Abstract
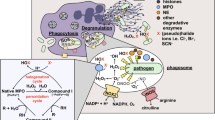
Reactive oxygen species (ROS) are cytotoxic agents produced by many phagocytic cells in response to membrane perturbations such as receptor-ligand interactions and phagocytosis. These agents are used to defend against infectious diseases by virtue of their antimicrobial properties. The initial ROS generated, the superoxide anion (O2-), is spontaneously or enzymatically converted to hydrogen peroxide (H2O2), which can give rise to even more toxic products, such as hydroxyl radical (•0H), hypochlorous acid (HOCI), and singlet oxygen (1O2). The ROS are toxic in their own right or may exert increased antimicrobial activity in concert with lysosomal hydrolases and/or reactive nitrogen species (RNS). Phagocyte activation triggers the assembly of a membrane-associated enzyme NADPH oxidase, which produces O from molecular oxygen and NADPH. Over-production of ROS by blood cells can overwhelm antioxidant defenses leading to various manifestations of oxidative stress. Human beings with genetic abnormalities in NADPH oxidase present with recurrent bacterial and fungal infections, granulomatous infiltration of many organs, and early deaths characteristic of chronic granulomatous disease (Bridges, Berendes and Good, 1959).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Adema C.M., van der Knaap W.P.W., Sminia T. Molluscan hemocyte-mediated cytotoxicity: the role of reactive oxygen intermediates. Rev. Aquat. Sci. 1991;4:201–223.
Adema C.M., van Deutekom-Mulder E.C., van der Knapp W.P.W., Smina T. NADPH-oxidase activity: the probable source of reactive oxygen intermediate generation in hemocytes of the gastropod Lymnea stagnalis. J. Leukoc. Biol. 1993;54:379–383.
Albrecht D., Jungi T.W. Luminol-enhanced chemiluminescence induced in peripheral blood-derived human phagocytes: obligatory requirement of myeloperoxidase exocytosis by monocytes. J. Leukoc. Biol. 1993;54:300–306.
Anderson R.S. “Modulation of blood cell mediated oxyradical production in aquatic species: implications and applications.” In Aquatic Toxicology: Molecular,Biochemical, and Cellular Perspectives, D.C. Malins and G.K. Ostrander, eds. Boca Raton: Lewis Publishers, 1994.
Anderson R.S. “Production of reactive oxygen intermediates by invertebrate hemocytes: immunological significance.” In New Directions in Invertebrate Immunology, K. Söderhäll, G. Vasta and S. Iwanaga, eds. Fair Haven, NJ: SOS Publications, 1996.
Anderson R.S. Perkinsus marinus secretory products modulate superoxide anion production by oyster (Crassostrea virginica) hemocytes. Fish Shellfish Immunol. 1999;9:51–60.
Anderson R.S. Lack of hemocyte chemiluminescence stimulation by Perkinsus marinus in oysters Crassostrea virginica with dermo disease. J. Aquat. Animal Health. In press.
Anderson R.S., Brubacher L.L., Ragone Calvo L.M., Burreson E.M., Unger M.A. Effect of in vitro exposure to tributyltin on generation of oxygen metabolites by oyster hemocytes. Environ. Res. 1997;74:84–90.
Anderson R.S., Brubacher L.L., Ragone-Calvo L., Unger M.A., Burreson E.M. Effects of tributyltin and hypoxia on the progression of Perkinsus marinus infections and host defence mechanisms in Crassostrea virginica (Gmclin). J. Fish Dis. 1998;21:371–379.
Anderson R.S., Burreson E.M., Paynter K.T. Defense responses of hemocytes withdrawn from Crassostrea virginica infected with Perkinsus marinus. J. Invertebr. Pathol. 1995;66:82–89.
Anderson R.S., Mora L.M., Thomson S.A. Exposure of oyster macrophages to particulate brass suppresses luminol-augmented chemiluminescence. Toxicologist 1994a;12:391.
Anderson R.S., Mora L.M., Thomson S.A. Modulation of oyster (Crassostrea virginica) hemocyte immune function by copper, as measured by luminol-enhanced chemiluminescence. Comp. Biochem. Physiol. 1994b;108C:215–220.
Anderson R.S., Oliver L.M., Jacobs D. Immunotoxicity of cadmium for the eastern oyster (Crassostrea virginica [Gmelin, 1791]): effects on hemocyte chemiluminescence. J. Shellfish Res. 1992;11:31–35.
Anderson R.S., Paynter K.T., Burreson E.M. Increased reactive oxygen intermediate production by hemocytes withdrawn from Crassostrea virginica infected with Perkinsus marinas. Biol. Bull. 1992;183:476–481.
Anderson R.S., Unger M.A., Burreson E.M. Enhancement of Perkinsus marinus disease progression in TBT-exposed oysters (Crassostrea virginica). Mar. Environ. Res. 1996;42:177–180.
Austin K.A., Paynter K.T. Characterization of the chemiluminescence measured in hemocytes of the Eastern oyster, Crassostrea virginica. J. Exp. Zool. 1995;273:461–471.
Babior B.M. Oxidants from phagocytes: agents of defense and destruction. Blood 1984;64:959–966. Baier-Anderson C., Anderson R.S. The effect of pentachlorophenol on NADPH production in oyster hemocytes: immunomodulatory consequences. J. Shellfish Res. 1996;15:498–499.
Baier-Anderson C., Anderson R.S. The effect of pentachlorophenol on pyridine nucleotide production in oyster hemocytes: NADPH and immunomodulatio. J. Shellfish Res. 1997;16:111–114.
Bramble L.H., Anderson R.S. A comparison of the chemiluminescent response of Crassostrea virginica and Morone saxatilis phagocytes to zymosan and viable Listonella anguillarum. Dev. Comp. Immunol. 1998;22:55–61.
Bramble L.H., Anderson R.S. Lack of involvement of reactive oxygen species in the bactericidal activity of Crassostrea virginica hacmocytes in contrast to Morone saxatilis phagocytes. Fish Shellfish Immunol. 1999;9:109–123.
Bridges R.A., Berendes H., Good R.A. A fatal granulomatous disease of childhood. Amer. J. Dis. Child. 1959;97:387.
Bushek D., Allen S.K., Alcox K.A., Gustafson R., Ford S. Dose response of the eastern oyster, Crassostrea virginica, to cultured cells of Perkinsus marinus, the agent of Dermo disease. J. Shellfish Res. 1994;13:313.
Chu F.-L.E., Hale R.C. Relationship between pollution and susceptibility to infectious disease in the Eastern oyster, Crassostrea virginica. Mar. Environ. Res. 1994;38:243–256.
Chu F.-L.E., Volety A.K., Lingenfelser J.T., Hale R.C. Modulation of hemocyte activities in oysters (Crassostrea virginica) upon exposure to PAHs. SETAC Abstract Book, 17th Annual Meeting, SETAC Press, Pensacola, FL. p 150.
Coles J.A., Farley S.R., Pipe R.K. Alterations of the immune response of the common marine mussel Mytilus edulis resulting from exposure to Cadmium. Dis. Aquat. Org. 1995;22:59–65.
Connors V.A., Lodes M.J., Yoshino T.P. Identification of a Schistosoma mansoni sporocyst execretorysecretory antioxidant molecule and its effect on superoxide production by Biomphalaria glabrata hemocytes. J. Invertebr. Pathol. 1991;58:387–395.
Connors V.A., Yoshino T.P. In vitro effect of larval Schistosoma mansoni cxecretory-secretory products on phagocytosis-stimulated superoxide production in hemocytes from Biomphalaria glabrata. J. Parasitol. 1990;76:895–902.
Dyrynda E.A., Law R.J., Dyrynda P.E.J., Kelly C.A., Pipe R.K., Graham K.L., Ratcliffe N.A. Modulations in cell-mediated immunity of Mytilus edulis following the `Sea Empress’ oil spill. J. Mar. Biol. Assoc. UK 1997;77:281–284.
Dyrynda E.A., Pipe R.K., Burt G.R., Ratcliffe N.A. Modulations in the immune defenses of mussels (Mytilus edulis) from contaminated sites in the UK. Aquat. Toxicol. 1998;42:169–185.
Fisher W.S., Oliver L.M., Sutton E.B., Manning C.S., Walker W.W. Exposure of eastern oysters to tributyltin increases the severity of Perkinsus marinas disease. J. Shellfish Res. 1995;14:265–266.
Fisher W.S., Wishkovsky A., Chu F.-L.E. Effects of tributyltin on defense-related activities of oyster hemocytes. Arch. Environ. Contam. Toxicol. 1990;19:354–360.
Hervio D., Bachère E., Mialhe E., Grizel H. Chemiluminescent responses of Ostrea edulis and Crassostrea gigas to Bonamia ostrea (Ascetospora). Dev. Comp. Immunol. 1989;13:449.
LaPeyre J.F. (1993): Studies on the oyster pathogen Perkinsus marinus (Apicomplexa): interactions with host defenses of C. virginica and C. gigas, and in vitro propagation. Ph.D. Dissertation, The College of William and Mary, Gloucester Point, VA.
LaPeyre J.F., Chu F.-L.E., Vogelbein W.K. In vitro interaction of Perkinsus marinus with hemocytes from eastern and Pacific oysters, Crassostrea virginica and Crassostrea gigas. J. Shellfish Res. 1992;11:200.
Larson K.G., Roberson B.S., Hetrick F.M. Effect of environmental pollutants on the chemiluminescence of hemocytes from the American oyster Crassostrea virginica. Dis. Aquat. Org. 1989;6:131–136.
LeGall G., Bachère E., Mailhe E. Chemiluminescence analysis of the activity of Pecten maximus hemocytes stimulated with zymosan and host-specific Rickettsiales-like organisms. Dis. Aquat. Org. 1991;11:181–186.
LeGall G., Bachère E., Mialhe E., Grizel H. Zymosan and specific-rickettsia activation of oxygen free radicals production in Pecten maximus hemocytes. Dev. Comp. Immunol. 1989;13:448.
Noël D., Bachère E., Mialhe E. Phogocytosis associated chemiluminescence of hemocytes in Mytilus edulis (Bivalvia). Dev. Comp. Immunol. 1993;17:483–493.
Oliver L.M., Sutton E.B., Fisher W.S. Effects of tributyltin exposure on oyster (Crassostrea virginica) defense functions. J. Shellfish Res. 1995;14:274.
Pipe R.K., Coles J.A. Environmental contaminants influencing immune function in marine bivalve molluscs. Fish Shellfish Immunol. 1995;5:581–595.
Pipe R.K., Coles J.A., Thomas M.E., Fossato V.U., Pulsford A.L. Evidence for environmentally derived immunomodulation in mussels from the Venice Lagoon. Aquat. Toxicol. 1995;32:59–73.
Roszell L.E., Anderson R.S. Effects of pentachlorophenol on the chemiluminescent response of phagocytes from two estuarine species. Toxicologist 1992;12:392.
Tam P.E., Hinsdill R.D. Screening for immunomodulators: effects of xenobiotics on macrophage chemiluminescence in vitro. Fund. Appl. Toxicol. 1990;14:542–553.
Volety A.K., Chu F.-L.E. Suppression of chemiluminescence of eastern oyster (Crassostrea virginica) hemocytes by the protozoan parasite Perkinsus marinus. Dev. Comp. Immunol. 1995;19:135–142.
Winstead J.T., Couch J.A. Enhancement of protozoan pathogen Perkinsus marinus infections in American oysters Crassostrea virginica exposed to the chemical carcinogen n-nitrosodicthylamine (DENA). Dis. Aquat. Org. 1988;5:205–213.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2001 Springer Science+Business Media New York
About this chapter
Cite this chapter
Anderson, R.S. (2001). Reactive Oxygen Species and Antimicrobial Defenses of Invertebrates: A Bivalve Model. In: Beck, G., Sugumaran, M., Cooper, E.L. (eds) Phylogenetic Perspectives on the Vertebrate Immune System. Advances in Experimental Medicine and Biology, vol 484. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-1291-2_12
Download citation
DOI: https://doi.org/10.1007/978-1-4615-1291-2_12
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-5481-9
Online ISBN: 978-1-4615-1291-2
eBook Packages: Springer Book Archive