Abstract
Among carrier-free immobilization techniques, cross-linked enzyme aggregates (CLEA) have been extensively described for a great number of diverse enzymes. During the last two decades, numerous efforts have been devoted to identify and understand the main variables involved in CLEA’s preparation process leading to robust immobilized biocatalysts. Since every enzyme immobilized as CLEA requires specific conditions and protocols, herein we provide a general preparation strategy where main parameters are highlighted and correlated with a possible desired improved enzyme feature.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dewan SS (2017) Global markets for enzymes in industrial applications. A BBC research report, Wesley, MD
Robinson PK (2015) Enzymes: principles and biotechnological applications. Essays Biochem 59:1–41
Stepankova V, Bidmanova S, Koudelakova T, Prokop Z, Chaloupkova R, Damborsky J (2013) Strategies for stabilization of enzymes in organic solvents. ACS Catal 3(12):2823–2836
Bommarius AS, Paye MF (2013) Stabilizing biocatalysts. Chem Soc Rev 42(15):6534–6565
Iyer PV, Ananthanarayan L (2008) Enzyme stability and stabilization—aqueous and non-aqueous environment. Process Biochem 43(10):1019–1032
Cao L, Van Rantwijk F, Sheldon RA (2000) Cross-linked enzyme aggregates: a simple and effective method for the immobilization of penicillin acylase. Org Lett 2(10):1361–1364
Talekar S, Joshi A, Joshi G, Kamat P, Haripurkar R, Kambale S (2013) Parameters in preparation and characterization of cross linked enzyme aggregates (CLEAs). RSC Adv 3(31):12485–12511
Cui JD, Jia SR (2015) Optimization protocols and improved strategies of cross-linked enzyme aggregates technology: current development and future challenges. Crit Rev Biotechnol 35(1):15–28
Sheldon RA (2011) Characteristic features and biotechnological applications of cross-linked enzyme aggregates (CLEAs). Appl Microbiol Biotechnol 92(3):467–477
Velasco-Lozano S, López-Gallego F, Mateos Diaz JC, Favela-Torres E (2015) Cross-linked enzyme aggregates (CLEA) in enzyme improvement—a review. Biocatalysis 1:166–177
Cruz J, Barbosa O, Rodrigues RC, Fernandez-Lafuente R, Torres R, Ortiz C (2012) Optimized preparation of CALB-CLEAs by response surface methodology: the necessity to employ a feeder to have an effective crosslinking. J Mol Catal B Enzym 80:7–14
Zerva A, Antonopoulou I, Enman J, Iancu L, Jütten P, Rova U, Christakopoulos P (2018) Optimization of transesterification reactions with clea-immobilized feruloyl esterases from Thermothelomyces thermophila and Talaromyces wortmannii. Molecules 23(9):2403
Rajendhran J, Gunasekaran P (2007) Application of cross-linked enzyme aggregates of Bacillus badius penicillin G acylase for the production of 6-aminopenicillanic acid. Lett Appl Microbiol 44(1):43–49
Ba S, Haroune L, Cruz-Morató C, Jacquet C, Touahar IE, Bellenger JP, Legault CY, Jones JP, Cabana H (2014) Synthesis and characterization of combined cross-linked laccase and tyrosinase aggregates transforming acetaminophen as a model phenolic compound in wastewaters. Sci Total Environ 487(1):748–755
Talekar S, Desai S, Pillai M, Nagavekar N, Ambarkar S, Surnis S, Ladole M, Nadar S, Mulla M (2013) Carrier free co-immobilization of glucoamylase and pullulanase as combi-cross linked enzyme aggregates (combi-CLEAs). RSC Adv 3(7):2265–2271. https://doi.org/10.1039/c2ra22657j
Dalal S, Kapoor M, Gupta MN (2007) Preparation and characterization of combi-CLEAs catalyzing multiple non-cascade reactions. J Mol Catal B Enzym 44(3):128–132
Nguyen LT, Yang KL (2017) Combined cross-linked enzyme aggregates of horseradish peroxidase and glucose oxidase for catalyzing cascade chemical reactions. Enzym Microb Technol 100:52–59
Schoevaart R, Wolbers MW, Golubovic M, Ottens M, Kieboom APG, Van Rantwijk F, Van Der Wielen LAM, Sheldon RA (2004) Preparation, optimization, and structures, of cross-linked enzyme aggregates (CLEAs). Biotechnol Bioeng 87(6):754–762
Talekar S, Nadar S, Joshi A, Joshi G (2014) Pectin cross-linked enzyme aggregates (pectin-CLEAs) of glucoamylase. RSC Adv 4(103):59444–59453
Zerva A, Antonopoulou I, Enman J, Iancu L, Rova U, Christakopoulos P (2018) Cross-linked enzyme aggregates of feruloyl esterase preparations from Thermothelomyces thermophila and Talaromyces wortmannii. Catalysts 8(5):208
Velasco-Lozano S, López-Gallego F, Vázquez-Duhalt R, Mateos-Díaz JC, Guisán JM, Favela-Torres E (2014) Carrier-free immobilization of lipase from candida rugosa with polyethyleneimines by carboxyl-activated cross-linking. Biomacromolecules 15(5):1896–1903
Wilson L, Illanes A, Soler L, Henríquez MJ (2009) Effect of the degree of cross-linking on the properties of different CLEAs of penicillin acylase. Process Biochem 44(3):322–326
Majumder AB, Mondal K, Singh TP, Gupta MN (2008) Designing cross-linked lipase aggregates for optimum performance as biocatalysts. Biocatal Biotransformation 26(3):235–242
Kim MH, Park S, Kim YH, Won K, Lee SH (2013) Immobilization of formate dehydrogenase from Candida boidinii through cross-linked enzyme aggregates. J Mol Catal B Enzym 97:209–214
Velasco-Lozano S, López-Gallego F, Rocha-Martin J, Guisán JM, Favela-Torres E (2016) Improving enantioselectivity of lipase from Candida rugosa by carrier-bound and carrier-free immobilization. J Mol Catal B Enzym 130:32–39
Wang A, Zhang F, Chen F, Wang M, Li H, Zeng Z, Xie T, Chen Z (2011) A facile technique to prepare cross-linked enzyme aggregates using p-benzoquinone as cross-linking agent. Korean J Chem Eng 28(4):1090–1095
Valdés EC, Soto LW, Arcaya GA (2011) Influence of the pH of glutaraldehyde and the use of dextran aldehyde on the preparation of cross-linked enzyme aggregates (CLEAs) of lipase from Burkholderia cepacia. Electron J Biotechnol 14(3). https://doi.org/10.2225/vol14-issue3-fulltext-1
Yu HW, Chen H, Wang X, Yang YY, Ching CB (2006) Cross-linked enzyme aggregates (CLEAs) with controlled particles: application to Candida rugosa lipase. J Mol Catal B Enzym 43(1–4):124–127
Kumar S, Mohan U, Kamble AL, Pawar S, Banerjee UC (2010) Cross-linked enzyme aggregates of recombinant Pseudomonas putida nitrilase for enantioselective nitrile hydrolysis. Bioresour Technol 101(17):6856–6858
Dinh TH, Jang NY, McDonald KA, Won K (2015) Cross-linked aggregation of glutamate decarboxylase to extend its activity range toward alkaline pH. J Chem Technol Biotechnol 90:2100–2105
Mateo C, Palomo JM, Van Langen LM, Van Rantwijk F, Sheldon RA (2004) A new, mild cross-linking methodology to prepare cross-linked enzyme aggregates. Biotechnol Bioeng 86(3):273–276
Zhen Q, Wang M, Qi W, Su R, He Z (2013) Preparation of β-mannanase CLEAs using macromolecular cross-linkers. Catal Sci Technol 3(8):1937–1941
Šulek F, Fernández DP, Knez Ž, Habulin M, Sheldon RA (2011) Immobilization of horseradish peroxidase as crosslinked enzyme aggregates (CLEAs). Process Biochem 46(3):765–769
Arsenault A, Cabana H, Jones JP (2011) Laccase-based CLEAs: Chitosan as a novel cross-linking agent. Enzyme Res 2011:376015, 10 pages
Yang Xe ZP, Ni Y, Sun Z (2012) Highly efficient biosynthesis of sucrose-6-acetate with cross-linked aggregates of Lipozyme TL 100 L. J Biotechnol 161(1):27–33
Cui JD, Sun LM, Li LL (2013) A simple technique of preparing stable cleas of phenylalanine ammonia lyase using co-aggregation with starch and bovine serum albumin. Appl Biochem Biotechnol 170(8):1827–1837
Miletic N, Loos K (2009) Over-stabilization of chemically modified and cross-linked Candida antarctica lipase B using various epoxides and diepoxides. Aust J Chem 62(8):799–805
Shah S, Sharma A, Gupta MN (2006) Preparation of cross-linked enzyme aggregates by using bovine serum albumin as a proteic feeder. Anal Biochem 351(2):207–213
Yamaguchi H, Miyazaki M, Asanomi Y, Maeda H (2011) Poly-lysine supported cross-linked enzyme aggregates with efficient enzymatic activity and high operational stability. Catal Sci Technol 1(7):1256–1261
Wilson L, Fernández-Lorente G, Fernández-Lafuente R, Illanes A, Guisán JM, Palomo JM (2006) CLEAs of lipases and poly-ionic polymers: a simple way of preparing stable biocatalysts with improved properties. Enzym Microb Technol 39(4):750–755
Tirunagari H, Basetty S, Rode HB, Fadnavis NW (2018) Crosslinked enzyme aggregates (CLEA) of phytase with soymilk proteins. J Biotechnol 282:67–69
Goetze D, Foletto EF, da Silva HB, Silveira VCC, Dal Magro L, Rodrigues RC (2017) Effect of feather meal as proteic feeder on combi-CLEAs preparation for grape juice clarification. Process Biochem 62:122–127
Vaidya BK, Kuwar SS, Golegaonkar SB, Nene SN (2012) Preparation of cross-linked enzyme aggregates of l-aminoacylase via co-aggregation with polyethyleneimine. J Mol Catal B Enzym 74(3–4):184–191
Li X, Yu Z, Bian Z, Xu J, Zhang L, Qiao M (2018) Physiochemical characterization of α-amylase as crosslinked enzyme aggregates. Catalysts 8(8):299
Cabana H, Jones JP, Agathos SN (2007) Preparation and characterization of cross-linked laccase aggregates and their application to the elimination of endocrine disrupting chemicals. J Biotechnol 132(1):23–31
Li XD, Wu J, Jia DC, Wan YH, Yang N, Qiao M (2016) Preparation of cross-linked glucoamylase aggregates immobilization by using dextrin and xanthan gum as protecting agents. Catalysts 6(6):77
Pan J, Kong XD, Li CX, Ye Q, Xu JH, Imanaka T (2011) Crosslinking of enzyme coaggregate with polyethyleneimine: a simple and promising method for preparing stable biocatalyst of Serratia marcescens lipase. J Mol Catal B Enzym 68(3–4):256–261
Gupta P, Dutt K, Misra S, Raghuwanshi S, Saxena RK (2009) Characterization of cross-linked immobilized lipase from thermophilic mould Thermomyces lanuginosa using glutaraldehyde. Bioresour Technol 100(18):4074–4076
López-Serrano P, Cao L, Van Rantwijk F, Sheldon RA (2002) Cross-linked enzyme aggregates with enhanced activity: application to lipases. Biotechnol Lett 24(16):1379–1383
Guimarães JR, de Lima Camargo Giordano R, Fernandez-Lafuente R, Tardioli PW (2018) Evaluation of strategies to produce highly porous cross-linked aggregates of porcine pancreas lipase with magnetic properties. Molecules 23(11):2993
Wang M, Jia C, Qi W, Yu Q, Peng X, Su R, He Z (2011) Porous-CLEAs of papain: application to enzymatic hydrolysis of macromolecules. Bioresour Technol 102(3):3541–3545
Rodrigues RC, Ortiz C, Berenguer-Murcia A, Torres R, Fernández-Lafuente R (2013) Modifying enzyme activity and selectivity by immobilization. Chem Soc Rev 42(15):6290–6307
Fishman A, Cogan U (2003) Bio-imprinting of lipases with fatty acids. J Mol Catal B Enzym 22(3–4):193–202
Cui JD, Liu RL, Li LL, Cui JD (2015) Imprinted cross-linked enzyme aggregate (iCLEA) of phenylalanine ammonia lyase: a new stable biocatalyst. In: Zhang TC, Nakajima M (eds) Advances in applied biotechnology. Lecture notes in electrical engineering, vol 332. Springer, Berlin, Heidelberg
Sampath C, Belur PD, Iyyasami R (2018) Enhancement of n-3 polyunsaturated fatty acid glycerides in Sardine oil by a bioimprinted cross-linked Candida rugosa lipase. Enzym Microb Technol 110:20–29
De Winter K, Soetaert W, Desmet T (2012) An imprinted cross-linked enzyme aggregate (iCLEA) of sucrose phosphorylase: combining improved stability with altered specificity. Int J Mol Sci 13(9):11333
Amaral-Fonseca M, Kopp W, Giordano RLC, Fernández-Lafuente R, Tardioli PW (2018) Preparation of magnetic cross-linked amyloglucosidase aggregates: solving some activity problems. Catalysts 8(11):496
Kopp W, Da Costa TP, Pereira SC, Jafelicci M Jr, Giordano RC, Marques RFC, Araújo-Moreira FM, Giordano RLC (2014) Easily handling penicillin G acylase magnetic cross-linked enzymes aggregates: catalytic and morphological studies. Process Biochem 49(1):38–46
Lee J, Na HB, Kim BC, Lee JH, Lee B, Kwak JH, Hwang Y, Park JG, Gu MB, Kim J, Joo J, Shin CH, Grate JW, Hyeon T, Kim J (2009) Magnetically-separable and highly-stable enzyme system based on crosslinked enzyme aggregates shipped in magnetite-coated mesoporous silica. J Mater Chem 19(42):7864–7870
Matijošyte I, Arends IWCE, de Vries S, Sheldon RA (2010) Preparation and use of cross-linked enzyme aggregates (CLEAs) of laccases. J Mol Catal B Enzym 62(2):142–148
Wine Y, Cohen-Hadar N, Freeman A, Frolow F (2007) Elucidation of the mechanism and end products of glutaraldehyde crosslinking reaction by X-ray structure analysis. Biotechnol Bioeng 98(3):711–718
Okuda K, Urabe I, Yamada Y, Okada H (1991) Reaction of glutaraldehyde with amino and thiol compounds. J Ferment Bioeng 71(2):100–105
Guauque Torres MP, Foresti ML, Ferreira ML (2013) Cross-linked enzyme aggregates (CLEAs) of selected lipases: a procedure for the proper calculation of their recovered activity. AMB Express 3(1):1–11
Acknowledgments
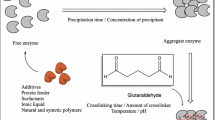
S. Velasco acknowledges the Mexican Council of Science and Technology (CONACyT) for the received postdoctoral fellowship, as well as the scientific illustrator Daniela Velasco (https://danielavelasco.myportfolio.com/scientific-illustration), for illustrating the Fig. 1.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Velasco-Lozano, S. (2020). Immobilization of Enzymes as Cross-Linked Enzyme Aggregates: General Strategy to Obtain Robust Biocatalysts. In: Guisan, J., Bolivar, J., López-Gallego, F., Rocha-Martín, J. (eds) Immobilization of Enzymes and Cells. Methods in Molecular Biology, vol 2100. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0215-7_23
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0215-7_23
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0214-0
Online ISBN: 978-1-0716-0215-7
eBook Packages: Springer Protocols