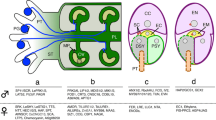
Abstract
The mechanisms of pollen tube growth have been studied in a wide variety of plant species. Since the 1990s, with the explosion of molecular genetic analyses in Arabidopsis thaliana, most studies started to focus on this model plant. However, because of their particular characteristics, plant species other than Arabidopsis are still used to reveal physiological mechanisms and identify novel molecules relating to pollen tube growth, including, for example, lily, tobacco, Nicotiana alata, tomato, rice, maize, Brassica spp., corn poppy and Torenia (Table 1). Here, we designate all of these relatively common experimental plants as “biological models” for the study of pollen tube growth. These models sometimes provide a good first step in the identification of novel physiological mechanisms and molecules. As genome sequencing technologies become more advanced, the difficulty of performing molecular analyses in these biological models will decrease. Thus, a better understanding of these biological models will allow researchers to perform unique studies of pollen tube growth. In this chapter, we compare the characteristics of biological models, focusing on in vitro systems, to facilitate the use of these biological models for in vitro analyses.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Aida R, Yoshida K, Kondo T, Kishimoto S, Shibata M (2000) Copigmentation gives bluer flowers on transgenic torenia plants with the antisense dihydroflavonol-4-reductase gene. Plant Sci 160:49–56
Arabidopsis Genome Initiative (2000) Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 408:796–815
Barinova I, Zhexembekova M, Barsova E, Lukyanov S, Heberle-Bors E, Touraev A (2002) Antirrhinum majus microspore maturation and transient transformation in vitro. J Exp Bot 53:1119–1129
Birky CW Jr (1995) Uniparental inheritance of mitochondrial and chloroplast genes: mechanisms and evolution. Proc Natl Acad Sci USA 92:11331–11338
Brewbaker JL (1967) The distribution and phylogenetic significance of binucleate and trinucleate pollen grains in the angiosperms. Am J Bot 54:1069–1083
Brewbaker JL, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50:859–865
Chen YF, Matsubayashi Y, Sakagami Y (2000) Peptide growth factor phytosulfokine-alpha contributes to the pollen population effect. Planta 211:752–755
Chen CY, Wong EI, Vidali L, Estavillo A, Hepler PK, Wu HM, Cheung AY (2002) The regulation of actin organization by actin-depolymerizing factor in elongating pollen tubes. Plant Cell 14:2175–2190
Cheung AY, Wang H, Wu HM (1995) A floral transmitting tissue-specific glycoprotein attracts pollen tubes and stimulates their growth. Cell 82:383–393
Cheung AY, Chen CYH, Glaven RH, de Graaf BHJ, Vidali L, Hepler PK, Wu HM (2002) Rab2 GTPase regulates vesicle trafficking between the endoplasmic reticulum and the Golgi bodies and is important to ddpollen tube growth. Plant Cell 14:945–962
Derksen J, Knuiman B, Hoedemaekers K, Guyon A, Bonhomme S, Pierson ES (2002) Growth and cellular organization of Arabidopsis pollen tubes in vitro. Sex Plant Reprod 15:133–139
Dong J, Kim ST, Lord EM (2005) Plantacyanin plays a role in reproduction in Arabidopsis. Plant Physiol 138:778–789
Engel ML, Chaboud A, Dumas C, McCormick S (2003) Sperm cells of Zea mays have a complex complement of mRNAs. Plant J 34:697–707
Fauré S, Noyer JL, Carreel F, Horry JP, Bakry F, Lanaud C (1994) Maternal inheritance of chloroplast genome and paternal inheritance of mitochondrial genome in bananas (Musa acuminata). Curr Genet 25:265–269
Faure JE, Rotman N, Fortune P, Dumas C (2002) Fertilization in Arabidopsis thaliana wild type: developmental stages and time course. Plant J 30:481–488
Forsthoefel NR, Bohnert HJ, Smith SE (1992) Discordant inheritance of mitochondrial and plastid DNA in diverse alfalfa genotypes. J Hered 83:342–345
Franklin-Tong VE, Lawrence MJ, Franklin FCH (1988) An in vitro bioassay for the stigmatic product of the self-incompatibility gene in Papaver rhoeas L. New Phytol 110:109–118
Harushima Y, Nakagahra M, Yano M, Sasaki T, Kurata N (2001) A genome-wide survey of reproductive barriers in an intraspecific hybrid. Genetics 159:883–892
Harushima Y, Nakagahra M, Yano M, Sasaki T, Kurata N (2002) Diverse variation of reproductive barriers in three intraspecific rice crosses. Genetics 160:313–322
Heslop-Harrison J, Heslop-Harrison Y (1986) Pollen-tube chemotropism: fact or delusion? In: Cresti M, Dallai R (eds) Biology of reproduction and cell motility in plant and animals. University of Siena, Siena, Italy, p 169–174
Heslop-Harrison Y, Shivanna KR (1977) Receptive surface of angiosperm stigma. Ann Bot 41:1233–1258
Higashiyama T (2002) The synergid cell: attractor and acceptor of the pollen tube for double fertilization. J Plant Res 115:149–160
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1997) Kinetics of double fertilization in Torenia fournieri based on direct observations of the naked embryo sac. Planta 203:101–110
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (1998) Guidance in vitro of the pollen tube to the naked embryo sac of Torenia fournieri. Plant Cell 10:2019–2032
Higashiyama T, Kuroiwa H, Kawano S, Kuroiwa T (2000) Explosive discharge of pollen tube contents in Torenia fournieri. Plant Physiol 122:11–14
Higashiyama T, Yabe S, Sasaki N, Nishimura Y, Miyagishima S, Kuroiwa H, Kuroiwa T (2001) Pollen tube attraction by the synergid cell. Science 293:1480–1483
Higashiyama T, Kuroiwa H, Kuroiwa T (2003) Pollen-tube guidance: beacons from the female gametophyte. Curr Opin Plant Biol 6:36–41
Hodgkin T (1983) A medium for germinating Brassica pollen in vitro. Cruciferae Newslett 8:62–63
Holdaway-Clarke TL, Weddle NM, Kim S, Robi A, Parris C, Kunkel JG, Hepler PK (2003) Effect of extracellular calcium, pH and borate on growth oscillations in Lilium formosanum pollen tubes. J Exp Bot 54:65–72
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Hülskamp M, Schneitz K, Pruitt RE (1995) Genetic evidence for a long-range activity that directs pollen tube guidance in Arabidopsis. Plant Cell 7:57–64
International Rice Genome Sequencing Project (2005) The map-based sequence of the rice genome. Nature 436:793–800
Itoh J, Nonomura K, Ikeda K, Yamaki S, Inukai Y, Yamagishi H, Kitano H, Nagato Y (2005) Rice plant development: from zygote to spikelet. Plant Cell Physiol 46:23–47
Jahnen W, Lush WM, Clarke AE (1989) Inhibition of in vitro pollen tube growth by isolated S-glycoproteins of Nicotiana alata. Plant Cell 1:501–510
Janson J (1993) Placental pollination in Lilium longiflorum thunb. Plant Sci 90:105–115
Kao TH, Tsukamoto T (2004) The molecular and genetic bases of S-RNase-based self-incompatibility. Plant Cell 16:S72–83
Kariya K (1989) Sterility caused by cooling treatment at the flowering stage in rice plants. 3. Establishment of a method of in vitro pollen germination. Japanese J Crop Sci 58:96–102
Kasahara RD, Portereiko MF, Sandaklie-Nikolova L, Rabiger DS, Drews GN (2005) MYB98 is required for pollen tube guidance and synergid cell differentiation in Arabidopsis. Plant Cell 17:2981–2992
Khatun S, Flowers TJ (1995) The estimation of pollen viability in rice. J Exp Bot 46:151–154
Kim S, Mollet JC, Dong J, Zhang K, Park SY, Lord EM (2004) Chemocyanin, a small basic protein from the lily stigma, induces pollen tube chemotropism. Proc Natl Acad Sci USA 100:16125–16130
Li H, Lin Y, Heath RM, Zhu MX, Yang Z (1999) Control of pollen tube tip growth by a Rop GTPase-dependent pathway that leads to tip-localized calcium influx. Plant Cell 11:1731–1742
Loomis WD, Durst RW (1992) Chemistry and biology of boron. Biofactors 3:229–239
Lush WM (1999) Whither chemotropism and pollen tube guidance? Trends Plant Sci 4:413–418
Lush WM, Grieser F, Wolters-Arts M (1998) Directional guidance of Nicotiana alata pollen tubes in vitro and on the stigma. Plant Physiol 118:733–741
Malhó R, Trewavas AJ (1996) Localized apical increases of cytosolic free calcium control pollen tube orientation. Plant Cell 8:1935–1949
Márton ML, Cordts S, Broadhvest J, Dresselhaus T (2005) Micropylar pollen tube guidance by egg apparatus 1 of maize. Science 307:573–576
Mascarenhas JP, Machlis L (1962a) Chemotropic response of Antirrhinum majus pollen to calcium. Nature 196:292–293
Mascarenhas JP, Machlis L (1962b) The hormonal control of the directional growth of pollen tubes. Vitamins and Hormones 20:347–372
Mascarenhas JP, Machlis L (1962c) The pollen-tube chemotropic factor from Antirrhinum majus: bioassay, extraction, and partial purification. Am J Bot 49:482–489
McClure BA, Haring V, Ebert PR, Anderson MA, Simpson RJ, Sakiyama F, Clarke AE (1989) Style self-incompatibility gene products of Nicotiana alata are ribonucleases. Nature 342:955–957
McCormick S, Yang H (2005) Is there more than one way to attract a pollen tube? Trends Plant Sci 10:260–263
Miki H (1954) A study of tropism of pollen tubes to the pistil. I. Tropism in Lilium. Bot Mag Tokyo 67:143–147
Mòl R, Matthys-Rochon E, Dumas C (1994) The kinetics of cytological events during double fertilization in Zea mays L. Plant J 5:197–206
Mori T, Kuroiwa H, Higashiyama T, Kuroiwa T (2003) Identification of higher plant GlsA, a putative morphogenesis factor of gametic cells. Biochem Biophys Res Commun 306:564–569
Negre F, Kish CM, Boatright J, Underwood B, Shibuya K, Wagner C, Clark DG, Dudareva N (2003) Regulation of methylbenzoate emission after pollination in snapdragon and petunia flowers. Plant Cell 15:2992–3006
Nitsch JP (1951) Growth and development in vitro of excised ovaries. Am J Bot 38:566–577
Palanivelu R, Brass L, Edlund AF, Preuss D (2003) Pollen tube growth and guidance is regulated by POP2, an Arabidopsis gene that controls GABA levels. Cell 114:47–59
Preuss D, Lemieux B, Yen G, Davis RW (1993) A conditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signaling during fertilization. Genes Dev 7:974–985
Raghavan V (2003) Some reflections on double fertilization, from its discovery to the present. New Phytologist 159:565–583
Ray SM, Park SS, Ray A (1997) Pollen tube guidance by the female gametophyte. Development 124:2489–2498
Read M, Bacic A, Clarke AE (1992) Pollen tube growth in culture. I. Control of morphology and generative cell division in cultured pollen tubes of Nicotiana. In: Ottaviano E, Mulcahy DL, Sari Gorla M, Bergamini Mulcahy G (eds) Angiosperm pollen and ovules. Springer-Verlag, New York, p 162–167
Read SM, Clarke AE, Bacic A (1993) Requirements for division of the generative nucleus in cultured pollen tubes of Nicotiana. Protoplasma 174:101–115
Romagnoli S, Cai G, Cresti M (2003) In vitro assays demonstrate that pollen tube organelles use kinesin-related motor proteins to move along microtubules. Plant Cell 15:251–269
Rudd JJ, Franklin-Tong VE (2003) Signals and targets of the self-incompatibility response in pollen of Papaver rhoeas. J Exp Bot 54:141–148
Russell SD (1984) Ultrastructure of the sperm of Plumbago zeylanica: 2. Quantitative cytology and 3-dimensional organization. Planta 162:385–391
Russell SD (1985) Preferential fertilization in Plumbago zeylanica: ultrastructural evidence for gamete-level recognition in an angiosperm. Proc Natl Acad Sci USA 82:6129–6132
Saito C, Nagata N, Sakai A, Mori K, Kuroiwa H, Kuroiwa T (2002) Angiosperm species that produce sperm cell pairs or generative cells with polarized distribution of DNA-containing organelles. Sex Plant Reprod 15:167–178
Schreiber DN, Dresselhaus T (2003) In vitro pollen germination and transient transformation of Zea mays and other plant species. Plant Mol Biol Rep 21:31–41
Shimizu KK, Okada K (2000) Attractive and repulsive interactions between female and male gametophytes in Arabidopsis pollen tube guidance. Development 127:4511–4518
Shimizu KK, Cork JM, Caicedo AL, Mays CA, Moore RC, Olsen KM, Ruzsa S, Coop G, Bustamante CD, Awadalla P, Purugganan MD (2004) Darwinian selection on a selfing locus. Science 306:2081–2084
Shirasaki R, Katsumata R, Murakami F (1998) Change in chemoattractant responsiveness of developing axons at an intermediate target. Science 279:105–107
Sodmergen, Suzuki T, Kawano S, Nakamura S, Tano S, Kuroiwa T (1992) Behaviour of organelle nuclei (nucleoids) in generative and vegetative cells during maturation of pollen in Lilium longiflorum and Pelargonium zonale. Protoplasma 168:73–82
Sodmergen, Chen GH, Hu ZM, Guo FL, Guan XL (1995) Male gametophyte development in Plumbago zeylanica: cytoplasm localization and cell determination in the early generative cells. Protoplasma 186:79–86
Sogo A, Tobe H (2005) Intermittent pollen-tube growth in pistils of alders (Alnus). Proc Natl Acad Sci USA 102:8770–8775
Stern H (1985) Initiation of meiosis in lily and mouse: some molecular considerations. Arch Anat Microsc Morphol Exp 74:10–13
Takayama S, Isogai A (2003) Molecular mechanism of self-recognition in Brassica self-incompatibility. J Exp Bot 54:149–156
Tang W, Ezcurra I, Muschietti J, McCormick S (2002) A cysteine-rich extracellular protein, LAT52, interacts with the extracellular domain of the pollen receptor kinase LePRK2. Plant Cell 14:2277–2287
Tansengco ML, Imaizumi-Anraku H, Yoshikawa M, Takagi S, Kawaguchi M, Hayashi M, Murooka Y (2004) Pollen development and tube growth are affected in the symbiotic mutant of Lotus japonicus, crinkle. Plant Cell Physiol 45:511–520
Tupy J, Rihova L (1984) Changes and growth effect of pH in pollen-tube culture. J Plant Physiol 115:1–10
Twell D, Klein TM, Fromm ME, McCormick S (1989) Transient expression of chimeric genes delivered into pollen by microprojectile bombardment. Plant Physiol 91:1270–1274
Ueda K, Tanaka I (1994) The basic-proteins of male gametic nuclei isolated from pollen grains of Lilium longiflorum. Planta 192:446–452
Van Tieghem MP (1869) Végétation libre du pollen et de l'ovule. Ann Sci Nat Botan 12:312–328
Van Went JL, Willemse MTM (1984) Fertilization. In: Johri BM (ed) Embryology of angiosperms. Springer, Berlin, p 273–317
Vasil IK (1987) Physiology and culture of pollen. Int Rev Cytol 107:127–174
Vervaeke I, Parton E, Maene L, Deroose R, De Proft MP (2002) Pollen tube growth and fertilization after different in vitro pollination techniques of Aechmea fasciata. Euphytica 124:75–83
Vidali L, McKenna ST, Hepler PK (2001) Actin polymerization is essential for pollen tube growth. Mol Biol Cell 12:2534–2545
Walden DB (1993) In vitro pollen germination. In: Freeling M, Walbot V (eds) The maize hand book. Springer-Verlag, New York, p 723–724
Welk SM, Millington WF, Rosen WG (1965) Chemotropic activity and the pathway of the pollen tube in lily. Am J Bot 52:774–781
Weterings K, Russell SD (2004) Experimental analysis of the fertilization process. Plant Cell 16:S107–118
Wolters-Arts M, Lush WM, Mariani C (1998) Lipids are required for directional pollen-tube growth. Nature 392:818–821
Zhang HQ, Croes AF (1982) A new medium for pollen germination in vitro. Acta Bot Neerl 31:113–119
Author information
Authors and Affiliations
Corresponding author
Editor information
Rights and permissions
About this chapter
Cite this chapter
Higashiyama, T., Inatsugi, R. Comparative Analysis of Biological Models used in the Study of Pollen Tube Growth. In: Malhó, R. (eds) The Pollen Tube. Plant Cell Monographs, vol 3. Springer, Berlin, Heidelberg. https://doi.org/10.1007/7089_053
Download citation
DOI: https://doi.org/10.1007/7089_053
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-31121-8
Online ISBN: 978-3-540-34276-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)