Abstract
Polylactones are important biodegradable and biocompatible environmentally friendly polyesters widely used for many applications and more particularly for biomedical applications. This review covers recent advances dealing with their synthesis by ring-opening polymerization (ROP). First, lactones polymerized by ROP will be reviewed with special attention paid to the effect of the ring size on polymerizability. Aliphatic polyesters synthesized by the ROP of lactones can also be obtained by polycondensation. The advantages of ROP compared with polycondensation will be highlighted. The second section is devoted to the different mechanisms used to carry out ROP, such as anionic, coordination, cationic, enzymatic, and organocatalytic polymerization. Special attention will be paid to the control imparted to the polymerization by the use of catalysts and initiators. The polymerization of lactones substituted by functional groups will be shown to afford functionalized aliphatic polyesters. The final section will focus on the synthesis of different architectures such as star-shaped, graft, hyperbranched, and macrocyclic polylactones in the frame of macromolecular engineering.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Aliphatic polyester
- Biodegradable polymer
- Functionalized polymer
- Lactone
- Living polymerization
- Macromolecular engineering
- Ring-opening polymerization
1 Introduction
Aliphatic polyesters occupy a key position in the field of polymer science because they exhibit the remarkable properties of biodegradability and biocompatibility, which opens up a wide range of applications as environmentally friendly thermoplastics and biomaterials. Three different mechanisms of polymerization can be implemented to synthesize aliphatic polyesters: (1) the ring-opening polymerization (ROP) of cyclic ketene acetals, (2) the step-growth polymerization of lactones, and (3) the ROP of lactones (Fig. 1).
The first route relies on the ROP of cyclic ketene acetals [1–3]. The electron-rich double bond is prone to react with radicals and electrophiles. Therefore, this class of monomers undergoes cationic and radical polymerization. For example, radical initiators react with the double bond to provide a new tertiary radical (Fig. 2). Two distinct mechanisms of polymerization can then take place: direct vinyl polymerization or indirect ring opening of the cycle accompanied by the formation of a new radical, which is the propagating species (Fig. 2). The ester function is formed during the ring-opening step. The formation of the C=O double bond, approximately 50 kcal more stable than a C=C double bond, and the release of the ring strain are important driving forces of the reaction [2]. The selectivity of the ring opening versus the vinyl polymerization depends on ring size, substituents, and temperature. For instance, the radical ROP of the seven-membered ketene acetal 2-methylene-1,3-dioxepane yields a 100% ring opening at room temperature [4]. Conversely, as far as five- and six-membered cyclic acetals are concerned, ring opening is generally accompanied by direct vinyl polymerization [2]. Nevertheless, the presence of substituents prone to stabilize the radical formed by ring opening does not favor direct vinyl polymerization. For instance, 2-methylidene-4-phenyl-1,3-dioxolane undergoes a 100% ring-opening event even though this monomer is a five-membered cycle [2]. Although this approach has been known for a long time, its development remains limited, most probably due to the low selectivity of the polymerization and to the difficult synthesis of this class of monomer. Nevertheless, it is worth noting the unusual low crystallinity of aliphatic polyesters synthesized by the ROP of cyclic ketene acetals due to a high content of branching [5, 6].
The second approach, i.e., step-growth polymerization, is far more used than the ROP of cyclic ketene acetals. The step-growth polymerization relies on the esterification reaction of diacids and diols or, more directly, on the esterification of hydroxy-acids. A main advantage of this technique is the easy availability of a very wide range of acid and alcohol precursors of aliphatic polyesters. Nevertheless, this polymerization presents severe limitations. It is mandatory to reach conversion very close to 100% to reach high degrees of polymerization. The esterification reaction has to be carried out at high temperature. Moreover, it is also difficult to predetermine the molar mass, and it is polydispersity index is quite broad. Although some important progress has been made, the synthesis of aliphatic polyesters with a high molar mass is still challenging.
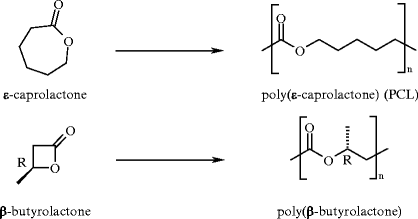
All these limitations were overcome by implementing a third approach based on the ROP of lactones [7, 8]. Indeed, many examples of living or/and controlled polymerization with fast initiation are reported using this technique. High molar mass aliphatic polyesters with low polydispersity indexes can be thus be synthesized. In terms of the availability of the monomers, this approach occupies an intermediate position between step-growth polymerization and ROP of ketene acetals, and a wide range of polymerizable lactones have been polymerized. The most typical lactone is ε-caprolactone (εCL), whose the polymerization was reported in 1934 by Carothers (Fig. 3) [9]. Nowadays, the polymerization of εCL into poly(ε-caprolactone) (PCL) is implemented at the industrial scale. Unlike polyesters synthesized by ROP of cyclic ketene acetals, PCL is highly semicrystalline with a melting temperature around 60 °C and a glass transition temperature of −60 °C. PCL is commonly used in the polyurethane industry to impart good water, oil, solvent, and chlorine resistance. PCL is soluble in many organic solvents and is very easily processable. Moreover, PCL is highly biocompatible and is accordingly used for biomedical applications, as recently reviewed [10]. The degradation of PCL is slow, making it a suitable carrier for long-term drug delivery applications [11]. Interestingly, PCL is miscible with a set of polymers such as poly(vinyl chloride) (PVC).
Another interesting example of lactones are the β-hydroxyalkanoates, whose ROP affords poly(β-hydroxyalkanoate)s (PHAs), a class of aliphatic polyesters naturally produced by bacteria (Fig. 3) [12, 13]. Poly(3-(R)-hydroxybutyrate) (PHB) is a typical example. PHB is a stiff thermoplastic material with relatively poor impact strength, but the incorporation of other monomers can improve the mechanical properties.
This review aims at reporting on the synthesis of aliphatic polyesters by ROP of lactones. It is worth noting that lactones include cyclic mono- and diesters. Typical cyclic diesters are lactide and glycolide, whose polymerizations provide aliphatic polyesters widely used in the frame of biomedical applications. Nevertheless, this review will focus on the polymerization of cyclic monoesters. It will be shown that the ROP of lactones can take place by various mechanisms. The polymerization can be initiated by anions, organometallic species, cations, and nucleophiles. It can also be catalyzed by Bronsted acids, Lewis acids, enzymes, organic nucleophiles, and bases. The number of processes reported for the ROP of lactones is so huge that it is almost impossible to describe all of them. In this review, we will focus on the more representative examples and on the more widely used processes. The polymerization of unsubstituted lactones will be reported first. The last section will deal with the macromolecular engineering of poly(lactone)s. It will be shown that a wide range of aliphatic polyesters can be synthesized by the ROP of lactones bearing substituents, functionalized or not. Finally, special attention will be paid to the synthesis of aliphatic polyesters with different topologies such as star-shaped, graft, macrocyclic, and crosslinked polyesters.
2 ROP of Unsubstituted Lactones
2.1 Nomenclature
The IUPAC organization proposed a systematic nomenclature for organic compounds. Nevertheless, this official nomenclature is not very popular in the case of lactones. The usual names are based on the name of the hydroxyacids as precursors and the Greek letter refers to the position of the hydroxy group in relation to the acid group. This letter directly indicates the ring size. For instance, β-lactones are four-membered lactones. The IUPAC and usual names of main lactones are shown in Table 1. In this review, the usual names will be used for the sake of simplification. Nevertheless, as far as substituted lactones are concerned, different authors can give different names for a single lactone. Whenever such confusion is possible, the IUPAC nomenclature will be systematically preferred in this chapter, especially in the case of substituted lactones.
2.2 Polymerizability of Lactones
In order to determine whether the ROP of lactones into the corresponding aliphatic polyesters is possible, thermodynamics have to be taken into account. The ROP follows the micro-reversibility rule according to (1):
The polymerization is only possible if the free enthalpy is negative (ΔG p < 0). The free enthalpy is given by (2), where ΔH p and ΔS p stand for the enthalpy and the entropy of polymerization, respectively. The monomer concentration at equilibrium ([M]eq) and the ceiling temperature (T c) are given by (3) and (4), respectively. ΔH p, ΔS p, [M]eq and T c are shown in Table 2 for the polymerization of a set of lactones of different sizes [14].
As shown in Table 2, the ROP of lactones is accompanied by a decrease of entropy (ΔS p < 0). Accordingly, the polymerization is an enthalpy-driven process, the negative polymerization enthalpy stemming from the release of the ring strain during the ring opening. As a rule, high molecular weight polyesters can thus be synthesized by ROP of four-, six- and seven-membered cyclic esters, as witnessed by the low equilibrium monomer concentration and the low ceiling temperature. The polymerization of five-membered lactones is far more difficult, as shown by the very high ceiling temperature. The main reason for this particular behavior is the very fast reverse reaction (i.e., the unzipping reaction compared to propagation) and the equilibrium monomer concentration is then very high. The ROP of this monomer was so difficult that some authors even reported that this polymerization is impossible. This view was nevertheless revised because it was shown that low molecular weight oligomers can be obtained under suitable conditions and because γ-butyrolactone is also able to co-polymerize with other lactones [14].
2.3 Main Mechanisms of Polymerization of Lactones by Ring Opening
A very broad range of initiators and catalysts are reported in the scientific literature to polymerize lactones. The polymerization mechanisms can be roughly divided into five categories, i.e., anionic polymerization, coordination polymerization, cationic polymerization, organocatalytic polymerization, and enzymatic polymerization.
2.3.1 Anionic Polymerization
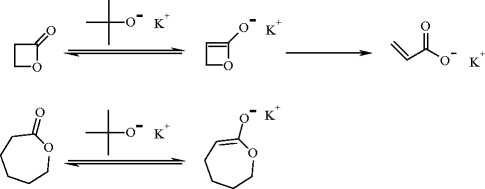
The polymerization of lactones is initiated by nucleophilic metal alkoxides. It is worth noting that bulky alkoxides are not nucleophilic enough and react as bases. For example, potassium tert-butoxide deprotonates β-propionolactone and εCL into new anionic species, which are anionic initiators for the polymerization of lactones [8] (Fig. 4). As a rule, carboxylic salts are less nucleophilic than the corresponding alkoxides and are, in general, not efficient initiators for the polymerization of lactones. Nevertheless, β-lactones are exceptions because their polymerization can be initiated by carboxylic salts [8].
The more usual mechanism of the anionic ROP of lactones relies on the addition of alkoxides onto the ester group followed by the cleavage of the acyl–oxygen bond (Fig. 5).
Nevertheless, it is worth noting that a second mechanism was observed in the particular case of the ROP of β-lactones. This mechanism takes place by the scission of the alkyl–oxygen bond into a new carboxylate, as shown in Fig. 6. Indeed, the polymerization of β-lactones by the first mechanism based on the cleavage of the acyl–oxygen bond is disfavored, which was accounted for by stereo-electronic effects [15].
In order to be living, according to Szwarc a polymerization has to take place without any transfer and termination reactions [16]. All chains remain active during the polymerization and remain indefinitely able to grow. Under these conditions, the concentration of active species is kept constant once the initiation is over, and first-order kinetics in monomer is observed. Furthermore, the degree of polymerization is determined by the ratio of the amount of monomer and initiator. Finally, after complete conversion, the addition of a new feed of monomer results in a quantitative resumption of polymerization. As long as the anionic ROP is carried out in the absence of protic species, termination reactions are minor events. The loss of control of polymerization is then mainly caused by transfer reactions. Indeed, alkoxides are prone to react not only with the ester function of the cyclic monomer but also with ester functions present all along the polyester chains, resulting in transesterification reactions (Figs. 7 and 8). On the one hand, intramolecular transesterification reactions bring about a decrease of the molar mass and result in the formation of cyclic oligomers (Fig. 7). The extent of the loss of polymerization control is quantified by the selectivity factor β, which is the ratio of the rate of propagation and the rate of the transfer by intramolecular transesterification reactions (β = k p/k tr1). On the other hand, intermolecular transesterification reactions reshuffle the length of polyester chains until a polydispersity index of 2 is reached (Fig. 8). The extent of the loss of polymerization control is quantified by a second selectivity factor γ, which is the ratio of the rate of propagation and the rate of transfer by intermolecular transesterification reactions (β = k p/k tr1).
Control of the molecular parameters can be improved by disfavoring the transesterification reactions. This task is carried out by decreasing the reactivity of the initiator in order to let it react more selectively with the more reactive ester groups of the cyclic monomer and not with the less reactive ester groups along the chains. The decrease in the reactivity of the alkoxide can be achieved by using both steric and electronic effects. Indeed, the use of hindered ligands is a tool that allows lower reactivity, better selectivity, and thus better control of the polymerization to be achieved. Another possibility relies on the use of less electropositive metals. In this respect, a good control of the polymerization was observed using alkoxides based on metals with d-orbitals of favorable energy as initiators. This family of initiators will be discussed in the section dealing with coordination polymerization.
It is worth noting that another transfer reaction to the monomer is reported in the case of the anionic polymerization of β-lactones, as shown in Fig. 9. This transfer reaction takes place during the polymerization of β-propionolactone initiated by carboxylates; even though molar masses up to 10,000 can be reached, even at room temperature [8]. The situation is even more critical in the case of the polymerization of substituted β-butyrolactones, as witnessed by a k p/k tr ratio equal to 102, which is the highest number average degree of polymerization that can be reached under these conditions [8].
2.3.2 Coordination Polymerization
In order to impart a better control of the polymerization, less reactive and thus more selective organometallic derivatives of metals with d-orbitals of favorable energy were substituted for anionic initiators [17]. The first works were carried out by Teyssié and coworkers using bimetallic μ-oxo-alkoxides [18, 19].
Control of the ROP of lactones was improved by using aluminum alkoxides instead of their anionic counterpart, as witnessed by the increase of the selectivity factor β, as shown for instance in Table 3 in the case of the polymerization of εCL. The lower reactivity of aluminum alkoxides compared to their anionic counterparts is shown by the decrease in the rate of propagation of the polymerization (k p).
A wide range of aluminum alkoxides can easily be synthesized by the reaction of alcohols with triethylaluminum. These alcohols can even be substituted by compatible functional groups such as bromides, olefins, and tertiary amines (Fig. 10) [20, 21]. An alternative route towards aluminum alkoxides relies on the reaction of the alcohols with aluminum isopropoxide in toluene. Isopropanol (iPr) formed during this reaction is withdrawn by the distillation of the azeotrope made up of toluene and isopropanol [20, 21].
Some initiators used for the coordinative ROP of lactones are known to form aggregates. Aluminum isoporopoxide is a typical example because it is known to exist as a mixture of trimers (A3) and tetramers (A4) (Fig. 11). If the polymerization of εCL is initiated at 0 °C by a mixture of A3 and A4, only A3 is prone to initiate the polymerization. Under these conditions, the interconversion of A4 into A3 is too slow compared to the propagation of the polymerization and, accordingly, A4 remained mostly untouched at the end of the polymerization. It is worth noting that A4 is slowly incorporated into polyester chains by transesterification reactions with ester functions present all along the chains, even after the end of the polymerization.
The propagation of the polymerization proceeds according to a coordination–insertion mechanism, as shown in Fig. 12. The first step relies on the coordination of the alkoxide (RO-M) to the carbonyl of the monomer followed by the addition of the nucleophilic alkoxide onto the electrophilic ester function. Thereafter, an elimination step results in the cleavage of the acyl–oxygen bond, the opening of the ring, and the formation of a new alkoxide, which is nothing but the propagating species.
Aggregation, already observed during initiation, can also take place during propagation depending upon the structure of the propagating species. These phenomena were evidenced by NMR spectroscopy and kinetic studies. The kinetic model shown in Fig. 13 was proposed by Duda and Penczek [22]. The model is based on several hypotheses. It is assumed that aggregated species are not reactive enough to react with the lactone and thus to propagate. Conversely, the dissociated species are the propagating species. Moreover, the equilibrium is shifted towards aggregated species.
Under these conditions, (5) is valid:
Equation (6) can then be deduced:
The logarithmic form of (6) gives (7):
Accordingly, the plot of \( \ln ([{k\prime_{\text{p}}}]) \) versus ln([I]0) is linear with a slope of 1/n. The aggregation state can thus be directly deduced from this plot. For instance, if the polymerization of εCL is initiated by A3, the three-arm growing species is an unimer. Conversely, when the same polymerization is initiated by Et2AlOR′, trimeric species are observed [21].
In the presence of aggregation, the polymerization cannot be strictly considered as living, at least according to the original definition given by Szwarc [16], because aggregation is a transfer reaction. Nevertheless, aggregated species can be considered as being in a dormant state and can be reactivated into active disaggregated species at any time. Interestingly, control of the molecular weight is maintained even though reversible aggregation is present, provided that the aggregation and dissociation rates are high enough compared to propagation.
Later, Kricheldorf and coworkers extended the concept of the aluminum alkoxide-initiated ROP of lactones to a set of other metal alkoxides such as tin(IV) [23–25], titanium, and zirconium alkoxides. As a rule, the polymerization takes place according to the same coordination–insertion mechanism shown in Fig. 12. A very wide range of range of initiators and catalysts are reported in the scientific literature. Their number is so high that it is practically impossible to describe all of them in an exhaustive review [17]. Nevertheless, special attention has to be paid to lanthanide alkoxides because of their higher activity compared to aluminum alkoxides while maintaining a good control of the polymerization. A very first example was reported by McLain and Drysdale [26]. It was shown that yttrium isopropoxide, which is an oxo alkoxide cluster with the Y5(O)(OCHMe2)13 stoichiometry, initiated the controlled polymerization of εCL. The polymerization turned out to be very fast and went to completion within 5 min at room temperature. It was mentioned that other lanthanides alkoxides (metal = Er, Sm, Dy, La) were also efficient initiators for the same polymerization [26]. Shen and coworkers showed that an increase of steric hindrance of the ligand did not favor transesterification reactions [27]. Yasuda and coworkers used SmOEt(C5Me5)2(OEt2), [YOMe(C5H5)2]2, and YOMe(C5Me5)2(THF) as lanthanide alkoxides to polymerize εCL [28]. In 1996, Feijen and coworkers generated yttrium isopropoxide in situ by reaction of isopropanol and yttrium tris(2,6-di-tert-butylphenolate [29, 30]. Later, Jérôme and coworkers synthesized in situ yttrium isopropoxide by reaction of isopropanol with Y[N(SiMe3)2]3 [31, 32]. A similar approach was implemented by Spitz and coworkers, who initiated the polymerization of εCL by Nd(OiPr)3 obtained from the reaction of isopropanol and Nd[N(SiMe3)2]3 [33]. In 2003, Soum and coworkers reported a study on the mechanism and kinetics of the polymerization of εCL by La(OiPr)3 [34]. It is worth noting that the reduction of lactones by hydrides affords alkoxides able to initiate the propagation of the polymerization. This route was implemented by Guillaume and coworkers, who polymerized εCL by M(BH4)3(THF)3 (M=Nd, La, Sm) into hydroxyl-telechelic PCL [35, 36]. The polymerization of εCL by lanthanide alkoxides was extended to other monomers such as β-propionolactone and δ-valerolactone, and was carried out using SmOEt(C5Me5)2(OEt2), [YOMe(C5H5)2]2, and YOMe(C5Me5)2(THF) as lanthanide alkoxides [28].
Currently, tin(II) bis-(2-ethylhexanoate), also referred as tin octoate, is the most widely used catalyst for the ROP of lactones. This popularity stems from its acceptance by the American Food and Drug Administration (FDA) for the formulation of polymer coatings in contact with food. Moreover, tin(II) bis-(2-ethylhexanoate) is less sensitivity towards water and other protic impurities than aluminum alkoxides, which facilitates its use in the laboratory and in industry.
The mechanism of the tin(II) bis-(2-ethylhexanoate)-mediated ROP of lactones remained a matter of controversy for many years, and many different mechanisms were proposed. Indeed, tin(II) bis-(2-ethylhexanoate) is not made up of alkoxides but of carboxylates, known as poor initiators for the ROP of lactones. In 1998, Penczek and coworkers made a major contribution in this field. They reported that, if the polymerization is carried out in THF at 80 °C, then tin(II) bis-(2-ethylhexanoate) is converted in situ into a new tin alkoxide by the reaction with either an alcohol, purposely added in the reaction medium, or with any other protic impurity present in the polymerization medium (Fig. 14) [37]. Tin alkoxides formed in situ are the real initiators of the polymerization, which takes place according the usual insertion–coordination mechanism shown in Fig. 12. This mechanism has been proved by experimental data such as kinetic measurements [37, 38], analysis of the chain-ends by MALDI-TOF [39], and the influence of proton trapping agents [40]. A dynamic equilibrium is established between the dormant alcohol and the active tin alkoxide. This equilibrium maintains the concentration of propagating tin alkoxides at a quite low level throughout the polymerization process. Nevertheless, each chain remains able to propagate and the polymerization is kept under control. Accordingly, the molecular weight of the aliphatic polyester is predetermined by the molar ratio of the monomer to the alcohol. Secondly, the structure of the chain-end is controlled. Finally, after the end of the polymerization, the addition of a new feed of monomer results in a complete resumption of polymerization. Nevertheless, too high a concentration of tin(II) bis-(2-ethylhexanoate) favors transesterification reactions, which limits the control of the polymerization.
Interestingly, salts other than tin(II) bis-(2-ethylhexanoate) such as scandium and tin trifluoromethanesulfonate [41–43], zinc octoate [44, 45], and aluminum acetyl acetonate [45] were reported to mediate the ROP of lactones. As far as scandium trifluoromethanesulfonate is concerned, the main advantage is the increase of its Lewis acidity enabling the polymerization to be carried out at low temperatures with acceptable kinetics. Later, faster kinetics were obtained by extending the process to scandium trifluoromethanesulfonimide [Sc(NTf2)3] and scandium nonafluorobutanesulfonimide [Sc(NNf2)3] and to other rare earth metal catalysts (metal=Tm, Sm, Nd) [46].
Although tin(II) bis-(2-ethylhexanoate) is accepted by the FDA, this catalyst turned out to be cytotoxic and it implementation to synthesize aliphatic polyesters for biomedical applications is questionable. It is worth noting that the tin-based residues are very difficult to remove from the polyester. Interestingly, Albertsson and coworkers reported an efficient process aiming at reducing the amount of metallic remnants left in aliphatic polyesters [47]. To this end, after a polymerization of εCL initiated by 1-di-n-butyl-1-stanna-2,5-dioxacyclopentane (Fig. 15) goes to completion, 1,2-ethanedithiol was added to the polymerization medium with the aim of replacing the Sn–O bonds by more stable Sn–S bonds. The resulting S-containing dibutyltin derivative was more soluble in organic solvents, and thus it was easier to get rid of tin residues through precipitation. Remarkably, the implementation of this process allowed the preparation of a sample of PCL contaminated by only 23 ppm of tin residues, which is very close to the limit of 20 ppm imposed by the FDA [47].
Coordination ROP can be carried out in bulk and in organic solvents. Particular attention has to be paid to supercritical carbon dioxide because it is a green, environmentally friendly medium. A first report dealing with the ROP of εCL in supercritical CO2 was published by Mingotaud and coworkers, but with little information about possible control [48, 49]. Later, Jérôme and coworkers observed that tin(IV) alkoxides are very efficient initiators for the controlled ROP of εCL in supercritical CO2 [50]. Nevertheless, kinetics of ROP are very slow in supercritical CO2, which was accounted for by the reversible reaction of alkoxides and CO2 into carbonated tin compounds [51]. The nature of the metal is important because less ionic alkoxides have a lesser reactivity towards CO2 [52]. It is worth noting that PCL is not soluble in supercritical CO2. In the presence of a suitable surfactant, nanoparticles were obtained [53]. Last but not least, the remarkable “gaslike” mass transfer properties of supercritical CO2 can be exploited to remove quantitatively any unconverted monomer and metallic remnants.
The main limitation of the coordination ROP of lactones remains the toxicity of the metal. For instance, aluminum derivatives are suspected to be involved in Alzheimer’s disease, and tin(II) bis-(2-ethylhexanoate) is cytotoxic. In order to overcome this drawback, many groups have investigated the replacement of tin and aluminum alkoxides by initiators based on less toxic metals such as magnesium [54, 55] and calcium [56, 57] alkoxides.
Another possible approach relies on the immobilization of the initiator on an insoluble support. Toward this end, Jérôme and coworkers grafted yttrium isopropoxide onto a porous silica surface [58, 59]. Two methods were reported for the immobilization. The first method relies on the reaction of surface hydroxyl groups with an excess of Y[N(SiMe3)2]3. The silylamido groups immobilized on the surface were then converted into yttrium alkoxides by the reaction with 2-propanol. In the second approach, a yttrium alkoxide was prepared by reacting Y[N(SiMe3)2]3 with less than three equivalents of 2-propanol and then grafted onto the silica surface. Using similar methods, Hamaide and coworkers supported Al, Zr, Y, Sm, and Nd alkoxides onto silica and alumina [60].
2.3.3 Cationic Polymerization
Cationic ROP of lactones has been known for a long time but is not very popular due to its poor control of the molecular parameters. In 1984, Penczek and coworkers reported the cationic polymerization of εCL and β-propionolactone mediated by acylating agents [61]. The cationic initiators and catalysts can be roughly divided in four main families: alkylating agents, acylating agents, Lewis acids, and protonic acids.
Until the middle of the 1980s, it was accepted that the mechanism for the cationic ROP initiated by alkylating agents was based on the reaction of the cation with the endocyclic oxygen, followed by cleavage of the acyl–oxygen bond (Fig. 16).
Nevertheless, in 1984, Penczek [62] and Kricheldorf [63] revised this mechanism. If alkylating agents are used as initiators, they proposed that the cation reacts with the exocyclic oxygen to form the dialkoxycarbocationic species, which reacts further by cleavage of the alkyl–oxygen bond as shown in Fig. 17. This mechanism was clearly proved by the presence of an ester function at the α-chain-end, whereas the first mechanism would result in the presence of an ether function. The propagation of polymerization takes then place by reaction of the exocyclic oxygen with the dioxocarbocation.
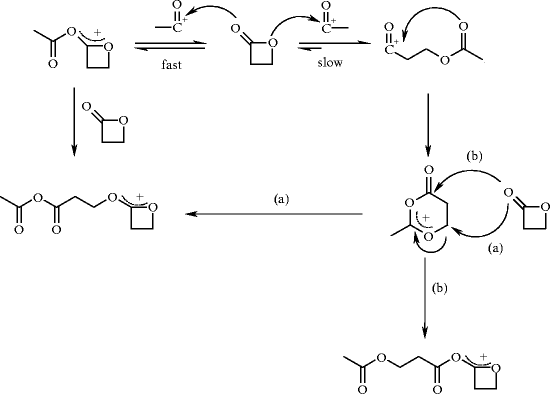
The mechanism is more complex when acylating agents are used as initiators because both mechanisms are observed (Fig. 18) [64].
Lewis acids were also screened for the ROP of lactones [65]. The polymerization takes place according to a cationic mechanism provided that the counterion is not too nucleophilic. Conversely, when Lewis acids with a nucleophilic counterion are used, several examples are reported where the polymerization takes place according to the usual coordination–insertion mechanism (Fig. 12). This coordination–insertion mechanism was indeed reported for the ROP initiated by ZnCl2 [66], TiCl4, and AlCl3 [67].
Another approach enabling cationic ROP relies on initiation by nucleophilic alcohols and amines in the presence of Bronsted acids as catalysts. The mechanism of initiation is based on the activation of lactones by protonation of the exocyclic oxygen, followed by the nucleophilic attack of the alcohol and the scission of the oxygen–acyl bond (Fig. 19). The mechanism of propagation is similar except that the hydroxyl function at the chain-end is the nucleophilic species. This activation monomer mechanism is more favorable than the mechanism shown in Fig. 17 owing to the lower nucleophilicity of the exocyclic oxygen of lactones compared to alcohols. In 2000, Endo and coworkers reported a first example of controlled cationic ROP of εCL and δ-valerolactone by using an alcohol as an initiator and HCl.Et2O as catalyst [68]. Nevertheless, the molar mass did not exceed 15,000, except for poly(δ-valerolactone), which has been prepared with molecular weight up to 50,000 [69]. Recently, it was shown that the cationic polymerization of εCL [70] is under control if trifluoromethanesulfonic is used as a catalyst. Molar masses up to 20,000 were obtained. Later, Bourrissou showed that trifluoromethanesulfonic acid can be replaced by the less acidic methanesulfonic acid [71]. Kakuchi and coworkers reported the polymerization of δ-valerolactone using 3-phenyl-1-propanol as the initiator and trifluoromethanesulfonimide as the catalyst in CH2Cl2 at 27 °C [72]. Interestingly, the polymerization was under control and various functionalized alcohols were used as initiators. Very recently, Takasu and coworkers extended this strategy to other perfluoroalkanesulfonates and perfluoroalkanesulfonimides to perform fast ROP [73]. As far as εCL is concerned, the fastest polymerization was obtained by using nonafluorobutanesulfonimide as a catalyst.
Organic compounds such as lactic acid [74, 75], citric acid [74], fumaric acid [76, 77], and amino acids [74] were also shown to catalyze the ROP of lactones, using alcohols and amines as initiators. Interestingly, an acid catalyst can be supported on a solid support such as porous silica [78]. Unfortunately, the polymerization is slow and reuse of the catalyst after its recovery and regeneration turned out to be unsuccessful. Finally, it is worth noting the particular behavior of amino acids, which are able to both catalyze and initiate the ROP of lactones [79].
2.3.4 Organocatalytic Polymerization
Many researchers have investigated the use of amines and alcohols as initiators for the ROP of lactones. As a rule, amines and alcohols are not nucleophilic enough to be efficient initiators, and it is then mandatory to use catalysts to perform the polymerization successfully. Nevertheless, highly reactive β-lactones exhibit a particular behavior because their polymerization can be initiated by nucleophilic amines in the absence of any catalyst. As far as tertiary amines are concerned, the initiation step implies the formation of a zwitterion made up of an ammonium cation and a carboxylate anion, as shown in Fig. 20. Authors coined the name “zwitterionic polymerization” for this process [80]. Nevertheless, this polymerization is not really new because the mechanism is mainly anionic. Interestingly, Kricheldorf and coworkers did not exclude the possibility that, at least at some stage of the polymerization, chain extension takes place by step-growth polycondensation [81]. The same authors did not observe intramolecular cyclization by the coupling reaction of the ammonium cation and the carboxylate anion, at least in their work [81]. The presence of elimination reactions of the ammonium salt should not be ruled out, at least at high temperatures.
For less reactive lactones, the initiation and propagation of the polymerization require a catalyst. Two strategies can be implemented: the catalysts can activate either the initiator or the monomer. Interestingly, dual catalysts can associate both mechanisms of activation. Some significant advances have been made in the last few years, especially under the impulse of the group of Hedrick, as recently reviewed [82, 83].
The activation of lactones by Bronsted acids was discussed in the section 2.3.3 dealing with cationic polymerization. An alternative relies on the use of nucleophilic species for the activation of lactones (Fig. 21).
N-Heterocyclic carbenes are an example of a family of nucleophilic catalysts [84–87]. For instance, the polymerization of β-butyrolactone was catalyzed by 1,3,4-triphenyl-4,5-dihydro-1H,1,2-triazol-5-ylidene in the presence of methanol as an initiator [86]. This reaction was carried out in toluene at 80 °C. Nevertheless, an undesired elimination (Fig. 4) reaction was observed and control of the polymerization was lost. This issue was overcome by using tert-butanol as a co-solvent, which reacts reversibly with the free carbene to form a new adduct. Owing to the decrease in the concentration of the free carbene, the elimination is disfavored and the polymerization is then under control provided that a degree of polymerization below 200 is targeted. As a rule, the reactivity of N-heterocyclic carbenes depends on their substituents. Hindered N-heterocyclic carbenes turned out to be not nucleophilic enough for the ROP of εCL. Recently, it was shown that unencumbered N-heterocyclic carbenes were more efficient catalysts [87].
1,5,7-Triazabicyclo[4.4.0]dec-5-ene (TBD) is another efficient organocatalyst for the ROP of lactones initiated by alcohols [88, 89]. Indeed, the polymerization of εCL and δ-valerolactone was successfully carried out, and the corresponding polyesters were obtained with predictable degrees of polymerization of up to 200 and low polydispersities (<1.16). Interestingly, Hedrick and coworkers proposed a mechanism based on the dual activation of both the monomer and the initiator according to Fig. 22. It is worth noting that β-butyrolactone did not polymerize in the presence of this catalyst.
When 1,8-diaza[5.4.0]bicycloundec-7-ene (DBU) and N-methylated TBD (MTBD) were used as catalysts instead of TBD, no polymerization was observed, even with catalyst loading of up to 20 mol% [89]. The lack of activity of these amines was accounted for by the absence of any activation of the lactone, and the activation of the alcohol turned out to be not sufficient. However, the polymerization was successfully carried out by the addition of a thiourea as a co-catalyst to activate lactones, as shown in Fig. 23. Again, β-butyrolactone was not reactive enough and was not polymerized [89].
The implementation of phosphazene bases as organocatalysts was also investigated in order to efficiently activate alcohols [90]. More particularly, 2-tertbutylimino-2-diethylamino-1,3-dimethylperhydrdro-1,3,2-diazaphosphorine (BEMP) was investigated due to its higher basicity (pK BH+ = 27.6) compared to DBU (pK BH+ = 24.3) and MTBD (pK BH+ = 25.4). The polymerization of εCL and δ-valerolactone was successfully carried out in the presence of BEMP by using 1-pyrernebutanol as an initiator (Fig. 24). Although the polymerization was under control, the kinetics were quite low, especially in the case of the polymerization of εCL, which required 10 days to reach a conversion of 14% [90].
2.4 Enzymatic Polymerization
Lipases are enzymes of the hydrolase family and, in nature, hydrolyze fatty acid esters in aqueous environment. It is worth recalling that the hydrolysis of esters is a reversible reaction. Chemists thus often use lipases to catalyze the reverse reaction, i.e., the esterification and the ROP of lactones. In 1993, the groups of Kobayashi [91] and Knani [92] reported independently the lipase-catalyzed ROP of εCL and δ-valerolactone. The aliphatic polyesters were functionalized by a carboxylic group at one chain-end and a hydroxyl group at the other chain-end. Accordingly, the polymerization was initiated and terminated by water present in the reaction media.
Since then, the process has been extended to a wide variety of lactones of different size and to several lipases, as recently reviewed [93–96]. Interestingly, large-membered lactones, which are very difficult to polymerize by usual anionic and coordination polymerizations due to the low ring strain, are successfully polymerized by enzymes. Among the different lipases available, that from Candida antarctica (lipase CA, CALB or Novozym 435) is the most widely used due to its high activity. An alcohol can purposely be added to the reaction medium to initiate the polymerization instead of water. The polymerization can be carried out in bulk, in organic solvents, in water, and in ionic liquids. Interestingly, Kobayashi and coworkers reported in 2001 the ROP of lactones by lipase CA in supercritical CO2 [97]. Later, Howdle and coworkers supported lipase CA on macroporous beads [98]. It is worth noting that the enzyme can be withdrawn and recycled by using supercritical CO2. The success of the polymerizations carried out in organic solvents stems directly from the sustained activity of several lipases in organic solvents. In this respect, it must be noted that water has a manifold influence on the course of the polymerization. On the one hand, water can initiate the polymerization. On the other hand, a minimum amount of water has to be bound to the surface of the enzyme to maintain its conformational flexibility, which is essential for its catalytic activity [94]. Lipase-mediated polymerization cannot therefore be achieved in strictly anhydrous conditions.
The mechanism of the enzymatic polymerization is shown in Fig. 25 and can be decomposed into three main steps. First, a complex is formed between the enzyme and the lactone. The second step is based on the nucleophilic activation of the lactone by the hydroxyl function belonging to a serine residue of the active site of the enzyme. An activated opened monomer is obtained, which is more reactive than lactone towards nucleophiles. Finally, the activated opened monomer reacts with nucleophilic hydroxyl-functionalized initiators and propagating species (Fig. 25). It is worth noting that this activation monomer mechanism is very similar to that shown in the section 2.3.4 dealing with ROP catalyzed by nucleophilic organocatalysts.
A key point should be to identify the rate-limiting step of the polymerization. Several studies indicate that the formation of the activated open monomer is the rate-limiting step. The kinetics of polymerization obey the usual Michaelis–Menten equation. Nevertheless, all experimental data cannot be accounted for by this theory. Other studies suggest that the nature of the rate-limiting step depends upon the structure of the lactone. Indeed, the reaction of nucleophilic hydroxyl-functionalized compounds with activated opened monomers can become the rate-limiting step, especially if sterically hindered nucleophilic species are involved.
Unlike chemical ROP, enzymatic ROP is not governed by the ring strain. Unexpectedly, several studies showed that macrolides polymerize faster than smaller ring lactones when lipases are used as catalysts. It was shown that macrolides exhibit a higher dipole moment and are thus more hydrophobic than smaller ring lactones [99]. This factor was invoked to shed light on the very particular reactivity of lactones in enzymatic ROP. Indeed, the faster kinetics of polymerization of macrolides was accounted for by their better recognition by lipases due to their higher hydrophobicity and to their shape. Indeed, macrolides have a chemical structure closer to the glycerol fatty acids esters hydrolyzed by lipases in nature.
The benefits of enzymes as catalysts for the ROP of lactones are manifold:
-
Enzymes are green catalysts obtained from renewable resources
-
Enzymes are easily separated from the polyesters
-
The polymerization proceeds under mild conditions in terms of pH, temperature and pressure
-
The polymerization can be carried out in bulk, in organic media, and at various interfaces
-
The polymerization is not carried out under strictly anhydrous conditions as is the case when aluminum and tin alkoxides are used as initiators
-
The polymerization can take place with high regio- and stereoselectivity
-
Enzymes are very efficient catalysts for the polymerization of large-size lactones, which are particularly difficult to polymerize by usual chemical catalysts and initiators owing to the low ring strain
Nevertheless, the use of enzymes presents some drawbacks. Enzymes are expensive and large amounts are needed for polymerization. Besides, it is still quite challenging to synthesize high molar mass polyesters. The control of polymerization remains less efficient with enzymes than with chemical initiators such as aluminum alkoxides.
3 Polymerization of Substituted Lactones
The polymerization of substituted lactones is an attractive strategy for extending the range of aliphatic polyesters and for tailoring important properties such as biodegradation rate, bioadherence, crystallinity, hydrophilicity, and mechanical properties [100]. Moreover, the substituent can bear a functional group, which can be very useful for the covalent attachment of drugs, probes, or control units.
The most direct route towards functionalized aliphatic polyesters is based on the functionalization of polyester chains. This approach is a very appealing because a wide range of functionalized aliphatic polyesters could then be made available from a single precursor. This approach was implemented by Vert and coworkers using a two-step process. First, PCL was metallated by lithium diisopropylamide with formation of a poly(enolate). Second, the poly(enolate) was reacted with an electrophile such as naphthoyl chloride [101], benzylchloroformate [101] acetophenone [101], benzaldehyde [101], carbon dioxide [102] tritiated water [103], α-bromoacetoxy-ω-methoxy-poly(ethylene oxide) [104], or iodine [105] (Fig. 26). The implementation of this strategy is, however, difficult because of a severe competition between chain metallation and chain degradation. Moreover, the content of functionalization is quite low (<30%), even under optimized conditions.
A less direct strategy relies on the synthesis and ROP of substituted and/or unreactive functionalized lactones [100]. Two approaches can be implemented. In the first approach, the unreactive functional group is inserted directly inside the ring. It is worth recalling that cyclic diesters such as lactide and glycolide, for which both esters can be involved in the polymerization, will not be covered by this review. The second approach is based on the synthesis and polymerization of lactones bearing a substituent, functionalized or not.
Lactones bearing functions inside the ring that do not interfere with the ROP mechanism, such an ether [106–113], an amine [114, 115], an amide [116–118], an unsaturation [119–122], or a ketone [123] are shown in Fig. 27. If the functional group is not tolerated by the polymerization mechanism, it has then to be protected as is the case for amines [114, 115].
It is worth noting that 6,7-dihydro-2(3H)-oxepinone is an unusual lactone because it can be polymerized by two distinct mechanisms: ROP of the cyclic esters by aluminum alkoxides, and the ring-opening metathesis polymerization (ROMP) of endocyclic olefins by the Schrock’s catalyst (Fig. 28) [121]. The metathesis route opens up new opportunities for the synthesis of new copolymers by copolymerizing 6,7-dihydro-2(3H)-oxepinone with other cyclic olefins such as norbornene, even though this approach has barely been exploited until now [121].
The second approach is based on the polymerization of lactones bearing functionalized substituents. A set of lactones bearing chloride [124–126], bromide [127, 128], iodide [129], alkene [130–134], or alkyne [135] were synthesized and (co)polymerized by tin octoate, tin(IV), and aluminum alkoxides (Fig. 29).
Unfortunately, this strategy presents severe drawbacks. Firstly, several of these lactones are not commercially available and several steps are often necessary for their synthesis. Moreover, it is mandatory to rigorously purify theses lactones before polymerization, especially if sensitive alkoxides are used as initiators, which is sometimes a difficult task. Accordingly, the global yield of the synthesis is sometimes low and the functionalized lactone is thus quite expensive. Another issue is the lack of chemoselectivity of the chemical polymerization. Several functional groups such as epoxides, alcohols, and carboxylic acids are not tolerated by propagating species such as aluminum and tin(IV) alkoxides. This drawback was successfully overcome by the protection of groups such as alcohols [15, 114, 136–140], amines [141–143] and carboxylic acids [15, 114, 139, 144–147] prior to polymerization (Fig. 30). Nevertheless, the protecting groups have to be removed after polymerization and it is not always easy to find deprotection conditions where no degradation takes place, especially when acidic conditions are used. Benzylic ethers and esters have been often used because they are typically deprotected by hydrogenation, under neutral conditions. In some particular cases, it was observed that the chemoselectivity depends on the nature of the initiator. For instance, the ketone function is not tolerated by aluminum alkoxides but is well tolerated by tin(IV) alkoxides for unclear reasons [123]. Accordingly, the polymerization of 5-oxepane-2-dione was successfully carried out by using 2,2-dibutyl-2-stanna-1,3-dioxepane (DSDOP) as an initiator. Conversely, control was completely lost when aluminum isopropoxide was used as an initiator [148] and it was then necessary to protect the ketone function in the form of a ketal group [149].
Last but not least, enzymatic polymerization is more chemoselective than chemical polymerization as witnessed, for instance, by the successful polymerization of functionalized lactones bearing unsaturations and epoxides (Fig. 31) [150].
Functionalized lactones chemoselectively polymerized by enzymes [150]
Many of these substituted lactones possess at least one chiral center and are thus present as a mixture of stereoisomers. The simplest case is based on the polymerization of lactones with one chiral center, which is thus a mixture of two R and S enantiomers. These stereochemical aspects have been completely overlooked in many works: the polymerization is carried out on a racemic mixture and no stereo selectivity is reported or even discussed. It is worth pointing out that several metal-based catalysts were successfully designed for the stereoselective polymerization of lactide, even though their description is beyond the scope of this review. Nevertheless, a few examples dealing with the stereoselective polymerization of cyclic monoesters are reported. For instance, Carpentier reported the stereoselective polymerization of racemic β-butyrolactone into syndiotactic poly(β-butyrolactone) by an yttrium-based catalyst (Fig. 32) [12, 151]. A chain-end control was proposed to account for the syndioselectivity [134]. Later, this syndiotactic polymerization was extended to 4-(but-3-en-1-yl)-β-butyrolactone [152].
Interestingly, enzymes are chiral catalysts and their potential for enantioselective polymerization has been investigated [93]. Several examples are reported where a racemic mixture of lactones is polymerized by enzymatic polymerization to afford the corresponding optically active polyester [93]. For instance, lipase CA (Novozym 435) catalyses the ROP of racemic 4-methyl-ε-caprolactone and 4-ethyl-ε-caprolactone in bulk at 45 °C and 60 °C to afford (S)-enriched poly(4-methyl-ε-caprolactone) and poly(4-ethyl-ε-caprolactone) with an enantiomeric purity higher than 95% [153].
4 Macromolecular Engineering by ROP of Lactones
A change of architecture is another route that enables diversification of the properties of aliphatic polyesters. This review will focus on star-shaped, graft, macrocyclic, and crosslinked aliphatic polyesters. It must be noted that the ROP of lactones has been combined with several other polymerization mechanisms such as ROP of other heterocyclic monomers, ionic polymerization, ROMP, and radical polymerization. Nevertheless, this review will not cover these examples and will focus on polymers exclusively made up of poly(lactone)s.
4.1 Star-Shaped Polyesters
Star-branched polyesters exhibit unique properties such as lower melt viscosities, lower crystallinity, and smaller hydrodynamic volume in solution by comparison with their linear counterparts. Two general strategies are possible for their synthesis: arm-first and core-first. The arm-first method is based on the reaction of living chains with multifunctional electrophiles carrying at least three reacting groups. The core-first method relies on the initiation of the polymerization by a multifunctional (>2) initiator.
An example of the synthesis of a three-arm star-shaped PCL by the arm-first method is shown in Fig. 33 [154]. First, the arms were synthesized by the ROP of εCL initiated by aluminum alkoxides. Then, the living polymers were reacted with a three-functional terminating agent, i.e., benzene-1,3,5-tricarbonyl trichloride. The implementation of this approach implies the coupling of chains. However, this reaction can be kinetically difficult to carry out, especially when the reactive sites at the origin of the coupling are buried, depending upon the conformation adopted by the polyester chains. As a rule, as the molar mass of the arms increases, the steric hindrance increases at the expense of the efficiency of the coupling reaction. In the example shown in Fig. 33, the molar mass is limited to 10,000 g/mol. Moreover, the steric hindrance will also increase when star-shaped polyesters with a higher number of arms are targeted. A non-quantitative coupling reaction would result in the formation of a mixture of linear and star-shaped polyesters with a variable number of arms. Unfortunately, the fractionation of this mixture to obtain pure well-defined star-shaped polyesters is a very tedious or even impossible.
The core-first approach is based on the initiation of polymerization by a multifunctional initiator. The number of arms is then defined by the number of functional units present on the core. In order to have a good control of the molecular structure of star-shaped polyesters, the initiation must be quantitative and fast. It is also mandatory to avoid possible side-reactions between the initiating species on the core.
To this end, a very widely used approach is ROP initiated by polyols (at least triols) in the presence of tin(II) bis-(2-ethylhexanoate) [155, 156]. By implementing this technique, alcohols are dormant species and have to be activated by reaction with tin(II) bis-(2-ethylhexanoate) into tin alkoxides to initiate or to propagate the polymerization. The alcohols are thus not activated at the same time and no side-reactions between them are observed. Besides, it is more appropriate to initiate the polymerization using polyols with all alcohols exhibiting a similar reactivity. Thus, polyols made up exclusively of primary alcohols are preferred, even though examples of initiators made up of a mixture of primary and secondary alcohols such as glycerol [157] are also reported. Some examples of initiators are reported in Fig. 34. A very nice example was provided by Hawker and Hedrick, who reported the synthesis of a 48-arm star-shaped PCL using a dendritic polyester functionalized by 48 alcohols [158]. It is worth noting that the initiation turned out to be less efficient than that of the corresponding hyperbranched polyester.
Multifunctional initiators made up of metal alkoxides rather than alcohols have been less used for the synthesis of star-shaped polyesters than have the tin(II) bis-(2-ethylhexanoate)/alcohol system. Nevertheless, Kricheldorf initiated the polymerization of εCL using a spiro-cyclic tin(IV) alkoxide to obtain a tin-containing height-shaped polyester whose final hydrolysis resulted in the formation of a star-shaped polyester (Fig. 35) [25, 159–161].
Interestingly, Hedrick and coworkers reported a metal-free approach for the synthesis of star-shaped copolymers. They synthesized star-shaped PCLs by the ROP of εCL initiated by polyols in the presence of unencumbered N-heterocyclic carbenes [87].
Finally, Lecomte and coworkers reported the synthesis of mikto-arm star-shaped aliphatic polyesters by implementing a strategy based on click chemistry (Fig. 36) [162]. Firstly, the polymerization of εCL was initiated by a diol bearing an alkyne function. The chain-ends were protected from any further undesired reaction by the esterification reaction with acetyl chloride. The alkyne was then reacted with 3-azidopropan-1-ol. The hydroxyl function located at the middle of the chain was then used to initiate the ROP of εCL and γ-bromo-ε-caprolactone. Finally, pendant bromides were reacted successfully with sodium azide and then with N,N-dimethylprop-2-yn-1-amine to obtain pendant amines. Under acidic conditions, pendant amines were protonated and the polymer turned out to exhibit amphiphilic properties.
4.2 Comb-Shaped and Graft Polyesters
Although many examples of syntheses of graft copolymers by the combination of the ROP of lactones with other polymerization mechanisms have been reported, there are only a few examples dealing with the synthesis of comb-shaped and graft polymers exclusively made up of poly(lactone)s [163]. Three main strategies are known for the synthesis of comb-shaped and graft polymers. The first approach, known as the “grafting through” process, relies on the polymerization of macromonomers, i.e., chains end-capped by a polymerizable moiety. In the second approach, the “grafting onto” process, end-reactive chains are coupled onto a mutually reactive polymer backbone. However, the coupling of chains might be kinetically difficult, depending on the conformation adopted by chains in the solvent used for the reaction and on the molar mass of the chains. Besides, the coupling is not quantitative so a mixture of polyesters is obtained, and the purification can be challenging. In the third approach, the “grafting from” process, graft copolymers can be prepared by initiating the polymerization using a macroinitiator. For instance, this approach was implemented by Jérôme and coworkers who reported the polymerization of εCL initiated by a polyester bearing pendant hydroxyl groups (Fig. 37) [137]. The macroinitiator was synthesized by the ROP of εCL, 5-triethylsilyloxy-ε-caprolactone, and 1,4,8-trioxaspiro[4.6]undecan-9-one (TOSUO), followed by the acetylation of the chain-end and the deprotection of the silyl ethers (Fig. 37) [137]. It is worth noting that the further conversion of the ketal groups into hydroxyl functions prone to initiate the ROP of a second monomer was carried out in order to synthesize heterograft copolyesters [137].
4.3 Hyperbranched Polymers
The interest in synthesizing highly branched aliphatic polyesters is driven by their unique mechanical and rheological properties. Dendrimers (from the Greek “dendra” for tree) are globular macromolecules that are characterized by both a multiplicity of reactive chain-ends and a highly branched structure, in which all bonds converge to a focal point or core. Dendrimers have a very regular structure and are synthesized step-by-step by convergent or divergent strategies. Hyperbranched polymers have a less regular structure but their synthesis is easier. The ROP of lactones was implemented to synthesize hyperbranched polyesters. The strategy relies on the polymerization of an ABx inimer. An inimer is a molecular made up of a unit prone to initiate the polymerization (A) and a polymerizable unit (B). Typical ABx inimer precursors of hyperbranched aliphatic polyesters are lactones substituted by unprotected alcohols, where x stands for the number of hydroxyl groups.
Fréchet and coworkers reported a first example of the synthesis of a hyperbranched aliphatic polyester by polymerization of an AB inimer, i.e., 4-(hydroxyethyl)-ε-caprolactone [164]. It is worth noting that the synthesis of this inimer is quite long. Since this pioneering work, other teams have reported the polymerization of a series of AB [165–167] and AB2 [168] inimers, which are shown in Fig. 38. The polymerization of these inimers can be carried out in the absence or presence of a comonomer such as εCL [167].
4.4 Macrocycles
There are only a few studies dealing with cyclic aliphatic polyesters owing to their difficult synthesis [169]. Until recently, most works were based on ring-expansion polymerization. In the pioneering work of Kricheldorf and coworkers, cyclic aliphatic polyesters were synthesized by the ROP of lactones initiated by cyclic tin(IV) alkoxides, typically 2,2-dibutyl-2-stanna-1,3-dioxepane (DSDOP) (Fig. 39) [24, 170]. Interestingly, no high dilution is needed and the polymerization can even be carried out in bulk. Unfortunately, the macrocyclic polyesters synthesized by this technique contain hydrolytically sensitive endocyclic tin–oxygen bonds and the hydrolysis brings about an opening of the macrocycle (Fig. 39). The hydrolytic stability was improved by end-capping the cycles with 1,3-dihtian-2-one (Fig. 39) [171]. Another approach relies on the insertion of a few lactones bearing pendant acrylic units, followed by an intramolecular crosslinking by UV irradiation (Fig. 39) [172].
Very recently, Hedrick and coworkers reported a very original process for the synthesis of macrocyclic poly(β-lactone)s by ring-expansion polymerization of β-lactones initiated by metal-free spirocyclic initiators [173]. Indeed, a saturated carbene, i.e., 1,3-dimesitylimidazolin-2-ylidene, mediated the ROP of β-lactones to afford the corresponding cyclic polyester through the mechanism shown in Fig. 40. The carbene reacted with the monomer to afford a spirocyclic adduct. This adduct turned out to be the initiator of the polymerization. The reaction with carbon disulfide (CS2) enabled release of the cyclic aliphatic polyester by an unclear mechanism. As far as the polymerization of optically active (R)-β-butyrolactone was concerned, cyclic poly[(R)-β-butyrolactone] was obtained with retention of configuration. This experimental observation implies that the polymerization took place by the scission of the acyl–oxygen bond and not the alkyl–oxygen bond, which would result in an inversion of configuration.
One of the more usual strategies aiming at synthesizing macrocycles is based on the direct coupling of the two chain-ends of a linear chain [169]. Undesired chain-extension reactions, which compete with the desired cyclization of a single chain, can be disfavored by using ultrahigh dilution conditions. Unlike ring-expansion polymerization, the entanglements of the chains results, after cyclization, in the formation of permanent knots and catenanes. Several reactions were investigated for carrying out the cyclization of the linear precursor. For poly(lactone)s, the coupling was carried out by ring-closing metathesis [174], ring-closing enyne metathesis [174], and click copper-catalyzed Huisgen’s cycloaddition [174–176]. Of these methods, the click copper-catalyzed Huisgen’s cycloaddition appeared a particularly efficient reaction [174]. For instance, Grayson and coworkers synthesized macrocyclic PCL by the procedure shown in Fig. 41 [176]. Firstly, 3-azidopropanol initiated the polymerization of εCL in the presence of tin octoate. Secondly, the hydroxylated chain-end of the resulting PCL was esterified by 4-pentynoic anhydride in pyridine and in the presence of 4-(dimethylamino) pyridine (DMAP) as a catalyst. Finally, the coupling of the chain-ends was carried out in dichloromethane in the presence of copper bromide as a catalyst and N,N,N′,N″,N″-pentamethyldiethylene triamine (PMDETA) as a ligand. Although more work is needed to assess the influence of the cyclic topology on properties, it is worth pointing out that the cyclic architectures yield a delay in the loss of molar mass during the acid-catalyzed degradation.
4.5 Networks
The crosslinking of polyesters is a widely used process for tailoring key properties such as crystallinity, biodegradation rate, and mechanical behavior. Interestingly, chemically crosslinked PCL exhibits remarkable shape-memory properties [177, 178]. One can distinguish intra- and intermolecular crosslinking. A nice example is provided by Hedrick and coworkers who implemented a thermal radical crosslinking of PCL bearing pendant acrylates [179]. Under ultradilute conditions, intramolecular crosslinking took place and nanoparticles with very small sizes were then prepared. Conversely, under concentrated conditions, the crosslinking is intermolecular and a three-dimensional network can then be obtained. Many other works report the synthesis of networks by the crosslinking of polyesters bearing pendant unsaturations. The crosslinking is usually carried out thermally in the presence of a source of radicals [179] or photochemically [133, 180]. A very similar approach relies on the crosslinking of polyesters bearing double bonds present directly inside the chains [181]. A simpler route towards crosslinked aliphatic polyesters relies on the synthesis and crosslinking of linear aliphatic polyesters end-capped by a polymerizable unsaturation at each chain-end [182]. The number of reactive chain-ends can be increased by using star-shaped rather than linear polyesters [183].
Another approach for the synthesis of networks relies on the polycondensation of multifunctionalized polyesters with the appropriate multifunctionalized agent, provided that one of the partner is at least tri-functionalized. Toward this end, several reaction have been reported, such as the Michael addition of amines onto acrylates [184], the coupling of ketones and oxyamines [185], the click copper(II)-catalyzed azide–alkyne cycloaddition [186], and esterification reactions [25, 159, 187]. Interestingly, if esterification reactions are used, the crosslinks are then degradable.
Albertsson and coworkers proposed another strategy based on the ROP of tetrafunctional bis-(ε-caprolactones), e.g., using 4,4′-bioxepane-7,7′-dione and 5,5′-propane-2,2-diyldioxepan-2-one as crosslinking agents (Fig. 42) [188, 189]. Networks were obtained by the ROP of lactones in the presence of these bis-lactones. Another approach was to transesterify aliphatic polyesters in the presence of the bis-lactone [188, 189]. It is worth noting than other bis-heterocycles polymerizable by ROP have been used as crosslinking agent, such as bis-carbonates [190] or lactones bearing pendant epoxides [178].
5 Conclusions
Owing to the outstanding control imparted to the ROP of lactones and the remarkable properties of biodegradable aliphatic polyesters, steadily increasing attention has been paid to its development over the last few decades. Among the various processes investigated so far, coordination polymerization initiated by suitable metal alkoxides turned out to be particularly efficient. A wide range of lactones, functionalized or not, have been synthesized and polymerized. The living character of the ROP of lactones has been exploited to synthesize aliphatic polyesters with various architectures. Although this field can be considered as mature, some important issues remained to be addressed. The toxicity of tin- and aluminum-based initiators is a severe limitation for many applications and especially for biomedical applications because metallic remnants are very difficult to get rid of. These drawbacks have prompted researchers to investigate new efficient processes based on less toxic metals than tin and aluminum, and on metal-free polymerization techniques. In this respect, the recent advent of efficient organocatalysts is very promising. Although more work is needed to assess the toxicity of these catalysts, they will obviously be more and more widely used in the future. Another challenge will be to search for more efficient processes for the synthesis of lactones, especially substituted and functionalized lactones. Although εCL is synthesized and polymerized at the industrial scale, the syntheses of many other functionalized lactones remain too expensive for many applications. Special attention will have to be paid to their synthesis by green processes, and ideally from renewable resources.
References
Sanda F, Endo T (2001) Radical ring-opening polymerization. J Polym Sci A Polym Chem 39:265–276
Bailey WJ (1985) Free-radical ring-opening polymerization. Polym J 17:85–95
Agarwal S (2010) Chemistry, chances and limitations of the radical ring-opening polymerization of cyclic ketene acetals for the synthesis of degradable polyesters. Polym Chem 1:953–954
Bailey WJ, Ni Z, Wu S-R (1982) Synthesis of poly-ε-caprolactone via a free radical mechanism. Free radical ring opening polymerization of 2-methylene-1,3-dioxepane. J Polym Sci A Polym Chem 20:3021–3030
Undin J, Plikk P, Finne-Wistrand A, Albertsson A-C (2010) Synthesis of amorphous aliphatic polyester-ether homo- and copolymers by radical polymerization of ketene acetals. J Polym Sci A Polym Chem 48:4965–4973
Jin S, Gonsalves KE (1997) A study of the mechanism of the free-radical ring-opening polymerization of 2-methylene-1,3-dioxepane. Macromolecules 30:3104–3106
Stridsberg KM, Ryner M, Albertsson A-C (2002) Controlled ring-opening polymerization: polymers with designed macromolecular architecture. Adv Polym Sci 157:42–139
Penczek S, Cypryk M, Duda A, Kubisa P, Slomkowski S (2007) Living ring-opening polymerizations of heterocyclic monomers. Prog Polym Sci 32:247–282
van Natta FJ, Hill JW, Carothers WH (1934) Studies of polymerization and ring formation. XXIII. ε-caprolactone and its polymers. J Am Chem Soc 56:455–457
Woodruff MA, Hutmacher W (2010) The return of a forgotten polymer-polycaprolactone in the 21st century. Prog Polym Sci 35:1217–1256
Sinha VR, Bansal K, Kaushik R, Kumria R, Trehan A (2004) Poly-ε-caprolactone microspheres and nanospheres: an overview. Int J Pharm 278:1–23
Carpentier J-F (2010) Discrete metal catalysts for stereoselective ring-opening polymerization of chiral racemic β-lactones. Macromol Rapid Commun 31:1696–1705
Lu J, Tappel RC, Nomura CT (2009) Mini-review: biosynthesis of poly(hydroxyalkanoates). J Macromol Sci, Part C: Polym Rev 49:226–248
Duda A, Libiszowski J, Mosnacek J, Penczek S (2005) Copolymerization of cyclic esters at the living polymer-monomer equilibrium. Macromol Symp 226:109–119
Coulembier O, Degée Ph, Hedrick JL, Dubois Ph (2006) From controlled ring-opening polymerization to biodegradable aliphatic polyester: especially poly(β-malic acid) derivatives. Prog Polym Sci 31:723–747
Szwarc M (1956) Living polymers. Nature 178:1168–1169
Lecomte Ph, Jérôme R (2004) Recent developments in controlled/living ring opening polymerization. In: Kroschwitz J (ed) Encyclopedia of polymer science and technology. Wiley, Hoboken, pp 547–565
Hamitou A, Jérôme R, Hubert AJ, Teyssié P (1973) A new catalyst for the ring-opening polymerization of lactones to polyesters. Macromolecules 6:651–652
Ouhadi T, Hamitou R, Jérôme R, Teyssié P (1976) Soluble bimetallic μ-oxoalkoxides. 8. Structure and kinetic behavior of the catalytic species in unsubstituted lactone ring-opening polymerization. Macromolecules 9:927–931
Dubois P, Jérôme R, Teyssié P (1989) Macromolecular engineering of polylactones and polylactides. I. End-functionalization of poly-ε-caprolactone. Polym Bull 22:475–482
Ropson N, Dubois P, Jérôme R, Teyssié P (1995) Macromolecular engineering of polylactones and polylactides. 20. Effect of monomer, solvent, and initiator on the ring-opening polymerization as initiated with aluminum alkoxides. Macromolecules 28:7589–7598
Duda A, Penczek S (1991) Anionic and pseudoanionic polymerization of ε-caprolactone. Makromol Chem, Macromol Symp 42/43:135–143
Kricheldorf HR, Stricker A, Langanke D (2001) Polylactones, 50. The reactivity of cyclic and noncyclic dibutyltin bisalkoxides as initiators in the polymerization of lactones. Macromol Chem Phys 202:2525–2534
Kricheldorf HR, Eggerstedt S (1998) Macrocycles 2. Living macrocyclic polymerization of ε-caprolactone with 2,2-dibutyl-2-stanna-1,3-dioxepane as initiator. Macromol Chem Phys 199:283–290
Kricheldorf HR (2004) Biodegradable polymers with variable architectures via ring-expansion polymerization. J Polym Sci A Polym Chem 42:4723–4742
McLain SJ, Drysdale NE (1992) Living ring-opening polymerization of ε-caprolactone by yttrium and lanthanide alkoxides. Polymer Preprints, American Chemical Society 33(1):174–175
Shen Y, Shen Z, Zhang Y, Yao K (1996) Novel rare earth catalysts for the living polymerization and block copolymerization of ε-caprolactone. Macromolecules 29:8289–8295
Yamashita M, Takemoto Y, Ihara E, Yasuda H (1996) Organolanthanide-initiated living polymerization of ε-caprolactone, δ-valerolactone, and β-propionolactone. Macromolecules 29:1798–1806
Stevels WM, Ankoné MJK, Dijkstra PJ, Feijen J (1996) A versatile and highly efficient catalyst system for the preparation of polyesters based on lanthanide tris(2,6-di-tert-butylphenolate)s and various alcohols. Macromolecules 29:3332–3333
Stevels WM, Ankoné MJK, Dijkstra PJ, Feijen J (1996) Kinetics and mechanism of ε-caprolactone polymerization using yttrium alkoxides as initiators. Macromolecules 29:8296–8303
Martin E, Dubois P, Jérôme R (2000) Controlled ring-opening polymerization of ε-caprolactone promoted by “in situ” formed yttrium alkoxides. Macromolecules 33:1530–1535
Martin E, Dubois P, Jérôme R (2003) "In situ" formation of yttrrium alkoxides: a versatile and efficient catalyst for the ROP of ε-caprolactone. Macromolecules 36:5934–5941
Tortosa K, Hamaide T, Boisson C, Spitz R (2001) Homogeneous and heterogeneous polymerization of ε-caprolactone by neodymium alkoxydes prepared in situ. Macromol Chem Phys 202:1156–1160
Save M, Schappacher M, Soum A (2002) Controlled ring-opening polymerization of lactones and lactides initiated by lanthanum isopropoxide, 1 general aspects and kinetics. Macromol Chem Phys 203:889–899
Guillaume SM, Schappacher M, Soum A (2003) Polymerization of ε-caprolactone by Nd(BH4)3(THF)3: synthesis of hydroxytelechelic poly(ε-caprolactone). Macromolecules 36:54–60
Palard I, Soum A, Guillaume SM (2005) Rare earth metal tris(borohydride) complexes as initiators for ε-caprolactone polymerization: general features and IR investigations of the process. Macromolecules 36:54–60
Kowalski A, Duda A, Penczek S (1998) Kinetics and mechanism of cyclic esters polymerization initiated with tin(II) octoate, 1 Polymerization of ε-caprolactone. Macromol Rapid Commun 19:567–572
Kowalski A, Duda A, Penczek S (2000) Kinetics and mechanism of cyclic esters polymerization initiated with tin(II) octoate. 3. Polymerization of L,L-dilactide. Macromolecules 33:7359–7370
Kowalski A, Duda A, Penczek S (2000) Mechanism of cyclic ester polymerization initiated with tin(II) octoate. 2. Macromolecules fitted with tin(II) alkoxide species observed directly in MALDI-TOF spectra. Macromolecules 33:689–695
Majerska K, Duda A, Penczek S (2000) Kinetics and mechanism of cyclic esters polymerization initiated with tin(II) octoate, 4. Influence of proton trapping agents on the kinetics of ε-caprolactone and L,L-dilatce polymerization. Macromol Rapid Commun 21:1327–1332
Möller M, Kange R, Hedrick JL (2000) Sn(OTf)2 and Sc(OTf)3: efficient and versatile catalysts for the controlled polymerization of lactones. J Polym Sci A Polym Chem 38:2067–2074
Möller M, Nederberg F, Lim LS, Kange R, Hawker CJ, Hedrick JL, Gu Y, Shah R, Abbott NL (2000) Sn(OTf)2 and Sc(OTf)3: efficient and versatile catalysts for the controlled polymerization of lactones. J Polym Sci A Polym Chem 38:2067–2074
Nomura N, Taira A, Tomioka T, Okada M (2000) A catalytic approach for cationic living polymerization: Sc(OTf)3-catalyzed ring-opening polymerization of lactones. Macromolecules 33:1497–1499
Libiszowki J, Kowalski A, Duda A, Penczek S (2002) Kinetics and mechanism of cyclic esters polymerization initiated with covalent metal carboxylates, 5. End-group studies in the model ε-caprolactone and L,L-dilactide/tin(II) and zinc octoate/butyl alcohol systems. Macromol Chem Phys 203:1694–1701
Kowalski A, Libiszowski J, Majerska K, Duda A, Penczek S (2007) Kinetics and mechanism of ε-caprolactone and L,L-lactide polymerization coinitiated with zinc octoate or aluminum acetylacetonate: The next proofs for the general alkoxide mechanism and synthetic applications. Polymer 48:3952–3960
Oshimura M, Takasu A (2010) Controlled ring-opening polymerization of ε-caprolactone catalyzed by rare-earth perfluoroalkanesulfonates and perfluoroalkanesulfonimides. Macromolecules 43:2283–2290
Stjerndahl A, Wistrand AF, Albertsson A-C (2007) Industrial utilization of tin-initiated resorbable polymers: synthesis on a large scale with a low amount of initiator residue. Biomacromolecules 8:937–940
Mingotaud A-F, Dargelas F, Cansell F (2000) Cationic and anionic ring-opening polymerization in supercritical CO2. Macromol Symp 153:77–86
Mingotaud A-F, Cansell F, Gilbert N, Soum A (1999) Cationic and anionic ring-opening polymerization in supercritical CO2. Preliminary results. Polym J 31:406–410
Stassin F, Halleux O, Jérôme R (2001) Ring-opening polymerization of ε-caprolactone in supercritical carbon dioxide. Macromolecules 34:775–781
Stassin F, Jérôme R (2003) Effect of pressure and temperature upon tin alkoxide-promoted ring-opening polymerisation of ε-caprolactone in supercritical carbon dioxide. Chem Commun:232–233
Bergeot V, Tassaing T, Besnard M, Cansell T, Mingotaud A-F (2004) Anionic ring-opening polymerization of ε-caprolactone in supercritical carbon dioxide: parameters influencing the reactivity. J Supercrit fluids 28:249–261
Stassin F, Jérôme R (2004) Contribution of supercritical CO2 to the preparation of aliphatic polyesters and materials thereof. Macromol Symp 217:135–146
Shueh ML, Wang Y-S, Huang B-H, Kuo C-Y, Lin C-C (2004) Reactions of 2,2’-methylenebis(4-chloro-6-isopropyl-3-methylphenol) and 2,2’-ethylenebis(4,6-di-tert-butylphenol)with MgnBr2: efficient catalysts for the ring-opening polymerization of ε-caprolactone and L-lactide. Macromolecules 37:5155–5162
Yu T-L, Wu C-C, Chen C-C, Huang B-H, Wu J, Lin CC (2005) Catalysts for the ring-opening polymerization of ε-caprolactone and L-lactide and the mechanistic study. Polymer 46:5909–5917
Zhong Z, Dijkstra PJ, Birg C, Westerhausen M, Feijen J (2001) A novel and versatile calcium-based initiator system for the ring-opening polymerization of cyclic esters. Macromolecules 34:3863–3868
Westerhausen M, Schneiderbauer S, Kneifel AN, Söltl Y, Mayer P, Nöth H, Zhong Z, Dijkstra PJ, Feijen J (2003) Organocalcium compounds with catalytic activity for the ring-opening polymerization of lactones. Eur J Inorg Chem:3432–3439
Martin E, Dubois P, Jérôme R (2003) Preparation of supported yttrium alkoxides as catalysts for the polymerization of lactones and oxirane. J Polym Sci A Polym Chem 41:569–578
Martin E, Dubois P, Jérôme R (2003) Polymerization of ε-caprolactone initiated by Y alkoxide grafted onto porous silica. Macromolecules 36:7094–7099
Miola-Delaite C, Colomb E, Pollet E, Hamaide T (2000) Macromol Symp 153:275–286
Hofman A, Szymanski R, Slomkowski S, Penczek S (1984) Structure of active species in the cationic polymerization of β-propiolactone and ε-caprolactone. Makromol Chem 185:655–667
Hoffman A, Szymanski R, Slomkowski S, Penczek S (1984) Structure of active species in the cationic polymerization of β-propiolactone and ε-caprolactone. Makromol Chem 185:655–667
Kricheldorf HR, Jonte JM, Dunsing R (1986) Polylactones. 7. The mechanism of cationic polymerization of β-propionolactone and ε-caprolactone. Makromol Chem 187:771–785
Slomkowski S, Szymanski R, Hofman A (1985) Formation of the intermediate cyclic six-membered oxonium ion in the cationic polymerization of β-propiolactone initiated with CH3CO+SbF -6 . Makromol Chem 186:2283–2290
Albertsson A-C, Palmgren R (1996) Cationic Polymerization of 1,5-dioxepan-2-one with Lewis acids in bulk and solution. J Macromol Sci: Pure Appl Chem A33:747–758
Abraham GA, Gallardo A, Lozano AE, San RJ (2000) ε-caprolactone/ZnCl2 complex formation: characterization and ring-opening polymerization mechanism. J Polym Sci A Polym Chem 38:1355–1365
Kricheldorf HR, Sumbél MV (1988) Polylactones, 15. Reactions of δ-valerolactone and ε-caprolactone with acidic metal bromides. Makromol Chem 185:317–331
Shibasiki Y, Sanada H, Yokoi M, Sanda F, Endo T (2000) Activated monomer cationic polymerization of lactones and the application to well-defined block copolymer synthesis with seven-membered cyclic carbonate. Macromolecules 33:4316–4320
Lou X, Detrembleur C, Jérôme R (2002) Living cationic polymerization of δ-valerolactone and synthesis of high molecular weight homopolymer and asymmetric telechelic and block copolymer. Macromolecules 35:1190–1195
Basko M, Kubisa P (2006) Cationic copolymerization of ε-caprolactone and L,L-lactide by an activated monomer mechanism. J Polym Sci A Polym Chem 44:7071–7081
Gazeau-Bureau S, Delcroix D, Martin-Vaca B, Bourissou D, Navarro C, Magnet S (2008) Organo-catalyzed ROP of ε-caprolactone: methanesulfonic acid competes with trifluoromethanesulfonic acid. Macromolecules 41:3782–3784
Kakuchi R, Tsuji Y, Chiba K, Fuchise K, Sakai R, Satoh T, Kakuchi T (2010) Controlled/living ring-opening polymerization of δ-valerolactone using triflylimide as an efficient cationic organocatalyst. Macromolecules 43:7090–7094
Oshimura M, Tang T, Takasu A (2011) Ring-opening polymerization of ε-caprolactone using perfluoroalkanesulfonates and perfuloroalkanesulfonimides as organic catalysts. J Polym Sci A Polym Chem 49:1210–1218
Casas J, Persson PV, Iversen T, Cordova A (2004) Direct organocatalytic ring-opening polymerizations of lactones. Adv Synth Catal 346:1087–1089
Persson PV, Casas J, Iversen T, Cordova A (2006) Direct organocatalytic chemoselective synthesis of a dendrimer-like star polyester. Macromolecules 39:2819–2822
Sanda F, Sanada H, Shibasaki Y, Endo T (2002) Star polymer synthesis from ε-caprolactone utilizing polyol/protonic acid initiator. Macromolecules 35:680–683
Zeng F, Lee H, Chidiac M, Allen C (2005) Synthesis and characterization of six-arm star poly(δ-valerolactone)-block-methoxy poly(ethylene glycol) copolymers. Biomacromolecules 6:2140–2149
Wilson BC, Jones CW (2004) A recoverable, metal-free catalyst for the green polymerization of ε-caprolactone. Macromolecules 37:9709–9714
Liu J, Liu L (2004) Ring-opening polymerization of ε-caprolactone initiated by natural amino acids. Macromolecules 37:2674–2676
Löfgren A, Albertsson A-C, Dubois P, Jérôme R (1995) Recent advances in ring-opening polymerization of lactones and related compounds. J Macromol Sci, Part C: Rev Macromol Chem Phys 35:379–418
Kricheldorf HR, Garaleh M, Schwarz G (2005) Tertiary amine-initiated zwitterionic polymerization of pivalolactone - a reinvestigation by means of MALDI-TOF mass spectrometry. J Macromol Sci, Part A: Pure Appl Chem 42:139–148
Kamber NE, Jeong W, Waymouth RM, Pratt RC, Lohmeijer BGG, Hedrick JL (2007) Organocatalytic ring-opening polymerization. Chem Rev 107:5813–5840
Kiesewetter MK, Shin EJ, Hedrick JL, Waymouth RM (2010) Organocatalysis: opportunities and challenges for polymer synthesis. Macromolecules 43:2093–2107
Connor EF, Nyce GW, Myers M, Möck A, Hedrick JL (2002) First example of N-heterocyclic carbenes as catalysts for living polymerization: organocatalytic ring-opening polymerization of cyclic esters. J Am Chem Soc 124:914–915
Nyce GW, Glauser T, Connor EF, Möck A, Waymouth RM, Hedrick JL (2003) In situ generation of carbenes: a general and versatile platform for organocatalytic living polymerization. J Am Chem Soc 125:3046–3056
Coulembier O, Lohmeijer BGG, Dove AP, Pratt RC, Mespouille L, Culkin DA, Benight SJ, Dubois P, Waymouth RM, Hedrick J (2006) Alcohol adducts of N-heterocyclic carbenes: latent catalysts for the thermally –controlled living polymerization of cyclic esters. Macromolecules 2006:5617–5628
Kamber NE, Jeong W, Gonzales S, Hedrick JL, Waymouth RM (2009) N-Heterocyclic carbenes for the organocatalytic ring-opening polymerization of ε-caprolactone. Macromolecules 42:1634–1639
Pratt RC, Lohmeijer BGG, Long DA, Waymouth RM, Hedrick JL (2006) Triazabicyclodecene: a simple bifunctional organocatalyst for acyl transfer and ring-opening polymerization of cyclic esters. J Am Chem Soc 128:4556–4557
Lohmeijer BGG, Pratt RC, Leibfarth F, Logan JW, Long DA, Dove AP, Nederberg F, Choi J, Wade C, Waymouth RM, Hedrick JL (2006) Guanidine and amidine organocatalysts for ring-opening polymerization of cyclic esters. Macromolecules 39:8574–8583
Zhang L, Nederberg F, Pratt RC, Waymouth RM, Hedrick JL, Wade CG (2007) Phosphazene bases: a new category of organocatalysts for the living ring-opening polymerization of cyclic esters. Macromolecules 40:4154–4158
Uyama H, Kobayashi S (1993) Enzymatic ring-opening polymerization of lactones catalyzed by lipase. Chem Lett:1149–1150
Knani D, Gutman AL, Kohn DH (1993) Enzymatic polyesterification in organic media. Enzyme-catalyzed synthesis of linear polyesters. I. Condensation polymerization of linear hydroxyester. II. Ring-opening polymerization of ε-caprolactone. J Polym Sci A Polym Chem 31:1221–1232
Kobayashi S, Makino A (2009) Enzymatic polymer synthesis: an opportunity for green polymer chemistry. Chem Rev 109:5288–5353
Gross RA, Kumar A, Kalra B (2001) Polymer synthesis by in vitro enzyme catalysis. Chem Rev 101:2097–2124
Varma IK, Albertsson A-C, Rajkhowa R, Srivistava RK (2005) Enzyme catalyzed synthesis of polyesters. Prog Polym Sci 30:949–981
Albertsson A-C, Srivastava RK (2008) Recent developments in enzyme-catalyzed ring-opening polymerization. Adv Drug Deliv Rev 60:1077–1093
Takamoto T, Uyama H, Kobayashi S (2001) Lipase-catalyzed synthesis of aliphatic polyesters in supercritical carbon dioxide. e-Polymers no.004
Loeker FC, Duxbury CJ, Kumar R, Gao W, Gross RA, Howdle SM (2004) Enzyme-catalyzed ring-opening polymerization of ε-caprolactone in supercritical carbon dioxide. Macromolecules 37:2450–2453
Duda A, Kowalski A, Penczek S, Uyama H, Kobayashi S (2002) Kinetics of the ring-opening polymerization of 6-, 7-, 9-, 12-, 13-, 16-, and 17-membered lactones. Comparison of chemical and enzymatic polymerizations. Macromolecules 35:4266–4270
Lou X, Detrembleur C, Jérôme R (2003) Novel aliphatic polyesters based on functional cyclic (di)esters. Macromol Rapid Commun 24:161–172
Ponsart S, Coudane J, Vert M (2000) A novel route to poly(ε-caprolactone)-based copolymers via anionic derivatization. Biomacromolecules 1:275–281
Gimenez S, Ponsart S, Coudane J, Vert M (2001) Synthesis, properties and in vitro degradation of carboxyl-bearing PCL. J Bioact Compat Polym 16:32–46
Ponsart S, Coudane J, Morgat J-L, Vert M (2001) Synthesis of 3H and fluorescence-labelled poly (dl-Lactic acid). J Labelled Comp Radiopharm 44:677–687
Ponsart S, Coudane J, McGrath J, Vert M (2002) Study of the grafting of bromoacetylated α-hydroxy-ω-methoxypoly(ethyleneglycol) onto anionically activated poly(ε-caprolactone). J Bioact Compat Polym 17:417–432
Nottelet B, Coudane J, Vert M (2006) Synthesis of an X-ray opaque biodegradable copolyester by chemical modification of poly(ε-caprolactone). Biomaterials 27:4948–4954
Mathisen T, Albertsson A-C (1989) Polymerization of 1,5-dioxepan-2-one. 1. Synthesis and characterization of the monomer 1,5-dioxepan-2-one and its cyclic dimer 1,5,8,12-tetraoxacyclotetradecane-2,9-dione. Macromolecules 22:3838–3842
Mathisen T, Masus K, Albertsson A-C (1989) Polymerization of 1,5-dioxepan-2-one. 2. Polymerization of 1,5-dioxepan-2-one and its cyclic dimer, including a new procedure for the synthesis of 1,5-dioxepan-2-one. Macromolecules 22:3842–3846
Löfgren A, Albertsson A-C, Dubois P, Jérôme R, Teyssié P (1994) Synthesis and characterization of biodegradable homopolymers and block copolymers based on 1,5-dioxepan-2-one. Macromolecules 27:5556–5562
Shirahama H, Mizuma K, Kawaguchi Y, Shomi M, Yasuda H (1993) Development of new biodegradable polymers. Kobunshi Ronbunshu 50:821–835
Shirahama H, Shomi M, Sakane M, Yasuda H (1996) Biodegradation of novel optically active polyesters synthesized by copolymerization of (R)-MOHEL with lactone. Macromolecules 29:4821–4828
Raquez J-M, Degée P, Narayan R, Dubois P (2000) “Coordination-insertion” ring-opening polymerization of 1,4-dioxan-2-one and controlled synthesis of diblock copolymers with ε-caprolactone. Macromol Rapid Commun 21:1063–1071
Raquez J-M, Degée P, Narayan R, Dubois P (2001) Some thermodynamic, kinetic, and mechanistic aspects of the ring-opening polymerization of 1,4-dioxan-2-one initiated by Al(OiPr)3 in bulk. Macromolecules 34:8419–8425
Kricheldorf HR, Damrau D-O (1998) Zn L-lactate-catalyzed polymerizations of 1,4-dioxan-2-one. Macromol Chem Phys 199:1089–1097
Trollsas M, Lee VY, Mecerreyes D, Löwenhielm P, Möller M, Miller RD, Hedrick JL (2000) Hydrophilic aliphatic polyesters: design, synthesis, and ring-opening polymerization of functional cyclic esters. Macromolecules 33:4619–4627
Kudoh R, Sudo A, Endo T (2009) Synthesis of eight-membered lactone having tertiary amine moiety by ring-expansion reaction of 1,3-benzoxazine and its anionic ring-opening polymerization behavior. Macromolecules 42:2327–2329
Feng Y, Knüfermann J, Klee D, Höcker H (1999) Lipase-catalyzed ring-opening polymerization of 3(S)-isopropylmorpholine-2,5-dione. Macromol Chem Phys 200:1506–1514
Feng Y, Klee D, Keul H, Höcker H (2000) Lipase-catalyzed ring-opening polymerization of morpholine-2,5-dione derivatives: a novel route to the synthesis of poly(ester amide)s. Macromol Chem Phys 201:2670–2675
Feng Y, Knüfermann J, Klee D, Höcker H (1999) Enzyme-catalyzed ring-opening polymerization of 3(S)-isopropylmorpholine-2,5-dione. Macromol Rapid Commun 20:88–90
Lou X, Detrembleur C, Lecomte P, Jérôme R (2001) Living ring-opening (co)polymerization of 6,7-dihydro-2(5H)-oxepinone into unsaturated aliphatic polyesters. Macromolecules 34:5806–5811
Lou X, Detrembleur C, Lecomte P, Jérôme R (2002) Controlled synthesis and chemical modification of unsaturated aliphatic (co)polyesters based on 6,7-dihydro-2(3H)-oxepinone. J Polym Sci A Polym Chem 40:2286–2297
Lou X, Detrembleur C, Lecomte P, Jérôme R (2002) Novel unsaturated ε-caprolactone polymerizable by ring-opening metathesis mechanisms. e-Polymers no 034
Pentzer EB, Gadzikwa T, Nguyen ST (2008) Substrate encapsulation: an efficient strategy for the RCM synthesis of unsaturated ε-lactones. Org Lett 10:5613–5615
Latere J-P, Lecomte P, Dubois P, Jérôme R (2002) 2-Oxepane-1,5-dione: a precursor of a novel class of versatile semicrystalline biodegradable (co)polyesters. Macromolecules 21:7857–7859
Lenoir S, Riva R, Lou X, Detrembleur C, Jérôme R, Lecomte P (2004) Ring-opening polymerization of α-chloro-ε-caprolactone and chemical modification of poly(α-chloro-ε-caprolactone) by atom transfer radical processes. Macromolecules 37:4055–4061
Liu X-Q, Wang M-X, Li Z-C, Li F-M (1999) Synthesis and ring-opening polymerization of α-chloromethyl-α-methyl-β-propiolactone. Macromol Chem Phys 200:468–473
Liu X-Q, Li Z-C, Du F-S, Li FM (1999) Ring-opening copolymerization of α-chloromethyl-α-methyl-β-propionolactone with ε-caprolactone. Macromol Rapid Commun 20:470–474
Detrembleur C, Mazza M, Halleux O, Lecomte P, Mecerreyes D, Hedrick JL, Jérôme R (2000) Ring-opening polymerization of γ-Bromo-ε-caprolactone: a novel route to functionalized aliphatic polyesters. Macromolecules 33:14–18
Mecerreyes D, Atthoff B, Boduch KA, Trollsas M, Hedrick JL (1999) Unimolecular combination of an atom transfer radical polymerization initiator and a lactone monomer as a route to new graft copolymers. Macromolecules 16:5175–5182
El Habnouni HS, Darcos V, Coudane J (2009) Synthesis and ring-opening polymerization of a new functional lactone, α-iodo-ε-caprolactone: a novel route to functionalized aliphatic polyesters. Macromol Rapid Commun 30:165–169
Parrish B, Quansah JK, Emrick T (2002) Functional polyesters prepared by polymerization of α-allyl(valerolactone) and its copolymerization with ε-caprolactone and δ-valerolactone. J Polym Sci A Polym Chem 40:1983–1990
Mecerreyes D, Miller RD, Hedrick JL, Detrembleur C, Jérôme R (2000) Ring-opening polymerization of 6-hydroxynon-8-enoic acid lactone: novel biodegradable copolymers containing allyl pendent groups. J Polym Sci A Polym Chem 38:870–875
Mecerreyes D, Humes J, Miller RD, Hedrick JL, Lecomte Ph, Detrembleur C, Jérôme R (2000) First example of an unsymmetrical difunctional monomer polymerizable by two living/controlled methods. Macromol Rapid Commun 21:779–784
Vaida C, Mela P, Keul H, Möller M (2008) 2D- and 3D-microstructured biodegradable polyester resins. J Polym Sci A Polym Chem 46:6789–6800
Ajellal N, Thomas CM, Carpentier J-F (2009) Functional syndiotacticpoly(β-hydroxyalkanoate)s via stereoselective ring-opening copolymerization of rac-β-butyrolactone and rac-allyl-β-butyrolactone. J Polym Sci A Polym Chem 47:3177–3189
Parrish B, Breitenkamp RB, Emrick T (2005) PEG- and peptide-grafted aliphatic polyesters by click chemistry. J Am Chem Soc 127:7404–7410
Pitt CG, Gu Z-H, Ingram P, Hendren RW (1987) The synthesis of biodegradable polymers with functional side chains. J Polym Sci A Polym Chem 25:955–966
Stassin F, Halleux O, Dubois P, Detrembleur C, Lecomte P, Jérôme R (2000) Ring-opening copolymerization of ε-caprolactone, γ-(triethylsilyloxy)- ε-caprolactone and γ-ethylene ketal-ε-caprolactone: a route to heterograft copolyesters. Macromol Symp 153:27–39
Gautier S, D’Aloia V, Halleux O, Mazza M, Lecomte P, Jérôme R (2003) Amphiphilic copolymers of ε-caprolactone and γ-substituted-ε-caprolactone. Synthesis and functionalization of poly(d,l-lactide) nanoparticles. J Biomater Sci Polym Ed 114:63–85
Bizzari R, Chiellini F, Solaro R, Chiellini E, Cammas-Marion S, Guerin P (2002) Synthesis and characterization of new malolactonate polymers and copolymers for biomedical applications. Macromolecules 35:1215–1223
Vert M (1998) Chemical routes to poly( β-malic acid) and potential applications to this water-soluble bioresorbable poly(β-hydroxy alkanoate). Polym Degrad Stab 59:169–175
Blanquer S, Tailhades J, Darcos V, Amblard M, Martinez J, Nottelet B, Coudane J (2010) Easy synthesis and ring-opening polymerization of 5-Z-Amino-δ-valerolactone: new degradable amino-functionalized (co)polyesters. J Polym Sci A Polym Chem 48:5891–5898
Flétier I, Le Borgne A, Spassky N (1990) Synthesis of functional polyesters derived from serine. Polym Bull 24:349–353
Yan J, Zhang Y, Xiao Y, Zhang Y, Lang MD (2010) Novel poly(ε-caprolactone)s bearing amino groups: Synthesis, characterization and biotinylation. React Funct Polym 70:400–407
Mahmud A, Xiong X-B, Lavasanifar A (2006) Novel self-associating poly(ethylene oxide)-block-poly(ε-caprolactone) block copolymers with functional side groups on the polyester block for drug delivery. Macromolecules 39:9414–9428
Mahmud A, Xiong X-B, Lavasanifar A (2006) Novel self-associating poly(ethylene oxide)-block-poly(ε-caprolactone) block copolymers with functional side groups on the polyester block for drug delivery. Macromolecules 39:9419–9428
Barbaud C, Fay F, Abdillah F, Randriamahefa S, Guérin P (2004) Synthesis of new homopolyester and copolyesters by anionic ring-opening polymerization of α, α', β-trisubstituted β-lactones. Macromol Chem Phys 205:199–207
De Winter J, Coulembier O, Gerbaux P, Dubois P (2010) High molecular weight poly(α, α', β-trisubstituted β-lactones) as generated by metal-free phosphazene catalysts. Macromolecules 43:10291–10296
Lecomte P, Stassin F, Jérôme R (2004) Recent developments in the ring-opening polymerization of ε-caprolactone and derivatives inititated by tin (IV) alkoxides. Macromol Symp 215:325–338
Tian D, Dubois P, Grandfils C, Jérôme R (1997) Ring-opening polymerization of 1,4,8-trioxaspiro[4.6]-9-undecanone: A new route to aliphatic polyesters bearing functional pendent groups. Macromolecules 30:406–409
Veld MJ, Palmans ARA, Meijer EW (2007) Selective polymerization of functional monomers with Novozym 435. J Polym Sci A Polym Chem 45:5968–5978
Amgoune A, Thomas CM, Ilinca S, Roisnel T, Carpentier J-F (2006) Highly active, productive, and syndiospecific yttrium initiators for the polymerization of racemic β-butyrolactone. Ang Chem Int Ed 45:2782–2784
Ajellal N, Bouyahyi M, Amgoune A, Thomas CM, Bondon A, Pillin I, Grohens Y, Carpentier J-F (2009) Syndiotactic-enriched Poly(3-hydroxybutyrate)s via stereoselective ring-opening polymerization of racemic β-butyrolactone with discrete yttrium catalysts. Macromolecules 42:987–993
Al-Azemi TF, Kondaveti L, Bisht KS (2002) Solventless enantioselective ring-opening polymerization of substituted-caprolactones by enzymatic catalysis. Macromolecules 35:3380–3386
Tian D, Dubois P, Jérôme R, Teyssié P (1994) Macromolecular engineering of polylactones and polylactides. 18. Synthesis of star-branched aliphatic polyesters bearing various functional end-groups. Macromolecules 27:4134–4144
Kricheldorf HR, Ahrensdorf K, Rost S (2004) Star-shaped homo- and copolyesters derived from ε-caprolactone, L,L-lactide and trimethylene carbonate. Macromol Chem Phys 205:1602–1610
Choi J, Kim I-K, Kwak C-Y (2005) Synthesis and characterization of a series of star-branched poly(ε-caprolactone)s with the variation in arm numbers and lengths. Polymer 46:9725–9735
Lang M, Wong RP, Chu C-C (2002) Synthesis and structural analysis of functionalized poly(ε-caprolactone)-based three arm star polymers. J Polym Sci A Polym Chem 40:1127–1141
Trollsas M, Hawker CJ, Remenar JF, Hedrick JL, Johansson M, Ihre H, Hult A (1998) J Polym Sci A Polym Chem 36:2793–2798
Kricheldorf HR, Fechner B (2002) Polylactones. 59. Biodegradable networks via ring-expansion polymerization of lactones and lactides. Biomacromolecules 3:691–695
Li H, Riva R, Kricheldorf HR, Jérôme R, Lecomte P (2008) Synthesis of eight and star-shaped poly(ε-caprolactone)s and their amphiphilic derivatives. Chem Eur J 14:358–368
Kricheldorf HR, Lee SR (1996) Polylactones. 40. Nanopretzels by macrocyclic polymerization of lactones via a spirocyclic tin initiator derived from pentaerythritol. Macromolecules 29:8669–8695
Riva R, Lazzari W, Billiet L, Du Prez F, Jérôme C, Lecomte P (2011) Preparation of pH-sensitive star-shaped aliphatic poplyesters as precursors of polymersomes. J Polym Sci A Polym Chem 49:1552–1563
Dai W, Zhu J, Shangguan A, Lang M (2009) Synthesis, characterization and degradability of the comb-type poly(4-hydroxyl-ε-caprolactone-co-ε-caprolactone)-g-poly(l-lactide). Eur Polym J 45:1659–1667
Liu M, Vladimirov N, Fréchet JMJ (1999) A new approach to hyperbranched polymers by ring-opening polymerization of an AB Monomer: 4-(2-hydroxyethyl)-ε-caprolactone. Macromolecules 32:6881–6884
Yu X-h, Feng J, Zhuo RX (2005) Preparation of hyperbranched aliphatic polyester derived from functionalized 1,4-dioxan-2-one. Macromolecules 38:6244–6247
Parzuchowski PG, Grabowska M, Tryznowski M, Rokicki G (2006) Synthesis of glycerol based hyperbranched polyesters with primary hydroxyl groups. Macromolecules 39:7181–7186
Tasaka F, Ohya Y, Ouchi T (2001) One-pot synthesis of novel branched polylactide through the copolymerization of lactide with mevalolactone. Macromol Rapid Commun 22:820–824
Trollsas M, Löwenhielm P, Lee VY, Möller M, Miller RD, Hedrick JL (1999) New approach to hyperbranched polyesters: self-condensing cyclic ester polymerization of bis(hydroxymethyl)-substituted ε-caprolactone. Macromolecules 32:9062–9066
Laurent BA, Grayson SM (2009) Synthetic approaches for the preparation of cyclic polymers. Chem Soc Rev 38:2202–2213
Kricheldorf HR, Lee SR (1995) Polylactones. 35. Macrocyclic and stereoselective polymerization of β-d,l-butyrolactone with cyclic dibutyltin initiators. Macromolecules 28:6718–6725
Kricheldorf HR, Lee S-R, Schittenhelm N (1998) Macrocyclic polymerization of (thio)lactones – stepwise ring expansion contraction. Macromol Chem Phys 199:273–282
Li H, Debuigne A, Jérôme R, Lecomte P (2006) Synthesis of macrocyclic poly(ε-caprolactone) by intramolecular cross-linking of unsaturated end groups of chains precyclic by the initiation. Angew Chem Int Ed 45:2264–2267
Jeong W, Hedrick JL, Waymouth RM (2007) Organic spirocyclic initiators for the ring-expansion polymerization of β-lactones. J Am Chem Soc 129:8414–8415
Xie M, Shi J, Ding L, Li J, Han H, Zhang Y (2009) Cyclic Poly(ε-caprolactone) synthesized by combination of ring-opening pPolymerization with ring-closing metathesis, ring closing enyne metathesis, or “click” reaction. J Polym Sci A Polym Chem 47:3022–3033
Misaka H, Kakuchi R, Zhang C, Sakai R, Satoh T, Kakuchi T (2009) Synthesis of well-defined macrocyclic poly(δ-valerolactone) by “click cyclization”. Macromolecules 42:5091–5096
Hiskins JN, Grayson JM (2009) Synthesis and degradation behavior of cyclic poly(ε-caprolactone). Macromolecules 42:6406–6413
Lendlein A, Schmidt AM, Schroeter M, Langer R (2005) Shape-memory polymer networks from oligo( ε-caprolactone)dimethacrylates. J Polym Sci A Polym Chem 43:1369–1381
Lowe JR, Tolman WB, Hillmyer MA (2009) Oxidized dihydrocarvone as a renewable multifunctional monomer for the synthesis of shape memory polyesters. Biomacromolecules 10:2003–2008
Mecerreyes D, Lee V, Hawker CJ, Hedrick JL, Wursch A, Volksen W, Magbitang T, Huang E, Miller RD (2001) A novel approach to functionalized nanoparticles: self-crosslinking of macromolecules in ultradilute solution. Adv Mater 13:204–208
Riva R, Schmeits S, Jérôme C, Jérôme R, Lecomte P (2007) Combination of ring-opening polymerization and “click chemistry”: toward functionalization and grafting of poly(ε-caprolactone). Macromolecules 40:796–803
Malberg S, Plikk P, Finne-Wistrand A, Albertsson A-C (2010) Design of elastomeric homo- and copolymer networks of functional aliphatic polyester for use in biomedical applications. Chem Mater 22:3009–3014
Kweon H, Yoo MK, Park IK, Kim TH, Lee HC, Lee HS, Oh JS, Akaike T, Cho CS (2003) A novel degradable polycaprolactone networks for tissue engineering. Biomaterials 24:801–808
Turunen MPK, Korhonen H, Tuominen J, Seppälä JV (2001) Synthesis, characterization and crosslinking of functional star-shaped poly(ε-caprolactone). Polym Int 51:92–100
Theiler S, Teske M, Keul H, Sternberg K, Möller M (2010) Synthesis, characterization and in vitro degradation of 3D-microstructured poly(ε-caprolactone) resins. Polym Chem 1:1215–1225
van Horn BA, Wooley KL (2007) Cross-linked and functionalized polyester materials constructed using ketoxime ether linkages. Soft Matter 3:1032–1040
Zednik J, Riva R, Lussis P, Jérôme C, Jérôme R, Lecomte P (2008) pH-responsive biodegradable amphiphilic networks. Polymer 49:697–702
Kricheldorf HR, Fechner B (2001) Polylactones. 51. Resorbable networks by combined ring-expansion polymerization and ring-opening polycondensation of ε-caprolactone or DL-lactide. Macromolecules 34:3517–3521
Palmgren R, Karlsson S, Albertsson A-C (1997) Synthesis of degradable crosslinked polymers based on 1,5-dioxepan-2-one and crosslinker of bis-ε-caprolactone type. J Polym Sci A Polym Chem 35:1635–1649
Albertsson A-C, Edlund U, Stridsberg K (2000) Controlled ring-opening polymerization of lactones and lactides. Macromol Symp 157:39–46
Grijpma DW, Kroeze E, Nijenhuis AJ, Pennings AJ (1993) Poly(L-lactide) crosslinked with spiro-bis-dimethylene-carbonate. Polymer 34:1496–1503
Acknowledgements
CERM is indebted to the “Belgian Science Policy” for general support in the frame of the “Interuniversity Attraction Poles Programme (IAP 6/27) – Functional Supramolecular Systems.” P.L. is Research Associate funded by the “Fonds National pour la Recherche Scientifique” (FRS-FNRS).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Lecomte, P., Jérôme, C. (2011). Recent Developments in Ring-Opening Polymerization of Lactones. In: Rieger, B., Künkel, A., Coates, G., Reichardt, R., Dinjus, E., Zevaco, T. (eds) Synthetic Biodegradable Polymers. Advances in Polymer Science, vol 245. Springer, Berlin, Heidelberg. https://doi.org/10.1007/12_2011_144
Download citation
DOI: https://doi.org/10.1007/12_2011_144
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-27153-3
Online ISBN: 978-3-642-27154-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)