Abstract
Hospitalized children are often treated with antibiotics. However, 30–75 % of antibiotic treatment in pediatric hospitals is administrated incorrectly or unreasonably. Implementation of Hospital’s Antibiotic Policy (HAP) should improve antibiotic consumption patterns in pediatric wards. The objective of this study was to determine the effectiveness of HAP by assessing antibiotic consumption in the General Pediatric Ward of an academic hospital in the city of Warsaw, Poland before and after this policy was introduced in the years 2012 and 2013, respectively. Antibiotic use was calculated in daily-defined doses (DDDs) per 100 patient-days and DDDs per 100 admissions. Antibiotics were ranked by the volume of DDDs and the number of antibiotics which accounted for 90 % and 100 % of the total volume: DU90% and DU100% (where DU stands for drug use). The total antibiotic consumption and significantly decreased after the implementation of HAP; DDDs were 2,177.5 before and 1,335.4 after implementation of HAP. The number of DDDs/100 patient-days was also lower; 36.3 vs. 24.9 before and after HAP, respectively. After implementation of HAP a decreased use of ceftriaxone and cefuroxime was observed. The most commonly used antibiotic was amoxicillin with clavulanic acid. The DU100% rates remained the same (8 antibiotics) and DU90% increased (from 3 in 2012 to 5 in 2013). We conclude that implementation of HAP resulted a decreased consumption of antibiotics in the General Pediatric Ward, despite the hardly changed number of children treated with antibiotics.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Antibiotics are among the drugs most commonly prescribed for children. Although the vast majority of antibiotics are consumed in primary care, the pressure to opt for antimicrobial drugs in hospitals appears to be even higher than in outpatient care. It has been estimated that 36–49 % of hospitalized children receive antibiotics (Potocki et al. 2003; van Houten et al. 1998). It should also be highlighted that 15–45 % of antibiotic treatment regimens for pediatric patients may be inappropriate (Principi et al. 1981; Schollenberg and Albritton 1980). The frequent use of antibiotics is considered to be the main reason for the high prevalence of antimicrobial resistance, which may also result in side effects and excessive costs (Berild et al. 2008; Shehab et al. 2008; de Man et al. 2000). Therefore, monitoring of antibiotic use in hospitals is important for identifying prescribing trends, linking results with antimicrobial resistance data and identifying areas for improvement (Atti et al. 2008). In 2007, the Infectious Diseases Society of America (IDSA) and the Society for Healthcare Epidemiology of America (SHEA) published guidelines for developing institutional antimicrobial stewardship programs. Initial efforts were focused on the adult patient population but efforts for the widespread implementation of formal pediatrics programs have occurred in recent years. In 2010, the Pediatric Infectious Disease Society (PIDS) and the American Academy of Pediatrics (AAP) recommended the implementation of the Hospital Antibiotic Policy (HAP) for healthcare establishments providing inpatient and outpatient pediatric care (Hyun et al. 2013). To address the growing problem of antibiotic overuse and resistance, the Polish Ministry of Health has also urged all hospitals, including pediatric establishments, to develop systems to monitor antibiotic use and have encouraged physicians to reduce inappropriate use (Hryniewicz and Ozorowski 2011). According to the Centers for Disease Control and Prevention (CDC), antibiotic stewardship strategies should include one or more of the core principles: timely management of antimicrobial therapy, appropriate selection of antimicrobials, appropriate administration and de-escalation of antimicrobial therapy, use of available expertise and resources at the point of care and transparent monitoring of antibiotic use data (CDC 2012). As two of the goals mentioned have already been achieved in our hospital (formation of a multidisciplinary antibiotic stewardship committee and auditing and monitoring of antibiotic use), we decided to evaluate the results of HAP introduction in the General Pediatric Ward in Warsaw, Poland. According to our best knowledge this is the first such analysis in Poland, while the amount of research in this field by central and eastern European countries is also very limited.
2 Methods
Approval from the local Ethics Committee was obtained prior to the study. A retrospective analysis of antibiotic consumption in the General Pediatric Ward before and after implementation of HAP was conducted. The period analyzed was the year 2012 (a year before HAP’s implementation) and the year 2013 (a year after HAP’s implementation). The General Pediatric Ward is a 21-bed primary level unit providing care for children aged 0–18 years in Warsaw, the capital of Poland. The total number of admissions at the General Pediatric Ward was 1083 in 2012 and 1,111 in 2013. The number of patient-days was 5,592 in 2012 and 5,349 in 2013, while the mean length of hospitalization was 5.5 days in 2012 and 4.48 in 2013. The majority of patients hospitalized were infants and children under 3 years of age (Table 1). In 2013, an extensive antibiotic policy program created by the members of the hospital’s infection control team (ICT) was rolled out at the hospital. The HAP was defined as written guidelines for prescribing antibiotics. Antibiotics used in the hospital were divided into three groups: first-line antibiotics (e.g., penicillin, 1st generation cephalosporins), second-line antibiotics (e.g., macrolides, 2nd and 3rd generation cephalosporins, amoxicillin with clavulanic acid, piperacillin with tazobactam), and third-line antibiotics (restricted antibiotics) only available after pre-authorization (or additional authorization) from a physician from the infection control team (e.g. glycopeptides, carbapenems). Data on the quantitative and qualitative use of antibiotics in 2012 and 2013 were reported by the hospital’s pharmacy. The data represented the dispensing of antibiotics from the hospital pharmacy to the General Pediatric Ward. The annual report on the total volume of antibiotics prescribed was analyzed and antibiotic use was calculated in daily-defined doses (DDDs) per 100 patient-days and DDDs per 100 admissions according to the Anatomical Therapeutic Chemical Classification (ATC) System, from the WHO, version 2009. Furthermore, antibiotics were ranked by volume of DDDs and the number of antibiotics, which accounted for 90 % and 100 % of the total volume (DU90% and DU100%, respectively, where DU stands for drug use). For statistical analysis, the chi-square test was used for categorical variables. The medical statistical calculator, available on www.medcalc3000.com, was used.
3 Results
The majority of patients were referred to the hospital by general practitioners or primary care pediatricians due to respiratory tract infections (RTI) (Table 2). Among RTI cases, the most common diagnoses were: pneumonia (82 %), bronchitis (10 %), bronchiolitis (2 %), tonsillitis (1 %), laryngitis (2 %), sinusitis (2 %), and otitis media (1 %). The second biggest cause of admission to the hospital was gastrointestinal infection: gastroenterocolitis of unknown etiology (51 %), gastroenterocolitis of rotavirus or adenovirus etiology (36 %), and gastroenterocolitis of bacterial etiology (13 %). After the implementation of HAP, the proportion of children with RTI treated with antibiotics significantly decreased (91 % vs. 86 %, p < 0.05). The proportions of patients receiving antibiotics for the treatment of other infections (e.g., urinary tract or gastrointestinal infections) remained unchanged (p > 0.05). The total proportion of patients treated with antibiotics was significantly reduced after the implementation of HAP (72 % vs. 65 %, p < 0.05) (Table 2).
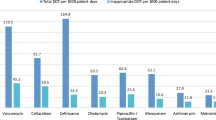
None of the restricted antibiotics (including glycopeptides or monobactams) were used either before or after HAP’s implementation (Table 3). After implementation of HAP, a decreased use of ceftriaxone and cefuroxime was observed. The most commonly used antibiotic was amoxicillin with clavulanic acid (Table 3). The DU100% remained the same before and after implementation of HAP (8 antibiotics), while DU90% increased after implementation of HAP (5 vs. 3 antibiotics) (Table 4).
4 Discussion
An important issue identified in our results is a high proportion of hospitalized children who were treated with antibiotics (65–72 %). This proportion is higher than observed previously in the eastern European countries (36–45 %) (Ang et al. 2008; Potocki et al. 2003; van Houten et al. 1998). However, some researchers recently found even a higher proportion of hospitalized children receiving antimicrobials (57 %) (Levy et al. 2012). Our data are similar to those obtained in pediatric intensive care units (70.8 %) (Blinova et al. 2013; Grohskopf et al. 2005). However, the present study was conducted in the general pediatric ward. The results indicate that Poland is a country with a high antimicrobial usage rate, which has also been reported by others (Rossignoli et al. 2007; Stichele et al. 2006).
We observed that the majority of patients were admitted due to respiratory tract infections. It is well known that a high proportion of pediatric respiratory tract infections are of a viral origin and do not require antibiotic therapy. We presume that antibiotics were overused in our group of patients. Several studies have demonstrated the overuse of antibiotics in clinical situations where antimicrobials are not indicated, including exacerbation of asthma, pharyngitis, viral respiratory infections, and bronchiolitis secondary to syncytial virus infection (de Boeck et al. 2011; Gaur et al. 2005; Stallworth et al. 2005; Benin et al. 2003). To decrease the risk of inappropriate use of antimicrobials, they should be prescribed less often empirically and more often after microbiological examination (Atti et al. 2008).
We reported that just 1 year after the implementation of the HAP, the total antibiotic consumption in the general pediatric ward measured in DDDs decreased. It can be explained by a shorter inpatient treatment and less often use of the combined therapy, i.e., two or three antibiotics in the same patient. This indicates that benefits from the HAP’s implementation can be expected very quickly, improving the quality of care for hospitalized children, preventing the emergence of resistance and lowering drug-related costs (Ohl and Luther 2011; di Pentima et al. 2011). It is also worth pointing out that the proportion of children with respiratory tract infection treated with antimicrobials decreased after HAP’s implementation, which is in agreement with other findings (Papaevangelou et al. 2012). The most commonly used antibiotic in the present study was amoxicillin with clavulanic acid followed by cefuroxime. It has been previously described that these antibiotics are overused in Polish hospitals (Bruce et al. 2009; Stichele et al. 2006; Cars et al. 2001). However, these are first-line antimicrobials in the treatment of community-acquired pneumonia (CAP) in young children according to the Polish recommendations (NPAP 2011). Amoxicillin with clavulanic acid was also found to be the most commonly used antibiotic in other pediatric wards in Europe (Svestina and Mozgis 2014; Atti et al. 2008). High pharmacological effectiveness against most local and systemic infections, a low incidence of side effects, and availability in many suitable dosage forms are the likely reasons behind the preferential prescribing of amoxicillin with clavulanic acid (Katakam et al. 2012). According to the Pediatric Infectious Disease Society of America (PIDS), the first-line antibiotics in the hospital treatment of uncomplicated CAP in fully-immunized children without any underlying medical conditions are ampicillin, macrolides, and beta-lactams when atypical pneumonia is suspected (Bradley et al. 2011). These differences in recommendations may be explained by different national immunization schedules (for example, in Poland the pneumococcal vaccination is not a universal one). After implementation of HAP in the general pediatric ward we observed a decreasing trend in the use of third generation cephalosporins which is in agreement with the national recommendations and the findings of others (di Pentima et al. 2011).
It should be noted that the number of antibiotics in the DU100% segment remained the same before and after HAP’s implementation (8 antibiotics). Interestingly, the number of antibiotics in the DU100% (8) and in the DU90% (3–5) was relatively low and comparable with Dutch data (Liem et al. 2010), but lower than reported in China (between 16 and 20) (Zhang et al. 2008), Russia (8, out of a total of 22 antibiotics used) (Hajdu et al. 2007), and Croatia (11, out of a total of 35) (Palcevski et al. 2004). The DU90% rate increased after the implementation of HAP (5 vs 3 antibiotics). The DU90% has been proven to be an important tool for assessing the quality of drug prescription. In addition to the number of drugs in the DU90% segment, the presence of treatment guidelines and adherence to them may serve as general quality indicators (Bergman et al. 1998). Our findings clearly suggest that the implementation of HAP in the general pediatric ward quickly resulted in a decrease in the total use of antimicrobials, with good adherence to the national recommendations.
The introduction of HAP in the General Pediatric Ward described in the present study demonstrated not only a positive change in the antibiotic consumption pattern but also a general decrease in the antibiotic use. These results are thus more advantageous than those described previously for the Special Care Neonatal Unit (SCNU) (Nitsch-Osuch et al. 2015), where just the consumption pattern of antibiotics was improved. The difference may be explained by several factors. Newborns hospitalized in SCNU are treated mainly for congenital intrauterine infections, so that antibiotics should be effective against Gram negative bacteria. On the other side, children hospitalized in the General Pediatric Ward are diagnosed with community acquired infections, so antimicrobials should be active mainly against Gram positive bacteria. Our results raise attention to the necessity of comparing the antibiotic consumption rates and patterns between similar hospital wards to make the comparison meaningful.
Our study has some limitations. Firstly, it was conducted in just one hospital and the results cannot be representative of the entire country. Secondly, we did not evaluate the appropriateness of the antibiotic prescriptions. Thirdly, antibiotic consumption was estimated with DDDs, while they are targeted at the average maintenance dose per day for a specific antimicrobial drug per adult population and do not take into consideration differences in dose-range and weight-based dosing. However, the alternative metrics, including days of therapy and doses administered, have not been fully evaluated yet (di Pentima et al. 2011). That is the reason why DDDs are still used for the assessment of antibiotic consumption in a pediatric population (Kuster et al. 2008). Additionally, we only compared two consecutive years and some epidemiologic variations in diseases patterns, e.g., influenza incidence, may have influenced antibiotic consumption.
5 Conclusions
The consumption of antibiotics in the General Pediatric Ward decreased after the implementation of the Hospital Antibiotic Policy, especially among children with respiratory tract infections. This intervention was effective in bringing about desirable positive changes in antibiotic consumption patterns. This successful experience in our General Pediatric Ward should encourage other pediatric centers to pursue similar programs.
Abbreviations
- AAP:
-
American Academy of Pediatrics
- ATC:
-
Anatomical Therapeutic Chemical Classification
- CAP:
-
Community-acquired pneumonia
- CDC:
-
Centers for Disease Control and Prevention
- DDDs:
-
Daily-defined doses
- DU90%:
-
90 % drug utilization
- DU100%:
-
100 % drug utilization
- IDSA:
-
Infectious Diseases Society of America
- HAP:
-
Hospital Antibiotic Policy
- NPAP:
-
National Program of Antibiotic Protection
- PIDS:
-
Pediatric Infectious Disease Society
- SHEA:
-
Society of Healthcare Epidemiology of America
- WHO:
-
World Health Organization
References
Ang L, Laskar R, Gray JW (2008) A point prevalence study of infection and antimicrobial use at a UK children’s hospital. J Hosp Infect 68:372–374
Atti ML, Raponi M, Tozzi E, Ciliento G, Ceradini J, Langiano T (2008) Point prevalence study of antibiotic use in a paediatric hospital in Italy. Eurosurevillance 13:1–4
Benin AL, Vitkauskas G, Thornquist E (2003) Improving diagnostic testing and reducing overuse of antibiotics for children with pharyngitis: a useful role for the electronic medical record. Pediatr Infect Dis J 22:1043–1047
Bergman U, Popa C, Tomson Y (1998) Drug utilization 90% – a simple method for assessing the quality of drug prescribing. Eur J Clin Pharmacol 54:113–118
Berild D, Abrahamsen TG, Andresen S, Bjørløw E, Haug O, Kossenko IM (2008) A controlled intervention study to improve antibiotic use in a Russian paediatric hospital. Int J Antimicrob Agents 31:478–483
Blinova E, Lau E, Bitnun A, Cox P, Schwartz S, Atenafu E, Yau Y, Streitenberger L, Parshuram C, Marshall J, Seto W (2013) Point prevalence survey of antimicrobial utilization in the cardiac and pediatric critical care. Pediatr Crit Care Med 14:280–288
Bradley JS, Byington CL, Shah SS (2011) The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis 53:e25–e76
Bruce CM, Kenzie FM, Cookson B, Mollison J, van der Meer J, Kracmery V, Dould I (2009) Antibiotic stewardship and consumption: findings from a pan-European hospital study. J Antimicrob Chemother 64:853–860
Cars O, Molstad S, Melander A (2001) Variation in antibiotic use in the European Union. Lancet 357:1851–1853
Centers for Disease Control and Prevention (CDC) (2012) Get smart: know when antibiotics work. For healthcare providers. Centers for Disease Control and Prevention. http://www.ded.gov/getsmart/specific-groups/hcp/index.html. Accessed on 1 Oct 2014
de Boeck K, Vermeulen F, Meyts I, Hutsebaut L, Franckaert D, Proesmans M (2011) Coprescription of antibiotics and asthma drugs in children. Pediatrics 127:1022–1026
de Man P, Verhoeven BA, Verbrugh HA, Vos MC, van den Anker JN (2000) An antibiotic policy to prevent emergence of resistant bacilli. Lancet 355:973–978
di Pentima M, Chan S, Hossain J (2011) Benefits of a pediatric antimicrobial stewardship program at a children’s hospital. Pediatrics 128:1062–1070
Gaur AH, Hare ME, Shorr RI (2005) Provider and practice characteristics associated with antibiotic use in children with presumed viral respiratory tract infections. Pediatrics 115:635–641
Grohskopf L, Huskins W, Sinkowitz-Cochran R, Levine G, Goldmann D, Jarvis W (2005) Use of antimicrobial agents in United States neonatal and pediatric intensive care patients. Pediatr Infect Dis 24:766–773
Hajdu A, Samodova OV, Carlsson TR, Voinova LV, Nazarenko SJ, Tjurikov AV (2007) A point prevalence survey of hospital-acquired infections and antimicrobial use in a paediatric hospital in north-western Russia. J Hosp Infect 66:378–384
Hryniewicz W, Ozorowski T (2011) Hospital antibiotic policy. Proposals for Polish hospitals. http://www.antybiotyki.edu.pl. Accessed on 12 July 2014
Hyun D, Hersh A, Namtu K, Palazzi D, Maples D, Newland J, Saiman L (2013) Antimicrobial stewardship in pediatrics. How every pediatrician can be a steward. J Am Med Assoc Pediatr 167:859–866
Katakam P, Elfituri A, Ramadan Z, Abadi O (2012) A retrospective study on antibiotic use in different clinical departments of a teaching hospital in Zawiya, Libya. Ibnosina J Med Biomed Sci 12:13–19
Kuster SP, Ruef C, Ledergerber B (2008) Quantitative antibiotic use in hospitals: comparison of measurements, literature review, and recommendations for a standard of reporting. Infection 36:549–559
Levy E, Swami S, Dubois S, Wendt R, Banerjee R (2012) Rates and appropriateness of antimicrobial prescribing at the academic children’s hospital, 200702010. Infect Control Hosp Epidemiol 33:346–353
Liem TB, Krediet T, Fleer A, Agberts T, Rademaker C (2010) Variation in antibiotic use in neonatal intensive care units in the Netherlands. J Antimicrob Chemother 14:245–247
National Programme for Antibiotic Prevention (NPAP) (2011) http://www.antybiotyki.edu.pl/pdf/RekomendacjeA42009.pdf. Accessed on 12 Oct 2014
Nitsch-Osuch A, Kurpas D, Kuchar E, Życińska K, Wardyn K (2015) Antibiotic consumption pattern in the neonatal special care unit before and after implementation of the Hospital’s Antibiotic Policy. Adv Exp Med Biol 835:45–51
Ohl C, Luther V (2011) Antimicrobial stewardship for inpatient facilities. J Hosp Med 11:S4–S15
Palcevski G, Ahel V, Vlahovic-Palcevski V (2004) Antibiotic use profile at paediatric clinics in two transitional countries. Pharmacoepidemiol Drug Saf 13:181–185
Papaevangelou V, Rousounides A, Hadjipanagis A, Katsioulis A, Theodoridou M, Hadjichristodoulou C (2012) Decrease of antibiotic consumption in children with upper respiratory tract infections after implementation of an intervention program in Cyprus. Antimicrob Agent Chemother 56:1658–1661
Potocki M, Goette J, Szucs TD, Nadal D (2003) Prospective survey of antibiotic utilization in pediatric hospitalized patients to identify targets for improvement of prescription. Infection 31:398–403
Principi N, Marchisio P, Sher D, Boccazzi A, Moresco RC, Viola G, Sereni F (1981) Control of antibiotic therapy in paediatric patients. II. Appropriateness of antibiotic choice in selected diseases. Eur J Clin Pharmacol 20:119–121
Rossignoli A, Clavenna A, Bonati M (2007) Antibiotic prescription and prevalence rate in the outpatient pediatric population: analysis of surveys published during 2000–2005. Eur J Clin Pharmacol 63:1099–1106
Schollenberg E, Albritton WL (1980) Antibiotic misuse in a pediatric teaching hospital. Can Med Assoc J 122:49–52
Shehab N, Patel PR, Srinivasan A, Budnitz DS (2008) Emergency department visits for antibiotic-associated adverse events. Clin Infect Dis 47:735–743
Stallworth LE, Fick DM, Ownby DR, Waller JL (2005) Antibiotic use in children who have asthma: results of retrospective database analysis. J Manag Care Pharmacol 11:657–666
Stichele RH, Elseviers MM, Ferech M, Blot S, Goossens H, European Surveillance of Antibiotic Consumption (ESAC) Project Group (2006) Hospital consumption of antibiotics in 15 European countries: results of the ESAC Retrospective Data Collection (1997–2002). J Antimicrob Chemother 58:159–167
Svestina I, Mozgis D (2014) Antimicrobial use among hospitalized children in Latvia: a neonatal and pediatric antimicrobial point prevalence study. Medicine 12:111–119
van Houten MA, Luinge K, Laseur M, Kimpen JL (1998) Antibiotic utilization for hospitalized paediatric patients. Int J Antimicrob Agents 10:161–164
Zhang W, Shen X, Bergman U (2008) Drug utilization 90% (DU90%) profiles of antibiotics in five Chinese children’s hospitals (2002–2006). Int J Antimicrob Agents 32:250–255
Conflicts of Interest
The authors declare no conflicts of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Nitsch-Osuch, A., Kuchar, E., Życińska, K., Gyrczuk, E., Miśkiewicz, K., Korzeniewski, K. (2015). Implementation of Hospital’s Antibiotic Policy Decreases Antimicrobial Use in the General Pediatric Ward. In: Pokorski, M. (eds) Pulmonary Infection. Advances in Experimental Medicine and Biology(), vol 857. Springer, Cham. https://doi.org/10.1007/5584_2015_124
Download citation
DOI: https://doi.org/10.1007/5584_2015_124
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-17457-0
Online ISBN: 978-3-319-17458-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)