Abstract
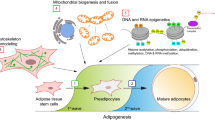
Adipose tissue regulates metabolic homeostasis by acting as an endocrine organ and energy reservoir. Adipose tissue development and functional maintenance are dependent on adipocyte differentiation, in which autophagy plays an important role. It has been shown that autophagy deficiency dampens adipocyte differentiation, compromises adipose tissue development, dysregulates adipocytokine secretion, and even causes sudden death in young animals. Therefore, accurate assessment of autophagy in adipocyte is critical for the study of adipose biology or pathology of metabolic diseases. In this chapter, we described the procedure of autophagy analysis during adipocyte differentiation, and discussed the power of steady-state autophagy protein (e.g., beclin 1, LC3, and p62) levels versus autophagy flux to reflect autophagy activity.
Similar content being viewed by others
References
Kershaw EE, Flier JS (2004) Adipose tissue as an endocrine organ. J Clin Endocrinol Metab 89:2548–2556
Galic S, Oakhill JS, Steinberg GR (2010) Adipose tissue as an endocrine organ. Mol Cell Endocrinol 316:129–139
Slutsky N, Vatarescu M, Haim Y et al (2016) Decreased adiponectin links elevated adipose tissue autophagy with adipocyte endocrine dysfunction in obesity. Int J Obes (Lond) 40:912–920
Rutkowski JM, Stern JH, Scherer PE (2015) The cell biology of fat expansion. J Cell Biol 208:501–512
Crewe C, An YA, Scherer PE (2017) The ominous triad of adipose tissue dysfunction: inflammation, fibrosis, and impaired angiogenesis. J Clin Invest 127:74–82
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741
Green DR, Levine B (2014) To be or not to be? How selective autophagy and cell death govern cell fate. Cell 157:65–75
Liu L, Tao Z, Zheng LD et al (2016) FoxO1 interacts with transcription factor EB and differentially regulates mitochondrial uncoupling proteins via autophagy in adipocytes. Cell Death Dis 2:16066
Liu L, Zheng LD, Zou P et al (2016) FoxO1 antagonist suppresses autophagy and lipid droplet growth in adipocytes. Cell Cycle 15:2033–2041
Singh R, Xiang Y, Wang Y et al (2009) Autophagy regulates adipose mass and differentiation in mice. J Clin Invest 119:3329–3339
Zhang Y, Goldman S, Baerga R, Zhao Y, Komatsu M, Jin S (2009) Adipose-specific deletion of autophagy-related gene 7 (atg7) in mice reveals a role in adipogenesis. Proc Natl Acad Sci U S A 106:19860–19865
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140:313–326
Klionsky DJ, Abdelmohsen K, Abe A et al (2016) Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 12:1–222
Liu L, Zheng LD, Donnelly SR, Emont MP, Wu J, Cheng Z (2017) Isolation of mouse stromal vascular cells for monolayer culture. Methods Mol Biol 1566:9–16
Liu L, Zou P, Zheng L et al (2015) Tamoxifen reduces fat mass by boosting reactive oxygen species. Cell Death Dis 6:e1586
Zou P, Liu L, Zheng L et al (2014) Targeting FoxO1 with AS1842856 suppresses adipogenesis. Cell Cycle 13:3759–3767
Acknowledgment
This work was supported in part by USDA National Institute of Food and Agriculture Hatch Project 1007334 (ZC).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Science+Business Media New York
About this protocol
Cite this protocol
Tao, Z., Liu, L., Zheng, L.D., Cheng, Z. (2017). Autophagy in Adipocyte Differentiation. In: Turksen, K. (eds) Autophagy in Differentiation and Tissue Maintenance. Methods in Molecular Biology, vol 1854. Humana Press, New York, NY. https://doi.org/10.1007/7651_2017_65
Download citation
DOI: https://doi.org/10.1007/7651_2017_65
Published:
Publisher Name: Humana Press, New York, NY
Print ISBN: 978-1-4939-8747-4
Online ISBN: 978-1-4939-8748-1
eBook Packages: Springer Protocols