Abstract
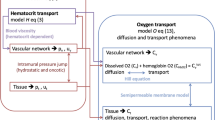
We describe recent models of oxygen transport in tissue along the pathwayfrom the hemoglobin molecule to the mitochondria and illustrate their applications. Microvasculature is the major site of exchange between blood and parenchymal cells for gases (O2, CO2, CO, NO), nutrients, metabolic products, and drugs. These exchange processes are affected by the architecture of the microvessels and the surrounding cells; distribution of blood flow; transport characteristics of blood, cells, and interstitial space; and rates of various chemical reactions associated with the transport processes. These processes operate at multiple levels of biological organization, from the molecular to the organ levels. Quantitative understanding of molecular transport in cells and tissues, specifically of oxygen transport, is the prerequisite for understanding the mechanisms of many diseases and for designing effective therapies. Mathematical and computational models provide a powerful set of tools for studies of these complex phenomena.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Krogh A. The number and distribution of capillaries in muscles with calculations of theoxygen pressure head necessary for supplying the tissue. J Physiol (London) 1919;52:409.
Krogh A. The Anatomy and Physiology of Capillaries, New Haven: Yale University Press, 1922;Pages.
Popel AS. Theory of oxygen transport to tissue. Crit Rev Biomed Eng 1989;17:257–321.
Hellums JD, Nair PK, Huang NS, Ohshima N. Simulation of intraluminal gas transport processes in the microcirculation. Ann Biomed Eng 1996;24:1–24.
Ellsworth ML, Ellis CG, Popel AS, Pittman RN. Role of Microvessels in oxygen supply to tissue. News Physiol. Sci. 1994;9:119–123.
Roy TK, Popel AS. Theoretical predictions of end-capillary P02 in muscles of athletic and nonathletic animals at VO2max. Am J Physiol 1996; 271:H721–737.
Eggleton CD, Vadapalli AR, Roy TK, Popel AS. Calculations of intracapillary oxygen tension distributions in muscle. Math Biosci 2000; 167:123–143.
Henry CB, Duling BR. Permeation of the luminal capillary glycocalyx is determined by hyaluronan. Am J Physiol 1999;277:H508–514.
Secomb TW, Hsu R, Pries AR. A model for red blood cell motion in glycocalyx-lined capillaries. Am J Physiol 1998;274:H1016–1022.
Wang CH, Popel AS. Effect of red blood cell shape on oxygen transport in capillaries. Math Biosci 1993;116:89–110.
Hsia CC, Johnson RL, Jr., Shah D. Red cell distribution and the recruitment of pulmonary diffusing capacity [see comments]. J Appl Physiol 1999;86:1460–1467.
Dutta A, Popel AS. A theoretical analysis of intracellular oxygen diffusion. J Theor Biol 1995;176:433–445.
Liu CY, Eskin SG, Hellums JD. The oxygen permeability of cultured endothelial cell monolayers. Adv Exp Med Biol 1994;345:723–730.
Vadapalli A, Pittman RN, Popel AS. Estimating oxygen transport resistance of the microvascular wall. Am J Physiol 2000;279:H657–H671.
Buerk DG, Goldstick TK. Arterial wall oxygen consumption rate varies spatially. Am J Physiol 1982;243: H948–958.
Tsai AG, Friesenecker B, Mazzoni MC, Kerger H, Buerk DG, Johnson PC, Intaglietta M. Microvascular and tissue oxygen gradients in the rat mesentery. Proc Natl Acad Sci USA 1998;95:6590–6595.
Baranov VI, Belichenko VM, Shoshenko KA. [The oxygen diffusion coefficient in isolated skeletal muscle fibers]. Fiziol Zh SSSR Im I M Sechenova 1991;77:29–34.
Groebe K. An easy-to-use model for O2 supply to red muscle. Validity of assumptions, sensitivity to errors in data [published erratum appears in Biophys J 1995 69:288]. Biophys J 1995;68:1246–1269.
Papadopoulos S, Jurgens KD, Gros G. Diffusion of myoglobin in skeletal muscle cells--dependence on fibre type, contraction and temperature. Pflugers Arch 1995;430:519–525.
Conley KE, Jones C. Myoglobin content and oxygen diffusion: model analysis of horse and steer muscle. Am J Physiol 1996;271:C2027–2036.
Conley KE, Ordway GA, Richardson RS. Deciphering the mysteries of myoglobin in striated muscle. Acta Physiol Scand 2000;168:623–34.
Honig CR, Gayeski TE, Federspiel W, Clark A, Jr., Clark P. Muscle O2 gradients from hemoglobin to cytochrome: new concepts, new complexities. Adv Exp Med Biol 1984;169:23–38.
Clark A, Jr., Clark PA, Connett RJ, Gayeski TE, Honig CR. How large is the drop in pO2 between cytosol and mitochondrion? Am J Physiol 1987;252:C583–587.
Mathieu-Costello O. Comparative aspects of muscle capillary supply. Annu Rev Physiol 1993;55:503–525.
Pries AR, Secomb TW, Gessner T, Sperandio MB, Gross JF, Gaehtgens P. Resistance to blood flow in microvessels in vivo. Circ Res 1994;75:904–915.
Hudetz AG. Blood flow in the cerebral capillary network: a review emphasizing observations with intravital microscopy. Microcirculation 1997; 4:233–252.
Ellis CG, Ellsworth ML, Pittman RN, Burgess WL. Application of image analysis for evaluation of red blood cell dynamics in capillaries. Microvasc Res 1992;44:214–225.
Damiano ER. The effect of the endothelial-cell glycocalyx on the motion of red blood cells through capillaries. Microvasc Res 1998;55:77–91.
Kiani MF, Pries AR, Hsu LL, Sarelius IH, Cokelet GR. Fluctuations in microvascular blood flow parameters caused by hemodynamic mechanisms. Am J Physiol 1994;266:H1822–1828.
Page TC, Light WR, Hellums JD. Prediction of microcirculatory oxygen transport by erythrocyte/hemoglobin solution mixtures. Microvasc Res 1998;56:113–126.
Eggleton CD, Roy TK, Popel AS. Predictions of capillary oxygen transport in the presence of fluorocarbon additives. Am J Physiol 1998; 275:H2250–2257.
Sharan M, Popel AS, Hudak ML, Koehler RC, Traystman RJ, Jones MD, Jr. An analysis of hypoxia in sheep brain using a mathematical model. Ann Biomed Eng 1998;26:48–59.
Ye GF, Jaron D, Buerk DG, Chou MC, Shi W. O2-Hb reaction kinetics and the Fahraeus effect during stagnant, hypoxic, and anemic supply deficit. Ann Biomed Eng 1998;26:6075.
Vaughn MW, Kuo L, Liao JC. Estimation of nitric oxide production and reaction rates in tissue by use of a mathematical model. Am J Physiol 1998;274:H2163–2176.
Vaughn MW, Kuo L, Liao JC. Effective diffusion distance of nitric oxide in the microcirculation. Am J Physiol 1998;274:H1705–1714.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2003 Springer Science+Business Media New York
About this chapter
Cite this chapter
Popel, A.S., Goldman, D., Vadapalli, A. (2003). Modeling of Oxygen Diffusion from the Blood Vessels to Intracellular Organelles. In: Dunn, J.F., Swartz, H.M. (eds) Oxygen Transport to Tissue XXIV. Advances in Experimental Medicine and Biology, vol 530. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-0075-9_46
Download citation
DOI: https://doi.org/10.1007/978-1-4615-0075-9_46
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-4912-9
Online ISBN: 978-1-4615-0075-9
eBook Packages: Springer Book Archive