Abstract
Isoprenoids are a class of compounds derived from the five carbon precursors, dimethylallyl diphosphate, and isopentenyl diphosphate. These molecules present incredible natural chemical diversity, which can be valuable for humans in many aspects such as cosmetics, agriculture, and medicine. However, many terpenoids are only produced in small quantities by their natural hosts and can be difficult to generate synthetically. Therefore, much interest and effort has been directed toward capturing the genetic blueprint for their biochemistry and engineering it into alternative hosts such as plants and algae. These autotrophic organisms are attractive when compared to traditional microbial platforms because of their ability to utilize atmospheric CO2 as a carbon substrate instead of supplied carbon sources like glucose. This chapter will summarize important techniques and strategies for engineering the accumulation of isoprenoid metabolites into higher plants and algae by choosing the correct host, avoiding endogenous regulatory mechanisms, and optimizing potential flux into the target compound. Future endeavors will build on these efforts by fine-tuning product accumulation levels via the vast amount of available “-omic” data and devising metabolic engineering schemes that integrate this into a whole-organism approach. With the development of high-throughput transformation protocols and synthetic biology molecular tools, we have only begun to harness the power and utility of plant and algae metabolic engineering.
Graphical Abstract

Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Isoprenoids
- Terpenoids
- Metabolism
- Genetic engineering
- Metabolic engineering
- Plants
- Algae
- Sub-cellular
- Tissue-specific
- Mevalonic acid pathway
- Methylerythritol phosphate pathway
- Plastid
- Mitochondria
- Terpene synthase
- Prenyl transferase
- Synthetic biology
- Genetically modified organisms
- Natural products
- Carbon neutral
- Biotechnology
1 Introduction
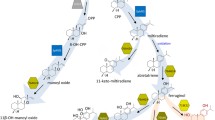
Isoprenoids constitute one of the most structurally diverse classes of compounds found in nature and are synthesized from two C5 isomeric precursors: isopentenyl diphosphate (IPP) and dimethylallyl diphosphate (DMAPP). These compounds have many roles in eukaryotic biology with special importance in plants. These roles are both critical (i.e., hormones, aliphatic membrane anchors, maintaining membrane structure) and unique (i.e., defense compounds, insect/animal attractants). There are tens of thousands of specialized metabolites (metabolites that are nonessential for normal cell homeostasis) and a significant proportion of these are isoprenoids, with well over 55,000 examples [178, 189]. Many excellent reviews have summarized the biosynthesis and roles of these compounds; the general model for isoprenoid biosynthesis is formation of the above-mentioned C5 precursors by either the mevalonate (MVA) pathway which is present in the cytoplasm/endoplasmic reticulum (ER) of plants (and is the route of isoprenoid biosynthesis ubiquitous in all eukaryotes) or the methylerythritol phosphate (MEP) pathway that is present in prokaryotes and the plastid compartment of plants (Fig. 1) [5, 12, 79, 161, 171, 189]. Briefly, these C5 precursors are then conjugated together in an additive fashion to generate prenyl diphosphates of varying chain lengths (in increments of five carbons) which are then cyclized and/or modified by terpene synthases, and can then be further decorated by downstream enzymes such as cytochrome P450s [80]. In plants, the MVA pathway is the primary pathway for the biosynthesis of sesqui- (C15) and triterpenes (C30), both generated from the C15 intermediate farnesyl diphosphate (FPP). The MEP pathway is the primary pathway for the biosynthesis of hemi- (C5), mono- (C10), di- (C20), tetra- (C40), and polyterpenes (>C50) which are generated from additions to the 10 carbon starting molecule geranyl diphosphate (GPP). Figure 1 summarizes the isoprenoid biosynthetic pathways operating in higher plants and highlights important enzymes and regulatory steps within the typical higher plant cell.
Summary of the two isoprenoid biosynthetic routes in plant cells. The mevalonic acid (MVA) pathway is shown starting with two acetyl-coenzyme A (CoA) molecules and proceeds through two steps (not shown) in the cytosol at microbodies to produce 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) which is reduced by HMG-CoA reductase (HMGR) at the ER interface to produce MVA. Three more steps (not shown) produce IPP and it is interconverted to its isomer DMAPP by isopentenyl diphosphate isomerase (IDI). Two molecules of IPP and one molecule of DMAPP are condensed to produce farnesyl pyrophosphate (FPP) by farnesyl pyrophosphate synthase (FPS). FPP serves as a precursor for sesquiterpenes, which are formed by terpene synthases and can be decorated by other various enzymes. Two FPP molecules are condensed by squalene synthase (SQS) at the ER face to produce squalene the precursor for triterpenes and sterols, which are generated by oxidosqualene cyclases (OSC) and are modified by various downstream enzymes. The methylerythritol phosphate (MEP) pathway present in the plastid compartment initiates its isoprenoid biosynthesis with pyruvate and d-glyceraldehyde 3-phosphate (G3P) condensed into 1-deoxy-d-xylulose 5-phosphate (DXP) by DXP synthase (DXS). Glycolysis and the Calvin cycle provide the precursors for the MEP pathway and recent studies have elucidated plastid importers of pyruvate (BASS2) and phosphoenolpyruvate (PEP) [47, 52]. DXP is then converted to MEP by DXP reductoisomerase (DXR). Five steps (not shown) convert MEP into IPP, which is also interconverted to DMAPP by the plastid IDI enzyme. DMAPP/IPP are also the precursor for hemiterpenes. One molecule of IPP and one molecule of DMAPP are condensed to form geranyl pyrophosphate (GPP) by GPP synthase (GPS). GPP is the immediate precursor for monoterpenes. Further condensations of two molecules of IPP to GPP forms geranylgeranyl pyrophosphate (GGPP) by GGPP synthase (GGPS). GGPP is the precursor for diterpenes and 2 GGPPs can be condensed by phytoene synthase (PSY) to form phytoene, the immediate precursor for carotenoids and tetraterpenes. Mitochondria also contain nuclear encoded, catalytically active isoforms of IDI, GGPS, and FPS. Mitochondrial FPP is critical for ubiquinone. Transport of precursors and prenyl diphosphates between membranes and sites of biosynthesis are represented by dashed arrows, while direct catalytic reactions are depicted by bold arrows. Enzymes that catalyze important regulatory steps or are important engineering targets are indicated in bold dark red. Figure adapted from information in [20, 52, 57, 123]
Initially, the identification of the MVA pathway suggested that it was probably the conserved evolutionary path for the biosynthesis of isoprenoids in all organisms. However, pioneering work starting in the 1980s showed that the MEP pathway was the bacterial method of isoprenoid production and that it was also primarily responsible for isoprenoid production in plant plastids [94, 150, 151]. Each pathway’s catalytic enzymes have been well described and several important regulatory elements are known [171]. Canonically, in the MVA pathway, 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGR) is one of the key regulatory steps controlling the flux of acetyl-CoA into the isoprenoid precursors in the MVA pathway, and 1-deoxy-D-xylulose 5-phosphate synthase (DXS) and 1-deoxy-D-xylulose 5-phosphate reductoisomerase (DXR) are key regulatory elements in the MEP pathway (Fig. 1) [62]. For some time evidence suggested the important role of DXS in controlling flux into the MEP pathway [42]. The role of DXS has been further underlined by several studies looking at MEP pathway carbon flux using 13C labeling experiments [54, 179]. These studies corroborate the findings by Estévez et al. [42], showing that both increased and decreased DXS activity directly appears to influence MEP pathway flux. However, overexpression of DXS alone did not seem to increase downstream isoprenoid accumulation highly, indicating other critical regulatory steps at downstream enzymes. It is important to note that DXS activity does not directly correlate with increased DXS transcription and posttranscriptional regulation of DXS activity has also been described [134]. The regulatory system of DXS appears typical for both isoprenoid pathways: control of flux is complex, occurring at multiple points in each pathway, in response to many different types of stimuli, and through varying methods across all levels of regulation (i.e., transcriptionally, posttranscriptionally, and posttranslationally). Untangling these regulatory networks is further complicated by the presence of gene families for each coding enzyme of the plant isoprenoid pathways. Furthermore, these gene families have varying compositions from species to species with specialized functions (e.g., active for specific developmental regimes) for the various isozyme members in each [62].
This subcellular division of labor in higher plants is an important biochemical facet that can be capitalized upon using metabolic engineering that will become apparent. The division effectively partitions major biochemical control over two areas within the cell, each of which may be exploited to avoid the endogenous regulation that controls production in either the cytoplasm or plastid. During the elucidation of the plastid MEP pathway, several clues hinted at the independence of plastid isoprenoids from the cytosolic MVA pathway. Labeling studies using MVA precursors indicated they were not effectively incorporated into carotenoids, diterpenes, or monoterpenes. Furthermore, chlorophylls and carotenoids were not affected in plants treated with the MVA pathway inhibitor, mevinolin, and initially it was speculated that the inhibitor was not able to permeate through the plastid envelope. Interestingly though, chloroplasts were able to incorporate exogenous IPP [150]. Mitochondria are also able to import IPP; however, mitochondria are void of an endogenous pathway that synthesizes the basic five-carbon prenyl unit, and must obtain IPP from the cytosolic MVA pathway [35]. This is critical for the production of polyprenols such as those present in ubiquinone [62]. It is appreciated that flux through the MEP pathway is more robust than flux through the MVA pathway. This is due to the plastid synthesizing large amounts of carotenoids and chlorophyll polyprenyl chains primarily from photosynthetically obtained CO2. The MVA pathway precursor, acetyl-CoA, is a common intermediate in several metabolic processes within the cell and is derived from the breakdown of carbohydrate (glycolysis), fatty acids (ß-oxidation), and amino acid recycling [62].
Each pathway appears to control its flux independently, however, they do have some basal level of intermediate sharing and this may be more prevalent during specific times of growth [63]. Continued study of various isoprenoid compounds indicates that the separation of these two pathways and the isolation of their respective prenyldiphosphate precursors do not follow any generalizable rules, and that noncanonical sharing of substrates across pathway boundaries does appear to occur. Whether these exchanges are facilitated by transporters or through another biophysical mechanism (such as intermediate sharing of tocopherol precursors as described by Mehrshahi et al. [111]) is not known, and may be a combination of methods [62].
Many of the isoprenoids produced by plants are of high value to humans. They have uses in medicine (e.g., the anticancer diterpene Taxol, the antimalarial sesquiterpene artemisinin), cosmetics (e.g., the triterpene squalene, the many fragrant monoterpenes), industry (e.g., the monoterpenes and diterpene resin acids for chemical feedstock), and agriculture (e.g., sesquiterpenes involved in complex ecological trophic interactions and isoprenoid-derived plant hormones such as the gibberellins [10]). However, many of these compounds are chemically complex with organic synthesis sometimes being prohibitively costly and complicated for large-scale production. Thus, much effort has been focused on producing these compounds in biological hosts using metabolic/genetic engineering technologies that have become possible within the past few decades. Although biological production has mostly focused on production using microbial fermentation systems, these require their own expensive infrastructure, feedstock (usually sugars), and purification methods. Compared to microbial systems, plants are especially appealing because they require a simple infrastructure, including water and fertilizer, are easily amenable to variable production scales, and are environmentally friendly by fixing atmospheric carbon into desired compounds.
However, plants are complex multicellular organisms, which have unique evolutionary adaptations that make them more difficult to engineer genetically when compared to microbes. Although efforts have indicated that metabolically engineering plants is feasible, further work will continue to refine and identify the most successful strategies in developing plant production platforms. The purpose of this chapter is to summarize important considerations, technologies, and strategies that should be considered when designing a metabolic engineering effort in higher plants, and also touches on work and the state of metabolic engineering in green algae. These important considerations begin at the macroscale in deciding upon an appropriate plant host in which the desired engineering might prove most effective, and continue to discussions of how tissue-specific (and cell-specific) targeting can be used in particular circumstances. Illustrative case examples where these engineering approaches have been successful are highlighted. Microscale and molecular approaches are also considered, with specific examples presented when applicable. Specific molecular techniques and engineering approaches that have been successful, or may be successful in the future, are also discussed.
Overall, we stress three essential components that underwrite any engineering strategy: (1) manipulating gene(s) expression (e.g., using heterologous genes from a species unrelated to the host under a promoter that drives the desired temporal and/or spatial expression patterns, or altering endogenous gene expression to change flux towards the desired pathway); (2) avoiding endogenous regulation (e.g., using specific hosts and targeting the introduced pathway to specific tissues and subcellular compartments); and (3) combining the first two considerations in unique and novel ways with the available technologies (e.g., using metabolic models, synthetic biology to ensure enzyme cooperation, using transporters to sequester accumulated compounds). In addition to the choice of plant host, these are all variables that can be controlled by construct design and are best summarized in Fig. 2.
A schematic blueprint illustrating important variables and approaches to metabolic engineering in plants. This figure shows a typical transgene design that can capitalize upon various permutations to yield the desired flux of carbon to the biosynthesis of unique end-products. The process(es) controlled by each arrow are defined within the inset figure legend
Before summarizing the technologies and efforts to engineer terpene metabolism in plants, an important distinction that arises from the literature needs to be mentioned: there is a difference between efforts to characterize genes, proteins, and biochemical pathways using transgenic plants as a tool for basic research versus efforts to introduce transgenes aimed towards the accumulation of large amounts of desired compound(s). As expected, many of the initial efforts to alter terpene metabolism in plants were proof-of-concept experiments [64, 90, 122, 163, 172, 190] and the first dedicated efforts to engineer particular isoprenoid compounds into plants soon followed [9, 12, 39, 73, 90, 100–102, 181]. Several recent review articles summarize many of these efforts in the field quite well [4, 87, 117, 160] and it is not the aim of this chapter to review comprehensively all the efforts to engineer isoprenoid compound accumulation in plants. Instead, we focus on the strategies important to obtain high levels of desired isoprenoid compounds.
2 Host Choice
The choice of host plant for metabolic engineering should be based upon relevant scientific criteria such as whether the endogenous flux of carbon through a desired pathway is high enough and flexible enough to accommodate the funneling of precursor metabolites towards a desired product. Additional criteria include how amenable the plant host is to genetic transformation, will the host produce enough biomass to obtain the desired amount of compound(s), and is the host devoid (or need to be made devoid) of endogenous enzymes that may modify or metabolize the desired end-product compound(s). Most of the current work has been done in model species that are amenable to transformation, have short life-spans (allowing for efficient genetic experiments), and for whom we generally have a more thorough understanding of their biochemistry (e.g., Arabidopsis and Nicotiana). Host choice should also consider the endogenous biochemical milieu of the target, especially if there exists the potential to modify the engineered compound chemically or sequester it via an undesired biochemical transformation. One way to avoid these latter concerns has been to choose a host for which the engineered metabolism is completely foreign (i.e., there is no endogenous amount of that compound produced naturally). However, this may not be possible either due to the compound’s ubiquity (e.g., squalene [180]) or the desire to alter the balance of compounds natively produced in a particular species (e.g., tocopherols). However, even introduction of foreign metabolism may not be enough to prevent further compound derivatization, as seen in work by Lücker et al. [100], where they saw modification of engineered S-linalool to S-linalyl-β-d-glucopyranoside in engineered Petunia. In that study, control plants did not natively produce S-linalool, but transgenic plants did emit S-linalool into the headspace from leaves, yet did not have any detectable S-linalool in extracted leaf tissue. Upon treating transgenic tissue with hydrolyzing enzymes, free linalool was detected. Using HPLC-MS/MS and multidimensional gas chromatography–mass spectrometry with comparison to standards, they were able to elucidate the conjugated linalool as S-linalyl-β-d-glucopyranoside. However, the main problem with model species is that they generally do not produce much biomass (e.g., Arabidopsis thaliana or Brachypodium distachyon), and therefore are of little applied use, besides proving that the particular engineering can be accomplished at a genetic and biochemical level.
When thinking of substantial biomass we generally think of crop plants (e.g., maize, rice, soybean, sugarcane, etc.), many of which have proven to be more recalcitrant to genetic transformation and thus have fewer successful reports of engineering efforts. However, with recent developments in increasing the efficiency of transformation for previously low-throughput species (as in the work done by Sahoo et al. [156] and Zhang et al. [186]), it is reasonable to speculate that the introduction of transgenes will not be the limiting factor in conducting metabolic engineering experiments in plants in the years to come. Therefore, host selection should revolve around the desired engineering outcome and how a particular plant can match those needs (i.e., whether the plant displays substantial tissue biomass for tissue-specific accumulation, if desired). Examining host choice from a more applied and agronomic sense, it may prove worthwhile to consider the choice of a particular line in light of where the plant would actually be grown. Selecting a line having already been bred to display resistance to pathogens and/or particular climate nuances would undoubtedly produce higher product accumulation. Perhaps most importantly, the host to be chosen should be a high-performing line, meaning it should display robust growth characteristics in a field setting with an emphasis on yield of desired engineered tissues.
3 Organ- and Tissue-Specific Engineering
It’s been well known that certain plant cells and tissues have unique chemical profiles, especially for the case of specialized metabolites (e.g., avenacins found in oat roots [106], alkaloids in the latex of poppy [43], and sesquiterpenes in glands of cotton Elzen et al. [40]) for which recent technologies and methods in metabolomics and transcriptomic studies have provided more understanding of the genes and enzymes involved in the production of unique metabolites [118, 119, 149, 161]. Often these unique metabolites are of special value and many of them are terpenoids (or terpenoid-derivatives). Therefore, it could be beneficial to hijack the biochemical flux through these specific cellular pathways without the potential disruption of plant homeostasis that often occurs when constitutive promoters are used. In an engineering strategy designed for large amounts of product accumulation, cell-/tissue-/organ-specific targeting would probably have the best results in one of four main cell/tissue/organ types: seeds, fruits, trichomes, and leaves. There are other cell/tissue types that are specific to certain plant species, such as resin glands in gymnosperms or laticifers in Hevea species and plants in the Asteraceae and Euphorbiaceae, which could also serve as excellent platforms in which to divert endogenous carbon flux [75]. However, engineering efforts into these cell types are limited by our incomplete understanding of the overall biology of these specialized cell types. The ability to target single cell types or even specific tissues is really limited by the number of promoters fully characterized for a specific pattern of gene expression/cell specificity. Future production platforms might also benefit from the development of synthetic promoters that could allow for more tailoring, tunable, optimal gene expression [71].
3.1 Leaves
With regard to organ specificity, leaf-specific targeting does not seem to be a goal towards which much effort has been expended. This is partly due to the historical use of constitutive promoters that drive ubiquitous expression and thus, leaf expression. Because leaves usually are the most accessible and easy to work with of plant tissues, they generally serve as the evaluation platform for testing new constructs or strategies using constitutive promoters to drive construct expression. Leaves are generally the default tissue engineered product accumulation is assayed within due to their biomass that they serve as the basic model in which plant biochemistry and metabolism are considered. This is intuitive because the leaf serves as the main site of photosynthesis and contains all of the primary metabolites derived from photosynthate that could be diverted into heterologous compounds. Presented here are a few cases showing how the separation of leaf-specific and constitutive expression most likely depends on the desired product outcome, and that it is difficult to anticipate the phenotypic effect that a particular compound may elicit in planta.
Plant isoprenoid production as a result of pathogen attack has been well documented [77, 128, 169, 170]. These elicited compounds can function to prevent further pathogen attack and recruit predators of the attacking pathogen. Recent studies have continued to describe the functions of isoprenoids in these roles [69]. Thus, engineering accumulation (or reduction) of these compounds to protect plants against pathogens is an intriguing proposition. Rodríguez et al. [147] engineered an antisense limonene synthase into oranges (under the CaMV35S promoter) and found reduced limonene (and other monoterpenes, sesquiterpenes, and monoterpene aldehydes), increased monoterpene alcohols, and most interestingly a marked resistance to infection by Penicillium digitatum, Xanthomonas citri subsp. Citri, and less attraction to the fruits by the insect Ceratitis capitata. Although introduction of the antisense limonene synthase changed the terpenoid profile of the orange fruits, the transgenic plants were phenotypically identical to wild-type (WT) plants. Engineering of maize plants with an oregano (E)-ß-caryophyllene (EbC) synthase resulted in an increase in aboveground herbivory during field growth, and olfactory assays showed a preference by Spodoptera frugiperda towards the transgenic lines [145]. These EbC-expressing lines did not repress S. frugiperda growth any more than WT lines. Thus, engineering the accumulation (or reduction) of the terpene constituents can affect the resistance characteristics of plants to pathogens in unexpected ways. Although, as previously reported, constitutive EbC expression in maize did decrease root damage by the economically relevant western corn rootworm, Diabrotica virgifera virgifera LeConte, probably through attraction of entomopathogenic nematodes [32, 145].
These EbC-expressing maize lines did show reduced root herbivory, however, the agronomic performance of the plants in a field setting was compromised. Although plant height and water content in the EbC-expressing lines were similar to WT, the leaf biomass was only 25 % of WT and yield was reduced compared to WT lines [145]. On average, the EbC-expressing maize plants emitted β-caryophyllene and α-humulene between 542–2,024 pg/h* plant and these values did not significantly change after attack by the S. frugiperda larvae [145]. These results suggest two possibilities for the reduced plant biomass: carbon flux into the engineered compounds is so robust that it is stealing isoprenoid precursors away from downstream products that are essential for plant growth (e.g., chlorophyll), or there is a certain amount of accumulating monoterpene that is directly harmful to plant cell growth. Inasmuch as tissue accumulation of β-caryophyllene or α-humulene was not presented in these studies [32, 145] the first possibility cannot be definitively ruled out. Other work has supported the latter possibility.
Work by Wu et al. [181] showed that plants engineered to produce patchoulol within the plastid accumulated high levels in leaf tissue and also emitted the sesquiterpene into the headspace (emission was at a higher level than the monoterpenes in maize presented above, 50–100 ng/h*g fresh weight, [FW]), and that some of these transgenic lines showed vein clearing and chlorosis of the leaves, indicating precursor funneling from plastid-produced pigments to the engineered terpene. Wu et al. [180] also showed that transgenic tobacco lines producing high amounts of triterpenes in the plastid (using a multigene approach) also had reduced biomass (Fig. 3). In addition, Aharoni et al. [2, 3] saw stunted growth with Arabidopsis engineered with a S-linalool/[3S]-E-nerolidol synthase (FaNES1) targeted to the plastid. Similarly, Aharoni et al. [4] saw stunted growth and leaf necrosis in potato plants engineered with FaNES1 under control of the strong chrysantheum Rubisco small subunit promoter [127].
Phenotypic effects of high accumulation of plastid-targeted triterpene production in tobacco. Plants engineered to express plastid-targeted FPS and SQS under putative trichome specific promoters (far left) and constitutive promoters (middle) have varying degrees of compromised growth compared to WT (far right). Transgenic plants presented are homozygous (T2 generation). Taken from Wu et al. [180]
In contrast to these results, Cahoon et al. [12] engineered tocotrienol accumulation into Arabidopsis leaves using barley homogentisate geranylgeranyl transferase (HGGT) under the CaMV35S promoter, and reported elevated tocopherols and tocotrienols increased to 700–900 µg/g dry weight (DW) from <100 µg/g DW with tocotrienols accounting for ~85 % of the total content. These transgenic plants had tocopherol levels approximately equal to the WT plants, but no changes in the levels of chlorophyll or carotenoids. Sitbon and Jonsson [164] engineered Nicotiana tabacum with sterol methyltransferase genes one and two (SMT1 and SMT2) under a TET1/35S promoter and saw reduced biomass in only SMT2 engineered plants (Table 1). Interestingly, they did not observe changes in total sterol content in the transgenic lines versus WT, but did observe changes in the sterol profile of the transgenic lines versus the WT. These results suggest that unknown or unappreciated levels of metabolite control can act as compensatory mechanisms to maintain homeostasis and simultaneously allow high accumulation of the desired product. This also reiterates the point that the choice of the enzyme(s) in a pathway where attempts to push or pull carbon downstream towards the desired endpoint is of crucial importance.
3.2 Seeds
Although leaf biomass can be prolific in some plants (e.g., tobacco), seeds are probably the oldest target of selection/engineering, usually to increase biomass (yield) for animal/human nutritional value, and in oilseeds, to increase oil content for both industrial and nutritional value. There have been successful efforts in diverting carbon flux into terpenoid compounds in seeds, perhaps the best known being the introduction of β-carotene biosynthesis into rice endosperm, producing the titular “Golden Rice” of the engineered plant [183]. Ye et al. [183] did this by engineering in heterologous versions of PSY, phytoene desaturase, and lycopene β-cyclase. These enzymes were targeted to the plastid (the site of carotenoid biosynthesis) and segregating lines showed β-carotene accumulation of varying levels with the highest presented line producing 1.6 µg/g endosperm. However, expressing only the endogenous PSY under the Brassica napus NAPIN1 (BcNA1) seed-specific promoter in Arabidopsis saw an increase in carotenoids and chlorophyll, with the largest change being a 43-fold increase in β-carotene (260 µg/g FW) compared to WT (6 µg/g FW). Fujisawa et al. [51] introduced seven genes into the seeds of B. napus for the production of ketocarotenoids, including a synthetic IDI to increase the availability of upstream IPP. Engineered B. napus seeds produced 19–30 times higher carotenoids (412–657 µg/g FW) compared to the WT, with 60–190 µg/g FW being ketocarotenoids. Importantly, they also noticed that introduction of heterologous genes altered the expression of endogenous carotenoid biosynthesis genes. Similar changes to endogenous carotenoid gene expression were also seen in Gutensohn et al. [57] when tomato fruits were engineered with a GPS small-subunit (GPS-SSU) from snapdragon.
Holmberg et al. [58, 66] showed elevated sterols when seeds were targeted to express heterologous sterol methyltransferases, one of the rate-limiting steps into sterol biosynthesis. Savidge et al. [159] showed elevated levels of total seed tocopherols, up to 60 % increases, when engineered with Arabidopsis homogentisate phytyl transferase (HPT1) under the expression of the BcNA1 seed-specific promoter. Cahoon et al. [12] engineered tocotrienol accumulation into Zea mays embryos using barley HGGT under the embryo-specific Zea mays 16 kDa oleosin gene promoter and found increased whole seed tocotrienol production at least 20-fold compared to WT whole seeds (Fig. 4). These efforts indicate that altering terpenoid flux in developing seeds is possible and even more important, that they are generated in a natural storage repository that can be harvested/extracted at a later time. The fact that seeds can serve as storage for produced compounds with little biochemical alteration once the seed is dormant, is of special interest, especially if the site of production and extraction are physically separated or long-term storage is desired.
Zea mays seeds engineered with barley HGGT under a seed-specific promoter. Tocotrienol production was increased at least 20-fold compared to WT. Modified from Cahoon et al. [12]
3.3 Fruits
Like seeds, fruits also have the potential to be natural storage tissues and targeting to fruits is desirable for compounds that have beneficial impacts on human nutrition or could deter pathogens (avoiding the application of pesticides that could be detrimental to the environment and human/animal health). Work has been done in both of these arenas to varying degrees of success. Like the efforts to increase the nutritional content of seeds by engineering in carotenoid accumulation, similar, early work was done in tomato fruits, which are naturally rich in carotenoids [48, 154]. Introduction of a PSY gene increased total tomato fruit carotenoid accumulation approximately 2-fold (over 2 mg/g DW in transgenic fruits vs. 1.3 mg/g DW for WT plants).
In contrast, when a GPS-SSU gene was introduced, it caused a 5-fold reduction in overall carotenoid accumulation in tomato fruits (~215 μg/g FW in WT vs. an average of ~41 μg/g FW in the transgenic lines). Heterologous expression of the snapdragon GPS-SSU was previously observed to control prenyl-chain length formation of endogenous GGPS enzymes in transgenic tobacco, resulting in more GPP and less GGPP accumulation [126]. Hence, the decrease in carotenoid formation is most likely due to the heterologous expressed GPS-SSU saturating the endogenous GGPS enzymes, causing a reduction in the biosynthesis of the carotenoid precursor GGPP. Interestingly, the concentrations of some of the lycopene-derived carotenoids (β-carotene, zeaxanthin, and lutein) were not significantly decreased in the GPS-SSU transgenic fruits [57]. This indicates a much more complex regulatory network controlling the flux of GGPP into carotenoid compounds, and suggests the existence of multiple branch pathways to specific carotenoid end-products.
3.4 Trichomes
Trichomes encompass a group of specialized cells that originate from the epidermis of plant tissues and are differentiated on the basis of their biochemical capabilities. A recent review by Lange and Turner [89] summarizes the current knowledge of isoprenoid biosynthesis in trichomes and touches upon how our current understanding in trichome biology might be harnessed for use in metabolic engineering efforts. Briefly, there are several types of trichomes but glandular trichomes (GTs) are the most important in terms of specialized metabolism capacity due to their seemingly dedicated role as metabolite production factories. The presence, number, and type of trichomes vary between plant species. Thus, the capacity to engineer them does not exist for every engineering project (although, this could be an important factor in choosing a particular production host). Equally important, glandular trichomes can secrete their products onto the leaf surface, facilitating collection, or the glandular head synthesized compounds may accumulate in cavities/sacs associated with the metabolically active cells. Trichomes secreting hydrophobic compounds including isoprenoids generally have interesting intracellular features such as an extensive smooth ER network that maintains contact with nonpigmented leucoplasts. The leucoplasts appear to have a nonuniform shape that could be implicated in increasing the contact surface area with the smooth ER [89]. These extensive connections between intracellular membrane networks may be critical for transport of large amounts of hydrophobic compounds. A recent study has also implicated a possible role of a lipid transfer protein in the export of isoprenoids from Nicotiana tall GTs [17].
The species in which isoprenoid biology of the trichomes has been best studied is the pathway leading to the production of (−)-menthol in Mentha x piperita (peppermint), which occurs exclusively in this plant’s trichomes. The biosynthesis and subcellular organization of (−)-menthol production has been well described [24]. The production of this monoterpene through the MEP pathway (Fig. 1) illustrates the robust carbon flux through trichome plastids. Further support for a high carbon flux through the MEP pathway in trichomes is the high level of Δ9-tetrahydrocannabinolic acid accumulation in Cannabis sativa. Tetrahydrocannabinolic acid contains an integrated GPP molecule attached to a phenolic precursor that allows for further carbon- and lactone-cyclization yielding the final product [89]. Trichome-specific promoters have also been identified that enable trichome-specific transgene targeting and novel biotechnology applications (summarized in [168]). The natural capacity for trichomes to function as specialized biochemical factories, in fact, makes them intriguing targets for the redirection of flux into heterologous, high-valued compounds [180].
3.5 Roots
Targeting compound accumulation in roots may pose special technical challenges as well as physical limitations on harvesting. However, compound accumulation in roots can be beneficial for defense compounds that prevent pathogen attack in the soil and for nutritional enhancement in tubers or other root vegetables. As noted above, Degenhardt et al. [32] and Robert et al. [145] showed that root emissions of β-caryophyllene and α-humulene attracted nematodes that helped prevent root herbivory by western corn rootworms.
Work by Diretto et al. [34] also demonstrated how the chemical profile of roots was amenable to manipulation by genetic engineering. Silencing the lycopene epsilon cyclase (LCY-e) increased β-carotene levels >13-fold and total carotenoids were increased >2-fold in transgenic tubers relative to WT. The highest accumulating tubers showed a mild yellow phenotype. Follow-up work by Diretto et al. [33] reported engineering PSY, phytoene desaturase/carotene isomerase, and lycopene β-cyclase, all expressed under tuber-specific promoters, caused a greater than 3600-fold increase in β-carotene (47.4 µg/g DW) compared to WT. Total tuber carotenoid content was also increased >19-fold (up to ~114 µg/g DW) in these lines. These high carotenoid producing transgenic lines showed a distinct yellow color (compare wild-type to engineered lines P-YBI-17 and P-YBI-30; Fig. 5). These same investigators also transformed potato plants with the same three bacterial genes, but only placed the PSY gene under a tuber-specific promoter, whereas the remaining two genes were driven by the constitutive 35S promoter (K-YBI-41). Transgenic lines with expression of the PSY gene under the tuber-specific promoter definitely prevented severe chlorosis/growth retardation as previously reported by Fray et al. [50].
Transgenic Solanum tuberosum engineered with multiple carotenoid biosynthetic genes. Plants were engineered with phytoene synthase, phytoene desaturase/carotene isomerase, and lycopene β-cyclase. P-YBI-17 and P-YBI-30 transgenic lines had all genes expressed under tuber-specific promoters. K-YBI-41 plants had only phytoene synthase under a tuber-specific promoter, while the other two genes were expressed constitutively under the direction of the 35S promoter. a Transgenic tubers show a yellow pigmentation. b Note the apparent decreased leaf pigmentation of line K-YBI-41 compared to the WT and the tuber-specific P-YBI-17 line. Taken from Diretto et al. [33]
The transgenic lines constitutively expressing bacterial phytoene desturase/carotene isomerase and lycopene β-cyclase (but tuber-specific PSY) showed hampered regeneration of transgenic plantlets. The authors attributed this to a reduced ability to produce pigments necessary for photosynthesis, which was correlated with a chlorotic phenotype (compare middle leaf, to the WT and the tuber-specific expression line, P-YBI-17 in Fig. 5). These negative phenotypes were not observed when all three genes were placed under tuber-specific promoters. Indeed, these pK-YBI-41 plants showed approximately half the leaf chlorophyll accumulation of WT plants [33]. When these constructs were transformed into Solanum tuberosum L. cv. Désirée, which has an endogenous tuber carotenoid content of approximately 5.6 µg/g DW, the transgenic lines accumulated 6-fold more carotenoid, up to 35 µg/g DW. However, when the same bacterial PSY gene was transformed into the S. tuberosum cv. Mayan Gold (which has an endogenous tuber carotenoid content of approximately 20 µg/g DW), the highest carotenoid accumulating lines had nearly four times the amount of total carotenoid (78 µg/g DW) as found in the nontransgenic parental line [37]. This illustrates the importance of choosing a plant cultivar that naturally exhibits a robust metabolic flow through the desired pathway in order to generate the highest accumulating lines.
Other work utilized the Or transcription factor, originally identified as the gene controlling orange pigmentation in cauliflower mutants by causing the formation of chromoplast structures and the increased accumulation of carotenoid content [98]. Transformation of the Or gene into S. tuberosum cv. Désirée under the control of the primarily tuber-specific starch synthase promoter resulted in orange pigmented potato tubers containing >5-fold higher carotenoid content than the nontransgenic parental line (~31 µg/g DW) [92, 97].
Altogether, these observations have demonstrated that carotenoid content can be manipulated by the overexpression of either genes encoding for biosynthetic enzymes or relevant trans-acting regulatory factors. An interesting question remaining unanswered is if overexpression of the Or gene in combination with the bacterial carotenoid biosynthetic genes could enhance carotenoid accumulation even more in potato than either of the separate engineering strategies?
3.6 Other Specialized Cell Types
Many plants contain specialized cells that serve as endogenous synthesis and storage compartments for specialized metabolites. Often these cells are utilized to sequester defense compounds that would otherwise impose their cytotoxic activities onto the host plant. An example of this cell type is the resin ducts formed in conifer species upon herbivore or fungal attack [76]. These cells produce prodigious amounts of monoterpenes and diterpene resin acids along with smaller amounts of sesquiterpenes [75]. The formation and biochemistry of these terpenoid factory cells is still a matter of investigation. In one study, for instance, introducing a Pinus abies bifunctional isoprenyl diphosphate synthase (IDS; IDS catalyzes the biosynthesis of GPP and GGPP) into Pinus glauca did not reveal an increase in any of the monoterpenes, sesquiterpenes, diterpene resin acids, sterols, or carotenoids in any tissue. The prenyl diphosphate intermediates, GPP and GGPP, were increased in needles but not bark, and higher levels of esters of geranylgeraniol with fatty acids were noted [120]. This was unexpected because IDS expression was driven by the constitutive Zea mays ubiquitin 1 promoter, and although transcript accumulation was much lower in the bark than in needles, no real metabolic changes were observed in the bark tissue. This is indicative of unique control mechanisms that might need further elucidation before transgenic conifers could be used to increase production of the valuable oleoresins.
In contrast to the induced formation of resin ducts in conifers, lactifers are reticulated, interconnected cells present in particular plant species and often in particular tissues and that generate and accumulate latex. Latex is a combination of many specialized compounds, including phenolics, proteins, alkaloids, terpenes, and the main constituent being polyisoprenoids, that is, natural rubber which can contain polyisoprene chains with >18,000 isoprenyl subunits [1, 8]. Human utilization of latex has been important for thousands of years, with an expanding role more recently for its use in industrial production of diverse rubber products. Interestingly, not all the steps in rubber biosynthesis have been elucidated [18], which has definitely limited the prospects for engineering greater rubber production capacities [8].
Specialized plant cell cultures might also serve as production platforms for important terpenoid compounds [11, 146]. These systems have been especially important for high-value pharmaceuticals that tend to accumulate to low levels in planta, such as providing intermediates from Taxus baccata for the semisynthetic production of Taxol, portending similar opportunities for terpene indole alkaloids (TIAs) from Catharanthus roseus and artemisinin from Artemisia annua cultures.
4 Engineering Isoprenoid Metabolism in Plant Organelles
As noted earlier, metabolic flux in isoprenoid biosynthesis revolves around a complex network that involves multiple subcellular compartments within the plant cell: the cytosolic MVA pathway, the plastid-localized MEP pathway, localization of many sequential isoprenoid biosynthetic enzymes arrayed on the ER membrane or targeted to the mitochondria and microbodies, and the possible participation of the vacuole as a storage/sequestration organelle (Fig. 1). Hence, targeting of enzymes and biosynthetic capacity to an appropriate subcellular location is a prerequisite for successful metabolic engineering in plants, which must overcome several layers of complicated regulation [61, 87]. Early metabolic engineering attempts achieved less success in elevating terpene production by introducing enzymes into the cytoplasm without considering the regulatory complexity of the MVA pathway [181]. Now, engineering terpene metabolism into organelles to overcome these innate regulatory constraints has become an important strategy, as illustrated by the following two examples.
It is well established that the biosynthesis of distinct classes of isoprenoids take place in distinct organelles (Fig. 1). For example, the major steps of monoterpene, diterpene, and carotenoid biosynthesis are known to operate in higher plant chloroplasts, so most of the engineering efforts to enhance biosynthesis of these compounds have used heterologous enzymes fused with a plastid-targeting signal peptide to direct them to their endogenous site of biosynthesis (or by introducing the transgene into the plastid genome directly). This type of “straightforward” genetic engineering has been conducted in various species of higher plants, extensively reviewed (6, 45, 49, 88, 7, 114, 116, 182), and is not reiterated in this section other than to note that this type of engineering utilizes pre-existing substrate biosynthesis, which is captured and diverted to produce compounds that may or may not be normally present within a particular organelle. Hence, this type of strategy does not completely avoid endogenous regulatory elements that may respond directly to the synthesized compound or indirectly through changes in intermediates and large amounts of target compound accumulation. It is also necessary to be aware of any inherent regulatory systems (e.g., allosteric sites) of the introduced enzymes that may inhibit their activity.
In order to avoid this, targeting introduced enzymes to particular organelles to divert available substrate(s) for the formation of a novel pathway that is foreign to the organelle is becoming a commonplace strategy. In an early study, a strawberry linalool/nerolidol synthase FaNES1, was targeted to mitochondria by fusing a mitochondrial targeting signal sequence to the amino terminus of the FaNES1 protein, leading to generation of two novel sesquiterpenes in transgenic Arabidopsis, (3S)-(E)-nerolidol and (E)-DMNT, neither of which is found in WT lines [73]. In another recent study [44], amorphadiene-4,11-diene synthase (ADS) targeted to the mitochondria was coexpressed with a cytochrome P450 (CYP71AV1), a cytochrome P450 reductase (CPR), an artemisinic aldehyde reductase (DBR2) from A. annua, and a truncated and deregulated HMGR from yeast in N. tabacum. This resulted in artemisinin accumulation of 5–7 μg/g DW, which is 8-fold more than when ADS was targeted to the cytoplasm [44]. These results provide strong evidence that mitochondrial targeted FaNES1 and ADS have access to FPP in the mitochondria and convert this intermediate into (3S)-(E)-nerolidol and amorpha-4,11-diene, respectively. These reaction intermediates were then were further converted by native (and unknown enzymes in Arabidopsis) or engineered enzymes (CYP71AV1, CPR, DBR2) present in cytosol to the final products (E)-DMNT and artemisinin, respectively. Although it is not clear how the intermediate product (3S)-(E)-nerolidol and amorpha-4,11-diene are shuttled between the mitochondria and cytosol, the results show that the heterologous terpene synthases can efficiently use the FPP pool in the mitochondria for novel sesquiterpene production. In contrast, free pools of FPP in chloroplasts and the cytoplasm must be less available in these species because simply introducing a FPP-dependent synthase does not yield appreciable novel terpene accumulation [3, 44, 73, 181].
Wu et al. [181] furthered this approach by applying it to chloroplasts. Accumulation greater than ~25 µg/g FW of the nonnative sesquiterpenes patchoulol and amorpha-4,11-diene, was achieved in transgenic N. tabacum when an avian FPS gene and nonnative sesquiterpene synthase, ADS or patchoulol synthase (PTS), genes were transformed into the nuclear genome with plastid targeting signal sequences appended to the amino-terminus of encoded proteins. Using the same strategy, up to ~500 µg/g FW of the triterpenes squalene and botryococcene were achieved when an avian FPS gene in combination with either a yeast squalene synthase (SQS) or an engineered algal botryococcene synthase (BS) were targeted to the chloroplast compartment [180] and Jiang et al., before unpublished). Conceptually, the plastid-targeted FPS diverts the IPP and DMAPP intermediates from the MEP pathway towards the accumulation of free FPP, the substrate for sesquiterpene and triterpene production. The biosynthesis of FPP, sesquiterpenes, and triterpenes are foreign to the chloroplast (Fig. 1), and introduction of these two-step biochemical pathways allows for elevated nonnative terpene production in the plastid compartment, sheltering these nonnative biosynthetic pathways from any native regulatory mechanisms and hence allowing for an unlimited flux of carbon to a desired terpene. Kumar et al. [84] corroborated this notion in an independent study where the entire yeast MVA pathway (a total of six enzymes) was introduced into the chloroplast genome of tobacco. The resulting homoplasmic transgenic lines accumulated multiple isoprenoid products, including mevalonate, carotenoids, sterols, squalene, and interestingly, triacylglycerides.
When engineering novel biosynthetic capacity into the chloroplast, there is an important choice to be made between plastidic transformation versus nuclear transformation. Plastidic transformation offers several advantages over nuclear transformation: homologous recombination methodology exists, expression of transgene operons could improve coordinated gene expression, transgene inheritance should only pass from maternal tissue, nuclear epigenetic effects should not present any difficulties, and the translation of expressed transgenes into protein is likely to be higher compared to nuclear transgene expression [27, 29, 84]. However, there are no direct comparisons of isoprenoid production by plastid genome transformations versus nuclear genome transformations expressing the same enzymes within or targeted to the chloroplast. Moreover, the higher level of protein expression does not always positively correlate with a higher level of terpene production and reflects the need to consider many factors, such as: protein (enzyme) activity, substrate availability, flux control within the pathway, and other regulatory complexities that may exist in the plastid. Thus, the best transformation strategy, nuclear versus plastidic, will probably vary on a case-by-case basis.
Metabolic engineering of higher plant chloroplasts offers additional potential for high yields of isoprenoid production: (1) there are many plastids within each higher plant cell (up to 50) that have polyploidy genomes (up to 1,000 copies per plastid); (2) because they are the site of photosynthesis, carbon flux in chloroplasts is robust and theoretically could provide for an unlimited supply of precursors; (3) chloroplasts may offer a good environment for exogenous protein folding, expression, and activity; (4) chloroplasts appear especially suitable for heterologous isoprenoid production due to lax endogenous regulation of the MEP pathway as compared to the MVA pathway operating in the cytosol; and (5) there is now the possibility to improve photosynthetic efficiency, which could increase production of engineered compounds [74].
A recent review by Heinig et al. [61] discussed the current challenges in conducting subcellular targeting in plant metabolic engineering. They suggested important considerations needed to be given to ensure substrate availability, whether this be overcome by co-introducing transporters, upstream catalytic enzymes to increase pathway flux, or suppressing endogenous pathways that bleed away precursors or cofactors. Although the most successful subcellular targeting efforts so far have used nuclear-encoded propeptides, which include transit peptides that direct the final protein to its intracellular destination, further development and optimization of plastid-encoded enzymes could allow for even higher titers of products. Engineering genetic constructs into the chloroplast has been possible since the late twentieth century [28, 82, 110], and high protein titers have been obtained, however, engineering chloroplast-encoded catalytically active enzymes has been less successful. Hence, there appears to be an inherent limitation in the chloroplast for the biosynthesis of high levels of catalytically competent enzymes, a problem that currently does not seem to be receiving considerable attention.
5 Altering Expression Patterns of Endogenous Genes
5.1 Downregulation
Downregulation or knock-out of endogenous biosynthetic genes is another important strategy commonly used to regulate/redirect metabolic pathway flux. These efforts attempt to suppress or abolish gene expression of a possible competing enzyme, thus flux can be redirected into the desired enzyme/pathway. However, this technique has not been used substantially and effectively in plant metabolic engineering compared with efforts in microbial systems. This is, in part, because of the difficulties in obtaining specific plant mutants and the low efficiency in obtaining appropriate amounts of downregulation of gene expression in plants (due mainly to large unknown genomes and/or redundant genes). Nevertheless, a number of studies have reported that antisense RNA and RNAi (RNA interference) techniques have been successfully used in manipulating plant terpene metabolism to increase terpenoid production.
Monoterpene essential oil production was elevated (61 % yield increase over WT plants) in transgenic peppermint expressing peppermint antisense (+)-menthofuran synthase (MFS) with simultaneous overexpression of DXR [88]. Downregulation of MFS alone was shown to decrease the level of side-product (+)-menthofuran, and redirected carbon flux to desirable monoterpene oil production, leading to an increased oil yield of roughly 35 % [104].
Tuber-specific expression of antisense fragments for either LCY-e or β-carotene hydroxylase, the genes encoding the enzymes that compete for lycopene and further metabolism of β-carotene, respectively, led to significantly increased levels of β-carotene (up to 14-fold and 38-fold, respectively) and total carotenoids (up to 2.5-fold and 4.5-fold, respectively) in potato tuber [33, 34]. When LCY-e was suppressed by an RNAi approach, increased carotenoid content in B. napus seeds was reported [185]. RNAi was also used in several studies to increase the content of artemisinin in A. annua by downregulating SQS and β-caryophyllene synthase, both enzymes that compete for FPP, which is a key intermediate in the artemisinin biosynthetic pathway [15, 46, 187]. When the C. roseus gene encoding 7-deoxyloganic acid 7-hydroxylase (CrDL7H), which is involved in secologanin biosynthesis, was suppressed by virus-induced gene silencing the accumulation of secologanin was reduced by at least 70 %. Critically, the accumulation of the intermediate, 7-deoxyloganic acid (the substrate for CrDL7H), was 4 mg/g FW in silenced plants whereas this compound is normally undetectable in WT plants [157].
Transgenic oranges with reduced levels of limonene caused by an antisense downregulation of the (+)-limonene synthase gene, were shown to be resistant to economically important pathogens [147]. Exactly how the reduced level of limonene in fruits activates a defense response has yet to be determined, but reduced limonene accumulation does correlate with increased levels of GGPS which could provide substrate for the formation of diterpene antimicrobial compounds that inhibit pathogen infection. The authors of this study suggested that the link between limonene accumulation and pathogen attack could play an ecological role in facilitating seed dispersal by allowing frugivores easier access to the fruit pulp [148].
5.2 Transcription Factors
Most specialized metabolites in plants accumulate when plants respond to acute developmental or environmental signals [14]. Therefore, it seems reasonable that their biosynthetic genes would be regulated in a coordinated manner by transcriptional factors. Although transcriptional regulation of the isoprenoid biosynthetic pathways is not well characterized, metabolic engineering of certain regulatory genes (the transcriptional factors) provides a novel approach to enhance terpene production in plants [129].
Recent studies have reported transcription factors in A. annua that appear to regulate artemisinin biosynthesis. AaWRKY1 was characterized as a transcription factor that regulates the native ADS gene in A. annua. Transient expression of AaWRKY1 also led to increased transcript accumulation of a majority of artemisinin biosynthetic genes [103]. Two jasmonate-responsive (AaERF1 and AaERF2) and a trichome-specific (AaORA) AP2/ERF transcription factors were also characterized as positive regulators for artemisinin biosynthesis in A. annua. Overexpressing either transcription factor resulted in increased accumulation of artemisinin and artemisinic acid [99, 184]. In contrast and contrary to expectations, constitutive expression of an Arabidopsis blue light receptor, CRY1, gene in A. annua increased the abundance of the FPS, ADS, and CYP71AV1 transcripts, three important enzymes in artemisinin biosynthesis, and led to increased accumulation of artemisinin and anthocyanins [67].
Catharanthine accumulation was improved up to 6.5-fold higher than WT in C. roseus hairy roots by coexpression of the ORCA3 transcription factor and the gene encoding for geraniol 10-hydroxylase, an enzyme involved in the TIA biosynthetic pathway [173]. Likewise, overexpression of the Arabidopsis transcription factor Agamous-like 12 in C. roseus suspension cells promoted enhanced accumulation of ajmalicine, a TIA with antihypertensive properties [115]. Arabidopsis transcription factors MYC2 and MYB21 have also been reported to regulate the expression of sesquiterpene synthase genes TPS11 and TPS21. When mutated, the Arabidopsis myc2 and myb21 mutants emit less sesquiterpene volatiles from their flowers than the WT plants, which was correlated with reduced levels of the TPS11 and TPS21 mRNAs [68, 142].
6 Understanding and Utilizing Compartmentalization
Understanding the flux of metabolites throughout the plant cell and attempts to use in silico algorithms to predict how specific changes in one metabolite may affect overall cell homeostasis has been an active area of interest. Efforts in creating integrative maps for comprehending the systemic biology of an organism is a rather new field that has become more accessible with the proliferation of available bioinformatics data and computing power. Full-scale system simulations are not available yet, partially due to our lack of understanding of full reaction dynamics and kinetics. Current genome-scale models have four main constituents: a list of enzymes and their reactions, the types and constraints on those reactions (e.g., reversibility, capacity), gene–protein–reaction linkages, and functions that dictate cellular activity, that is, the stipulations by which the listed reactions will be constrained within the cellular model [162]. Databases defining a biological system in terms of its biochemical, genetic, and genomic data have been generated for several types of species, including plants. Plants that have had genome-scale models generated for them include: Arabidopsis [16, 113, 125, 132, 137, 177], Zea mays [26, 155], Sorghum bicolor [26], Saccharum officinarum [26], Hordeum vulgare [56, 112, 152], Oryza sativa [85], and B. napus [59, 60, 131].
In the case of Arabidopsis, one of the metabolic networks incorporated 5,253 gene-reaction inputs with a total of 1,567 unique reactions and 1,748 metabolites, and it also considered the subcellular divisions of the typical plant cell (e.g., plastids, mitochondria, peroxisomes) but was limited in the consideration of cellular metabolic pathways that it models [26]. These models are still being refined and so far only consider primary metabolism, thus they have limited utility in understanding flux through the isoprenoid pathways especially when the system is altered by heterologous enzymes, although they can give insight into disconnects between available precursors and experimentally observed flux. However, recent mathematical models for understanding important regulatory components of essential oil production in peppermint have received experimental support [143, 144]. The reader is directed to two recent reviews discussing metabolic modeling for further details [124, 162].
One of the annotations that is important in generating genome-scale models across the subcellular locations of a plant cell, is the role of transporters in allowing accessibility of specific metabolites to the various enzymes of specialized cellular compartments. Metabolite transporters for terpenoid compounds probably exist to facilitate interorganelle sharing of intermediates (Fig. 1). However, identification of these transporters has been lacking. Doshi et al. [36] conducted analyses on bacterial homologues (including some mutated versions) of the ATP-binding cassette (ABC) exporter, MsbA, which is responsible for exporting the lipid A-moiety of lipopolysaccharide from the inner to outer leaflet of the E. coli inner membrane. They found that heterologous expression of some of these MsbA homologues allowed an increase in carotenoid excretion from E. coli cells also engineered for novel carotenoid biosynthesis. Although these transporters could export the hydrophobic carotenoid compounds, natural transporters for all types of isoprenoids have yet to be elucidated. There is ongoing research into this area and recently a plasma membrane ABC transporter from N. tabacum, NtPDR1, was described that is involved in diterpene export across the plasma membrane. Expression of NtPDR1 was observed across several tissue types and induced in response to several stimuli [25]. ABC transporters for the secretion of hydrophobic cuticular waxes in epidermal cells have been identified [158] as well as a plastid-localized ABC transporter important for importing ER-synthesized lipids [153]. In light of this, it seems reasonable to speculate that specialized transporters for intra- and extracellular movement of isoprenoid compounds may exist. Alteration of isoprenoid exchange within the cell and deposition to the exterior of the cell could also be altered using engineered lipid transfer proteins (LTPs).
As stated previously, a recently identified tobacco trichome LTP showed increased trichome exudate of alkanes and diterpenes when overexpressed [17]. Other recent studies have identified additional putative lipid transfer proteins from B. napus and Zea mays which remain to be biochemically validated [93, 191]. However, even if these LTPs do not normally transport isoprenoid substrates, it may be possible to engineer them to recognize certain isoprenoid compounds. Using transporters within a metabolic engineering strategy could allow for sequestration of produced molecules within a specialized structure away from the site of synthesis, allowing for higher titers by removing the product from the site of synthesis, decreasing possible negative feedback on the biosynthetic enzyme(s), and promoting continued pathway flux via simple mass-action principles.
The notion of using synthetic biology to increase flux through a pathway has been a topic of considerable discussion. Synthetic protein scaffolds, where successive acting catalytic enzymes are tethered via specific epitopes to a protein scaffold, in theory allow for the creation of a synthetic metabolon in vivo. These synthetic metabolons can be designed so that the stoichiometry of the pathway components is optimized. This is exemplified by Dueber et al. [38] where a protein scaffold was used to tether three MVA pathway enzymes in a stoichiometrically optimized ratio, thus allowing for a 77-fold increase in MVA accumulation in scaffold-containing E. coli cells versus cells without the scaffold.
7 Algae Engineering
Algae are a polyphyletic group of water-dwelling, eukaryotic single and multicellular organisms with diverse morphological, physiological, and biochemical features. As do higher plants, algae fix CO2 through photosynthesis and can ultimately sequester this carbon in numerous downstream products such as terpenoids. Mass algae cultures can produce large amounts of terpenoids that are used in nutraceutical [188], pharmaceutical [174], and renewable fuel [130] applications without competing for arable farmland used in food production. Moreover, algae can achieve higher cellular densities than plants, enabling them to produce more compound per unit of land area [53]. Several species are already cultivated on a large scale for production of carotenoids with useful antioxidant and pigmentation properties [58]. Higher titers of these and other terpenoids are always desirable and metabolic engineering offers one route to accomplish this using genetic engineering strategies [53, 55]. Currently, no one phylum can be labeled superior for terpenoid metabolic engineering; too little is known about the incredible diversity that exists. From a biotechnological standpoint members of chlorophyta, particularly Chlamydomonas reinhardtii, have received the greatest attention because they are the best studied “omically,” physiologically, and biochemically, and because most terpenoid metabolic engineering efforts have been attempted in green algae.
Fundamental concepts for metabolic engineering of terpenoids in algae are largely built around strategies developed for higher plants; these include but are not limited to increasing substrate levels/access, deregulation of rate-limiting steps, reduction/elimination of competing pathways, and so on (see [31] for review). Accomplishing such feats first requires consideration of the pathway(s) that supply terpenoid precursors in the algae species of interest. Depending on phylogeny, the MVA and MEP biosynthetic pathways responsible for supplying IPP are differentially dispersed. Primary endosymbiotic algae such as glaucophyta and rhodophyta (red algae) typically possess both the MVA and MEP pathways whereas chlorophyta (green algae) only harbor the plastidic MEP pathway; secondary endosymbiotic algae phyla euglenophyta, chlorarachniophyta, and heterokontophyta generally utilize both pathways [96].
As noted for higher plants, metabolic engineering of terpenes in chlorophyta can be approached differentially by expressing transgenes from either the nuclear or chloroplast genome. Methodology exists for both types of genetic manipulations in Chlamydomonas reinhardtii, Haematococcus pluvialis, Chlorella ellipsoidea, Chlorella sorokiniana, Chlorella kessleri, Volvox carteri, and Gonium pectorale [133, 175]. Recombinant DNA is integrated randomly in the nuclear genome presumably through double-stranded chromosome breakage followed by nonhomologous end joining; homologous recombination has only been described in the nuclear genome of one nonchlorophyte algae, Nannochloropsis sp. [79]. As in higher plants, algal chloroplast transformations proceed through homologous recombination lending better genetic precision and yielding more reliable titers of protein, however, no significant manipulations of chloroplast terpenoid metabolism have been reported, possibly because of protein misfolding or the lack of posttranslational modifications that are needed for obtaining catalytically active enzyme [135]. Nuclear engineering offers alternative means for protein folding and posttranslation modifications. However, this approach has been plagued by low-level expression of transgenes [83, 139] due to transgene silencing [166] and positional effects [121]; hence, many independent transgenic lines per gene construct must be assessed before high producers are identified. Other important aspects of algae terpenoid metabolic engineering include promoter/terminator choice, intron inclusion, codon optimization, exogenous versus endogenous isoforms of catalytic enzymes, transformation method, and metabolite assessment (colorimetric, spectroscopic, chromatographic, cytometric) [53, 55, 70, 175]. However, despite all the advances in algal biotechnology [140], no precedent-setting examples have been published for algae metabolically engineered to produce high levels of terpenoids or any other specialized metabolites from nuclear encoded transgenes [21–23, 91, 95, 165].
Lackluster results for algal terpenoid engineering insinuate underlying biological issues not yet fully appreciated. One area that has been investigated extensively is construct design for nuclear transformation. Transgene promoter analyses suggest that nuclear gene silencing occurs via a chromatin-based, epigenetic mechanism (histone methylation/acylation) that can be bypassed if proper cis-elements are placed upstream of a strong constitutive promoter [166]. One study demonstrated successful nuclear transgene expression by using an endogenous nuclear promoter from the highly expressed, intronless psaD gene, but also noted the importance of a transcriptional terminator element as well [83]. These investigators also determined that transformation with linear DNA improved transformant recovery and transgene expression [83].
An alternative approach to promoter/terminator choice has been constructing inframe chimeras with transgenes fused to an antibiotic resistance enzyme via a 2A linker. When this chimera is expressed, two separate polypeptides are produced due to ribosomal skipping of a peptide bond in the 2A linker sequence. In effect, the cell is forced to express both proteins for survival because of the antibiotic present [139–141]. This has been utilized successfully to express active enzymes and could be used to target enzymes to various subcellular locales such the ER or chloroplasts [139–141].
In addition to construct design and transformation methodology, C. reinhardtii strains (UVM4 and UVM11) have been developed for higher efficiency expression of nonselectable transgenes [121]. The open-reading frame for C. reinhardtii SQS has been integrated into the nuclear genome of the UVM strains and although appreciable levels of the corresponding protein were detected, enzymatic activity was not assessed [81]. Thus, using the C. reinhardtii UVM strains to express nuclear transgenes inframe with the 2A linker and antibiotic resistance gene under control of the highly expressed, intronless psaD promoter and terminator may be beneficial in future attempts to engineer terpene metabolism in algae. Overcoming the nuclear transgene expression problem in C. reinhardtii, and presumably other green algae, is one hurdle that needs to be addressed before complex genetic engineering efforts such as expressing the entire MVA pathway in the cytosol of a green alga or adding extra copies of the MEP pathway enzymes can be contemplated.
Another key area for future algal metabolic engineering efforts will be to understand algae-specific oleaginous traits. New “omics” studies have suggested there are different genes and proteins contributing to the ability of a species to accumulate their respective lipids. Specifically, in the heterokont Nannochloropsis sp., gene dosage (13 copies of DGAT vs. 4 in C. reinhardtii) seems to be extremely important for the accumulation of high levels of TAGs [81]. Moreover, B. braunii race B constitutively expresses three distinct genes for DXS, presumably to promote high carbon flux through the MEP pathway and into the abundant triterpene molecules that it accumulates [107]. In silica predictions such as these are needed to guide the design of synthetic mechanisms and provide the testable hypotheses that will be necessary to uncover the fundamental principles controlling terpene metabolism in algae and higher plants [109].
High titers of naturally occurring algal terpenoids are possible as observed for Dunaliella salina [138] and Botryococcus braunii race B [41]. However, an overarching question is how do these algae accomplish this and what are the prospects for capturing these mechanisms and deploying them in other algae or plant species? B. braunii race B accumulates 30 % of its mass in triterpene oils, albeit at the cost of growth rate [41]. Increased growth rates can be achieved under various culture conditions while maintaining high levels of hydrocarbon production [78], but genetically engineered strains of this colony-forming algae have not been reported. Moreover, B. braunii hydrocarbon production and secretion into the extracellular matrix seem to be intimately linked with the cell cycle [167, 176], making prospects for the deployment of this mechanism into other algae more daunting. Thus, using B. braunii as a design template for terpenoid metabolic engineering approaches may not be as fruitful as other algae such as Dunaliella salina. This halophilic, marine green alga can accumulate 10–15 % of its mass in β-carotene stored in plastid globules; it can also form cytoplasmic globules of TAGs produced during stressful conditions [19, 30, 86, 136, 138]. Supposing that the carotenogenic enzymes could be knocked down/out in this alga, the available IPP/DMAPP in the chloroplast might be increased.; this could be bolstered by additional nuclear copies of DXS targeted to the plastid. Nuclear copies of isopentenyl diphosphate isomerase, prenyl phosphate synthase (such as FPS), and triterpene synthases including BS could then be targeted to the cytoplasm. Hypothetically, this could create an environment where the high levels of substrates produced in the chloroplast are drawn into the cytoplasm by mass-action effects and channeled into botryococcene (foreign molecule, no feedback regulation) that could be stored in pre-existing TAG cytoplasmic globules.
Currently, green algae are blank slates for testing new strategies to engineer higher terpenoid accumulations metabolically. Emerging information about specialized green algae metabolism will buttress systems/synthetic biology approaches where carbon fluxes are optimized among cellular pathways in order to achieve maximum growth and hydrocarbon production rates. Coupled with improved genetic engineering methodology and thorough bioinformatics, machine learning, and network flux analyses, ideal strategies will be revealed for manipulating terpenoid metabolism in green algae.
8 Conclusions and Future Directions
Several important considerations regarding metabolic engineering efforts in plants have been covered here, as well as a look at concerns that should be addressed before advanced higher plant and algae engineering can be successfully accomplished. Once a goal has been identified, one must approach the engineering variables from a macro- to microscale. However, the main choices are which host to engineer and how the genetic construct design will be designed. Construct design is where one can fine tune expression to specific cell/tissue/organ types or developmental stages using specific promoters, as well as direct the introduced protein(s) to discrete subcellular locales. A useful analogy for viewing internal metabolite flux is that of a plumbing system (Fig. 6). It is important to remember that the metabolic pathways within the cell are interconnected and that changing one parameter (such as swapping a connecting pipe within Fig. 6) will also alter flow through the various other pipes and pathways within the scheme. However, as represented by the open pipes within Fig. 6, we do not always know what and where certain metabolic pathways meet with each other nor how they physically interact. Certainly, a limitation in our knowledge base is that we can currently only obtain such information through empirical experimentation.
Plumbing model and analogy for metabolic flux. One can imagine that carbon flux throughout the cell can be modeled as a system of connected pipes. The pipe junctions represent enzymes and the connecting pipes represent the reactions catalyzed by those enzymes—with thinner pipes representing rate-limiting steps. In this model the water collects in various cisterns which represent the final metabolic product. These cisterns have a certain maximum capacity (maximum amount of end product which can accumulate) and this is monitored by various sensors (feedback mechanisms), which can alter valves (blue tee-shaped objects) that represent regulatory proteins (e.g. transcription factors) controlling flux through the various connecting pipes or reactions. However, there are many steps designated by question marks for which we do not know how they might interact with our metabolic network
The future of metabolic engineering will need to embrace a holistic use of all available technologies presented. Early plant engineering efforts focused on proof-of-concept experiments using basic tools to ensure transgene expression and show that heterologous proteins can be expressed and be catalytically active (such as [64]). Now our primary challenges seem more dependent upon our ability to integrate vast amounts of genetic, proteomic, and metabolomic information to construct complex recombinant expression cassettes, and test these engineering designs in economically valuable hosts [72, 105, 108, 156, 186]. For instance, refinement of metabolic flux models will help to identify endogenous rate-limiting steps and control mechanisms that can be altered to increase desired product accumulation.
The continued improvement and success of future experiments will also rely on precise transgene tuning using synthetic biology components to introduce and integrate metabolic pathway(s) into seamless functional units and avoid unwanted feedback from homeostatic mechanisms. This may involve using synthetic promoters that allow transgene expression during an appropriate time in development and in coordination with suppression of endogenous pathway genes, using RNAi or repressive transcription factors, which would otherwise rob substrate from the introduced pathway. Engineering expression cassettes into specific genomic locales may also remove the necessity to screen large numbers of transgenic lines that have had transgenic DNA randomly inserted, and allow for a more rapid and direct comparison between engineering efforts in various cultivars of the same species.
Creating synthetic regulatory mechanisms that effectively partition precursor availability and provide robust flux to the desired product without allowing buildup of intermediates may be another way to utilize endogenous enzymes to boost overall productivity. Efforts that hope to generate plants applicable to actual commercial targets should also focus on obtaining the highest amount of product accumulation in high-biomass/yielding lines with low input requirements (e.g., fertilizer). Building better plant production platforms now only seems limited by our ability to assimilate all of the available biological and technological information into higher levels or more complex architectures taking into account the nuances of network interactions and connectivity.
References
Agrawal A, Konno K (2009) Latex: a model for understanding mechanisms, ecology, and evolution of plant defense against herbivory. Annu Rev Ecol Evol Syst 40:311–331
Aharoni A, Giri A, Deuerlein S (2003) Terpenoid metabolism in wild-type and transgenic Arabidopsis plants. Plant Cell 15:2866–2884
Aharoni A, Giri AP, Deuerlein S, Griepink F, de Kogel W-J, Verstappen FWA, Verhoeven HA, Jongsma MA, Schwab W, Bouwmeester HJ (2003) Terpenoid metabolism in wild-type and transgenic Arabidopsis plants. Plant Cell 15:2866–2884
Aharoni A, Jongsma MA, Kim T-Y, Ri M-B, Giri AP, Verstappen FWA, Schwab W, Bouwmeester HJ (2006) Metabolic engineering of terpenoid biosynthesis in plants. Phytochem Rev 5:49–58
Bach TJ (1995) Some new aspects of isoprenoid biosynthesis in plants–a review. Lipids 30:191–202
Bai C, Twyman RM, Farre G, Sanahuja G, Christou P, Capell T, Zhu C (2011) A golden era--pro-vitamin A enhancement in diverse crops. In Vitro Cell Dev Biol 47:205–221
Beyer P (2010) Golden rice and ‘golden’ crops for human nutrition. New Biotechnol 27:478–481
Van Beilen JB, Poirier Y (2008) Production of renewable polymers from crop plants. Plant J 54:684–701
Besumbes O, Sauret-Güeto S, Phillips MA, Imperial S, Rodríguez-Concepción M, Boronat A (2004) Metabolic engineering of isoprenoid biosynthesis in Arabidopsis for the production of taxadiene, the first committed precursor of Taxol. Biotechnol Bioeng 88:168–175
Bohlmann J, Keeling CI (2008) Terpenoid biomaterials. Plant J 54:656–669
Bonfill M, Malik S, Mirjalili M, Goleniowski M, Cusido R, Palazón J (2013) Production and genetic engineering of terpenoids production in plant cell and organ cultures. In: Ramawat KG, Mérillon J-M (eds) Natural products. Springer, Berlin, pp 2761–2796
Cahoon EB, Hall SE, Ripp KG, Ganzke TS, Hitz WD, Coughlan SJ (2003) Metabolic redesign of vitamin E biosynthesis in plants for tocotrienol production and increased antioxidant content. Nat Biotechnol 21:1082–1087
Chappell J (1995) Biochemistry and molecular biology of the isoprenoid biosynthetic pathway in plants. Annu Rev Plant Physiol Plant Mol Biol 46:521–547
Chappell J, Nable R (1987) Induction of sesquiterpenoid biosynthesis in tobacco cell suspension cultures by fungal elicitor. Plant Physiol 85:469–473
Chen J-L, Fang H-M, Ji Y-P, Pu G-B, Guo Y-W, Huang L-L, Du Z-G, Liu B-Y, Ye H-C, Li G-F et al (2011) Artemisinin biosynthesis enhancement in transgenic Artemisia annua plants by downregulation of the β-caryophyllene synthase gene. Planta Med 77:1759–1765
Cheung CYM, Williams TCR, Poolman MG, Fell DA, Ratcliffe RG, Sweetlove LJ (2013) A method for accounting for maintenance costs in flux balance analysis improves the prediction of plant cell metabolic phenotypes under stress conditions. Plant J 75:1050–1061
Choi YE, Lim S, Kim H-J, Han JY, Lee M-H, Yang Y, Kim J-A, Kim Y-S (2012) Tobacco NtLTP1, a glandular-specific lipid transfer protein, is required for lipid secretion from glandular trichomes. Plant J 70:480–491
Chow K-S, Mat-Isa M-N, Bahari A, Ghazali A-K, Alias H, Mohd-Zainuddin Z, Hoh C-C, Wan K-L (2012) Metabolic routes affecting rubber biosynthesis in Hevea brasiliensis latex. J Exp Bot 63:1863–1871
Cifuentes AS, Gonzalez M, Conejeros M, Dellarossa V, Parra O (1992) Growth and carotenogenesis in eight strains of Dunaliella saline Teodoresco from Chile. J Appl Phycol 4:111–118
Closa M, Vranova E, Bortolotti C, Bigler L, Arro M, Ferrer A, Gruissem W (2010) The Arabidopsis thaliana FPP synthase isozymes have overlapping and specific functions in isoprenoid biosynthesis, and complete loss of FPP synthase activity causes early developmental arrest. Plant J 63:512–525
Cordero BF, Couso I, León R, Rodríguez H, Vargas MA (2011) Enhancement of carotenoids biosynthesis in Chlamydomonas reinhardtii by nuclear transformation using a phytoene synthase gene isolated from Chlorella zofingiensis. Appl Microbiol Biotechnol 91:341–351
Couso I, Cordero BF, Vargas MÁ, Rodríguez H (2012) Efficient heterologous transformation of Chlamydomonas reinhardtii npq2 mutant with the zeaxanthin epoxidase gene isolated and characterized from Chlorella zofingiensis. Mar Drugs 10:1955–1976
Couso I, Vila M, Rodriguez H, Vargas MA, León R (2011) Overexpression of an exogenous phytoene synthase gene in the unicellular alga Chlamydomonas reinhardtii leads to an increase in the content of carotenoids. Biotechnol Prog 27:54–60
Croteau RB, Davis EM, Ringer KL, Wildung MR (2005) (−)-Menthol biosynthesis and molecular genetics. Naturwissenschaften 92:562–577
Crouzet J, Roland J, Peeters E, Trombik T, Ducos E, Nader J, Boutry M (2013) NtPDR1, a plasma membrane ABC transporter from Nicotiana tabacum, is involved in diterpene transport. Plant Mol Biol 82:181–192
Dal’Molin CGDO, Quek L-E, Palfreyman RW, Brumbley SM, Nielsen LK (2010) C4GEM, a genome-scale metabolic model to study C4 plant metabolism. Plant Physiol 154:1871–1885
Daniell H (2006) Production of biopharmaceuticals and vaccines in plants via the chloroplast genome. Biotechnol J 1:1071–1079
Daniell H, Datta R, Varma S, Gray S, Lee SB (1998) Containment of herbicide resistance through genetic engineering of the chloroplast genome. Nat Biotechnol 16:345–348
Daniell H, Kumar S, Dufourmantel N (2005) Breakthrough in chloroplast genetic engineering of agronomically important crops. Trends Biotechnol 23:238–245
Davidi L, Katz A, Pick U (2012) Characterization of major lipid droplet proteins from Dunaliella. Planta 236:19–33
Davies FK, Jinkerson RE, Posewitz MC (2014) Toward a photosynthetic microbial platform for terpenoid engineering. Photosynth Res. doi:10.1007/s11120-014-9979-6
Degenhardt J, Hiltpold I, Köllner TG, Frey M, Gierl A, Gershenzon J, Hibbard BE, Ellersieck MR, Turlings TCJ (2009) Restoring a maize root signal that attracts insect-killing nematodes to control a major pest. Proc Natl Acad Sci USA 106:13213–13218
Diretto G, Al-Babili S, Tavazza R, Papacchioli V, Beyer P, Giuliano G (2007) Metabolic engineering of potato carotenoid content through tuber-specific overexpression of a bacterial mini-pathway. PLoS One 2:e350
Diretto G, Tavazza R, Welsch R, Pizzichini D, Mourgues F, Papacchioli V, Beyer P, Giuliano G (2006) Metabolic engineering of potato tuber carotenoids through tuber-specific silencing of lycopene epsilon cyclase. BMC Plant Biol 6:13
Disch A, Hemmerlin A, Bach TJ, Rohmer M (1998) Mevalonate-derived isopentenyl diphosphate is the biosynthetic precursor of ubiquinone prenyl side chain in tobacco BY-2 cells. Biochem J 331:615–621
Doshi R, Nguyen T, Chang G (2013) Transporter-mediated biofuel secretion. Proc Natl Acad Sci USA 110:7642–7647
Ducreux LJM, Morris WL, Hedley PE, Shepherd T, Davies HV, Millam S, Taylor MA (2005) Metabolic engineering of high carotenoid potato tubers containing enhanced levels of beta-carotene and lutein. J Exp Bot 56:81–89
Dueber JE, Wu GC, Malmirchegini GR, Moon TS, Petzold CJ, Ullal AV, Prather KLJ, Keasling JD (2009) Synthetic protein scaffolds provide modular control over metabolic flux. Nat Biotechnol 27:753–759
Van Eenennaam AL, Lincoln K, Durrett TP, Valentin HE, Shewmaker CK, Thorne GM, Jiang J, Baszis SR, Levering CK, Aasen ED et al (2003) Engineering vitamin E content: from Arabidopsis mutant to soy oil. Plant Cell 15:3007–3019
Elzen GW, Williams HJ, Alois AB, Stipanovic RD, Vinson SB (1985) Quantification of volatile terpenes of glanded and glandless Gossypium hirsutum L. cultivars and lines by gas chromatography. J Agric Food Chem 33:1079–1082
Eroglu E, Okada S, Melis A (2011) Hydrocarbon productivities in different Botryococcus strains: comparative methods in product quantification. J Appl Phycol 23:763–775
Estévez JM, Cantero A, Reindl A, Reichler S, León P (2001) 1-Deoxy-D-xylulose-5-phosphate synthase, a limiting enzyme for plastidic isoprenoid biosynthesis in plants. J Biol Chem 276:22901–22909
Facchini PJ, De Luca V (2008) Opium poppy and Madagascar periwinkle: model non-model systems to investigate alkaloid biosynthesis in plants. Plant J 54:763–784
Farhi M, Marhevka E, Ben-Ari J, Algamas-Dimantov A, Liang Z, Zeevi V, Edelbaum O, Spitzer-Rimon B, Abeliovich H, Schwartz B et al (2011) Generation of the potent anti-malarial drug artemisinin in tobacco. Nat Biotechnol 29:1072–1074
Farre G, Bai C, Twyman RM, Capell T, Christou P, Zhu C (2011) Nutritious crops producing multiple carotenoids--a metabolic balancing act. Trends Plant Sci 16:532–54
Feng L-L, Yang R-Y, Yang X-Q, Zeng X-M, Lu W-J, Zeng Q-P (2009) Synergistic re-channeling of mevalonate pathway for enhanced artemisinin production in transgenic Artemisia annua. Plant Sci 177:57–67
Fischer K, Kammerer B, Gutensohn M, Arbinger B, Weber A, Hausler R, Flugge U-I (1997) A new class of plastidic phosphate translocators: a putative link between primary and secondary metabolism by the phosphoenolpyruvate/phosphate antiporter. The Plant Cell 9:453–462
Fraser PD, Romer S, Shipton CA, Mills PB, Kiano JW, Misawa N, Drake RG, Schuch W, Bramley PM (2002) Evaluation of transgenic tomato plants expressing an additional phytoene synthase in a fruit-specific manner. Proc Natl Acad Sci USA 99:1092–1097
Fraser PD, Enfissi EMA, Bramley PM (2009) Genetic engineering of carotenoid formation in tomato fruit and the potential application of systems and synthetic biology approaches. Arch Biochem Biophys 483:196–204
Fray RG, Wallace A, Fraser PD, Valero D, Hedden P, Bramley PM, Grierson D (1995) Constitutive expression of a fruit phytoene synthase gene in transgenic tomatoes causes dwarfism by redirecting metabolites from the gibberellin pathway. Plant J 8:693–701
Fujisawa M, Takita E, Harada H, Sakurai N, Suzuki H, Ohyama K, Shibata D, Misawa N (2009) Pathway engineering of Brassica napus seeds using multiple key enzyme genes involved in ketocarotenoid formation. J Exp Bot 60:1319–1332
Furumoto T, Yamaguchi T, Ohshima-Ichie Y, Nakamura M, Tsuchida-Iwata Y, Shimamura M, Ohnishi J, Hata S, Gowik U, Westhoff P, Brautigam A, Weber APM, Izui K (2011) A plastidial sodium-dependent pyruvate transporter. Nature 476:472–475
Georgianna DR, Mayfield SP (2012) Exploiting diversity and synthetic biology for the production of algal biofuels. Nature 488:329–335
Ghirardo A, Wright LP, Bi Z, Rosenkranz M, Pulido P, Rodríguez-Concepción M, Niinemets Ü, Brüggemann N, Gershenzon J, Schnitzler J-P (2014) Metabolic flux analysis of plastidic isoprenoid biosynthesis in poplar leaves emitting and nonemitting isoprene. Plant Physiol 165:37–51
Gimpel JA, Specht EA, Georgianna DR, Mayfield SP (2013) Advances in microalgae engineering and synthetic biology applications for biofuel production. Curr Opin Chem Biol 17:489–495
Grafahrend-Belau E, Junker A, Eschenröder A, Müller J, Schreiber F, Junker BH (2013) Multiscale metabolic modeling: dynamic flux balance analysis on a whole-plant scale. Plant Physiol 163:637–647
Gutensohn M, Orlova I, Nguyen TTH, Davidovich-Rikanati R, Ferruzzi MG, Sitrit Y, Lewinsohn E, Pichersky E, Dudareva N (2013) Cytosolic monoterpene biosynthesis is supported by plastid-generated geranyl diphosphate substrate in transgenic tomato fruits. Plant J 75:351–363
Gómez PI, Inostroza I, Pizarro M, Pérez J (2013) From genetic improvement to commercial-scale mass culture of a Chilean strain of the green microalga Haematococcus pluvialis with enhanced productivity of the red ketocarotenoid astaxanthin. AoB Plants 5:plt026
Hay J, Schwender J (2011) Computational analysis of storage synthesis in developing Brassica napus L. (oilseed rape) embryos: flux variability analysis in relation to 13C metabolic flux analysis. Plant J 67:513–525
Hay J, Schwender J (2011) Metabolic network reconstruction and flux variability analysis of storage synthesis in developing oilseed rape (Brassica napus L.) embryos. Plant J 67:526–541
Heinig U, Gutensohn M, Dudareva N, Aharoni A (2013) The challenges of cellular compartmentalization in plant metabolic engineering. Curr Opin Biotechnol 24:239–246
Hemmerlin A, Harwood JL, Bach TJ (2012) A raison d’être for two distinct pathways in the early steps of plant isoprenoid biosynthesis? Prog Lipid Res 51:95–148
Hemmerlin A, Hoeffler J-F, Meyer O, Tritsch D, Kagan IA, Grosdemange-Billiard C, Rohmer M, Bach TJ (2003) Cross-talk between the cytosolic mevalonate and the plastidial methylerythritol phosphate pathways in tobacco bright yellow-2 cells. J Biol Chem 278:26666–26676
Hohn TM, Ohlrogge JB (1991) Expression of a fungal sesquiterpene cyclase gene in transgenic tobacco. Plant Physiol 97:460–462
Holmberg N, Harker M, Wallace AD, Clayton JC, Gibbard CL, Safford R (2003) Co-expression of N-terminal truncated 3-hydroxy-3-methylglutaryl CoA reductase and C24-sterol methyltransferase type 1 in transgenic tobacco enhances carbon flux towards end-product sterols. Plant J 36:12–20
Holmberg N, Harker M, Gibbard CL, Wallace AD, Clayton JC, Rawlins S, Hellyer A, Safford R (2002) Sterol C-24 methyltransferase type 1 controls the flux of carbon into sterol biosynthesis in tobacco seed. Plant Physiol 130:303–311
Hong G-J, Hu W-L, Li J-X, Chen X-Y, Wang L-J (2009) Increased accumulation of Artemisinin and Anthocyanins in Artemisia annua expressing the arabidopsis blue light receptor CRY1. Plant Mol Biol Report 27:334–341
Hong G-J, Xue X-Y, Mao Y-B, Wang L-J, Chen X-Y (2012) Arabidopsis MYC2 interacts with DELLA proteins in regulating sesquiterpene synthase gene expression. Plant Cell 24:2635–2648
Huang M, Sanchez-Moreiras AM, Abel C, Sohrabi R, Lee S, Gershenzon J, Tholl D (2012) The major volatile organic compound emitted from Arabidopsis thaliana flowers, the sesquiterpene (E)-β-caryophyllene, is a defense against a bacterial pathogen. New Phytol 193:997–1008
Hyka P, Lickova S, Přibyl P, Melzoch K, Kovar K (2012) Flow cytometry for the development of biotechnological processes with microalgae. Biotechnol Adv 31:2–16
Jackson MA, Sternes PR, Mudge SR, Graham MW, Birch RG (2014) Design rules for efficient transgene expression in plants. Plant Biotechnol J 12:925–933
Jaganath B, Subramanyam K, Mayavan S, Karthik S, Elayaraja D, Udayakumar R, Manickavasagam M, Ganapathi A (2014) An efficient in planta transformation of Jatropha curcas (L.) and multiplication of transformed plants through in vivo grafting. Protoplasma 251:591–601
Kappers IF, Aharoni A, van Herpen TWJM, Luckerhoff LLP, Dicke M, Bouwmeester HJ (2005) Genetic engineering of terpenoid metabolism attracts bodyguards to Arabidopsis. Science 309(80):2070–2072
Kebeish R, Niessen M, Thiruveedhi K, Bari R, Hirsch H-J, Rosenkranz R, Stäbler N, Schönfeld B, Kreuzaler F, Peterhänsel C (2007) Chloroplastic photorespiratory bypass increases photosynthesis and biomass production in Arabidopsis thaliana. Nat Biotechnol 25:593–599
Keeling CI, Bohlmann J (2006) Diterpene resin acids in conifers. Phytochemistry 67:2415–2423
Keeling CI, Bohlmann J (2006) Genes, enzymes and chemicals of terpenoid diversity in the constitutive and induced defence of conifers against insects and pathogens. New Phytol 170:657–675
Kessler A, Baldwin IT (2001) Defensive function of herbivore-induced plant volatile emissions in nature. Science 291:2141–2144
Khatri W, Hendrix R, Niehaus T, Chappell J, Curtis WR (2014) Hydrocarbon production in high density Botryococcus braunii race B continuous culture. Biotechnol Bioeng 111:493–503
Kilian O, Benemann CSE, Niyogi KK, Vick B (2011) High-efficiency homologous recombination in the oil-producing alga Nannochloropsis sp. Proc Natl Acad Sci 108:21265–21269
Kirby J, Keasling JD (2009) Biosynthesis of plant isoprenoids: perspectives for microbial engineering. Annu Rev Plant Biol 60:335–355
Kong F, Yamasaki T, Ohama T (2014) Expression levels of domestic cDNA cassettes integrated in the nuclear genomes of various Chlamydomonas reinhardtii strains. J Biosci Bioeng 117:613–616
Kota M, Daniell H, Varma S, Garczynski SF, Gould F, Moar WJ (1999) Overexpression of the Bacillus thuringiensis (Bt) Cry2Aa2 protein in chloroplasts confers resistance to plants against susceptible and Bt-resistant insects. Proc Natl Acad Sci USA 96:1840–1845
Kumar A, Falcao VR, Sayre RT (2013) Evaluating nuclear transgene expression systems in Chlamydomonas reinhardtii. Algal Res 2:321–332
Kumar S, Hahn FM, Baidoo E, Kahlon TS, Wood DF, McMahan CM, Cornish K, Keasling JD, Daniell H, Whalen MC (2012) Remodeling the isoprenoid pathway in tobacco by expressing the cytoplasmic mevalonate pathway in chloroplasts. Metab Eng 14:19–28
Lakshmanan M, Zhang Z, Mohanty B, Kwon J-Y, Choi H-Y, Nam H-J, Kim D-I, Lee D-Y (2013) Elucidating rice cell metabolism under flooding and drought stresses using flux-based modeling and analysis. Plant Physiol 162:2140–2150
Lamers PP, van de Laak CCW, Kaasenbrood PS, Lorier J, Janssen M, De Vos RCH, Bino RJ, Wijffels RH (2010) Carotenoid and fatty acid metabolism in light-stressed Dunaliella salina. Biotechnol Bioeng 106:638–648
Lange BM, Ahkami A (2013) Metabolic engineering of plant monoterpenes, sesquiterpenes and diterpenes—current status and future opportunities. Plant Biotechnol J 11:169–196
Lange BM, Mahmoud SS, Wildung MR, Turner GW, Davis EM, Lange I, Baker RC, Boydston RA, Croteau RB (2011) Improving peppermint essential oil yield and composition by metabolic engineering. Proc Natl Acad Sci USA 108:16944–16949
Lange BM, Turner GW (2013) Terpenoid biosynthesis in trichomes–current status and future opportunities. Plant Biotechnol J 11:2–22
Lavy M, Zuker A, Lewinsohn E, Larkov O, Ravid U, Weiss D (2002) Linalool and linalool oxide production in transgenic carnation flowers expressing the Clarkia breweri linalool synthase gene. Mol Breed 9:103–112
León R, Couso I, Fernández E (2007) Metabolic engineering of ketocarotenoids biosynthesis in the unicelullar microalga Chlamydomonas reinhardtii. J Biotechnol 130:143–152
Li L, Van Eck J (2007) Metabolic engineering of carotenoid accumulation by creating a metabolic sink. Transgenic Res 16:581–585
Li J, Gao G, Xu K, Chen B, Yan G, Li F, Qiao J, Zhang T, Wu X (2014) Genome-wide survey and expression analysis of the putative non-specific lipid transfer proteins in Brassica rapa L. PLoS One 9:e84556
Lichtenthaler HK, Schwender J, Disch A, Rohmer M (1997) Biosynthesis of isoprenoids in higher plant chloroplasts proceeds via a mevalonate-independent pathway. FEBS Lett 400:271–274
Liu J, Gerken H, Huang J, Chen F (2013) Engineering of an endogenous phytoene desaturase gene as a dominant selectable marker for Chlamydomonas reinhardtii transformation and enhanced biosynthesis of carotenoids. Process Biochem 48:788–795
Lohr M, Schwender J, Polle JEW (2012) Isoprenoid biosynthesis in eukaryotic phototrophs: a spotlight on algae. Plant Sci 185–186:9–22
Lopez AB, Van Eck J, Conlin BJ, Paolillo DJ, O’Neill J, Li L (2008) Effect of the cauliflower or transgene on carotenoid accumulation and chromoplast formation in transgenic potato tubers. J Exp Bot 59:213–223
Lu S, Van Eck J, Zhou X, Lopez AB, O’Halloran DM, Cosman KM, Conlin BJ, Paolillo DJ, Garvin DF, Vrebalov J et al (2006) The cauliflower Or gene encodes a DnaJ cysteine-rich domain-containing protein that mediates high levels of beta-carotene accumulation. Plant Cell 18:3594–3605
Lu X, Zhang L, Zhang F, Jiang W, Shen Q, Zhang L, Lv Z, Wang G, Tang K (2013) AaORA, a trichome-specific AP2/ERF transcription factor of Artemisia annua, is a positive regulator in the artemisinin biosynthetic pathway and in disease resistance to Botrytis cinerea. New Phytol 198:1191–1202
Lücker J, Bouwmeester HJ, Schwab W, Blaas J, van der Plas LH, Verhoeven HA (2001) Expression of Clarkia S-linalool synthase in transgenic petunia plants results in the accumulation of S-linalyl-beta-D-glucopyranoside. Plant J 27:315–324
Lücker J, Schwab W, Franssen MCR, Van Der Plas LHW, Bouwmeester HJ, Verhoeven HA (2004) Metabolic engineering of monoterpene biosynthesis: two-step production of (+)-trans-isopiperitenol by tobacco. Plant J 39:135–145
Lücker J, Schwab W, Van Hautum B, Blaas J, Van Der Plas LHW, Bouwmeester HJ, Verhoeven HA (2004) Increased and altered fragrance of tobacco plants after metabolic engineering using three monoterpene synthases from lemon. Plant Physiol 134:510–519
Ma D, Pu G, Lei C, Ma L, Wang H, Guo Y, Chen J, Du Z, Wang H, Li G et al (2009) Isolation and characterization of AaWRKY1, an Artemisia annua transcription factor that regulates the amorpha-4,11-diene synthase gene, a key gene of artemisinin biosynthesis. Plant Cell Physiol 50:2146–2161
Mahmoud SS, Croteau RB (2001) Metabolic engineering of essential oil yield and composition in mint by altering expression of deoxyxylulose phosphate reductoisomerase and menthofuran synthase. Proc Natl Acad Sci USA 98:8915–8920
Mallón R, Vieitez AM, Vidal N (2013) High-efficiency Agrobacterium-mediated transformation in Quercus robur: selection by use of a temporary immersion system and assessment by quantitative PCR. Plant Cell Tissue Organ Cult 114:171–185
Mary W, Crombie L, Crombie L (1986) Distribution of avenacins A-1, A-2, B-1 and B-2 in oat roots: their fungicidal activity towards “take-all” fungus. Phytochemistry 25:2069–2073
Matsushima D, Jenke-Kodama H, Sato Y, Fukunaga Y, Sumimoto K, Kuzuyama T, Matsunaga S, Okada S (2012) The single cellular green microalga Botryococcus braunii, race B possesses three distinct 1-deoxy-D-xylulose 5-phosphate synthases. Plant Sci 185–186:309–320
Mayavan S, Subramanyam K, Arun M, Rajesh M, Kapil Dev G, Sivanandhan G, Jaganath B, Manickavasagam M, Selvaraj N, Ganapathi A (2013) Agrobacterium tumefaciens-mediated in planta seed transformation strategy in sugarcane. Plant Cell Rep 32:1557–1574
Mayfield S (2013) The green revolution 2.0: the potential of algae for the production of biofuels and bioproducts. Genome 56:551–555
McBride KE, Svab Z, Schaaf DJ, Hogan PS, Stalker DM, Maliga P (1995) Amplification of a chimeric Bacillus gene in chloroplasts leads to an extraordinary level of an insecticidal protein in tobacco. Biotechnology (NY) 13:362–365
Mehrshahi P, Stefano G, Andaloro JM, Brandizzi F, Froehlich JE, DellaPenna D (2013) Transorganellar complementation redefines the biochemical continuity of endoplasmic reticulum and chloroplasts. Proc Natl Acad Sci USA 110:12126–12131
Melkus G, Rolletschek H, Fuchs J, Radchuk V, Grafahrend-Belau E, Sreenivasulu N, Rutten T, Weier D, Heinzel N, Schreiber F et al (2011) Dynamic 13C/1 H NMR imaging uncovers sugar allocation in the living seed. Plant Biotechnol J 9:1022–1037
Mintz-Oron S, Meir S, Malitsky S, Ruppin E, Aharoni A, Shlomi T (2012) Reconstruction of Arabidopsis metabolic network models accounting for subcellular compartmentalization and tissue-specificity. Proc Natl Acad Sci USA 109:339–344
Misawa N (2009) Pathway engineering of plants toward astaxanthin production. Plant Biotechnol 26:93–99
Montiel G, Breton C, Thiersault M, Burlat V, Jay-Allemand C, Gantet P (2007) Transcription factor Agamous-like 12 from Arabidopsis promotes tissue-like organization and alkaloid biosynthesis in Catharanthus roseus suspension cells. Metab Eng 9:125–132
Morandini P (2013) Control limits for accumulation of plant metabolites: brute force is no substitute for understanding. Plant Biotechnol J 11:253–267
Moses T, Pollier J, Thevelein JM, Goossens A (2013) Bioengineering of plant (tri)terpenoids: from metabolic engineering of plants to synthetic biology in vivo and in vitro. New Phytol 200:27–43
Murata J, De Luca V (2005) Localization of tabersonine 16-hydroxylase and 16-OH tabersonine-16-O-methyltransferase to leaf epidermal cells defines them as a major site of precursor biosynthesis in the vindoline pathway in Catharanthus roseus. Plant J 44:581–594
Murata J, Roepke J, Gordon H, De Luca V (2008) The leaf epidermome of Catharanthus roseus reveals its biochemical specialization. Plant Cell 20:524–542
Nagel R, Berasategui A, Paetz C, Gershenzon J, Schmidt A (2013) Overexpression of an isoprenyl diphosphate synthase in spruce leads to unexpected terpene diversion products that function in plant defense. Plant Physiol 164:555–569
Neupert J, Karcher D, Bock R (2009) Generation of Chlamydomonas strains that efficiently express nuclear transgenes. Plant J 57:1140–1150
Ohara K, Ujihara T, Endo T, Sato F, Yazaki K (2003) Limonene production in tobacco with Perilla limonene synthase cDNA. J Exp Bot 54:2635–2642
Okada K, Saito T, Nakagawa T, Kawamukai M, Kamiya Y (2000) Five geranylgeranyl diphosphate synthases expressed in different organs are localized into three subcellular compartments in Arabidopsis. Plant Physiol 122:1045–1056
De Oliveira Dal’Molin CG, Nielsen LK (2013) Plant genome-scale metabolic reconstruction and modelling. Curr Opin Biotechnol 24:271–277
De Oliveira Dal’Molin CG, Quek L-E, Palfreyman RW, Brumbley SM, Nielsen LK (2010) AraGEM, a genome-scale reconstruction of the primary metabolic network in Arabidopsis. Plant Physiol 152:579–589
Orlova I, Nagegowda DA, Kish CM, Gutensohn M, Maeda H, Varbanova M, Fridman E, Yamaguchi S, Hanada A, Kamiya Y et al (2009) The small subunit of snapdragon geranyl diphosphate synthase modifies the chain length specificity of tobacco geranylgeranyl diphosphate synthase in planta. Plant Cell 21:4002–4017
Outchkourov NS, Peters J, de Jong J, Rademakers W, Jongsma MA (2003) The promoter-terminator of chrysanthemum rbcS1 directs very high expression levels in plants. Planta 216:1003–1012
Pare PW, Tumlinson JH (1999) Plant volatiles as a defense against insect herbivores. Plant Physiol 121:325–331
Patra B, Schluttenhofer C, Wu Y, Pattanaik S, Yuan L (2013) Transcriptional regulation of secondary metabolite biosynthesis in plants. Biochim Biophys Acta 1829:1236–1247
Peralta-Yahya PP, Ouellet M, Chan R, Mukhopadhyay A, Keasling JD, Lee TS (2011) Identification and microbial production of a terpene-based advanced biofuel. Nat Commun 2:483
Pilalis E, Chatziioannou A, Thomasset B, Kolisis F (2011) An in silico compartmentalized metabolic model of Brassica napus enables the systemic study of regulatory aspects of plant central metabolism. Biotechnol Bioeng 108:1673–1682
Poolman MG, Miguet L, Sweetlove LJ, Fell DA (2009) A genome-scale metabolic model of Arabidopsis and some of its properties. Plant Physiol 151:1570–1581
Potvin G, Zhang Z (2010) Strategies for high-level recombinant protein expression in transgenic microalgae: a review. Biotechnol Adv 28:910–918
Pulido P, Toledo-Ortiz G, Phillips MA, Wright LP, Rodríguez-Concepción M (2013) Arabidopsis J-protein J20 delivers the first enzyme of the plastidial isoprenoid pathway to protein quality control. Plant Cell 25:4183–4194
Purton S, Szaub JB, Wannathong T, Young R, Economou CK (2013) Genetic engineering of algal chloroplasts: progress and prospects. Russ J Plant Physiol 60:491–499
Rabbani S, Beyer P, Lintig J, Hugueney P, Kleinig H (1998) Induced beta-carotene synthesis driven by triacylglycerol deposition in the unicellular alga Dunaliella bardawil. Plant Physiol 116:1239–1248
Radrich K, Tsuruoka Y, Dobson P, Gevorgyan A, Swainston N, Baart G, Schwartz J-M (2010) Integration of metabolic databases for the reconstruction of genome-scale metabolic networks. BMC Syst Biol 4:114
Ramos AA, Polle J, Tran D, Cushman JC, Jin E-S, Varela JC (2011) The unicellular green alga Dunaliella salina Teod. as a model for abiotic stress tolerance: genetic advances and future perspectives. Algae 26:3–20
Rasala BA, Barrera DJ, Ng J, Plucinak TM, Rosenberg JN, Weeks DP, Oyler GA, Peterson TC, Haerizadeh F, Mayfield SP (2013) Expanding the spectral palette of fluorescent proteins for the green microalga Chlamydomonas reinhardtii. Plant J 74:545–556
Rasala BA, Chao S-S, Pier M, Barrera DJ, Mayfield SP (2014) Enhanced genetic tools for engineering multigene traits into green algae. PLoS One 9:e94028
Rasala BA, Lee PA, Shen Z, Briggs SP, Mendez M, Mayfield SP (2012) Robust expression and secretion of Xylanase1 in Chlamydomonas reinhardtii by fusion to a selection gene and processing with the FMDV 2A peptide. PLoS One 7:e43349
Reeves PH, Ellis CM, Ploense SE, Wu M-F, Yadav V, Tholl D, Chételat A, Haupt I, Kennerley BJ, Hodgens C et al (2012) A regulatory network for coordinated flower maturation. PLoS Genet 8:e1002506
Rios-Estepa R, Lange I, Lee JM, Lange BM (2010) Mathematical modeling-guided evaluation of biochemical, developmental, environmental, and genotypic determinants of essential oil composition and yield in peppermint leaves. Plant Physiol 152:2105–2119
Rios-Estepa R, Turner GW, Lee JM, Croteau RB, Lange BM (2008) A systems biology approach identifies the biochemical mechanisms regulating monoterpenoid essential oil composition in peppermint. Proc Natl Acad Sci USA 105:2818–2823
Robert CAM, Erb M, Hiltpold I, Hibbard BE, Gaillard MDP, Bilat J, Degenhardt J, Cambet-Petit-Jean X, Turlings TCJ, Zwahlen C (2013) Genetically engineered maize plants reveal distinct costs and benefits of constitutive volatile emissions in the field. Plant Biotechnol J 11:628–639
Roberts SC (2007) Production and engineering of terpenoids in plant cell culture. Nat Chem Biol 3:387–395
Rodríguez A, San Andrés V, Cervera M, Redondo A, Alquézar B, Shimada T, Gadea J, Rodrigo MJ, Zacarías L, Palou L et al (2011) Terpene down-regulation in orange reveals the role of fruit aromas in mediating interactions with insect herbivores and pathogens. Plant Physiol 156:793–802
Rodríguez A, Shimada T, Cervera M, Alquézar B, Gadea J, Gómez-Cadenas A, De Ollas CJ, Rodrigo MJ, Zacarías L, Peña L (2014) Terpene down-regulation triggers defense responses in transgenic orange leading to resistance against fungal pathogens. Plant Physiol 164:321–339
Rogers ED, Jackson T, Moussaieff A, Aharoni A, Benfey PN (2012) Cell type-specific transcriptional profiling: implications for metabolite profiling. Plant J 70:5–17
Rohmer M (1999) The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat Prod Rep 16:565–574
Rohmer M, Knani M, Simonin P, Sutter B, Sahm H (1993) Isoprenoid biosynthesis in bacteria: a novel pathway for the early steps leading to isopentenyl diphosphate. Biochem J 295(2):517–524
Rolletschek H, Melkus G, Grafahrend-Belau E, Fuchs J, Heinzel N, Schreiber F, Jakob PM, Borisjuk L (2011) Combined noninvasive imaging and modeling approaches reveal metabolic compartmentation in the barley endosperm. Plant Cell 23:3041–3054
Roston RL, Gao J, Murcha MW, Whelan J, Benning C (2012) TGD1, -2, and -3 proteins involved in lipid trafficking form ATP-binding cassette (ABC) transporter with multiple substrate-binding proteins. J Biol Chem 287:21406–21415
Römer S, Fraser PD, Kiano JW, Shipton CA, Misawa N, Schuch W, Bramley PM (2000) Elevation of the provitamin a content of transgenic tomato plants. Nat Biotechnol 18:666–669
Saha R, Suthers PF, Maranas CD (2011) Zea mays iRS1563: a comprehensive genome-scale metabolic reconstruction of maize metabolism. PLoS One 6:e21784
Sahoo KK, Tripathi AK, Pareek A, Sopory SK, Singla-pareek SL (2011) An improved protocol for efficient transformation and regeneration of diverse indica rice cultivars. Plant Methods 7:49
Salim V, Yu F, Altarejos J, De Luca V (2013) Virus-induced gene silencing identifies Catharanthus roseus 7-deoxyloganic acid-7-hydroxylase, a step in iridoid and monoterpene indole alkaloid biosynthesis. Plant J 76:754–765
Samuels L, Kunst L, Jetter R (2008) Sealing plant surfaces: cuticular wax formation by epidermal cells. Annu Rev Plant Biol 59:683–707
Savidge B, Weiss JD, Wong YH, Lassner MW, Mitsky TA, Shewmaker CK, Post-Beittenmiller D, Valentin HE (2002) Isolation and characterization of homogentisate phytyltransferase genes from Synechocystis sp. PCC 6803 and Arabidopsis. Plant Physiol 129:321–332
Sawai S, Saito K (2011) Triterpenoid biosynthesis and engineering in plants. Front Plant Sci 2:25
Schilmiller AL, Miner DP, Larson M, McDowell E, Gang DR, Wilkerson C, Last RL (2010) Studies of a biochemical factory: tomato trichome deep expressed sequence tag sequencing and proteomics. Plant Physiol 153:1212–1223
Seaver SMD, Henry CS, Hanson AD (2012) Frontiers in metabolic reconstruction and modeling of plant genomes. J Exp Bot 63:2247–2258
Shintani D, DellaPenna D (1998) Elevating the vitamin E content of plants through metabolic engineering. Science 282(80):2098–2100
Sitbon F, Jonsson L (2001) Sterol composition and growth of transgenic tobacco plants expressing type-1 and type-2 sterol methyltransferases. Planta 212:568–572
Steinbrenner J, Sandmann G (2006) Transformation of the green alga Haematococcus pluvialis with a phytoene desaturase for accelerated astaxanthin biosynthesis. Appl Environ Microbiol 72:7477–7484
Strenkert D, Schmollinger S, Schroda M (2013) Heat shock factor 1 counteracts epigenetic silencing of nuclear transgenes in Chlamydomonas reinhardtii. Nucleic Acids Res 41:5273–5289
Suzuki R, Ito N, Uno Y, Nishii I, Kagiwada S, Okada S, Noguchi T (2013) Transformation of lipid bodies related to hydrocarbon accumulation in a green alga, Botryococcus braunii (Race B). PLoS One 8:e81626
Tissier A (2012) Trichome specific expression: promoters and their applications. In Çiftçi YÖ (ed) Transgenic plants-advances and limitations, March 2012. INTECH, pp 353–378
Unsicker SB, Kunert G, Gershenzon J (2009) Protective perfumes: the role of vegetative volatiles in plant defense against herbivores. Curr Opin Plant Biol 12:479–485
Vickers CE, Gershenzon J, Lerdau MT, Loreto F (2009) A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat Chem Biol 5:283–291
Vranová E, Coman D, Gruissem W (2012) Structure and dynamics of the isoprenoid pathway network. Mol Plant 5:318–333
Wallaart TE, Bouwmeester HJ, Hille J, Poppinga L, Maijers NC (2001) Amorpha-4,11-diene synthase: cloning and functional expression of a key enzyme in the biosynthetic pathway of the novel antimalarial drug artemisinin. Planta 212:460–465
Wang C-T, Liu H, Gao X-S, Zhang H-X (2010) Overexpression of G10H and ORCA3 in the hairy roots of Catharanthus roseus improves catharanthine production. Plant Cell Rep 29:887–894
Wang G, Tang W, Bidigare RR (2005) Terpenoids as therapeutic drugs and pharmaceutical agents. In: Zhang L, Demain AL (eds) Natural products. Humana Press, Totowa, pp 197–227
Wang B, Wang J, Zhang W, Meldrum DR (2012) Application of synthetic biology in cyanobacteria and algae. Front Microbiol 3:344
Weiss TL, Roth R, Goodson C, Vitha S, Black I, Azadi P, Rusch J, Holzenburg A, Devarenne TP, Goodenough U (2012) Colony organization in the green alga Botryococcus braunii (Race B) is specified by a complex extracellular matrix. Eukaryot Cell 11:1424–1440
Williams TCR, Poolman MG, Howden AJM, Schwarzlander M, Fell DA, Ratcliffe RG, Sweetlove LJ (2010) A genome-scale metabolic model accurately predicts fluxes in central carbon metabolism under stress conditions. Plant Physiol 154:311–323
Wink M (2003) Evolution of secondary metabolites from an ecological and molecular phylogenetic perspective. Phytochemistry 64:3–19
Wright LP, Rohwer JM, Ghirardo A, Hammerbacher A, Ortiz-Alcaide M, Raguschke B, Schnitzler J-P, Gershenzon J, Phillips MA (2014) Deoxyxylulose 5-phosphate synthase controls flux through the methylerythritol 4-phosphate pathway in Arabidopsis. Plant Physiol 165:1488–1504
Wu S, Jiang Z, Kempinski C, Eric Nybo S, Husodo S, Williams R, Chappell J (2012) Engineering triterpene metabolism in tobacco. Planta 236:867–877
Wu S, Schalk M, Clark A, Miles RB, Coates R, Chappell J (2006) Redirection of cytosolic or plastidic isoprenoid precursors elevates terpene production in plants. Nat Biotechnol 24:1441–1447
Wurtzel ET, Cuttriss A, Vallabhaneni R (2012) Maize provitamin a carotenoids, current resources, and future metabolic engineering challenges. Frontiers in Plant Science 3:29
Ye X, Al-Babili S, Klöti A, Zhang J, Lucca P, Beyer P, Potrykus I (2000) Engineering the provitamin A (beta-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 287:303–305
Yu Z-X, Li J-X, Yang C-Q, Hu W-L, Wang L-J, Chen X-Y (2012) The jasmonate-responsive AP2/ERF transcription factors AaERF1 and AaERF2 positively regulate artemisinin biosynthesis in Artemisia annua L. Mol Plant 5:353–365
Yu B, Lydiate DJ, Young LW, Schäfer UA, Hannoufa A (2008) Enhancing the carotenoid content of Brassica napus seeds by downregulating lycopene epsilon cyclase. Transgenic Res 17:573–585
Zhang W-J, Dewey RE, Boss W, Phillippy BQ, Qu R (2013) Enhanced Agrobacterium-mediated transformation efficiencies in monocot cells is associated with attenuated defense responses. Plant Mol Biol 81:273–286
Zhang L, Jing F, Li F, Li M, Wang Y, Wang G, Sun X, Tang K (2009) Development of transgenic Artemisia annua (Chinese wormwood) plants with an enhanced content of artemisinin, an effective anti-malarial drug, by hairpin-RNA-mediated gene silencing. Biotechnol Appl Biochem 52:199–207
Zhang Y, Li Y, Guo Y, Jiang H, Shen X (2009) A sesquiterpene quinone, dysidine, from the sponge Dysidea villosa, activates the insulin pathway through inhibition of PTPases. Acta Pharmacol Sin 30:333–345
Zhao L, Chang W, Xiao Y, Liu H, Liu P (2013) Methylerythritol phosphate pathway of isoprenoid biosynthesis. Annu Rev Biochem 82:497–530
Zook M, Hohn T, Bonnen A, Tsuji J, Hammerschmidt R (1996) Characterization of novel sesquiterpenoid biosynthesis in tobacco expressing a fungal sesquiterpene synthase. Plant Physiol 112:311–318
Zou H-W, Tian X-H, Ma G-H, Li Z-X (2013) Isolation and functional analysis of ZmLTP3, a homologue to Arabidopsis LTP3. Int J Mol Sci 14:5025–5035
Acknowledgments
Work from the Chappell laboratory mentioned in this chapter has been supported by the USDA, DOE, NSF, and by Sapphire Energy, Inc. The authors are also grateful for the many insightful discussions with past and current members of the Chappell laboratory.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Kempinski, C., Jiang, Z., Bell, S., Chappell, J. (2015). Metabolic Engineering of Higher Plants and Algae for Isoprenoid Production. In: Schrader, J., Bohlmann, J. (eds) Biotechnology of Isoprenoids. Advances in Biochemical Engineering/Biotechnology, vol 148. Springer, Cham. https://doi.org/10.1007/10_2014_290
Download citation
DOI: https://doi.org/10.1007/10_2014_290
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-20106-1
Online ISBN: 978-3-319-20107-8
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)