Abstract
Cardiovascular diseases are the leading causes of morbidity and mortality in industrialized countries worldwide, despite highly effective preventive treatments available. As a difference continues to exist between the estimated and true number of events, further improvement of risk stratification is an essential part of cardiovascular research.
Among hypertensive patients measurement of arterial stiffness parameters, like carotid-femoral pulse wave velocity (cfPWV) or brachial-ankle pulse wave velocity (baPWV) can contribute to the identification of high-risk subpopulation of patients. This is a hot topic of vascular research including the possibility of the non-invasive measurement of central hemodynamics, wave reflections and recently, 24-h arterial stiffness monitoring as well. This chapter discusses the past and the present of this area including the scientific achievements with cfPWV, baPWV and other measures, provides a short overview of methodologies and the representation of arterial stiffness parameters in guidelines.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Hypertension has many aspects and a wild range of medical professions are involved in its research and treatment, like cardiology, angiology, endocrinology, nephrology, neurology or psychiatry. In this chapter we focus on the arteries, especially their mechanical properties, that can be non-invasively measured with the identification and analysis of pulse wave curves leading to the evaluation of arterial stiffness, wave reflection and central hemodynamic parameters. The spread of this research area and its implantation into clinical practice might lead to the development of a new profession, which could be called “arteriology”.
The palpation of the pulse is a fundamental part of physical examination since the early ages of the Greek and Chinese medicine (Kuriyama 1999). The first European milestone of vascular research is dated back to 1628, when William Harvey described the basics of circulation in his classical text “de Motu Cordis…” (Harvey 1628; O’Rourke et al. 2001). Pulse wave analysis is rooted in the nineteenth century, when after the theoretical basis of Marey (Marey 1860), Frederick Akbar Mahomed developed sphygmograph, described normal radial pressure waveform and demonstrated differences from carotid wave (Mahomed 1872). In the middle of the twentieth century McDonald enlightened that this difference is based on wave reflection (McDonald 1960) and Womersley introduced transfer functions to characterize vascular beds in the frequency domain, an invention, that led to the development of the modern pulse wave analysis (O’Rourke et al. 2001; Womersley 1957). The history of arterial stiffness measurement begins in 1985, when Levy, Targett and their co-workers described in two articles the first device and computer program to automatically record and calculate pulse wave velocity (PWV) (Levy et al. 1985; Targett et al. 1985).
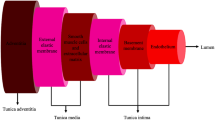
The shape of pulse wave curves is changing with age-associated arterial stiffening due to increased wave reflections, as it is demonstrated in Fig. 1. The pathopysiological background of this phenomenon is complex. It is thought, that with ageing, the chronic cyclical stress on the wall of large arteries leads to elastin fracturing and thinning, which is accelerated in the presence of hypertension (Nichols and O’Rourke 2005). Since in adulthood the production of elastin is not possible, this process leads to the irreversible change of the elastin/collagen ratio, which causes the stiffening of the large arteries (Powell et al. 1992). Among the autocrine, paracrine and neuroendocrine effects leading to an increase in arterial stiffening, the role of the renin-angiotensin-aldosterone system have been extensively studied. Its activation stimulate multiple inflammatory pathways, such as tissue growth factor-β and NF-kB, promoting reactive oxygen species production with reduction in nitric oxide bioavailability (Usui et al. 2000; Gibbons et al. 1992; Fok and Cruickshank 2015). In hypertension, systemic arterial compliance was strongly and negatively correlated with plasma aldosterone level (Blacher et al. 1997). Other deleterious effects of inflammatory processes are the impair of the balance between the production of proteases and their inhibitors and the promotion of the synthesis of advanced glycation end-products (AGEs). Matrix metalloproteases are proteases that are responsible for the accelerated breakdown of elastin and destruction of the molecular folding of collagen. AGEs promote the irreversible cross-linking of collagen, which together with overexpression of MMPs, eventuate in a stiff extracellular matrix (Fok and Cruickshank 2015). Sodium is also an important player in the process of arterial stiffening. High sodium concentration itself leads to the hypertrophy of vascular smooth muscle cells (Gu et al. 1998). In sodium-sensitive, borderline-hypertensive patients large artery compliance was found reduced compared to age-matched sodium-resistant subjects which suggests alterations in the viscoelastic properties of arterial wall characteristics in sodium-sensitive patients (Draaijer et al. 1993).
It remains a conundrum, whether arterial stiffening is a cause or a consequence of hypertension? It was a widely accepted belief, that increased arterial stiffness is a consequence of hypertension. But in contrast to this dogma, in treated hypertensive patients baseline arterial stiffness measures were found to be associated with longitudinal increases of systolic blood pressure, mean arterial pressure and pulse pressure (Coutinho et al. 2014). Moreover, in an analysis of the Framingham Heart Study higher arterial stiffness was associated with blood pressure progression and incident hypertension 7 years later, but higher blood pressure at the initial examination was not associated with progressive arterial stiffening (Kaess et al. 2012).
What is might mostly approaching the truth is, that arterial stiffening is both a cause and a consequence of hypertension in a sort of vicious circle. Elastic and muscular type arteries adapt differently to hemodynamic changes caused by the left ventricule construction. The ability of large and elastic arteries to accommodate for the nonlinear pressure-volume changes caused by stroke volume (conduit and Windkessel function) can be characterized by several functional and structural parameters, especially compliance, distensibility, that provides information about the extrinsic features of the arteries, meanwhile i.e. elastic modulus (Young’s) describe the intrinsic elastic properties of the vessel walls. The conduit and Windkessel functions of the aorta balance the effect of pressure-volume overload that generate central pulse pressure. It transfers toward the peripheral muscular vessels those of smaller inner diameter, thinner and stiffer vessel wall (London and Pannier 2010). The altered geometry and structure of muscular type arteries protect small vessels against higher pulse pressure from the central arteries that is called phenomenon of impedance mismatch. During aging the elasticity of the arterial wall decreases and become stiffer. Thus after a prime age small vessels can not be protected against the pressure load, that lead to increased shear stress and endothelial damage in the periphery. Physiologically, the transfer of pulse pressure is displayed as pulse wave that is reflected back from the points of vessel junctions and reach the heart at the diastolic phase. Stiffer arteries – transition from elastic to muscular type arteries and that of resistance arteries – can reflect the pulse wave earlier, thus the augmented pressure approach the heart cycle during the systolic phase causing pressure burden on the left ventricle.
Structural damage and functional deterioration of arteries reinforce the hemodynamic vicious circle of stiffness and high blood pressure, while highlighting the chicken and the egg debate of the possible origin of hypertension per se. These findings also underscore the importance of the better understanding of the pathophysiology of arterial stiffening, with the hope of providing a new potential targets for the prevention of hypertension.
2 Aortic and Carotid-Femoral Pulse Wave Velocity
The most accepted and wildly used arterial stiffness parameter is pulse wave velocity (PWV), derived from less flow velocity, but rather diameter and pressure waveforms recorded at different points of the arterial tree. PWV, as the Bramwell-Hill equation demonstrates, is a functional measurement of distensibility, which is defined by a volume change in proportion to a change in pressure relative to the initial volume (Bramwell and Hill 1922):
where PWV is the pulse wave velocity, V is the volume, P is the pressure and ρ is the blood density. Consequently, PWV is closely related to changes in volume and arterial pressure as well. Arterial wall properties are also important in determining PWV, and these are described by the Moens-Kroteweg equation (Nichols and O’Rourke 2005):
where PWV is the pulse wave velocity, E inc is the Young’s elastic modulus of arterial wall (a measure of the arterial wall mechanical properties), h is the arterial wall thickness, ρ is the blood density and D is the vessel diameter. An increase in wall stiffness (E inc ) and/or in wall thickness is accompanied with an increase in PWV and arterial calibre is inversely proportional to PWV. Taken together, arterial stiffness can be modulated directly by the changes in vascular tone, arterial wall mechanical properties and thickness and also indirectly, by changes in blood pressure (Fok and Cruickshank 2015).
As the thoracic and abdominal aorta gives the largest contribution to the arterial buffering function (Isnard et al. 1989), aortic PWV (aPWV) is an arterial stiffness parameter of high priority. As the exact evaluation of aPWV requires invasive intervention or MRI, methodologies with limited possibility of involvement into epidemiological studies, surrogate methodologies are used for approximations of aPWV. Among them, carotid-femoral PWV (cfPWV), the velocity of pulse as it travels from the heart to the carotid and to the femoral artery, is the most commonly applied non-invasive method and considered as the “gold standard” measurement of arterial stiffness (Laurent et al. 2006). cfPWV is usually evaluated using the “foot-to-foot” velocity method from a number of waveforms. Surface tonometry probes are usually applied at the right common carotid artery and the right femoral artery. cfPWV is calculated using the following formula:
where cfPWV is carotid-femoral pulse wave velocity, D is the distance between the two recording sites and Dt is the time delay between the “foot” of the carotid and the femoral waveforms. The “foot” of the wave is defined at the end of diastole, when the steep rise of the waveform begins (Vlachopoulos et al. 2015). The unit of cfPWV is meter/second (m/s).
From this formula it is clear, that a crucial point of cfPWV evaluation is the correct measurement of the travelled distance. Different measurement methodologies can lead to different PWV values which can also have different prognostic significances (Nemeth et al. 2011). For a long time there has been no agreement in this field until 2012, when consensus document was published recommending the use of the 80 % of the direct carotid to femoral distance as it provides only 0.4 % difference with MRI-calculated value (Van Bortel et al. 2012). Another important point of view in respect of methodological considerations is the accuracy of distance measurement between the carotid and femoral sites. In women the breast contour and in both sexes obesity can limit the use of tape measure, so it is recommended to apply sliding caliper instead of tape (Bossuyt et al. 2013).
Several devices are commercially available to measure directly cfPWV. The first was the Complior System (Alam Medical, Vincennes, France), which is based on the simultaneous recording of arterial pulse waves at carotid and femoral sites, through mechanotransducer probes (Asmar et al. 1995). The next was the SphygmoCor system (AtCor, Sydney, Australia), which uses a large-band piezoelectronic probe (applanation tonometer) and records consecutively carotid and femoral arterial pulse waves, with both signals being synchronized to the same ECG R wave (Wilkinson et al. 1998). The PulsePen (Diatecne, Milano, Italy) is based also on applanation tonometry and uses successive carotid and femoral pulse waves synchronized with ECG (Salvi et al. 2004). The Vicorder (Skidmore Medical Limited, Bristol, United Kingdom) is based on oscillometric technique to measure cfPWV through the inflation of a neck pad and a cuff around the thigh (van Leeuwen-Segarceanu et al. 2010). Moreover, cfPWV can also be measured by pulsed Doppler ultrasound with a Linear Array, with ECG gating, as it was demonstrated by Calabia et al. (2011). However, among these devices Complior and SpygmoCor are the most frequently used ones, also in epidemiologic studies.
Before implementation of a novel biomarker into clinical practice the applicant must fulfill different criteria, as Table 1. demonstrates. From this point of view cfPWV fulfill almost all requirements.
A number of studies have proven, that cfPWV is associated with different cardiovascular pathophysiological conditions and has strong prognostic value. Apart from the dominant effect of ageing (McEniery et al. 2005), hypertension was found to be another main contributor to enhanced arterial stiffening (Simon et al. 1985). In uncomplicated essential hypertension the independent predictive value of cfPWV from classic cardiovascular risk factors was clearly demonstrated (Boutouyrie et al. 2002; Laurent et al. 2001, 2003). Moreover, its predictive value was also confirmed in end-stage renal disease (Blacher et al. 1999), in patients after ischemic stroke (Gasecki et al. 2012), in elderly subjects (Meaume et al. 2001; Sutton-Tyrrell et al. 2005) and in the general population (Mattace-Raso et al. 2006). The independent predictive value of cfPWV was even confirmed by a recent meta-analysis, in which Ben-Shlomo et al. nicely demonstrated that after the adjustment of additional risk factors an increase in 1 SD change in log cfPWV is related to 30 %, 28 % and 17 % increase in cardiovascular (CV) events, CV mortality and all-cause mortality, respectively (Ben-Shlomo et al. 2014). In the practical interpretation of the results, for a 60-year-old man who is a non-smoker, non-diabetic, normotensive and normolipemic, a 1 m/s increase in cfPWV leads to a 7 % increase of the hazard for CV events (Ben-Shlomo et al. 2014; Vlachopoulos et al. 2014). The independent association with all-cause mortality suggests that the impact of arterial stiffening extends beyond the diseases of CV system.
The clinical utility of cfPWV measurement was confirmed by two studies and a meta-analysis demonstrating that patients at intermediate risk could be reclassified into a higher or lower CV risk category when cfPWV is measured (Mattace-Raso et al. 2006; Sehestedt et al. 2009; Mitchell et al. 2010). In the Framingham study for individuals at intermediate CV risk, addition of cfPWV resulted in upward reclassification of 14.3 % of participants who experienced a CV event and downward reclassification of 1.4 % of participants who did not experience a CV event, yielding a net reclassification of 15.7 % (Mitchell et al. 2010). Based on the above-mentioned results it is obvious, the cfPWV fulfills the requirements of the 1–4 points of Table 1.
According to point 5., the influence of cfPWV modification for clinical outcome, there is only one study available so far, in which in patients with end-stage renal disease the lack of the decrease of cfPWV in response to blood pressure medication was a predictor of all-cause and cardiovascular mortality (Guerin et al. 2001). A randomized clinical trial called Statégie de Prévention Cardiovasculaire Basée sur la Rigidité Arterielle Study (SPARTE) was started in 2012 in 40 French clinical centres with the planned involvement of 3000 hypertensive patients and with the follow-up period of 4 years, aiming to test the hypothesis that a therapeutic strategy that targets the normalization of arterial stiffness is more effective in preventing CV events than usual care. In the control group, the target is blood pressure, while in the active group the target is aortic stiffness with the aim of normalizing it to <10 m/s (Laurent et al. 2012). Without the result of this and other future trials with similar setups, there is a lack of evidence on broader patient population that the normalization of cfPWV or any other arterial stiffness parameter has positive impact on CV outcome above that of reaching the blood pressure target values.
No data are currently available about the cost-effectiveness of cfPWV measurement. Potentially, the spread of the technology could reduce device prices, however no marked reduction was observed in the latest years. The main expense is the salary of the examiner, which geographically can differ markedly. Considering the high reclassification rate of patients and the comparison of cost with other powerful, but costly biomarkers, like coronary calcium score, cfPWV is probably a cost-effective risk stratification methodology.
The accurate and reproducible measurement of cfPWV requires moderate expertise. It is easy in most of the cases, but obesity and picnic stature may render measurements challenging. A true disadvantage of cfPWV measurement is the manipulation in the inguinal region which can be uncomfortable for some patients.
An expert consensus document on the measurement of aortic stiffness on the daily practice using cfPWV is available involving such scientific communities, like the Artery Society, the European Society of Hypertension and the European Network for Noninvasive Investigation of Large Arteries (Van Bortel et al. 2012).
Reference values of cfPWV have been established in 2010 involving 1455 healthy subjects together with 11,092 patients with different CV risk factors (Arterial Stiffness’ Collaboration 2010). However, this paper was published before the consensus document on distance measurement (Van Bortel et al. 2012), but cfPWV values were calculated according to the later accepted 80 % of the direct carotid-femoral distance. Table 2. demonstrates the distribution of cfPWV according to the age category in apparently healthy population.
As a consequence that cfPWV fulfills almost all the nine requirements of Table 1 to be an accepted biomarker, it has already been involved into some guidelines. It was first recommended as a marker of subclinical organ damage with the value >12 m/s in 2007, in the guideline of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC), for the management of arterial hypertension (Mancia et al. 2007). In the next ESH-ESC hypertension guideline in 2013 cfPWV is recommended to be evaluated as a measure of asymptomatic organ damage with the strength of class IIa, level of evidence B (Mancia et al. 2013). In the recent ESC- Artery Society position paper, the evaluation of cfPWV as a vascular biomarker is recommended as a class IIa, level of evidence A method (Vlachopoulos et al. 2015). Pulse wave velocity measurement is also recommended in the recent ESC- European Association for the Study of Diabetes guideline, as a useful cardiovascular marker, adding predictive value to the usual risk estimate (Ryden et al. 2013). In 2010, in the American College of Cardiology Foundation (ACCF)/American Heart Association (AHA) task force document the measurement of cfPWV was not recommended to be used in asymptomatic adults, outside of research settings (Greenland et al. 2010) and cfPWV measurement was even not mentioned in the next ACC/AHA guideline for the assessment of cardiovascular risk (Goff et al. 2014). However, in a recently published AHA scientific statement it is declared that it is reasonable to measure arterial stiffness to provide incremental information beyond standard CV disease risk factors in the prediction of future CV disease events (class: IIa, level of evidence: A) (Townsend et al. 2015). With this statement document the position of cfPWV measurement in the cardiovascular risk stratification has become similar both in Europe and in America.
3 Oscillometric Approximations of Aortic Pulse Wave Velocity
As cfPWV measurement is partly operator-dependent and the manipulation in the inguinal region can cause discomfort for the subject, oscillometric approximations of aortic PWV through a brachial cuff can have perspectives in the future. Mobil-o-Graph (Wassertheurer et al. 2010), Arteriograph (Horvath et al. 2010) and Vasotens (Ageenkova and Purygina 2011) are such devices, and all of them are able per se or have versions developed for 24-h blood pressure and arterial stiffness monitoring as well (Laszlo et al. 2015). So far none of these new techniques is involved in the recommendations as an alternative of cfPWV (Vlachopoulos et al. 2015; Mancia et al. 2013; Townsend et al. 2015). As in case of these new methodologies in validation studies the determination coefficients (R2) in comparison with gold-standard methods are mostly between 0.4 and 0.7, which reflect very imperfect agreement, experts do not recommend their involvement into prospective studies (Boutouyrie et al. 2014a). This fact can lead to a catch-22, as a manufacturer company per se rarely have enough funds to perform large population-based investigations. A solution for this discrepancy were the use of gold-standard and oscillometric methods parallel in prospective studies, which could provide both answers for clinical questions and validations of these alternative methodologies.
4 Brachial-Ankle Pulse Wave Velocity and Other Promising Parameters
In this paragraph we would like to provide an overview of an arterial stiffness parameter which is widely accepted and used in Japan and China and the cumulating data with this methodology also enabled its involvement into guidelines. Brachial-ankle pulse wave velocity (baPWV) is a simple-to-assess stiffness marker of the large and middle-size arteries. It is measured with a volume-plethysmographic device (eg VP1000, VP2000, OMRON Health Care Co. Ltd., Kyota, Japan) using four cuffs placed on both arms (brachial) and ankles, connected to plethysmographic and oscillometric sensors, recording the brachial and posterior tibial pressure waveforms (Vlachopoulos et al. 2015; Tomiyama et al. 2006). Travel distance is calculated using the path lengths from the suprasternal notch to the brachium (Lb) and from the suprasternal notch to the ankle (La) with a correction for the height of the individual using validated equations. baPWV is calculated with the following equation:
where \( \varDelta T b a \) is the time interval between the wavefront of the brachial waveform and that of the ankle waveform (Vlachopoulos et al. 2015; Tomiyama et al. 2006).
It has been demonstrated, that baPWV is closely correlated with cfPWV and invasively assessed aortic PWV and the presence of CV risk factors is linked with its elevated value (Yamashina et al. 2002, 2003; Tomiyama and Yamashina 2010). Prospective studies have confirmed that baPWV is a useful predictor of future CV events not only in essential hypertension (Munakata et al. 2012), but also in general population (Ninomiya et al. 2013), in end-stage renal disease (Kitahara et al. 2005), in diabetes (Maeda et al. 2014), in patients with acute coronary syndrome (Tomiyama et al. 2005) and heart failure (Meguro et al. 2009). In a meta-analysis it was demonstrated that a 1 m/s increase in baPWV corresponds with an increase of 12 %, 13 % and 6 % in total cardiovascular events, cardiovascular mortality and all-cause mortality, respectively (Vlachopoulos et al. 2012). It seems, that an optimal cutoff value of baPWV is 18 m/s in the assessment of high risk for CV disease (Ninomiya et al. 2013; Tomiyama et al. 2005). Moreover, as baPWV can predict the development of hypertension or stage III chronic kidney disease (Yambe et al. 2007; Takase et al. 2011; Chen et al. 2011; Tomiyama et al. 2010), in healthy subjects under the age of 60, and baPWV values between 14 and 18 m/s lifestyle modifications are recommended by some experts of this field (Yamashina et al. 2003; Tomiyama et al. 2016).
Although lots of achievements have been succeeded in respect of baPWV to be an accepted CV biomarker, but there are still some incomplete requirements. No consensus document is available on the measurement methodology, only the manufacturer’s instructions. Reference values have been published only in Chinese populations (Ai et al. 2011; Wang et al. 2009), data are missing in Caucasian or other races. So far, the potential clinical advantage of baPWV over traditional risk scores have not been proven. Although it has been demonstrated that baPWV improves for the treatment of hypertension, dyslipidemia, diabetes or lifestyle modifications (Toyama et al. 2012, 2016), only one study reported so far, that improvement of baPWV obtained after 6 months of conventional therapy was a reliable marker of a better prognosis in patients with coronary artery disease (Orlova et al. 2010).
Based on these findings, in the recent ESC position paper on vascular biomarkers baPWV is recommended for primary and secondary CV disease prevention with the class of IIb, level of evidence B (Vlachopoulos et al. 2015). The recent AHA recommendation states that baPWV is useful in cardiovascular outcome predictions in Asian populations, but longitudinal studies in the United States and Europe by these methods are lacking (Class I; Level of Evidence B) (Townsend et al. 2015).
Unfortunately the limited extent of this book chapter does not permit the detailed description of other parameters that also can have future perspectives. Parameters of pulse wave analysis and central hemodynamics can be estimated alone or connected with cfPWV measurement. Other parameters can be evaluated with specific devices. Some of these measures are already mentioned in the recommendations. The recent ESC/ESH hypertension guideline states that augmentation index and central blood pressure can be helpful risk stratification tools in young patients with isolated systolic hypertension, however, more data are needed before central hemodynamic indices are recommended for routine use in hypertensive patients in general (Mancia et al. 2013). In the recent ESC biomarker position paper the usefulness of the measurement of central hemodynamics/wave reflections for primary and secondary CV disease prevention is judged as IIb/B (recommendation/level of evidence) (Vlachopoulos et al. 2015). The AHA scientific statement declares that the use of wave separation analysis is recommended when investigations are focused specifically on the role of wave reflection as either an exposure for CV outcome or a target for intervention (class I, level of evidence B). The same document states that similarly to baPWV, the measurement of cardiac ankle vascular stiffness index is useful in CV outcome prediction in Asian populations, but longitudinal studies in the United States and Europe are lacking (class I, level of evidence B) (Townsend et al. 2015). The measurement of carotid stiffness parameters is also promising. A recent meta-analysis demonstrated that greater carotid stiffness is associated with a higher incidence of stroke independently of cfPWV and modestly improved risk prediction of stroke beyond Framingham stroke risk score factors and cfPWV (van Sloten et al. 2015). In summary, this research area is far not limited only for cfPWV or baPWV, the complexity is growing with new candidates with further potential in helping risk stratification and individual therapy adjustment.
5 How Can We Improve the Arterial Stiffness of Our Patients?
Many non-pharmacological interventions can improve arterial stiffness and/or wave reflection, like dietary changing including weight loss and salt reduction (Pase et al. 2011; Dengo et al. 2010), aerobic exercise training (Edwards et al. 2004; Edwards and Lang 2005), passive vibration (Sanchez-Gonzalez et al. 2012) and enhanced external counterpulsation treatment (Nichols et al. 2006). For maximal cardiovascular benefits, these interventions must be initially introduced immediately and continued over an extended period of time (Townsend et al. 2015).
In respect of pharmaceutical interventions, it is demonstrated, that different kind of blood pressure medications have beneficial effect for arterial stiffening (Fok and Cruickshank 2015). The reduction of aortic PWV was confirmed with the administration of renin inhibitor (Virdis et al. 2012), angiotensin converting enzyme (ACE) inhibitors/angiotensin AT1 receptor blockers (ARBs) (Mitchell et al. 2002, 2005; Karalliedde et al. 2008; Boutouyrie et al. 2014b), an endothelin-A receptor antagonist (Dhaun et al. 2009), with spironolactone and hydrochlorotiazide monotherapy in elderly (Kithas and Supiano 2010), or with ACE inhibitor/ARBs in combination with spironolactone (Edwards et al. 2009). The problem with these interventions, that the destiffening effect of a blood pressure medication can hardly be divided from the effect of blood pressure reduction per se, however, some authors state that the observed destiffening effect is at least partly independent of blood pressure reduction (Karalliedde et al. 2008; Boutouyrie et al. 2014b; Edwards et al. 2009). One compound with a direct destiffening effect was tested so far, but the advanced glycation end-products crosslink breaker alagebrium after promising initial results (Kass et al. 2001; Zieman et al. 2007) unfortunately did not get through all the clinical pharmacological phases, probably due to the financial problems of the developing company.
Numerous nutritional supplements have been found to improve arterial stiffening like flavonoids (Lilamand et al. 2014), omega-3 and soy isoflavone (Pase et al. 2011) or tetrahydrobiopterin (Moreau et al. 2012; Pierce et al. 2012). As these interventions are also often accompanied with blood pressure reduction, their blood-pressure independent destiffening effect is not unambiguous as well. So the clinical importance of blood pressure-independent destiffening is still a pending question and such studies like the above-mentioned SPARTE (Laurent et al. 2012) are needed to give us answers.
6 Conclusions, Future Directions
In hypertensive patients the measurement of arterial stiffening, especially cfPWV is already a recommended method to detect target organ damage both in Europe and in America. Its role in risk stratification seems to be clarified, but the potential benefit in cardiovascular outcome from treating hypertensive patients until a certain cfPWV goal value is not confirmed yet. Besides cfPWV, other measures of arterial stiffness are getting closer to be recommended in clinical use, like baPWV, or different wave reflection parameters. Recently devices measuring 24-h ambulatory arterial stiffness have become available on the market, opening a new field of research interest. These findings confirm that the study of large artery structure and function is becoming an essential part of hypertension care.
References
Ageenkova OA, Purygina MA (2011) Central aortic blood pressure, augmentation index, and reflected wave transit time: reproducibility and repeatability of data obtained by oscillometry. Vasc Health Risk Manag 7:649–656, Pubmed Central PMCID: PMC3225346, Epub 2011/12/06. eng
Ai ZS, Li J, Liu ZM, Fan HM, Zhang DF, Zhu Y et al (2011) Reference value of brachial-ankle pulse wave velocity for the eastern Chinese population and potential influencing factors. Braz J Med Biol Res = Revista brasileira de pesquisas medicas e biologicas/Sociedade Brasileira de Biofisica [et al] 44(10):1000–1005, Epub 2011/08/31. eng
Arterial Stiffness’ Collaboration (2010) Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘establishing normal and reference values’. Eur Heart J 31(19):2338–2350, Pubmed Central PMCID: PMC2948201. Epub 2010/06/10. eng
Asmar R, Benetos A, Topouchian J, Laurent P, Pannier B, Brisac AM et al (1995) Assessment of arterial distensibility by automatic pulse wave velocity measurement. Validation and clinical application studies. Hypertension 26(3):485–490, Epub 1995/09/01. eng
Ben-Shlomo Y, Spears M, Boustred C, May M, Anderson SG, Benjamin EJ et al (2014) Aortic pulse wave velocity improves cardiovascular event prediction: an individual participant meta-analysis of prospective observational data from 17,635 subjects. J Am Coll Cardiol 63(7):636–646, Epub 2013/11/19. eng
Blacher J, Amah G, Girerd X, Kheder A, Ben Mais H, London GM et al (1997) Association between increased plasma levels of aldosterone and decreased systemic arterial compliance in subjects with essential hypertension. Am J Hypertens 10(12 Pt 1):1326–1334, Epub 1998/01/27. eng
Blacher J, Guerin AP, Pannier B, Marchais SJ, Safar ME, London GM (1999) Impact of aortic stiffness on survival in end-stage renal disease. Circulation 99(18):2434–2439, Epub 1999/05/11. eng
Bossuyt J, Van De Velde S, Azermai M, Vermeersch SJ, De Backer TL, Devos DG et al (2013) Noninvasive assessment of carotid-femoral pulse wave velocity: the influence of body side and body contours. J Hypertens 31(5):946–951, Epub 2013/03/21. eng
Boutouyrie P, Tropeano AI, Asmar R, Gautier I, Benetos A, Lacolley P et al (2002) Aortic stiffness is an independent predictor of primary coronary events in hypertensive patients: a longitudinal study. Hypertension 39(1):10–15, Epub 2002/01/19. eng
Boutouyrie P, Fliser D, Goldsmith D, Covic A, Wiecek A, Ortiz A et al (2014a) Assessment of arterial stiffness for clinical and epidemiological studies: methodological considerations for validation and entry into the European Renal and Cardiovascular Medicine registry. Nephrol Dial Transpl 29(2):232–239, Epub 2013/10/03. eng
Boutouyrie P, Beaussier H, Achouba A, Laurent S (2014b) Destiffening effect of valsartan and atenolol: influence of heart rate and blood pressure. J Hypertens 32(1):108–114, Epub 2013/11/28. eng
Bramwell JCH, Hill AV (1922) Velocity of transmission of the pulse wave. Lancet 199(5149):891–892
Calabia J, Torguet P, Garcia M, Garcia I, Martin N, Guasch B et al (2011) Doppler ultrasound in the measurement of pulse wave velocity: agreement with the Complior method. Cardiovasc Ultrasound 9:13, Pubmed Central PMCID: PMC3098145, Epub 2011/04/19. eng
Chen SC, Chang JM, Liu WC, Tsai YC, Tsai JC, Hsu PC et al (2011) Brachial-ankle pulse wave velocity and rate of renal function decline and mortality in chronic kidney disease. Clin J Am Soc Nephrol 6(4):724–732, Pubmed Central PMCID: PMC3069362, Epub 2011/04/02. eng
Coutinho T, Bailey KR, Turner ST, Kullo IJ (2014) Arterial stiffness is associated with increase in blood pressure over time in treated hypertensives. J Am Soc Hypertens 8(6):414–421, Pubmed Central PMCID: PMC4103613, Epub 2014/06/24. eng
Dengo AL, Dennis EA, Orr JS, Marinik EL, Ehrlich E, Davy BM et al (2010) Arterial destiffening with weight loss in overweight and obese middle-aged and older adults. Hypertension 55(4):855–861, Pubmed Central PMCID: PMC2859827, Epub 2010/03/10. eng
Dhaun N, Macintyre IM, Melville V, Lilitkarntakul P, Johnston NR, Goddard J et al (2009) Blood pressure-independent reduction in proteinuria and arterial stiffness after acute endothelin-a receptor antagonism in chronic kidney disease. Hypertension 54(1):113–119, Epub 2009/06/10. eng
Draaijer P, Kool MJ, Maessen JM, van Bortel LM, de Leeuw PW, van Hooff JP et al (1993) Vascular distensibility and compliance in salt-sensitive and salt-resistant borderline hypertension. J Hypertens 11(11):1199–1207, Epub 1993/11/01. eng
Edwards DG, Lang JT (2005) Augmentation index and systolic load are lower in competitive endurance athletes. Am J Hypertens 18(5 Pt 1):679–683, Epub 2005/05/11. eng
Edwards DG, Schofield RS, Magyari PM, Nichols WW, Braith RW (2004) Effect of exercise training on central aortic pressure wave reflection in coronary artery disease. Am J Hypertens 17(6):540–543, Epub 2004/06/05. eng
Edwards NC, Steeds RP, Stewart PM, Ferro CJ, Townend JN (2009) Effect of spironolactone on left ventricular mass and aortic stiffness in early-stage chronic kidney disease: a randomized controlled trial. J Am Coll Cardiol 54(6):505–512, Epub 2009/08/01. eng
Fok H, Cruickshank JK (2015) Future treatment of hypertension: shifting the focus from blood pressure lowering to arterial stiffness modulation? Curr Hypertens Rep 17(8):67, Epub 2015/07/15. eng
Gasecki D, Rojek A, Kwarciany M, Kubach M, Boutouyrie P, Nyka W et al (2012) Aortic stiffness predicts functional outcome in patients after ischemic stroke. Stroke J Cereb Circ 43(2):543–544, Epub 2011/11/15. eng
Gibbons GH, Pratt RE, Dzau VJ (1992) Vascular smooth muscle cell hypertrophy vs. hyperplasia. Autocrine transforming growth factor-beta 1 expression determines growth response to angiotensin II. J Clin Invest 90(2):456–461, Pubmed Central PMCID: PMC443121, Epub 1992/08/01. eng
Goff DC Jr, Lloyd-Jones DM, Bennett G, Coady S, D’Agostino RB, Gibbons R et al (2014) 2013 ACC/AHA guideline on the assessment of cardiovascular risk: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 129(25 Suppl 2):S49–S73, Epub 2013/11/14. eng
Greenland P, Alpert JS, Beller GA, Benjamin EJ, Budoff MJ, Fayad ZA et al (2010) 2010 ACCF/AHA guideline for assessment of cardiovascular risk in asymptomatic adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 122(25):e584–e636, Epub 2010/11/26. eng
Gu JW, Anand V, Shek EW, Moore MC, Brady AL, Kelly WC et al (1998) Sodium induces hypertrophy of cultured myocardial myoblasts and vascular smooth muscle cells. Hypertension 31(5):1083–1087, Epub 1998/05/12. eng
Guerin AP, Blacher J, Pannier B, Marchais SJ, Safar ME, London GM (2001) Impact of aortic stiffness attenuation on survival of patients in end-stage renal failure. Circulation 103(7):987–992, Epub 2001/02/22. eng
Harvey W (1628) de Motu Cordis et Sanguinis Animalibus. William Fitzer Frankfurt
Horvath IG, Nemeth A, Lenkey Z, Alessandri N, Tufano F, Kis P et al (2010) Invasive validation of a new oscillometric device (Arteriograph) for measuring augmentation index, central blood pressure and aortic pulse wave velocity. J Hypertens 28(10):2068–2075, Epub 2010/07/24. eng
Isnard RN, Pannier BM, Laurent S, London GM, Diebold B, Safar ME (1989) Pulsatile diameter and elastic modulus of the aortic arch in essential hypertension: a noninvasive study. J Am Coll Cardiol 13(2):399–405, Epub 1989/02/01. eng
Kaess BM, Rong J, Larson MG, Hamburg NM, Vita JA, Levy D et al (2012) Aortic stiffness, blood pressure progression, and incident hypertension. Jama 308(9):875–881, Pubmed Central PMCID: PMC3594687, Epub 2012/09/06. eng
Karalliedde J, Smith A, DeAngelis L, Mirenda V, Kandra A, Botha J et al (2008) Valsartan improves arterial stiffness in type 2 diabetes independently of blood pressure lowering. Hypertension 51(6):1617–1623, Epub 2008/04/23. eng
Kass DA, Shapiro EP, Kawaguchi M, Capriotti AR, Scuteri A, de Groof RC et al (2001) Improved arterial compliance by a novel advanced glycation end-product crosslink breaker. Circulation 104(13):1464–1470, Epub 2001/09/26. eng
Kitahara T, Ono K, Tsuchida A, Kawai H, Shinohara M, Ishii Y et al (2005) Impact of brachial-ankle pulse wave velocity and ankle-brachial blood pressure index on mortality in hemodialysis patients. Am J Kidney Dis 46(4):688–696, Epub 2005/09/27. eng
Kithas PA, Supiano MA (2010) Spironolactone and hydrochlorothiazide decrease vascular stiffness and blood pressure in geriatric hypertension. J Am Geriatr Soc 58(7):1327–1332, Pubmed Central PMCID: PMC3064882, Epub 2010/06/11. eng
Kuriyama S (1999) The expressiveness of the body and the divergence of Greek and Chinese medicine. Zone Books, New York
Laszlo A, Reusz G, Nemcsik J (2015) Ambulatory arterial stiffness in chronic kidney disease: a methodological review. Hypertens Res 3, Epub 2015/12/04. Eng
Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L et al (2001) Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 37(5):1236–1241, Epub 2001/05/23. eng
Laurent S, Katsahian S, Fassot C, Tropeano AI, Gautier I, Laloux B et al (2003) Aortic stiffness is an independent predictor of fatal stroke in essential hypertension. Stroke J Cereb Circ 34(5):1203–1206, Epub 2003/04/05. eng
Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D et al (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27(21):2588–2605, Epub 2006/09/27. eng
Laurent S, Briet M, Boutouyrie P (2012) Arterial stiffness as surrogate end point: needed clinical trials. Hypertension 60(2):518–522, Epub 2012/06/27. eng
Levy B, Targett RC, Bardou A, McIlroy MB (1985) Quantitative ascending aortic Doppler blood velocity in normal human subjects. Cardiovasc Res 19(7):383–393, Epub 1985/07/01. eng
Lilamand M, Kelaiditi E, Guyonnet S, Antonelli Incalzi R, Raynaud-Simon A, Vellas B et al (2014) Flavonoids and arterial stiffness: promising perspectives. Nutr Metab Cardiovasc Dis 24(7):698–704, Epub 2014/03/25. eng
London GM, Pannier B (2010) Arterial functions: how to interpret the complex physiology. Nephrol Dial Transpl 25(12):3815–3823, Epub 2010/10/16. eng
Maeda Y, Inoguchi T, Etoh E, Kodama Y, Sasaki S, Sonoda N et al (2014) Brachial-ankle pulse wave velocity predicts all-cause mortality and cardiovascular events in patients with diabetes: the Kyushu Prevention Study of Atherosclerosis. Diabetes Care 37(8):2383–2390, Epub 2014/06/06. eng
Mahomed F (1872) The physiology and clinical use of the sphygmograph. Med Times Gazette 1:62
Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G et al (2007) 2007 Guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 28(12):1462–1536, Epub 2007/06/15. eng
Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M et al (2013) 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 34(28):2159–2219, Epub 2013/06/19. eng
Marey E (1860) Recherches sur le pouls an moyen d’un nouvel appareil enregisteur: le sphygmographe. E Thunot et Cie, Paris
Mattace-Raso FU, van der Cammen TJ, Hofman A, van Popele NM, Bos ML, Schalekamp MA et al (2006) Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation 113(5):657–663, Epub 2006/02/08. eng
McDonald D (1960) Blood flow in arteries. Edward Arnold, London
McEniery CM, Yasmin, Hall IR, Qasem A, Wilkinson IB, Cockcroft JR (2005) Normal vascular aging: differential effects on wave reflection and aortic pulse wave velocity: the Anglo-Cardiff Collaborative Trial (ACCT). J Am Coll Cardiol 46(9):1753–1760, Epub 2005/11/01. eng
Meaume S, Benetos A, Henry OF, Rudnichi A, Safar ME (2001) Aortic pulse wave velocity predicts cardiovascular mortality in subjects >70 years of age. Arterioscler Thromb Vasc Biol 21(12):2046–2050, Epub 2001/12/18. eng
Meguro T, Nagatomo Y, Nagae A, Seki C, Kondou N, Shibata M et al (2009) Elevated arterial stiffness evaluated by brachial-ankle pulse wave velocity is deleterious for the prognosis of patients with heart failure. Circ J 73(4):673–680, Epub 2009/02/28. eng
Mitchell GF, Izzo JL Jr, Lacourciere Y, Ouellet JP, Neutel J, Qian C et al (2002) Omapatrilat reduces pulse pressure and proximal aortic stiffness in patients with systolic hypertension: results of the conduit hemodynamics of omapatrilat international research study. Circulation 105(25):2955–2961, Epub 2002/06/26. eng
Mitchell GF, Lacourciere Y, Arnold JM, Dunlap ME, Conlin PR, Izzo JL Jr (2005) Changes in aortic stiffness and augmentation index after acute converting enzyme or vasopeptidase inhibition. Hypertension 46(5):1111–1117, Epub 2005/10/19. eng
Mitchell GF, Hwang SJ, Vasan RS, Larson MG, Pencina MJ, Hamburg NM et al (2010) Arterial stiffness and cardiovascular events: the Framingham Heart Study. Circulation 121(4):505–511, Pubmed Central PMCID: PMC2836717, Epub 2010/01/20. eng
Moreau KL, Meditz A, Deane KD, Kohrt WM (2012) Tetrahydrobiopterin improves endothelial function and decreases arterial stiffness in estrogen-deficient postmenopausal women. Am J Phys Heart Circ Phys 302(5):H1211–H1218, Pubmed Central PMCID: PMC3311456, Epub 2012/01/17. eng
Munakata M, Konno S, Miura Y, Yoshinaga K (2012) Prognostic significance of the brachial-ankle pulse wave velocity in patients with essential hypertension: final results of the J-TOPP study. Hypertens Res 35(8):839–842, Epub 2012/04/27. eng
Nemeth ZK, Studinger P, Kiss I, Othmane Tel H, Nemcsik J, Fekete BC et al (2011) The method of distance measurement and torso length influences the relationship of pulse wave velocity to cardiovascular mortality. Am J Hypertens 24(2):155–161, Epub 2010/11/06. eng
Nichols WW, O’Rourke MF (2005) McDonald’s blood flow in arteries: theoretical, experimental and clinical applications, 5th edn. Hodder Arnold, London
Nichols WW, Estrada JC, Braith RW, Owens K, Conti CR (2006) Enhanced external counterpulsation treatment improves arterial wall properties and wave reflection characteristics in patients with refractory angina. J Am Coll Cardiol 48(6):1208–1214, Epub 2006/09/19. eng
Ninomiya T, Kojima I, Doi Y, Fukuhara M, Hirakawa Y, Hata J et al (2013) Brachial-ankle pulse wave velocity predicts the development of cardiovascular disease in a general Japanese population: the Hisayama Study. J Hypertens 31(3):477–483, discussion 83. Epub 2013/04/26. eng
O’Rourke MF, Pauca A, Jiang XJ (2001) Pulse wave analysis. Br J Clin Pharmacol 51(6):507–522, Pubmed Central PMCID: PMC2014492, Epub 2001/06/26. eng
Orlova IA, Nuraliev EY, Yarovaya EB, Ageev FT (2010) Prognostic value of changes in arterial stiffness in men with coronary artery disease. Vasc Health Risk Manag 6:1015–1021, Pubmed Central PMCID: PMC2988619, Epub 2010/12/04. eng
Pase MP, Grima NA, Sarris J (2011) The effects of dietary and nutrient interventions on arterial stiffness: a systematic review. Am J Clin Nutr 93(2):446–454, Epub 2010/12/15. eng
Pierce GL, Jablonski KL, Walker AE, Seibert SM, DeVan AE, Black SM et al (2012) Tetrahydrobiopterin supplementation enhances carotid artery compliance in healthy older men: a pilot study. Am J Hypertens 25(10):1050–1054, Pubmed Central PMCID: PMC3482981, Epub 2012/06/08. eng
Powell JT, Vine N, Crossman M (1992) On the accumulation of D-aspartate in elastin and other proteins of the ageing aorta. Atherosclerosis 97(2–3):201–208, Epub 1992/12/01. eng
Ryden L, Grant PJ, Anker SD, Berne C, Cosentino F, Danchin N et al (2013) ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD: the Task Force on diabetes, pre-diabetes, and cardiovascular diseases of the European Society of Cardiology (ESC) and developed in collaboration with the European Association for the Study of Diabetes (EASD). Eur Heart J 34(39):3035–3087, Epub 2013/09/03. eng
Salvi P, Lio G, Labat C, Ricci E, Pannier B, Benetos A (2004) Validation of a new non-invasive portable tonometer for determining arterial pressure wave and pulse wave velocity: the PulsePen device. J Hypertens 22(12):2285–2293, Epub 2004/12/23. eng
Sanchez-Gonzalez MA, Wong A, Vicil F, Gil R, Park SY, Figueroa A (2012) Impact of passive vibration on pressure pulse wave characteristics. J Hum Hypertens 26(10):610–615, Epub 2011/06/24. eng
Sehestedt T, Jeppesen J, Hansen TW, Rasmussen S, Wachtell K, Ibsen H et al (2009) Risk stratification with the risk chart from the European Society of Hypertension compared with SCORE in the general population. J Hypertens 27(12):2351–2357, Epub 2009/11/17. eng
Simon AC, Levenson J, Bouthier J, Safar ME, Avolio AP (1985) Evidence of early degenerative changes in large arteries in human essential hypertension. Hypertension 7(5):675–680, Epub 1985/09/01. eng
Sutton-Tyrrell K, Najjar SS, Boudreau RM, Venkitachalam L, Kupelian V, Simonsick EM et al (2005) Elevated aortic pulse wave velocity, a marker of arterial stiffness, predicts cardiovascular events in well-functioning older adults. Circulation 111(25):3384–3390, Epub 2005/06/22. eng
Takase H, Dohi Y, Toriyama T, Okado T, Tanaka S, Sonoda H et al (2011) Brachial-ankle pulse wave velocity predicts increase in blood pressure and onset of hypertension. Am J Hypertens 24(6):667–673, Epub 2011/02/19. eng
Targett RC, Levy B, Bardou A, McIlroy MB (1985) Simultaneous Doppler blood velocity measurements from aorta and radial artery in normal human subjects. Cardiovasc Res 19(7):394–399, Epub 1985/07/01. eng
Tomiyama H, Yamashina A (2010) Non-invasive vascular function tests: their pathophysiological background and clinical application. Circ J 74(1):24–33, Epub 2009/11/19. eng
Tomiyama H, Koji Y, Yambe M, Shiina K, Motobe K, Yamada J et al (2005) Brachial – ankle pulse wave velocity is a simple and independent predictor of prognosis in patients with acute coronary syndrome. Circ J 69(7):815–822, Epub 2005/07/01. eng
Tomiyama H, Hashimoto H, Hirayama Y, Yambe M, Yamada J, Koji Y et al (2006) Synergistic acceleration of arterial stiffening in the presence of raised blood pressure and raised plasma glucose. Hypertension 47(2):180–188, Epub 2005/12/29. eng
Tomiyama H, Tanaka H, Hashimoto H, Matsumoto C, Odaira M, Yamada J et al (2010) Arterial stiffness and declines in individuals with normal renal function/early chronic kidney disease. Atherosclerosis 212(1):345–350, Epub 2010/07/03. eng
Tomiyama H, Matsumoto C, Shiina K, Brachial-Ankle YA, PWV (2016) Current status and future directions as a useful marker in the management of cardiovascular disease and/or cardiovascular risk factors. J Atheroscler Thromb 23(2):128–146, Epub 2015/11/13. eng
Townsend RR, Wilkinson IB, Schiffrin EL, Avolio AP, Chirinos JA, Cockcroft JR et al (2015) Recommendations for improving and standardizing vascular research on arterial stiffness: a scientific statement from the American Heart Association. Hypertension 66(3):698–722, Pubmed Central PMCID: PMC4587661, Epub 2015/07/15. eng
Toyama K, Sugiyama S, Oka H, Iwasaki Y, Sumida H, Tanaka T et al (2012) Combination treatment of rosuvastatin or atorvastatin, with regular exercise improves arterial wall stiffness in patients with coronary artery disease. PLoS ONE 7(7):e41369, Pubmed Central PMCID: PMC3400658, Epub 2012/07/26. eng
Usui M, Egashira K, Tomita H, Koyanagi M, Katoh M, Shimokawa H et al (2000) Important role of local angiotensin II activity mediated via type 1 receptor in the pathogenesis of cardiovascular inflammatory changes induced by chronic blockade of nitric oxide synthesis in rats. Circulation 101(3):305–310, Epub 2000/01/25. eng
Van Bortel LM, Laurent S, Boutouyrie P, Chowienczyk P, Cruickshank JK, De Backer T et al (2012) Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens 30(3):445–448, Epub 2012/01/27. eng
van Leeuwen-Segarceanu EM, Tromp WF, Bos WJ, Vogels OJ, Groothoff JW, van der Lee JH (2010) Comparison of two instruments measuring carotid-femoral pulse wave velocity: Vicorder versus SphygmoCor. J Hypertens 28(8):1687–1691, Epub 2010/05/26. eng
van Sloten TT, Sedaghat S, Laurent S, London GM, Pannier B, Ikram MA et al (2015) Carotid stiffness is associated with incident stroke: a systematic review and individual participant data meta-analysis. J Am Coll Cardiol 66(19):2116–2125, Epub 2015/11/07. eng
Virdis A, Ghiadoni L, Qasem AA, Lorenzini G, Duranti E, Cartoni G et al (2012) Effect of aliskiren treatment on endothelium-dependent vasodilation and aortic stiffness in essential hypertensive patients. Eur Heart J 33(12):1530–1538, Epub 2012/03/28. eng
Vlachopoulos C, Aznaouridis K, Terentes-Printzios D, Ioakeimidis N, Stefanadis C (2012) Prediction of cardiovascular events and all-cause mortality with brachial-ankle elasticity index: a systematic review and meta-analysis. Hypertension 60(2):556–562, Epub 2012/06/27. eng
Vlachopoulos C, Aznaouridis K, Stefanadis C (2014) Aortic stiffness for cardiovascular risk prediction: just measure it, just do it! J Am Coll Cardiol 63(7):647–649, Epub 2013/11/19. eng
Vlachopoulos C, Xaplanteris P, Aboyans V, Brodmann M, Cifkova R, Cosentino F et al (2015) The role of vascular biomarkers for primary and secondary prevention. A position paper from the European Society of Cardiology Working Group on peripheral circulation: Endorsed by the Association for Research into Arterial Structure and Physiology (ARTERY) Society. Atherosclerosis 241(2):507–532, Epub 2015/06/29. eng
Wang X, Xie J, Zhang LJ, Hu DY, Luo YL, Wang JW (2009) Reference values of brachial-ankle pulse wave velocity for Northern Chinese. Chin Med J 122(18):2103–2106, Epub 2009/09/29. eng
Wassertheurer S, Kropf J, Weber T, van der Giet M, Baulmann J, Ammer M et al (2010) A new oscillometric method for pulse wave analysis: comparison with a common tonometric method. J Hum Hypertens 24(8):498–504, Pubmed Central PMCID: PMC2907506, Epub 2010/03/20. eng
Wilkinson IB, Fuchs SA, Jansen IM, Spratt JC, Murray GD, Cockcroft JR et al (1998) Reproducibility of pulse wave velocity and augmentation index measured by pulse wave analysis. J Hypertens 16(12 Pt 2):2079–2084, Epub 1999/01/14. eng
Womersley J (1957) The mathematical analysis of the arterial circulation in a state of oscillatory motion. Dayton, Ohio: Wright Air Development Center, Technical Report Wade-TR 56–614
Yamashina A, Tomiyama H, Takeda K, Tsuda H, Arai T, Hirose K et al (2002) Validity, reproducibility, and clinical significance of noninvasive brachial-ankle pulse wave velocity measurement. Hypertens Res 25(3):359–364, Epub 2002/07/24. eng
Yamashina A, Tomiyama H, Arai T, Hirose K, Koji Y, Hirayama Y et al (2003) Brachial-ankle pulse wave velocity as a marker of atherosclerotic vascular damage and cardiovascular risk. Hypertens Res 26(8):615–622, Epub 2003/10/22. eng
Yambe M, Tomiyama H, Yamada J, Koji Y, Motobe K, Shiina K et al (2007) Arterial stiffness and progression to hypertension in Japanese male subjects with high normal blood pressure. J Hypertens 25(1):87–93, Epub 2006/12/05. eng
Zieman SJ, Melenovsky V, Clattenburg L, Corretti MC, Capriotti A, Gerstenblith G et al (2007) Advanced glycation endproduct crosslink breaker (alagebrium) improves endothelial function in patients with isolated systolic hypertension. J Hypertens 25(3):577–583, Epub 2007/02/07. eng
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing AG
About this chapter
Cite this chapter
Nemcsik, J., Cseprekál, O., Tislér, A. (2016). Measurement of Arterial Stiffness: A Novel Tool of Risk Stratification in Hypertension. In: Islam, M.S. (eds) Hypertension: from basic research to clinical practice. Advances in Experimental Medicine and Biology(), vol 956. Springer, Cham. https://doi.org/10.1007/5584_2016_78
Download citation
DOI: https://doi.org/10.1007/5584_2016_78
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-44250-1
Online ISBN: 978-3-319-44251-8
eBook Packages: MedicineMedicine (R0)