Abstract
Taurine protects against tissue damage in a variety of models involving inflammation, especially the muscle. We set up a heavy exercise bout protocol for rats consisting of climbing ran on a treadmill to examine the effect of an intraabdominal dose of taurine (300 mg/kg/day) administered 1 h before heavy exercise for ten consecutive days. Each group ran on the treadmill at 20 m/min, 25% grade, for 20 min or until exhaustion within 20 min once each 10 days. Exhaustion was the point when an animal was unable to right itself when placed on its side. The muscle damage was associated with an increased accumulation of 8-nitroguanine and 8-OHdG in the nuclei of skeletal muscle cells. The immunoreactivities for NF-κB and iNOS were also increased in the exercise group. Taurine ameliorated heavy exercise-induced muscle DNA damage to a significant extent since it reduced the accumulation of 8-nitroguanine and 8-OHdG, possibly by down-regulating the expression of iNOS through a modulatory action on NF-κB signaling pathway. This study demonstrates for the first time that taurine can protect against intense exercise-induced nitrosative inflammation and ensuing DNA damage in the skeletal muscle of rats by preventing iNOS expression and the nitrosative stress generated by heavy exercise.
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Heavy exercise is thought to increase oxidative stress and to damage muscle tissue. Taurine protects against tissue damage in a variety of experimental models involving oxidative stress, especially during exercise. The mechanism of taurine protection is not well understood, but the ability of taurine to attenuate the toxic effects of HOCl/OCl via formation of taurine chloramine (TauCl) and its subsequent effects are thought to be important. Nitrosative stress-mediated activation of inflammatory mediators is currently being emphasized as an important factor mediating inflammation-related disorders. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) are capable of causing damage to various cellular constituents, such as nucleic acids, proteins and lipids. ROS can induce the formation of oxidative DNA lesion products, including 8-hydroxyldeoxyguanosine (8-OHdG) which is considered to be mutagenic. On the other hand, nitric oxide (NO) is generated specifically during inflammation via inducible nitric oxide synthase (iNOS) in inflammatory cells. Excess NO production plays a crucial role in an enormous variety of pathological processes, including apoptosis. NO can react with superoxide anion (O2 −) to form peroxynitrite (ONOO−), a highly reactive nitrogen species capable of causing nitrosative and oxidative DNA damage. In turn, ONOO−can mediate the formation of 8-OHdG and 8-nitroguanine, a marker of nitrosative DNA damage (Yermilov et al.1995). 8-Nitroguanine formed in DNA is chemically unstable and, thus, can be spontaneously released, resulting in the formation of an apurinic site (Yermilov et al.1995). The apurinic site can form a pair with adenine during DNA synthesis, leading to G → T transversions (Loeb and Preston1986). It has been demonstrated that 8-nitroguanine is formed via NO production associated with inflammation inHelicobacter pyloriinfected patients (Ma et al.2004). 8-Nitroguanine is considered to be not only a marker of inflammation, but also a potential mutagenic DNA lesion product-capable of mediating apoptosis and carcinogenesis. Nitrosative stress arises mainly from the large accumulation of NO following the overexpression of iNOS in damaged tissue to form ONOO−when NO reacts with O2 −to form 8-nitroguanine.
The aim of this study was to determine the cytoprotective role of taurine against nitrosative stress in intense exercise-induced damage of the skeletal muscle.
2 Methods
2.1 Animals and Experimental Design
All experimental protocols were approved by the Animal Ethics Committee of Suzuka University of Medical Science, Japan. In total, 18 male rats (24-months-old; 250–260 g bodyweight) were housed in cages (max. 6 per cage) with water and food ad libitum. The animals were randomly divided into the following groups: exercise plus saline (EX;n = 6); exercise plus taurine (EX-Tau;n = 6); and control (n = 6). All the animals were maintained under a 12-h light and 12-h dark cycle at 24°C.
2.2 Taurine Supplementation and Exercise Protocol
We set up a heavy exercise protocol which consisted of an inclined treadmill and examined the effect of intraabdominally administered taurine 1 h before heavy exercise at a dose of 300 mg/kg/day for ten consecutive days. All animals were initially acclimated to running on a motor-driven treadmill designed for rats (model MK-680, Muromachi Kikai, Tokyo, Japan), beginning at 8 m/min for 10 min during the week preceding the exercise experiments. This protocol accustomed the rats to the locomotion intended for the final exercise experiments without stimulating development of skeletal muscle as an adaptation to training. Each group ran on the treadmill at 20 m/min, 25% grade, for 20 min or until exhaustion once in 10 days. Exhaustion was determined as the point when an animal was unable to right itself when placed on its side. The workload was ∼75% of the rats’ maximal aerobic capacity (VO2max) (Brooks and White1978). To return to basal physiological conditions and to prevent the influence of acute exercise, the animals were killed by decapitation 48 h after the exercise session. Rats were deeply anesthetized with an IP injection of sodium pentobarbital and were perfused transcardially with a fixative that contained 4% paraformaldehyde in 0.01 M phosphate buffer, pH 7.4. After the perfusion, the soleus was dissected out and allowed to stand in the same fixative for 4 h. Then the tissue was rinsed several times with phosphate buffer, dehydrated with a graded alcohol series and acetone, and embedded in paraffin. Sections 6-μm in thickness were mounted on albumin-coated slides.
2.3 Immunofluorescence Study
Anti-8-nitroguanine rabbit polyclonal antibody was prepared as described earlier (Pinlaor et al.2004). The immunoreactivity of 8-nitroguanine and of other biomarkers in skeletal muscle fiber was assessed by single or double immunofluorescence labeling studies, as previously described (Pinlaor et al.2004). Briefly, deparaffinized and dehydrated sections (6 μm thickn) were incubated with 5% skim milk followed by incubation with rabbit polyclonal anti-8-nitroguanine antibody (2 μg/mL) or other antibody mouse monoclonal anti-8-OHdG (5 μg/mL; Japan Institute for Control of Aging, Fukoroi, Japan), mouse monoclonal anti-Oct3/4 (2 μg/mL, Santa Cruz Biotechnology, Santa Cruz, CA), mouse monoclonal anti-iNOS (1:400, Sigma, St. Louis, MO), rabbit polyclonal anti-iNOS (1:500, CalbiochemNovabiochem Corporation, Darmstadt, Germany), or mouse monoclonal anti-NF-κB p65 (2 μg/mL, Santa Cruz Biotechnology) antibody overnight at room temperature. Then, the sections were incubated for 3 h with Alexa 594-labeled goat antibody against rabbit IgG or Alexa 488-labeled goat antibody against mouse IgG (1:400; Molecular Probes, Eugene, OR). The stained sections were examined using a confocal laser scanning microscope (FV-1000, Olympus, Tokyo, Japan) or by fluorescence microscopy (BX53, Olympus, Tokyo, Japan).
2.4 Statistical Analysis
Data are presented as the mean ± S.E.M. values. The two-tailed Student’st-test was performed. Differences were considered statistically significant atp < 0.05.
3 Results
3.1 Analyses for 8-Nitroguanine and 8-OHdG Immunoreactivities in the Soleus Muscle
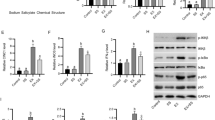
In each experimental group, the number of muscle cells positive for 8-nitroguanine and 8-OHdG were compared with the total number of muscle cells. The heavy exercise group had the highest percentage of 8-nitroguanine positive cells (35.1 ± 0.1%) followed by the heavy exercise plus taurine group (30.1 ± 0.1%) and control (23.1 ± 0.0%) groups. The highest 8-OHdG immunoreactivity was also observed with the heavy exercise group (34.8 ± 0.1%) followed by the heavy exercise plus taurine group (26.5 ± 0.1%) and the control (16.1 ± 0.0%) groups (Fig.20.1).
The mean diameter of muscle fibers positively and negatively immunoreactive for 8-nitroguanine was 26.2 ± 0.8 μm and 39.6 ± 1.3 μm in controls, 54.9 ± 4.8 μm and 76.9 ± 8.3 μm in the heavy exercise group, and 28.3 ± 1.1 μm and 39.7 ± 2.1 μm in the heavy exercise plus taurine group, respectively, showing that 8-nitroguanine-positive fibers were significantly smaller than negative ones. The mean diameter of muscle fibers showing positive and negative immunoreactivities for 8-OHdG was 27.3 ± 0.4 μm and 39.3 ± 2.5 μm in controls, 51.9 ± 2.8 μm and 76.5 ± 5.5 μm in the heavy exercise group, and 29.1 ± 1.5 μm and 38.9 ± 1.0 μm in the heavy exercise plus taurine group, respectively. These results demonstrate that the immunoreactive fibers were smaller than the non-immunoreactive ones (Fig.20.2).
3.2 -Nitroguanine and 8-OHdG Formation in the Soleus Muscle
The formation of 8-nitroguanine and 8-OHdG in fibers of soleus muscle is shown in Fig.20.3. Strong immunoreactivities for 8-nitroguanine (red) and 8-OHdG (green) were clearly observed in the nuclei of muscle fibers of all rats after heavy exercise; but they were decreased in the taurine-treated group, and became negative in the control (normal) group. While 8-nitroguanine and 8-OHdG were colocalized in almost all fibers, 8-nitroguanine was predominated than over 8-OHdG in the heavy exercise group (Fig.20.3).
3.3 -Nitroguanine and NF-κB Formation in the Soleus Muscle
8-Nitroguanine formation and NF-κB expression in fibers of soleus muscle are shown in Fig.20.4. 8-Nitroguanine immunoreactivity (red) was observed in the nuclei of the fibers whereas NF-κB expression (green) was strongly observed in both the cytoplasm and nucleus. The immunoreactivities of 8-nitroguanine and NF-κB were decreased significantly in the taurine-treated group. Little to no 8-nitroguanine and NF-κB expression were observed in control muscle fibers (Fig.20.4).
8-Nitroguanine formation and NF-κB immunoreactivities in fibers of the soleus muscle. In heavily exercised rats, double immunofluorescence staining of paraffin sections shows the localization of NF-κB and 8-nitroguanine and the immunoreactivities of 8-nitroguanine and NF-κB colocalized primarily in the nuclei of the muscle fibers
3.4 iNOS and NF-κB Formation in the Soleus Muscle
Strong immunoreactivities for iNOS and NF-κB were observed in soleus muscle fibers of all rats after heavy exercise. Intense staining of muscle cells was noted in both the nucleus and cytoplasm of the heavily exercised group. However, the taurine-treated group showed a marked reduction in the number of taurine-containing cells with stained nucleus and cytoplasm (Fig.20.5). The immunoreactivity for iNOS and NF-κB decreased significantly in the muscle cells of rats receiving taurine, and was either drastically decreased or absent in the muscle fibers of control rats.
iNOS and NF-κB immunoreacitivies in the soleus muscle. In heavily exercised rats, double immunofluorescence staining of paraffin sections shows the localization of iNOS and NF-κB and the immunoreactivity of 8-nitroguanine and NF-κB colocalizes primarily in the fibers of the extensor digitorum longus muscle
4 Discussion
In muscle cells, the levels of reactive oxygen species (ROS), constantly generated by cellular metabolic processes, are regulated by intracellular oxidative defense systems driven by antioxidant enzymes such as catalase, superoxide dismutase (SOD) (Silva et al.2011), and glutathione peroxidase (Sen et al.1997). Heavy exercise activates cellular metabolism and, thus, gives rise to a large number of ROS in the form of free radicals. An imbalance between the levels of ROS and antioxidant defenses results in oxidative and nitrosative stresses in the muscle fibers. Acute heavy exercise has been reported to activate NF-κB in skeletal muscle cells (Ji et al.2004). A study of the activation mechanism of NF-κB revealed that acute exercise promotes cellular oxidative stress and activation of NF-κB through oxidation of glutathione in muscle cells (Sen et al.1997).
In this study, oxidative and nitrosative stresses were induced in the skeletal muscle through heavy-load exercise (75% of VO2max) performed for 20 min or until exhaustion. Exercise activated inflammatory mediators such as iNOS and NF-κB in skeletal muscle cells and significantly increased the expression of the oxidative/nitrosative stress indicators 8-OHdG and 8-nitroguanine. However, administration of taurine prevented heavy exercise from activating NF-κB and reduced oxidative and nitrosative stresses.
The antioxidant action of taurine is related to its ability to scavenge free radicals generated by various enzymatic processes by directly causing reduction reactions (Aruoma et al.1988; Shi et al.1997). Taurine is also found to reduce protein carbonylation by effectively scavenging hypochlorous acid (HOCI) generated by myeloperoxidase (Prutz1996). Taurine functions like the NF-κB blocker sulphasalazine (Gurujeyalashmi et al.2000), and taurine chloramine (TauCl), a product of the condensation of taurine with HOCI, is reported to inhibit the activation of NF-κB (Barua et al.2001). In turn, TauCl can interact with hydrogen peroxide (H2O2), which is produced as an inflammatory response during exercise, to buffer the effects of H2O2before the appearance of oxidative stress. Consistent with the results of previous studies, those generated here suggest that taurine is cytoprotective by reducing both oxidative and nitrosative stresses.
ATP production and oxygen supply to the mitochondria need to be increased to activate skeletal muscles during exercise. An increase in oxygen consumption is also likely to increase the generation of superoxide anion radicals (O2 −). Taurine appears to reduce the production of O2 −via redox reaction, particularly in intracellular places prone to produce high levels of O2 −particularly the mitochondria (Hansen et al.2006). In this study, immunofluorescence analysis demonstrated that the diameter of 8-nitroguanine in immunoreactive muscle fibers was significantly smaller than that of nonimmunoreactive muscle fibers, suggesting that the immunoreactive muscle fibers were type I. Type I muscle fibers contain a large number of mitochondria and their aerobic metabolism consumes a large number of oxygen molecules to produce ATP during exercise. Therefore, it is likely that type I muscle fibers generate a high level of free radicals from oxygen and are vulnerable to the noxious effects of oxidative and nitrosative stress. Compared with type II muscle fibers, type I muscle fibers contain a higher level of polyunsaturated fatty acid that make them more susceptible to lipid peroxidation (Nikolaidis and Mougios2004; Nikolaidis et al.2006) as well as to other types of oxidative damage. Because type I muscle fibers also contain a higher number of mitochondria than type II muscle fibers, they may produce a higher level of free radical production during exercise and even at rest (Moyes2003). In fact, heavy exercise-induced muscle inflammation is accompanied by an increase formation of 8-nitrogauanine in the muscle fibers. In this study, taurine produced a decrease in NF-κB as well as in iNOS immunoreactivity in the muscle fibers of rats. This finding indicates that taurine is inhibiting proinflammatory factors by suppressing iNOS activity through an inhibitory effect on a signaling pathway activating NF-κB.
5 Conclusion
In conclusion, this study demonstrates for the first time that taurine offers a strong protective effect against intense exercise-induced nitrosative DNA damage in the skeletal muscle of rats. Since this sulfur-containing compound prevented iNOS expression, it may act as a modulator of nitrosative stress in the muscle during periods of heavy exercise. Hence, upregulated expression of iNOS in skeletal muscles could be responsible for the nitrosative muscle damage taking place during heavy exercise, probably through inflammatory damage mediated by a NF-κB-activating signaling pathway.
Abbreviations
- ROS:
-
Reactive oxygen species
- O2 − :
-
Superoxide anion
- NF-κB:
-
Nuclear factor kappa-B
- iNOS:
-
Inducible NO synthase
- 8-OHdG:
-
8-Hydroxydeoxyguanosine
References
Aruoma OI, Halliwell B, Hoey BM, Butler J (1988) The antioxidant action of taurine, hypotaurine and their metabolic precursors. Biochem J 256:251–255
Barua M, Liu Y, Quinn MR (2001) Taurine chloramine inhibits inducible nitric oxide synthase and TNF-alpha gene expression in activated alveolar macrophages: decreased NF-κB activation and IκB kinase activity. J Immunol 167:2275–2281
Brooks GA, White TP (1978) Determination of metabolic and heart rate responses of rats to treadmill exercise. J Appl Physiol 45:1009–1015
Gurujeyalashmi G, Wang Y, Giri SN (2000) Taurine and niacin block lung and fibrosis by down-regulating bleomycin-induced activation of transcription nuclear factor-κB in mice. J Pharmacol Exp Ther 293:82–90
Hansen SH, Andersen ML, Birkedal H, Cornett C, Wibrand F (2006) The important role of taurine in oxidative metabolism. Adv Exp Med Biol 583:129–135
Ji LL, Gomez-Cabrera MC, Steinhafel N, Vina J (2004) Acute exercise activates nuclear factor (NF)-κB signaling pathway in rat skeletal muscle. FASEB J 18:1499–1506
Loeb LA, Preston BD (1986) Mutagenesis by a apurinic/apyrimidinic sites. Annu Rev Genet 20:201–230
Ma N, Adachi Y, Hiraku Y, Horiki N, Horike S, Imoto I, Pinlaor S, Murata M, Semba R, Kawanishi S (2004) Accumulation of 8-nitroguanine in human gastric epithelium induced byHelicobacter pyloriinfection. Biochem Biophys Res Commun 319:506–510
Moyes CD (2003) Controlling muscle mitochondrial content. J Exp Biol 206:4385–4391
Nikolaidis MG, Mougios V (2004) Effects of exercise on the fatty-acid composition of blood and tissue lipids. Sports Med 34:1051–1076
Nikolaidis MG, Petridou A, Mougios V (2006) Comparison of the phospholipid and triacylglycerol fatty acid profile of rat serum, skeletal muscle and heart. Physiol Res 55:259–265
Pinlaor S, Hiraku Y, Ma N, Yongvanit P, Semba R, Oikawa S, Murata M, Sripa B, Sithithaworn P, Kawanishi S (2004) Mechanism of NO-mediated oxidative and nitrative DNA damage in hamsters infected with Opisthorchis viverrini: a model of inflammation-mediated carcinogenesis. Nitric Oxide 11:175–183
Prutz WA (1996) Hypochlorous acid interactions with thiols, nucleotides, DNA, and other biological substrates. Arch Biochem Biophys 332:110–120
Sen CK, Khanna S, Resznick AZ, Roy S, Packer L (1997) Glutathione regulation of tumor necrosis factor-α-induced NF-κB activation in skeletal muscle-derived L6 cells. Biochem Biophys Res Commun 237:645–649
Shi X, Flynn DC, Porter DW, Leonard SS, Vallyathan V, Castranova V (1997) Efficacy of taurine based compounds as hydroxyl radical scavengers in silica induced peroxidation. Ann Clin Lab Sci 27:365–374
Silva LA, Silveira PC, Ronsani MM, Souza PS, Scheffer D, Vieira LC, Benetti M, De Souza CT, Pinho RA (2011) Taurine supplementation decreases oxidative stress in skeletal muscle after eccentric exercise. Cell Biochem Funct 29:43–49
Yermilov V, Rubio J, Becchi M, Friesen MD, Pignatelli B, Ohshima H (1995) Formation of 8-nitroguanine by the reaction of guanine with peroxynitritein vitro. Carcinogenesis 16: 2045–2050
Acknowledgments
This work was supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this paper
Cite this paper
Sugiura, H. et al. (2013). Protection by Taurine Against INOS-Dependent DNA Damage in Heavily Exercised Skeletal Muscle by Inhibition of the NF-κB Signaling Pathway. In: El Idrissi, A., L'Amoreaux, W. (eds) Taurine 8. Advances in Experimental Medicine and Biology, vol 775. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6130-2_20
Download citation
DOI: https://doi.org/10.1007/978-1-4614-6130-2_20
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-6129-6
Online ISBN: 978-1-4614-6130-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)