems for ALR2 has allowed an examination of critical residues by site-directed mutagenesis (Bohren et al., 1991; Yamaoka et al., 1992).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Bhatnagar, A., Das, B. & Srivastava, S.K., 1987, Diethyl pyrocarbonate inactivation of human placental aldehyde reductase II of Texas Medical Branch, Galveston 77550, Biochim. Biophys. Acta 916: 179.
Bohren, K.M., Bullock, B., Wermuth, B. & Gabbay, K.H., 1989, The aldo-keto reductase superfamily. cDNAs and deduced amino acid sequences of human aldehyde and aldose reductases, J. Biol. Chem. 264: 9547.
Bohren, K.M., Page, J.L., Shankar, R., Henry, S.P. & Gabbay, K.H., 1991, Expression of human aldose and aldehyde reductases. Site-directed mutagenesis of a critical lysine 262, J. Biol. Chem. 266: 24031.
Carper, D., Nishimura, C., Shinohara, T., Dietzchold, B., Wistow, G., Craft, C., Kador, P. & Kinoshita, J.H., 1987, Aldose reductase and p-crystallin belong to the same protein superfamily as aldehyde reductase, FEBS. Lett. 220: 209.
Cromlish, J.A. & Flynn, T.G., 1983, Identity of pig muscle aldehyde reductase with pig lens aldose reductase and with the low KM aldehyde reductase of pig brain and pig kidney, J. Biol. Chem. 258: 3583.
Feinberg, A.P. & Vogelstein, B., 1983, A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity, Anal. Biochem. 132: 6.
Garcia-Perez, A., Martin, B., Murphy, H.R., Uchida, S., Murer, H., Cowley, B.D., Jr., Handler, J.S. & Burg, M.B., 1989, Molecular cloning of cDNA coding for kidney aldose reductase. Regulation of specific mRNA accumulation by NaCl-mediated osmotic stress, J. Biol. Chem. 264: 16815.
Grimshaw, C.E., Shahbaz, M. & Putney, C.G., 1990, Mechanistic basis for nonlinear kinetics of aldehyde reduction catalyzed by aldose reductase, Biochemistry 29: 9947.
Halder, A.B., James, M. & Crabbe, C., 1985, Chemical modification studies on purified bovine lens aldose reductase, Ophthalmic. Res. 17: 185.
Kubiseski, T.J., Hyndman, D.J., Morjana, N.A. & Flynn, T.G., 1992, Studies on pig muscle aldose reductase. Kinetic mechanism and evidence for a slow conformational change upon coenzyme binding, J. Biol. Chem. 267: 6510.
Liu, S., Bhatnagar, A., Das, B. & Srivastava, S.K., 1989, Functional cysteinyl residues in human placental aldose reductase, Arch. Biochem. Biophys. 275: 112.
Maniatis, T., Fritsch, E.F. & Sambrook, J. (1982) Molecular Cloning; A Laboratory Manual, Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
Morjana, N.A., Lyons, C. & Flynn, T.G., 1989, Aldose reductase from human psoas muscle. Affinity labeling of an active site lysine by pyridoxal 5’-phosphate and pyridoxal 5’-diphospho-5’-adenosine, J. Biol. Chem. 264: 2912.
Nihira, T., Toraya, T. & Fukui, S., 1981, Modification of tryptophanase with tetranitromethane, Eur. J. Biochem. 119: 273.
Nishimura, C., Matsuura, Y., Kokai, Y., Akera, T., Carper, D., Morjana, N., Lyons, C. & Flynn, T.G., 1990, Cloning and expression of human aldose reductase, J. Biol. Chem. 265: 9788.
Penefsky, S.H., 1971, A centrifuged-column procedure for the measurement of ligand binding by beef heart F1, Methods Enzymol. 56: 527.
Rondeau, J.-M., Tête-Favier, F., Podjarny, A., Reymann, J.-M., Barth, P., Biellmann, J.-F. & Moras, D., 1992, Novel NADPH-binding domain revealed by the crystal structure of aldose reductase, Nature 355: 469.
Saiki, R.K., Scharf, S., Faloona, F., Mullis, D.B., Horn, G.T., Erlich, H.A. & Arnhein, N., 1985, Enzymatic amplification of ß-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia, Science 230: 1350.
Schade, S.Z., Early, S.L., Williams, T.R., Kézdy, F.J., Heinrikson, R.L., Grimshaw, C.E. & Doughty, C.C., 1990, Sequence analysis of bovine lens aldose reductase, J. Biol. Chem. 265: 3628.
Stone, C.L., Li, T.-K. & Bosron, W.F., 1989, Stereospecific oxidation of secondary alcohols by human alcohol dehydrogenases, J. Biol. Chem. 264: 11112.
Wilson, D.K., Bohren, K.M., Gabbay, K.H. & Quiocho, F.A., 1992, An unlikely sugar substrate site in the 1.65 Å structure of the human aldose reductase holoenzyme implicated in diabetic complications, Science 257: 81.
Yamaoka, T., Matsuura, Y., Yamashita, K., Tanimoto, T. & Nishimura, C., 1992, Site-directed mutagenesis of His-42, His-188 and Lys-263 of human aldose reductase, Biochem. Biophys. Res. Commun. 183: 327.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1993 Springer Science+Business Media New York
About this chapter
Cite this chapter
Kubiseski, T.J., Green, N.C., Flynn, T.G. (1993). Location of an Essential Arginine Residue in the Primary Structure of Pig Aldose Reductase. In: Weiner, H., Crabb, D.W., Flynn, T.G. (eds) Enzymology and Molecular Biology of Carbonyl Metabolism 4. Advances in Experimental Medicine and Biology, vol 328. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-2904-0_28
Download citation
DOI: https://doi.org/10.1007/978-1-4615-2904-0_28
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6259-3
Online ISBN: 978-1-4615-2904-0
eBook Packages: Springer Book Archive