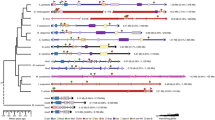
Abstract
The Hox gene clusters have been one of the most prominent paradigms within Developmental Biology. This stems from the great excitement that surrounded the discovery that the genes all contained the conserved homeobox motif and that the homologous genes were operating in broadly homologous ways in the development of organisms as phylogenetically widespread as flies and vertebrates. The sequence similarity between the genes based on the homeobox, and their specific genomic organization in both flies and vertebrates, immediately implied a particular mode of evolution of the Hox gene cluster by tandem duplication and, more intriguingly, a functional constraint on the organization of the cluster to conserve colinearity. This general picture still holds true, but our understanding of the nature and extent of the constraints on cluster organization have been modified in recent years as data has become available from a much wider selection of animal phyla.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Kmita M, Duboule D. Organizing Axes in time and space; 25 years of colinear tinkering. Science 2003; 301:331–333.
Lewis EB. Pseudoallelism and gene evolution. Cold Spring Harb Symp Quant Biol 1951; 16:159–174.
Lewis EB. Genes and developmental pathways. Am Zoologist 1963; 3:33–56.
Lewis EB. A gene complex controlling segmentation in Drosophila. Nature 1978; 276:565–570.
Sánchez-Herrero E, Vernós I, Marco R et al. Genetic organization of Drosophila bithorax complex. Nature 1985; 313:108–113.
Kaufman TC, Lewis R, Wakimoto B. Cytogenetic analysis of chromosome 3 in Drosophila melanogaster: The homeotic gene complex in polytene chromosome interval 84A-B. Genetics 1980; 94:115–133.
McGinnis W, Levine MS, Hafen E et al. A conserved DNA sequence in homeotic gene of the Drosophila Antennapedia and bithorax complexes. Nature 1984; 308:428–433.
Bürglin TR. Homeodomain proteins. In: Meyers RA, ed. Encyclopedia of Molecular Cell Biology and Molecular Medicine. Weinheim: Wiley-VCH Verlag GmbH and Co., 2005:179–222.
Gehring WJ, Affolter M, Bürglin T. Homeodomain proteins. Annu Rev Biochem 1994; 63:487–526.
Brooke NM, Garcia-Fernández J, Holland PWH. The ParaHox gene cluster is an evolutionary sister of the Hox gene cluster. Nature 1998; 392:920–922.
Kourakis MJ, Martindale MQ. Combined-method phylogenetic analysis of Hox and ParaHox genes of the Metazoa. J Exp Zoo (Mol Dev Evol) 2000; 288:175–191.
Ferrier DEK, Holland PWH. Ancient origin of the Hox gene cluster. Nature Rev Genet 2001; 2:33–38.
Finnerty JR, Martindale MQ. Ancient origins of axial patterning genes: Hox genes and ParaHox genes in the Cnidaria. Evol Dev 1999; 1(1):16–23.
Garcia-Fernández J. The genesis and evolution of homeobox gene clusters. Nature Rev Genet 2005; 6:1–12.
Collins AG. Evaluating multiple alternative hypotheses for the origin of Bilateria: An analysis of 18S rRNA molecular evidence. Proc Natl Acad Sci USA 1998; 95:15458–15463.
Peterson KJ, Eernisse DJ. Animal phylogeny and the ancestry of bilaterians: Inferences from morphology and 18S rDNA gene sequences. Evol Dev 2001; 3(3):170–205.
Schierwater B. My favorite animal, Trichoplax adhaerens. BioEssays 2005; 27:1294–1302.
Schierwater B, Kuhn K. Homology of Hox genes and the Zootype concept in early metazoan evolution. Mol Phyl Evol 1998; 9(3):375–381.
Gauchat D, Mazet F, Berney C et al. Evolution of Antp-class genes and differential expression of Hydra Hox/ParaHox genes in anterior patterning. Proc Natl Acad Sci USA 2000; 97:4493–4498.
Finnerty JR, Paulson D, Burton P et al. Early evolution of a homeobox gene: The ParaHox gene Gsx in the Cnidaria and Bilateria. Evol Dev 2003; 5(4):331–345.
Baguñá J, Riutort M. The dawn of bilaterian animals: The case of acoelomorph flatworms. BioEssays 2004; 26:1046–1057.
Garcia-Fernández J. Hox, ParaHox, ProtoHox: Facts and guesses. Heredity 2005; 94(2):145–152.
Pollard SL, Holland PWH. Evidence for 14 homeobox gene clusters in human genome ancestry. Curr Biol 2000; 10(17):1059–1062.
Minguillón C, Garcia-Fernández J. Genesis and evolution of the Evx and Mox genes and the extended Hox and ParaHox gene clusters. Genome Biol 2003; 4:R12.
Minguillón C, Gardenyes J, Serra E et al. No more than 14: The end of the amphioxusHox cluster. Int J Biol Sci 2005; 1:19–23.
Miller DJ, Miles A. Homeobox genes and the zootype. Nature 1993; 365:215–216.
Finnerty JR. Cnidarians reveal intermediate stages in the evolution of Hox clusters and axial complexity. Am Zoologist 2001; 41(3):608–620.
Cook CE, Jiménez E, Akam M et al. The Hox gene complement of acoel flatworms, a basal bilaterian clade. Evol Dev 2004; 6(3):154–163.
Aguinaldo AA, Turbeville JM, Linford LS et al. Evidence for a clade of nematodes, arthropods and other moulting animals. Nature 1997; 387:489–493.
Giribet G. Current advances in the phylogenetic reconstruction of metazoan evolution: A new paradigm for the Cambrian explosion? Mol Phyl Evol 2002; 24:345–357.
Telford MJ, Copley RR. Animal phylogeny: Fatal attraction. Curr Biol 2005; 15(8):R296–R299.
De Rosa R, Grenier JK, Andreeva T et al. Hox genes in brachiopods and priapulids and protostome evolution. Nature 1999; 399:772–776.
Balavoine G, de Rosa R, Adoutte A. Hox clusters and bilaterian phylogeny. Mol Phyl Evol 2002; 24:366–373.
Powers TP, Amemiya CT. Evidence for a Hox14 paralog group in vertebrates. Curr Biol 2004; 14(5):R183–R184.
Kobayashi M, Furuya H, Holland PWH. Dicyemids are higher animals. Nature 1999; 401:762–763.
Passamaneck YJ, Halanych KM. Evidence from Hox genes that bryozoans are lophotrochozoans. Evol Dev 2004; 6:275–281.
Telford MJ. Turning Hox “signatures” into synapomorphies. Evol Dev 2000; 2(6):360–364.
Telford MJ. Affinity for arrow worms. Nature 2004; 431:254–256.
Papillon D, Perez Y, Caubit X et al. Identification of chaetognaths as protostomes is supported by the analysis of their mitochondrial genome. Mol Biol Evol 2004; 21(11):2122–2129.
Helfenbein KG, Fourcade HM, Vanjani RG et al. The mitochondrial genome of Paraspadella gotoi is highly reduced and reveals that chaetognaths are a sister group to protostomes. Proc Natl Acad Sci USA 2004; 101:10639–10643.
Papillon D, Perez Y, Fasano L et al. Hox gene survey in the chaetognath Spadella cephaloptera: Evolutionary implications. Dev Genes Evol 2003; 213:142–148.
Van Auken K, Weaver DC, Edgar LG et al. Caenorhabditis elegans embryonic axial patterning requires two recently discovered posterior-group Hox genes. Proc Natl Acad Sci USA 2000; 97:4499–4503.
Halanych KM. Testing hypotheses of chaetognath origins: Long branches revealed by 18S ribosomal DNA. Syst Biol 1996; 45:223–246.
Ferrier DEK, Minguillón C, Holland PWH et al. The amphioxus Hox cluster: Deuterostome posterior flexibility and Hox14. Evol Dev 2000; 2(5):284–293.
Cameron RA, Rowen L, Nesbitt R et al. Unusual gene order and organization of the sea urchin Hox cluster. J Exp Zoo (Mol Dev Evol) 2005; 304B:1–14.
Martinez P, Rast JP, Arenas-Mena C et al. Organization of an echinoderm Hox gene cluster. Proc Natl Acad Sci USA 1999; 96:1469–1474.
Long S, Martinez P, Chen WC et al. Evolution of echinoderms may not have required modification of the ancestral deuterostome Hox gene cluster: First report of PG4 and PG5 Hox orthologues in echinoderms. Dev Genes Evol 2003; 213:573–576.
Mito T, Endo K. PCR survey of Hox genes in the crinoid and ophiuroid: Evidence for anterior conservation and posterior expansion in the echinoderm Hox gene cluster. Mol Phyl Evol 2000; 14(3):375–388.
Long S, Byrne M. Evolution of the echinoderm Hox gene cluster. Evol Dev 2001; 3(5):302–311.
Peterson KJ. Isolation of Hox and ParaHox genes in the hemichordate Ptychodera flava and the evolution of deuterostome Hox genes. Mol Phyl Evol 2004; 31:1208–1215.
Bourlat SJ, Nielsen C, Lockyer AE et al. Xenoturbella is a deuterostome that eats molluscs. Nature 2003; 424:925–928.
Duboule D. Temporal colinearity and the phylotypic progression: A basis for the stability of a vertebrate Bauplan and the evolution of morphologies through heterochrony. Development 1994; (Suppl):135–142.
Ferrier DEK, Holland PWH. Ciona intestinalis ParaHox genes: Evolution of Hox/ParaHox cluster integrity, developmental mode, and temporal colinearity. Mol Phyl Evol 2002; 24:412–417.
Ferrier DEK, Minguillón C. Evolution of the Hox/ParaHox gene clusters. Int J Dev Biol 2003; 47:605–611.
Crawford M. Hox genes as synchronized temporal regulators: Implications for morphological innovation. J Exp Zoo (Mol Dev Evol) 2003; 295B:1–11.
Patel NH. Time, space and genomes. Nature 2004; 431:28–29.
Seo HC, Edvardsen RB, Maeland AD et al. Hox cluster disintegration with persistent anteroposterior order of expression in Oikopleura dioica. Nature 2004; 431:67–71.
De Rosa R, Prud’homme B, Balavoine G. Caudal and even-skipped in the annelid Platynereis dumerilii and the ancestry of posterior growth. Evol Dev 2005; 7(6):574–587.
Pierce RJ, Wu W, Hirai H et al. Evidence for a dispersed Hox gene cluster in the platyhelminth parasite Schistosoma mansoni. Mol Biol Evol 2005; 22:2491–2503.
Arenas-Menas C, Martinez P, Cameron RA et al. Expression of the Hox gene complex in the indirect development of a sea urchin. Proc Natl Acad Sci USA 1998; 95:13062–13067.
Duboule D. Hox is in the hair: A break in colinearity? Genes Devel 1998; 12:1–4.
Roelen BAJ, de Graaff W, Forlani S et al. Hox cluster polarity in early transcriptional availability: A high order regulatory level of clustered Hox genes in the mouse. Mech Devel 2002; 119:81–90.
Suemori H, Noguchi S. HoxC cluster genes are dispensable for overall body plan of mouse embryonic development. Dev Biol 2000; 220:333–342.
Amores A, Suzuki T, Yan YL et al. Developmental roles of pufferfish Hox clusters and genome evolution in ray-finned fish. Genome Res 2004; 14:1–10.
Chambeyron S, Bickmore W. Chromatin decondensation and nuclear reorganization of the HoxB locus upon induction of transcription. Genes Devel 2004; 18:1119–1130.
Chambeyron S, Da Silva NR, Lawson KA et al. Nuclear reorganisation of the Hoxb complex during mouse embryonic development. Development 2005; 132:2215–2223.
Kmita M, van der Hoeven F, Zákány J et al. Mechanisms of Hox gene colinearity: Transposition of the anterior Hoxb1 gene into the posterior HoxD complex. Genes Devel 2000; 14:198–211.
Wada H, Garcia-Fernández J, Holland PWH. Colinear and segmental expression of amphioxus Hox genes. Dev Biol 1999; 213:131–141.
Duncan I. The Bithorax complex. Ann Rev Genet 1987; 21:285–319.
Averof M, Dawes R, Ferrier D. Diversification of arthropod Hox genes as a paradigm for the evolution of gene functions. Sem Cell Dev Biol 1996; 7:539–551.
Sharpe J, Nonchev S, Gould A et al. Selectivity, sharing and competitive interactions in the regulation of Hoxb genes. EMBO J 1998; 17(6):1788–1798.
Von Allmen G, Hogga I, Spierer A. Splits in fruitfly Hox gene complexes. Nature 1996; 380:116.
Negre B, Ranz JM, Casals F et al. A new split of the Hox gene complex in Drosophila: Relocation and evolution of the gene labial. Mol Biol Evol 2003; 20:2042–2054.
Lewis EB, Pfeiffer BD, Mathog DR et al. Evolution of the homeobox complex in the Diptera. Curr Biol 2003; 13(15):R587–R588.
Negre B, Casillas S, Suzanne M et al. Conservation of regulatory sequences and gene expression patterns in the disintegrating Drosophila Hox gene complex. Genome Research 2005; 15:692–700.
Powell JR, DeSalle R. Drosophila molecular phylogenies and their uses. Evol Biol 1995; 28:87–139.
Yasukochi Y, Ashakumary LA, Wu C et al. Organization of the Hox gene cluster of the silkworm, Bombyx mori: A split of the Hox cluster in a nonDrosophila insect. Dev Genes Evol 2004; 214:606–614.
Orlando V. Polycomb, epigenomes, and control of cell identity. Cell 2003; 112:599–606.
Kmita M, Kondo T, Duboule D. Targeted inversion of a polar silencer within the HoxD complex reallocates domains of enhancer sharing. Nature Genetics 2000; 26:451–454.
Belozerov VE, Majumder P, Shen P et al. A novel boundary element may facilitate independent gene regulation in the Antennapedia complex of Drosophila. EMBO J 2003; 22(12):3113–3121.
Ronshaugen M, Biemar F, Piel J et al. The Drosophila microRNA iab-4 causes a dominant homeotic transformation of halteres to wings. Genes Dev 2005; 19:2947–2952.
Stark A, Brennecke J, Bushati N et al. Animal microRNAs confer robustness to gene expression and have a significant impact on 3’UTR evolution. Cell 2005; 123:1133–1146.
Falciani F, Hausdorf B, Schröder R et al. Class 3 Hox genes in insects and the origin of zen. Proc Natl Acad Sci USA 1996; 93:8479–8484.
Stauber M, Jäckie H, Schmidt-Ott U. The anterior determinant bicoid of Drosophila is a derived Hox class 3 gene. Proc Natl Acad Sci USA 1999; 96:3786–3789.
Cameron CB, Garey JR, Swalla BJ. Evolution of the chordate body plan: New insights from phylogenetic anlyses of deuterostome phyla. Proc Natl Acad Sci USA 2000; 97:4469–4474.
Jakob W, Sagasser S, Dellaporta S et al. The Trox-2 Hox/ParaHox gene of Trichoplax (Placozoa) marks an epithelial boundary. Dev Genes Evol 2004; 214:170–175.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Copyright information
© 2007 Landes Bioscience and Springer Science+Business Media
About this chapter
Cite this chapter
Ferrier, D.E.K. (2007). Evolution of Hox Gene Clusters. In: HOX Gene Expression. Springer, New York, NY. https://doi.org/10.1007/978-0-387-68990-6_4
Download citation
DOI: https://doi.org/10.1007/978-0-387-68990-6_4
Publisher Name: Springer, New York, NY
Print ISBN: 978-0-387-68989-0
Online ISBN: 978-0-387-68990-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)