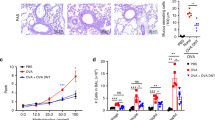
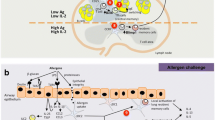
Abstract
Allergic diseases such as asthma, rhinitis and eczema are increasing in prevalence worldwide, in particular in industrialised countries affecting up to 20% of the population. Regulatory T-cells (Tregs) have been shown to be critical in T-cell homeostasis and in the maintenance of immune responses, such as prevention of autoimmunity and hampering allergic diseases. The so-called ‘natural’ CD4+CD25+ Tregs and/or IL-10-producing Tr1 cells have been shown to be responsible for the protection of immune tolerance and intact immune reactions following exposure to allergens such as aeroallergens or food allergens. In this regard, both cell-cell contact (through membrane bound TGF-β or via suppressive molecules such as CLTA-4) and soluble cytokine-(TGF-β and IL-10) dependent mechanisms have been shown to contribute to the ability of Tregs to operate effectively. The transcription factor Foxp3, a member of the forkhead-winged helix family, appears to be critical in the suppressive abilities of regulatory T-cells. Adoptive transfer of CD4+CD25+ Tregs from healthy to diseased animals corroborated and provided further evidence of the vital role of these populations in the prevention or cure of certain autoimmune conditions. Clinical improvement seen after allergen immunotherapy for allergic diseases such as rhinitis and asthma has also been associated with the induction of IL-10 and TGF-β producing Tr1 cells as well as Foxp3 expressing CD4+CD25+ T-cells, resulting in the suppression of Th2 cytokine milieu. Activation and expansion of antigen-specific CD4+CD25+ Tregs in vivo using adjuvants such as IL-10 or pharmacological agents such as low dose steroids or vitamin D3 could represent novel approaches to induce antigen-specific tolerance in immune-mediated conditions such as allergic asthma, autoimmune disease and the rejection of transplanted organs in man.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Sakaguchi S, Sakaguchi N, Asano M et al. Immunologic self-tolerance maintained by activated T-cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 1995; 155:1151–1164.
Asano M, Toda M, Sakaguchi N et al. Autoimmune disease as a consequence of developmental abnormality of a T-cell subpopulation. J Exp Med 1996; 184:387–396.
Ng WF, Duggan PJ, Ponchel F et al. Human CD4+CD25+ cells: a naturally occurring population of regulatory T-cells. Blood 2001; 98:2736–2744.
Jonuleit H, Schmitt E, Stassen M et al. Identification and functional characterization of human CD4+CD25+ T-cells with regulatory properties isolated from peripheral blood. J Exp Med 2001; 193:1285–1294.
Dieckmann D, Plottner H, Berchtold S et al. Ex vivo isolation and characterization of CD4+CD25+ T-cells with regulatory properties from human blood. J Exp Med 2001; 193:1303–1310.
Levings MK, Sangregorio R, Roncarolo MG. Human CD25+CD4+ T regulatory cells suppress naive and memory T-cell proliferation and can be expanded in vitro without loss of function. J Exp Med 2001; 193:1295–1302.
Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T-cells suppress polyclonal T-cell activation in vitro by inhibiting interleukin 2 production. J Exp Med 1998; 188:287–296.
Bayer AL, Yu A, Adeegbe D et al. Essential role for interleukin-2 for CD4+CD25+ T regulatory cell development during the neonatal period. J Exp Med 2005; 201:769–777.
Read S, Malmstrom V, Powrie F. Cytotoxic T-lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J Exp Med 2000; 19:295–302.
Graca L, Honey K, Adams E et al. Cutting edge: anti-CD 154 therapeutic antibodies induce infectious transplantation tolerance. J Immunol 2000; 165:4783–4786.
McHugh RS, Whitters MJ, Piccirillo CA et al. CD4+CD25+ immunoregulatory T-cells: gene expression analysis reveals a functional role for the glucocorticoid-induced TNF receptor. Immunity 2002; 1:311–323.
Liu W, Putnam AL, Xu-Yu Z et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ Treg cells. J Exp Med 2006; 203:1701–1711.
Nouri-Aria KT, Durham SR. Regulatory T-cells and allergic disease. Inflamm Allergy Drug Targets 2008; 7:237–52. Review.
Hori S, Nomura T, Sakaguchi S. Control of regulatory T-cell development by the transcription factor Foxp3. Science 2003; 299:1057–1061.
Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T-cells. Nat Immunol 2003; 4:330–336.
Brandenburg S, Takahashi T, de la Rosa M et al. IL-2 induces in vivo suppression by CD4+CD25+Foxp3+ regulatory T-cells. Eur J Immunol 2008; 38:1643–1653.
Lin W, Haribhai D, Relland LM et al. Regulatory T-cell development in the absence of functional Foxp3. Nat Immunol 2007; 8:359–368.
Khattri R, Cox T, Yasayko SA et al. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol 2003; 4:337–342.
Bennett CL, Christie J, Ramsdell F et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet 2001; 27:20–21.
Yagi H, Nomura T, Nakamura K et al. Crucial role of FOXP3 in the development and function of human CD25+CD4+ regulatory T. Int Immunol 2004; 16:1643–1656.
Ziegler SF. FOXP3: of mice and men. Annu Rev Immunol 2006; 24:209–226. Review.
Schmidt-Weber CB, Blaser K. The role of the FOXP3 transcription factor in the immune regulation of allergic asthma. Curr Allergy Asthma Rep 2005; 5:356–361.
Mantel PY, Ouaked N, Ruckert B et al. Molecular mechanisms underlying FOXP3 induction in human T-cells. J Immunol 2006; 176:3593–3602.
Zorn E, Nelson EA, Mohseni M et al. IL-2 regulates FOXP3 expression in human CD4+CD25+ regulatory T-cells through a STAT dependent mechanism and induces the expansion of these cells in vivo. Blood 2006; 108:1571–1579.
Fontenot JD, Rasmussen JP, Gavin MA et al. A function for interleukin 2 in Foxp3-expressing regulatory T-cells. Nat Immunol 2005; 6:1142–1151.
Janson PC, Winerdal ME, Marits P et al. FOXP3 promoter demethylation reveals the committed Treg population in humans. PLoS ONE 2008; 3:e1612.
Allan SE, Song-Zhao GX, Abraham T et al. Inducible reprogramming of human T-cells into Treg cells by a conditionally active form of FOXP3. Eur J Immunol 2008; 38:3282–3289.
Bardel E, Larousserie F, Charlot-Rabiega P. Human CD4+CD25+Foxp3+ regulatory T-cells do not constitutively express IL-35. J Immunol 2008; 181:6898–6905.
Collison LW, Workman CJ, Kuo TT et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature 2007; 450:566–569.
Cottrez F, Hurst SD, Coffman RL et al. T regulatory cells 1 inhibit a Th2-specific response in vivo. J Immunol 2000; 165:4848–4853.
Akdis M, Verhagen J, Taylor A et al. Immune responses in healthy and allergic individuals are characterized by a fine balance between allergen-specific T regulatory 1 and T helper 2 cells. J Exp Med 2004; 199:1567–1575.
Ling EM, Smith T, Dao Nguyen X et al. Relation of CD4+CD25+ regulatory T-cell suppression of allergen-driven T-cell activation to atopic status and expression of allergic disease. Lancet 2004; 363:608–615.
Karlsson MR, Rugtveit J, Brandtzaeg P. Allergen-responsive CD4+CD25+ regulatory T-cells in children who have outgrown cow’s milk allergy. J Exp Med 2004; 199:1679–1688.
Akdis M, Blaser K, Akdis CA. T regulatory cells in allergy. Chem Immunol Allergy 2006; 91:159–173. Review.
Sumi T, Fukushima A, Fukuda K et al. Thymus-derived CD4+ CD25+ T-cells suppress the development of murine allergic conjunctivitis. Int Arch Allergy Immunol 2007; 143:276–81.
Lin W, Truong N, Grossman WJ et al. Allergic dysregulation and hyperimmunoglobulinemia E in Foxp3 mutant mice. J Allergy Clin Immunol 2005; 116:1106–15.
Van Hove CL, Maes T, Joos GF et al. Prolonged inhaled allergen exposure can induce persistent tolerance. Am J Respir Cell Mol Biol 2007; 36:573–84.
Borish L. IL-10: evolving concepts. J Allergy Cli Immunol 1998; 101:293–297.
Scherf W, Burdach S, Hansen G. Reduced expression of transforming growth factor beta 1 exacerbates pathology in an experimental asthma model. Eur J Immunol 2005; 35:198–206.
Frossard CP, Hauser C, Eigenmann PA. Antigen-specificsecretory IgA antibodies in the gut are decreased in a mouse model of food allergy. J Allergy Clin Immunol 2004; 114:377–382.
Burchell JT, Wikstrom ME, Stumbles PA et al. Attenuation of allergen-induced Airway Hyperresponsiveness is mediated by airway regulatory T-cells. Am J Physiol Lung Cell Mol Physiol 2008. (Epub ahead of print)
Carson WF 4th, Guernsey LA, Singh A et al. Accumulation of regulatory T-cells in local draining lymph nodes of the lung correlates with spontaneous resolution of chronic asthma in a murine model. Int Arch Allergy Immunol 2008; 145:231–43.
Ostroukhova M, Seguin-Devaux C, Oriss TB et al. Tolerance induced by inhaled antigen involves CD4+ T-cells expressing membrane-bound TGF-beta and FOXP3. J Clin Invest 2004; 114:28–38.
Stock P, DeKruyff RH, Umetsu DT. Inhibition of the allergic response by regulatory T-cells. Curr Opin Allergy Clin Immunol 2006; 6:12–6. Review.
Francis JN, Till SJ, Durham SR. Induction of IL-10+CD4+CD25+ T-cells by grass pollen immunotherapy. J Allergy Clin Immunol 2003; 111:1255–1261.
Grindebacke H, Wing K, Andersson AC et al. Defective suppression of Th2 cytokines by CD4CD25 regulatory T-cells in birch allergies during birch pollen season. Clin Exp Allergy 2004; 3:1364–1372.
Xu G, Mou Z, Jiang H et al. A possible role of CD4+CD25+ T-cells as well as transcription factor Foxp3 in the dysregulation of allergic rhinitis. Laryngoscope 2007; 117:876–80.
Lee JH, Yu HH, Wang LC et al. The levels of CD4+CD25+ regulatory T-cells in paediatric patients with allergic rhinitis and bronchial asthma. Clin Exp Immunol 2007; 148:53–63.
Thunberg S, Akdis M, Akdis CA et al. Immune regulation by CD4+CD25+ T-cells and interleukin-10 in birch pollen-allergic patients and non-allergic controls. Clin Exp Allergy 2007; 37:1127–1136.
Maggi L, Santarlasci V, Liotta F et al. Demonstration of circulating allergen-specific CD4+CD25highFoxp3+ T-regulatory cells in both nonatopic and atopic individuals. J Allergy Clin Immunol 2007; 120:429–436.
Nagato T, Kobayashi H, Yanai M et al. Functional analysis of birch pollen allergen Bet v 1-specific regulatory T-cells. J Immunol 2007; 178:1189–1198.
Winkler B, Hufnagl K, Spittler A et al. The role of Foxp3+ T-cells in long-term efficacy of prophylactic and therapeutic mucosal tolerance induction in mice. Allergy 2006; 61:173–80.
Wilson MS, Taylor MD, Balic A et al. Suppression of allergic airway inflammation by helminth-induced regulatory T-cells. J Exp Med 2005; 202:1199–212.
Flohr C, Quinnell RJ, Britton J. Do helminth parasites protect against atopy and allergic disease? Clin Exp Allergy 2009; 39:20–32.
Schaub B, Liu J, Höppler S et al. Impairment of T-regulatory cells in cord blood of atopic mothers. J Allergy Clin Immunol 2008; 121:1491–9.
Smith M, Tourigny MR, Noakes P et al. Children with egg allergy have evidence of reduced neonatal CD4+CD25+CD127lo/− regulatory T-cell function. J Allergy Clin Immunol 2008; 121:1460–6.
Schaub B, Campo M, He H et al. Neonatal immune responses to TLR2 stimulation: influence of maternal atopy on Foxp3 and IL-10 expression. Respir Res 2006; 21:7:40–48.
Reefer AJ, Satinover SM, Solga MD et al. Analysis of CD25hiCD4+ “regulatory” T-cell subtypes in atopic dermatitis reveals a novel TH2-like population. J Allergy Clin Immunol 2008; 121:415–422.
Caproni M, Antiga E, Torchia D et al. FoxP3-expressing T regulatory cells in atopic dermatitis lesions. Allergy Asthma Proc 2007; 28:525–528.
de Boer OJ, van der Loos CM, Teeling P et al. Immunohistochemical analysis of regulatory T-cell markers FOXP3 and GITR on CD4+CD25+ T-cells in normal skin and inflammatory dermatoses. J Histochem Cytochem 2007; 55:891–898.
O’Garra A, Barrat FJ, Castro AG et al. Strategies for use of IL-10 or its antagonists in human disease. Immunol Rev 2008; 223:114–131.
Xystrakis E, Kusumakar S, Boswell S et al. Reversing the defective induction of IL-10-secreting regulatory T-cells in glucocorticoid-resistant asthma patients. J Clin Invest 2006; 116:146–155.
Hartl D, Koller B, Mehlhorn AT et al. Quantitative and functional impairment of pulmonary CD4+CD25hi regulatory T-cells in pediatric asthma. J Allergy Clin Immunol 2007; 119:1258–66.
Durham SR. Allergen immunotherapy (desensitisation) for allergic diseases. Clin Med 2006; 6:348–351.
Nelson HS. Advances in upper airway diseases and allergen immunotherapy. J Allergy Clin Immunol 2007; 119:872–880.
Noon L. Prophylactic inoculation against hay fever. Lancet 1911; 1:1572–1573.
Frew AJ, Powell RJ, Corrigan CJ et al. Efficacy and safety of specific immunotherapy with SQ allergen extract in treatment-resistant seasonal allergic rhinoconjunctivitis. J Allergy Clin Immunol 2006; 117:319–325.
Walker SM, Pajno GB, Lima MT et al. Grass pollen immunotherapy for seasonal rhinitis and asthma: a randomized, controlled trial. J Allergy Clin Immunol 2001; 107:87–93.
Akdis CA, Blesken T, Akdis M et al. Role of interleukin 10 in specific immunotherapy. J Clin Invest 1998; 102:98–106.
Durham SR, Walker SM, Varga EM et al. Long-Term Clinical Efficacy of Grass-Pollen Immunotherapy. New Engl J Med 1999; 341:468–475.
Wachholz PA, Nouri-Aria KT, Wilson DR et al. Grass pollen immunotherapy for hayfever is associated with increases in local nasal but not peripheral Th1:Th2 cytokine ratios. Immunology 2002; 105:56–62.
Till SJ, Francis JN, Nouri-Aria K et al. Mechanisms of immunotherapy. J Allergy Clin Immunol 2004; 113:1025–1034. Review.
Robinson DS, Larche M, Durham SR. Tregs and allergic disease. J Clin Invest 2004; 114:1389–1397. Review.
Verhagen J, Taylor A, Blaser K et al. T regulatory cells in allergen-specific immunotherapy. Int Rev Immunol 2005; 24:533–548. Review.
Ajduk J, Marinic I, Aberle N et al. Effect of house dust mite immunotherapy on transforming growth factor betal-producing T-cells in asthmatic children. Ann Allergy Asthma Immunol 2008; 100:314–22.
Mekala DJ, Alli RS, Geiger TL. IL-10-dependent infectious tolerance after the treatment of experimental allergic encephalomyelitis with redirected CD4+CD25+ T-Iymphocytes. Proc Natl Acad Sci USA 2005; 102:11817–11822.
Akdis CA, Joss A, Akdis M et al. A molecular basis for T-cell suppression by IL-10: CD28-associated IL-10 receptor inhibits CD28 tyrosine phosphorylation and phosphatidylinositol 3-kinase binding. FASEB J 2000; 14:1666–1668.
Nouri-Aria KT, Wachholz PA, Francis JN et al. Grass pollen immunotherapy induces mucosal and peripheral IL-10 responses and blocking IgG activity. J Immunol 2004; 172:3252–3259.
Pilette C, Nouri-Aria KT, Jacobson MR et al. Grass pollen immunotherapy induces an allergen-specific IgA2 antibody response associated with mucosal TGF-beta expression. J Immunol 2007; 178:4658–4666.
Jutel M, Akdis M, Budak F et al. IL-10 and TGF-beta cooperate in the regulatory T-cell response to mucosal allergens in normal immunity and specific immunotherapy. Eur J Immunol 2003; 33:1205–1214.
Radulovic S, Jacobson MR, Durham SR et al. Grass pollen immunotherapy induces Foxp3-expressing CD4+ CD25+ cells in the nasal mucosa. J Allergy Clin Immunol 2008; 121:1267–1272.
Mottet C, Uhlig HH, Powrie F. Cutting edge: cure of colitis by CD4+CD25+ regulatory T-cells. J Immunol 2003; 170:3939–3943.
Pereira-Santos MC, Baptista AP, Melo A et al. Expansion of circulating Foxp3+CD25bright CD4+ T-cells during specific venom immunotherapy. Clin Exp Allergy 2008; 38:291–297.
Akdis CA, Akdis M, Blesken T et al. Epitope-specific T-cell tolerance to phospholipase A2 in bee venom immunotherapy and recovery by IL-2 and IL-15 in vitro. J Clin Invest 1996; 98:1676–1683.
Taylor A, Verhagen J, Blaser K et al. Mechanisms of immune suppression by interleukin-10 and transforming growth factor-beta: the role of T regulatory cells. Immunology 2006; 117:433–442.
Croxford JL, Feldmann M, Chernajovsky Y et al. Different therapeutic outcomes in experimental allergic encephalomyelitis dependent upon the mode of delivery of IL-10: a comparison of the effects of protein, adenoviral or retroviral IL-10 delivery into the central nervous system. J Immunol 2001; 166:4124–4130.
Hansen G, Yeung VP, Berry G et al. Vaccination with heat-killed Listeria as adjuvant reverses established allergen-induced airway hyperreactivity and inflammation: role of CD8+ T-cells and IL-18. J Immunol 2000; 164:223–230.
Creticos PS, Schroeder JT, Hamilton RG. Immune Tolerance Network Group. Immunotherapy with a ragweed-toll-like receptor 9 agonist vaccine for allergic rhinitis. N Engl J Med 2006; 355:1445–1455.
Van Overtvelt L, Lombardi V, Razafindratsita A et al. IL-10-inducing adjuvants enhance sublingual immunotherapy efficacy in a murine asthma model. Int Arch Allergy Immunol 2008; 145:152–62.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Landes Bioscience and Springer+Business Media
About this chapter
Cite this chapter
Nouri-Aria, K.T. (2009). Foxp3 Expressing Regulatory T-Cells in Allergic Disease. In: Maiese, K. (eds) Forkhead Transcription Factors. Advances in Experimental Medicine and Biology, vol 665. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-1599-3_14
Download citation
DOI: https://doi.org/10.1007/978-1-4419-1599-3_14
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-1598-6
Online ISBN: 978-1-4419-1599-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)