Abstract
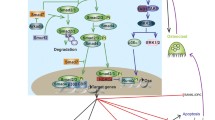
Development of the vertebrate skeleton is a complex process involving genes that control a series of cellular events such as proliferation, migration, mesenchymal condensations and eventually differentiation into osteoblasts and chondrocytes1. For osteoblast differentiation, an intricate and highly regulated interplay of growth factors, receptors/co-receptors, specific extracellular milieu and transcription factors is required2. Bone morphogenetic proteins (BMPs), members of the transforming growth factor-beta (TGF- ) superfamily, are multifunctional growth factors critically involved in osteoblast differentiation. The BMP functions are exerted through their interaction with specific cell surface receptors, BMP type I and II receptors, leading to the regulation of specific BMP target genes. The signaling is regulated at numerous steps at extracellular, cell surface and intracellular levels3, 4.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Bone Morphogenetic Protein
- Osteoblast Differentiation
- C2C12 Cell
- Osteogenic Marker
- Sodium Dodecyl Sulfate Sample Buffer
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
7.6. References
B. R. Olsen, A. M. Reginato, and W. Wang, Bone development, Annu Rev Cell Dev Biol 16, 191–220 (2000).
G. Xiao, R. Gopalakrishnan, D. Jiang, E. Reith, M. D. Benson, and R. T. Franceschi, Bone morphogenetic proteins, extracellular matrix, and mitogen-activated protein kinase signaling pathways are required for osteoblast-specific gene expression and differentiation in MC3T3-E1 cells, J Bone Miner Res 17(1), 101–110 (2002).
K. Miyazono, S. Maeda, and T. Imamura, BMP receptor signaling: transcriptional targets, regulation of signals, and signaling cross-talk, Cytokine Growth Factor Rev 16(3), 251–263 (2005).
P. ten Dijke, O. Korchynskyi, G. Valdimarsdottir, and M. J. Goumans, Controlling cell fate by bone morphogenetic protein receptors, Mol Cell Endocrinol 211(1–2), 105–113 (2003).
R. V. Iozzo, The biology of the small leucine-rich proteoglycans. Functional network of interactive proteins, J Biol Chem 274(27), 18843–18846 (1999).
L. Ameye, and M. F. Young, Mice deficient in small leucine-rich proteoglycans: novel in vivo models for osteoporosis, osteoarthritis, Ehlers-Danlos syndrome, muscular dystrophy, and corneal diseases, Glycobiology 12(9), 107R–116R (2002).
P. Bianco, L. W. Fisher, M. F. Young, J. D. Termine, and P. G. Robey, Expression and localization of the two small proteoglycans biglycan and decorin in developing human skeletal and non-skeletal tissues, J Histochem Cytochem 38(11), 1549–1563 (1990).
T. Xu, P. Bianco, L. W. Fisher, G. Longenecker, E. Smith, S. Goldstein, J. Bonadio, A. Boskey, A. M. Heegaard, B. Sommer, K. Satomura, P. Dominguez, C. Zhao, A. B. Kulkarni, P. G. Robey, and M. F. Young, Targeted disruption of the biglycan gene leads to an osteoporosis-like phenotype in mice, Nat Genet 20(1), 78–82 (1998).
X. D. Chen, S. Shi, T. Xu, P. G. Robey, and M. F. Young, Age-related osteoporosis in biglycan-deficient mice is related to defects in bone marrow stromal cells, J Bone Miner Res 17(2), 331–340 (2002).
Y. Bi, C. H. Stuelten, T. Kilts, S. Wadhwa, R. V. Iozzo, P. G. Robey, X. D. Chen, and M. F. Young, Extracellular matrix proteoglycans control the fate of bone marrow stromal cells, J Biol Chem 280(34), 30481–30489 (2005).
A. Hildebrand, M. Romaris, L. M. Rasmussen, D. Heinegard, D. R. Twardzik, W. A. Border, and E. Ruoslahti, Interaction of the small interstitial proteoglycans biglycan, decorin and fibromodulin with transforming growth factor beta, Biochem J 302 (Pt 2), 527–534 (1994).
Y. Hayashi, C. Y. Liu, J. J. Jester, M. Hayashi, I. J. Wang, J. L. Funderburgh, S. Saika, P. J. Roughley, C. W. Kao, and W. W. Kao, Excess biglycan causes eyelid malformation by perturbing muscle development and TGF-alpha signaling, Dev Biol 277(1), 222–234 (2005).
E. Tufvesson, and G. Westergren-Thorsson, Tumour necrosis factor-alpha interacts with biglycan and decorin, FEBS Lett 530(1–3), 124–128 (2002).
X. D. Chen, L. W. Fisher, P. G. Robey, and M. F. Young, The small leucine-rich proteoglycan biglycan modulates BMP-4-induced osteoblast differentiation, Faseb J 18(9), 948–958 (2004).
M. Moreno, R. Munoz, F. Aroca, M. Labarca, E. Brandan, and J. Larrain, Biglycan is a new extracellular component of the Chordin-BMP4 signaling pathway, Embo J 24(7), 1397–1405 (2005).
D. Parisuthiman, Y. Mochida, W. R. Duarte, and M. Yamauchi, Biglycan modulates osteoblast differentiation and matrix mineralization, J Bone Miner Res 20(10), 1878–1886 (2005).
T. Imamura, M. Takase, A. Nishihara, E. Oeda, J. Hanai, M. Kawabata, and K. Miyazono, Smad6 inhibits signalling by the TGF-beta superfamily, Nature 389(6651), 622–626 (1997).
T. Ebisawa, K. Tada, I. Kitajima, K. Tojo, T. K. Sampath, M. Kawabata, K. Miyazono, and T. Imamura, Characterization of bone morphogenetic protein-6 signaling pathways in osteoblast differentiation, J Cell Sci 112 (Pt 20), 3519–3527 (1999).
A. Nishihara, T. Watabe, T. Imamura, and K. Miyazono, Functional heterogeneity of bone morphogenetic protein receptor-II mutants found in patients with primary pulmonary hypertension, Mol Biol Cell 13(9), 3055–3063 (2002).
K. J. Livak, and T. D. Schmittgen, Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method, Methods 25(4), 402–408 (2001).
H. Itoh, M. Yamauchi, H. Kataoka, R. Hamasuna, N. Kitamura, and M. Koono, Genomic structure and chromosomal localization of the human hepatocyte growth factor activator inhibitor type 1 and 2 genes, Eur J Biochem 267(11), 3351–3359 (2000).
T. Katagiri, A. Yamaguchi, M. Komaki, E. Abe, N. Takahashi, T. Ikeda, V. Rosen, J. M. Wozney, A. Fujisawa-Sehara, and T. Suda, Bone morphogenetic protein-2 converts the differentiation pathway of C2C12 myoblasts into the osteoblast lineage, J Cell Biol 127(6 Pt 1), 1755–1766 (1994).
S. Maeda, M. Hayashi, S. Komiya, T. Imamura, and K. Miyazono, Endogenous TGF-beta signaling suppresses maturation of osteoblastic mesenchymal cells, Embo J 23(3), 552–563 (2004).
S. Harada, and G. A. Rodan, Control of osteoblast function and regulation of bone mass, Nature 423(6937), 349–355 (2003).
L. A. Sabourin, and M. A. Rudnicki, The molecular regulation of myogenesis, Clin Genet 57(1), 16–25 (2000).
J. M. Wozney, V. Rosen, A. J. Celeste, L. M. Mitsock, M. J. Whitters, R. W. Kriz, R. M. Hewick, and E. A. Wang, Novel regulators of bone formation: molecular clones and activities, Science 242(4885), 1528–1534 (1988).
T. Kirsch, W. Sebald, and M. K. Dreyer, Crystal structure of the BMP-2-BRIA ectodomain complex, Nat Struct Biol 7(6), 492–496 (2000).
J. Garcia Abreu, C. Coffinier, J. Larrain, M. Oelgeschlager, and E. M. De Robertis, Chordin-like CR domains and the regulation of evolutionarily conserved extracellular signaling systems, Gene 287(1–2), 39–47 (2002).
S. Hartwig, M. C. Hu, C. Cella, T. Piscione, J. Filmus, and N. D. Rosenblum, Glypican-3 modulates inhibitory Bmp2-Smad signaling to control renal development in vivo, Mech Dev 122(7–8), 928–938 (2005).
T. A. Samad, A. Rebbapragada, E. Bell, Y. Zhang, Y. Sidis, S. J. Jeong, J. A. Campagna, S. Perusini, D. A. Fabrizio, A. L. Schneyer, H. Y. Lin, A. H. Brivanlou, L. Attisano, and C. J. Woolf, DRAGON, a bone morphogenetic protein co-receptor, J Biol Chem 280(14), 14122–14129 (2005).
H. Nishitoh, H. Ichijo, M. Kimura, T. Matsumoto, F. Makishima, A. Yamaguchi, H. Yamashita, S. Enomoto, and K. Miyazono, Identification of type I and type II serine/threonine kinase receptors for growth/differentiation factor-5, J Biol Chem 271(35), 21345–21352 (1996).
A. Nohe, S. Hassel, M. Ehrlich, F. Neubauer, W. Sebald, Y. I. Henis, and P. Knaus, The mode of bone morphogenetic protein (BMP) receptor oligomerization determines different BMP-2 signaling pathways, J Biol Chem 277(7), 5330–5338 (2002).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer Science+Business Media, LLC
About this paper
Cite this paper
Mochida, Y., Parisuthiman, D., Yamauchi, M. (2006). Biglycan Is a Positive Modulator of BMP-2 Induced Osteoblast Differentiation. In: Fisher, J.P. (eds) Tissue Engineering. Advances in Experimental Medicine and Biology, vol 585. Springer, Boston, MA. https://doi.org/10.1007/978-0-387-34133-0_7
Download citation
DOI: https://doi.org/10.1007/978-0-387-34133-0_7
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-32664-1
Online ISBN: 978-0-387-34133-0
eBook Packages: EngineeringEngineering (R0)