Abstract
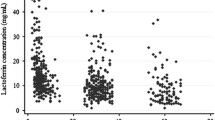
Changes in the lactoferrin and lysozyme concentration of human milk during lactation were determined by microparticle-enhanced nephelometric immunoassays of 360 milk samples collected from 64 lactating volunteers. These 360 samples were colostrum from days 1 to 5 postpartum (142 samples), transitional milk from days 6 to 14 (106 samples), and 112 mature milk samples obtained from days 15 to 28 (34 samples), from days 29 to 56 (50 samples) and from days 57 to 84 postpartum (28 samples). The concentration and percentage of lactoferrin vs. total protein were found to be significantly higher in colostrum (5.8 g/L, 27%) than in transitional milk (3.1 g/L, 22%) or day 15 to 28 mature milk (2 0 g/L, 19%), then increased in day 29 to 56 mature milk (2 2 g/L, 22%) and day 57 to 84 mature milk (3 3 g/L, 30%). The concentration of lysozyme decreased from colostrum (0.37 g/L) to transitional milk (0.27 g/L) and day 15 to 28 mature milk (0.24 g/L), then increased in day 29 to 56 mature milk (0 33 g/L) and was highest in day 57 to 84 mature milk (0 89 g/L). The percentage of lysozyme vs. total protein was found to be always rising during lactation: colostrum, 2%; transitional milk, 2%; days 15 to 28, 2%; days 29 to 56, 3%; and days 57 to 84 mature milk, 8%.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Anderson BF, Baker HM, Norris GE, Rice DW, Baker EN. Structure of human lactoferrin: Crystallo graphic structure analysis and refinement at 2.8 A resolution. J Mol Biol 1989;209:711–734.
Birgens HS, Hansen NE, Karle H, Kristensen LO. Receptor binding of lactoferrin by human monocytes. Brit J Haematol 1983;54:383–391.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–254.
Chandan RC, Parry RM, Shahani KM. Lysozyme, lipase, and ribonuclease in milk from various species. J Dairy Sci 1968;51:606–607.
Chang SJ. Antimicrobial proteins of maternal and cord sera and human milk in relation to maternal nutritional status. Am J Clin Nutr 1990;51:183–187.
Chipman DM, Sharon N. Mechanism of lysozyme action. Science 1969;165:454–465.
Cox TM, Mazurier J, Spik G, Montreuil J, Peters TJ. Iron binding proteins and influx of iron across the duodenal brush border. Evidence for specific lactotransferrin receptors in human intestine. Biochim Biophys Acta 1979;588:120–128.
Cuillière ML, Montagne P, Bessou T, El Omani R, Riochet D, Varcin P, Laroche P, Prud’homme P, Marchand J, Flecheux O, Pau B, Duheille J. Microparticle-enhanced nephelometric immunoassay (Néphélia®) for Immunoglobulins G, A, and M. Clin Chem 1991;37:20–25.
Eaton SS, Dubach J, Eaton GR, Thurman G, Ambruso DR. Electron spin echo envelope modulation evidence for carbonate binding to iron(III) and copper(II) transferrin and lactoferrin. J Biol Chem 1990;265:7138–7141.
Ekstrand B, Bjorck L. Fast protein liquid chromatography of antibacterial components in milk: Lactoperoxidase, lactoferrin and lysozyme. J Chromatogr 1986;358:429–433.
Fransson GB, Lönnerdal B. Iron in human milk. J Pediatr 1980;96:380–384.
Goldman AS, Garza C, Johnson CA, Nichols BL, Goldblum RM. Immunologic factors in human milk during the first year of lactation. J Pediatr 1982;100:563–567.
Hennart PF, Brasseur DJ, Delogne-Desnoeck JB, Dramaix MM, Robyn CE. Lysozyme, lactoferrin, and secretory immunoglobulin A content in breast milk: Influence of duration of lactation, nutrition status, prolactin status, and parity of the mother. Am J Clin Nutr 1991;53:32–39.
Johansson BG. Isolation of an iron-containing red protein from human milk. Acta Chem Scand 1960; 14:510–512.
Jolies J, Jolies P. Human tear and human milk lysozyme. Biochemistry 1967;6:411–417.
Kawakami H, Lönnerdal B. Isolation and function of a receptor for human lactoferrin in human fetal intestinal brush-border membranes. Am J Physiol 1991;261:841–846.
Marchand J, Varcin P, Riochet D, Montagne P, Cuillière ML, Duheille J, Pau B. Synthesis of new hydrophilic microspheres: Optimized carriers for microparticle-enhanced nephelometric immunoassay. Biopolymers 1992;32:971–980.
Masson PL, Heremans JE Metal-combining properties of human lactoferrin (red milk protein). The involvement of bicarbonate in the reaction. Eur J Biochem 1968;6:579–584.
Miranda R, Saravia NG, Ackerman R, Murphy N, Berman S, McMurray DN. Effect of maternal nutritional status on immunological substances in human colostrum and milk. Am J Clin Nutr 1983; 37:632–640.
Montagne P, Laroche P, Bessou T, Cuillière ML, Varcin P, Duheille J. Measurement of eleven serum proteins by microparticle-enhanced nephelometric immunoassay. J Clin Chem Clin Biochem 1992a; 30:217–222.
Montagne P, Varcin P, Cuillière ML, Duheille J. Microparticle-enhanced nephelometric immunoassay with microsphere-antigen conjugates. Bioconjugate Chem 1992b;3:187–193.
Montagne P, Cuillière ML, Marchai E, El Bari N, Montagne M, Benali M, Faure G, Duheille J, Humbert G, Linden G, Heurtaux N, Blesche JL, Gosselin D, Desmares A, Delahaye D. Application of microparticle-enhanced nephelometric immunoassay of alpha-, beta-and kappa-caseins to evaluate quality of milk, from the production to the valorization in cheese industry. Lait 1995;75:221–237.
Nichols BL, Mckee KS, Henry JF, Nichols VN. Human lactoferrin stimulates thymidine incorporation into DNA of rat crypt cells. Pediatr Res 1987;21:563–567.
Peters CW, Kruse U, Pollwein R, Grzeschik KH, Sippel AE. The human lysozyme gene. Sequence organization and chromosomal localization. Eur J Biochem 1989;182:507–516.
Rey MW, Woloshuk SL, De Boer HA, Pieper FR. Complete nucleotide sequence of human mammary gland lactoferrin. Nucleic Acids Res 1990;18:52–88.
Sanchez-Pozo A, Lopez J, Pita ML, Izquierdo A, Guerrero E, Sanchez-Medina F, Martinez Valverde A, Gil A. Changes in the protein fractions of human milk during lactation. Ann Nutr Metab 1986; 30:15–20.
Spik G, Strecker G, Fournet B, Bouquelet S, Montreuil J. Primary structure of the glycans from human lactotransferrin. Eur J Biochem 1982;121:413–419.
Stuart J, Norrel S, Harrington JP. Kinetic effect of human lactoferrin on the growth of Escherichia coli. J Biochem 1984;16:1043–1047.
Tomita M, Bellamy W, Takase M, Yamauchi K, Wakabayashi H. Potent antibacterial peptides generated by pepsin digestion of bovine lactoferrin. J Dairy Sci 1991;74:4137–4142.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2001 Springer Science+Business Media New York
About this chapter
Cite this chapter
Montagne, P., Cuillière, M.L., Molé, C., Béné, M.C., Faure, G. (2001). Changes in Lactoferrin and Lysozyme Levels in Human Milk During the First Twelve Weeks of Lactation. In: Newburg, D.S. (eds) Bioactive Components of Human Milk. Advances in Experimental Medicine and Biology, vol 501. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-1371-1_30
Download citation
DOI: https://doi.org/10.1007/978-1-4615-1371-1_30
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-5521-2
Online ISBN: 978-1-4615-1371-1
eBook Packages: Springer Book Archive