Abstract
Portal vein thrombosis (PVT) is usually associated with cirrhosis with reduced hepatic reserve. PVT sometimes has a natural history of spontaneous disappearance or shrinkage, but in other cases, PVT volume increases and portal vein blood flow is impaired, which reduces hepatic reserve causing portal hypertension, increased ascites, variceal exacerbation, and bleeding. Prognosis often does not differ between cases with and without PVT. In patients awaiting liver transplantation, the consensus recommendation for PVT is anticoagulant therapy, given that thrombus affects outcome and prognosis post-transplantation. Prophylactic low molecular weight heparin may prevent complicating PVT in patients with cirrhosis and delaying the progression to liver failure. However, it is not clear whether PVT affects prognosis directly. In terms of the effects of PVT on varicose veins (e.g., in the esophagus), variceal bleeding may occur and endoscopic treatment takes time. Thus, prevention and treatment of PVT may improve prognosis in patients with cirrhosis. Large-scale prospective studies of PVT and treatment are needed to clarify the types and effects of PVT on liver cirrhosis prognosis and identify good treatment targets.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Portal vein thrombosis
- Liver failure
- Ascites
- Esophageal varices
- Liver decompensation
- Anticoagulant therapy
- Prognosis
6.1 Impact of Portal Vein Thrombosis on Liver Decompensation and Prognosis
The appearance of portal vein thrombosis (PVT) is typically caused by three factors that may contribute to the progression of liver cirrhosis: stagnation of portal vein blood flow due to liver cirrhosis, abnormalities of various coagulation factors, and vascular endothelial damage. Alternatively, the appearance of thrombus may be a result of advanced liver cirrhosis or abnormal portal vein blood flow.
In Case 1, contrast-enhanced computed tomography (CECT) showed the appearance of PVT incidentally. There were no subjective symptoms, ascites, or changes in Child-Pugh score. The PVT disappeared after a short period of anticoagulant therapy (Fig. 6.1).
Case 2 was a patient with alcoholic liver cirrhosis and rupture of esophageal varices 1 year after endoscopic variceal ligation (EVL). CECT at that time revealed the appearance of PVT for the first time. At the same time, an increase in ascites was observed clinically. After hemostasis of the varicose vein, anticoagulant therapy was instituted, following which the PVT disappeared and ascites decreased (Fig. 6.2).
In Case 3, PVT was initially reduced following anticoagulant therapy, but the therapy was discontinued due to abnormal bleeding. Three years later, cavernous transformation occurred, pleural effusion and ascites worsened, and liver failure progressed (Fig. 6.3).
(a) Thrombus on initial appearance in the main trunk of the portal vein. (b) Thrombus disappeared after 3 months following anticoagulant therapy; (c) 3 years after anticoagulant therapy was discontinued due to abnormal bleeding, collateral circulation developed with evidence of pleural effusion and ascites
It can be seen then that the clinical course of PVT varies from case to case, and short-term observation of individual cases cannot conclusively determine whether PVT worsens the prognosis of liver cirrhosis. It is also unclear in the long term whether the appearance of PVT associated with advanced liver cirrhosis markedly influences the natural course of liver cirrhosis or whether it is itself the result of advanced liver cirrhosis. Opinions vary as to whether or not PVT affects the clinical course.
To clarify this issue, a prospective study was conducted in which 1243 cases of liver cirrhosis were thoroughly observed for about 4 years using Doppler ultrasound to detect the appearance of PVT and progression of liver disease. In total, 118 subjects had newly developed PVT, and the 5-year cumulative incidence was 10.7%. Seventeen developed completely obstructive PVT, and 14 progressed from partial thrombosis to complete obstruction. Eighty-seven were non-obstructive and partial thrombosis, which disappeared in 70% of cases at follow-up. Progression of liver disease was observed in 52 of the 118 cases, while exacerbation occurred before the appearance of thrombus in 23 cases. Conversely, the thrombus appeared at the same time in five cases, and exacerbation occurred after the appearance of the thrombus in 24 cases. It was concluded that progression of liver disease was independently associated with baseline age, body mass index, prothrombin time, serum albumin, and esophageal varices, but not PVT [1]. In a study of 150 patients with viral hepatitis associated with cirrhosis followed up using Doppler ultrasonography, 42 (28%) developed PVT, with a cumulative incidence of 12.8% per year and 20% in 5 years [2]. Thrombosis progressed in 7.2%, remained unchanged in 45.2%, and improved spontaneously in 47.6%. Cumulative survival rates were similar between the thrombotic and non-thrombotic groups. In a study of 42 patients with untreated partial extrahepatic PVT associated with cirrhosis and followed for an average of 27 months, PVT worsened in 48% of patients and improved in 45% [3]. There was no clear association between PVT progression and clinical outcome, and the baseline Child-Pugh score was the only independent predictor of survival or liver decompensation.
In each of these prospective observational studies, PVT was noted in some of the patients with cirrhosis. It has become clear that the volume of thrombus does not always increase; it may spontaneously shrink in some cases. In addition, individual studies concluded that the appearance of thrombus did not influence prognosis in patients with cirrhosis, and baseline liver reserve was associated with more advanced cirrhosis. Thus, PVT is believed to be the result, not the cause, of liver failure progression.
Conversely, the results of a meta-analysis involving 2436 cases in three studies reported that PVT was significantly associated with both mortality and decompensated ascites (Fig. 6.4) [4].
PVT and mortality. PVT is associated with an increased pooled risk of death in the absence of significant heterogeneity. (Reproduced from [4])
In the pathology of PVT, the original portal vein blood flow is finally occluded and collateral blood circulation, such as cavernous transformation, develops in cases where the lesion is partial (mural) and then disappears spontaneously. Previous studies include such cases, so the thrombus does not affect portal blood flow and cirrhosis to the same extent in all cases. As such, no unifying conclusions can be drawn from these studies.
In an interesting randomized controlled trial that examined the effectiveness of prophylactic anticoagulant therapy for primary prevention of PVT in patients with cirrhosis, the results suggest that the appearance of thrombus contributes to progression of cirrhosis [5]. Patients with cirrhosis with Child-Pugh scores 7–10 were randomly assigned to a treatment group (n = 34) administered the low molecular weight heparin enoxaparin twice daily for 48 weeks or an untreated control group (n = 36). The incidence of PVT was significantly lower in the enoxaparin group than in the control group, at 0% vs. 16.6% at 48 weeks, 0% vs. 27.7% at 96 weeks, and 8.8% vs. 27.7% at 192 weeks, respectively. The decompensation rate was 11.7% in the enoxaparin group and 59.4% in the control group, and the survival rate was also excellent in the enoxaparin group (Fig. 6.5). This study clarified that in patients with advanced liver cirrhosis, the absence of PVT had a better effect on delaying progression to liver failure and prognosis, even after short-term observation. However, the authors of the study hypothesized that differences in liver decompensation were not solely due to PVT, and that enoxaparin administration contributed to improved intestinal microcirculation and enterocytic damage, leading to reduced bacterial translocation. Whatever the cause, the fact that anticoagulant therapy prevented PVT, resulting in improved prognosis, had a major impact on subsequent treatment of PVT.
Actuarial probability of developing PVT or hepatic decompensation, and probability of survival according to treatment group. Probability of remaining free from (a) PVT and (b) hepatic decompensation, and (c) probability of survival. Dashed line: controls; continuous line: enoxaparin-treated patients. (Reproduced from [5])
Thus, prognosis for all cases with PVT was not necessarily worse than for those without PVT. However, it was confirmed that the appearance of PVT is associated with poor prognosis of liver cirrhosis. Therefore, a larger and more detailed clinical study is required to clarify the type of PVT cases that carries a poor prognosis.
6.2 Impact of PVT on Liver Cirrhosis-Related Complications
Esophagogastric varices are a typical complication of cirrhosis, and ruptured varicose veins have a prognostic impact. The presence of PVT is thought to increase portosystemic shunt flow and worsen varices. Because the bleeding rate from varices is higher when cirrhosis is complicated by PVT than when it is not, it is recommended that esophageal varices be evaluated by endoscopy [6]. In a meta-analysis by Loffredo et al. [7], four studies involving 158 patients showed significantly lower variceal bleeding rates in patients receiving anticoagulants for PVT than in controls, suggesting that the presence of thrombus may cause bleeding varicose veins (Fig. 6.6c). Regarding the outcomes of treating varices associated with PVT, a retrospective study examined the effect of endoscopic varicose vein ligation (EVL) [8]. Twenty-two cases of thrombosis required 50.9 days for varices to disappear, which was longer than the 43.4 days in cases without thrombosis. This suggests that EVL has no effect on recurrence of varices and can be performed without problems in patients with PVT.
(a) Meta-analysis of studies investigating complete recanalization of PVT according to anticoagulant treatment. (b) Meta-analysis of studies investigating progression of PVT according to anticoagulant treatment. (c) Meta-analysis of studies investigating variceal bleeding according to anticoagulant treatment. (Reproduced from [7])
Intestinal infarction is a rare complication associated with PVT, but it should be noted that it presents with serious symptoms. The occurrence of infarction is thought to be associated with complete occlusion of venous outflow and the contraction and occlusion of reflex arteries, in which the mesenteric arch does not function as a collateral pathway [9]. Many prospective studies of PVT do not report on the development of intestinal infarction. In a multicenter retrospective study that followed 173 cases of PVT for 2.5 years, intestinal ischemia or infarction led to intestinal resection in 3% of cases [10]. However, in this study, the majority of cases did not have liver disease, so intestinal infarction may occur less frequently in cases of liver cirrhosis. When symptoms of abdominal pain and diarrhea are observed in a case of PVT, it is necessary to evaluate intestinal blood flow using CECT. Special attention should be paid to cases of thrombosis that progress to rapid and complete obstruction or to cases of thrombosis in the distal mesenteric vein. In addition, because intestinal ischemia is a pathological condition with poor prognosis leading to multiple organ failure, patients with a history of intestinal ischemia may be eligible for continuous anticoagulation therapy [9, 11].
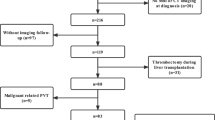
6.3 Impact of PVT on Liver Transplantation
Liver transplantation is an effective treatment for cirrhosis, but its outcome can be adversely affected by PVT, and in this respect the prognosis of liver cirrhosis can also be affected. A retrospective study of liver transplant cases from 2002 to 2013 found that 3321 out of 48,570 initial liver transplants, PVT occurred in 6.8%. Complications of PVT were independently associated with 90-day mortality as well as age and Model For End-stage Liver Disease score. PVT was also associated with graft failure after transplantation (Fig. 6.7). The presence of PVT was not associated with reduced transplant rates or death from exclusion from the transplant list. There were many cases in which PVT was confirmed for the first time during liver transplantation, and other cases in which thrombosis appeared during the waiting period. Fatty liver, obesity, diabetes, and ascites were risk factors for PVT during the waiting period [12]. In a meta-analysis of 44 studies on PVT and prognosis after liver transplantation, of 98,257 liver transplants, PVT occurred in 7257 (7.3%). The mortality rate at 30 days post-transplantation was 13% in patients with thrombosis and they had a poorer prognosis than the 7% of patients with no thrombosis. The mortality rate after 1 year was also similar; 13.5% of patients had thrombosis and a poorer prognosis than 9.9% with no thrombosis. Also, patients with complete thrombosis had significantly worse prognosis than those with partial thrombosis, especially in relation to the 30-day mortality rate [13]. The EASL guidelines recommend anticoagulant therapy for PVT while liver transplant candidates await transplantation [11].
Kaplan-Meier curves for (a) patient and (b) graft survival after liver transplantation in patients with PVT (black line) and without PVT (gray line), comparisons performed using the log-rank test. Vertical lines correspond to the 90-day timepoint after liver transplantation. (Reproduced from [12])
In order to prevent deterioration of liver transplantation outcomes due to PVT, it is necessary to establish measures against PVT while awaiting transplantation.
6.4 Effect of PVT Therapy on Liver Cirrhosis
There is still no clear evidence for the prognostic impact of treating PVT and its treatment remains controversial. Historically, evidence for the management of PVT in cirrhosis is of relatively poor quality, so there is urgent need for high levels of evidence for using high-risk anticoagulant therapy [14].
Therefore, while guidelines encourage consideration of anticoagulant therapy for cases awaiting liver transplantation and those with a history of bowel infarction, other treatment strategies and methods for PVT are still under discussion. The results of relatively small studies of anticoagulant therapies, such as warfarin and heparin and low molecular weight heparin, are well documented. In a meta-analysis of 353 cases included in eight studies comparing untreated and anticoagulant-treated cases, recanalization rates were 42% in the untreated group and 71% in the treated group, which was also complete recanalization [7]. The recanalization rate was also higher in the treatment group, at 53% compared with 33% in the untreated group (Fig. 6.6a, b). It has also been reported that there is no significant increase in hemorrhagic complications.
A study on the prevention of PVT with the anticoagulant enoxaparin was shown to yield good results in terms of hepatic reserve and prognosis [5]. However, the impact of treatment for evolving PVT on prognosis remains unclear. We investigated therapeutic efficacy and prognosis in 52 cases (Child-Pugh classification: A:13, B:25, C:14) who were administered with the low molecular weight heparin danaparoid sodium for 14 days [15]. Danaparoid sodium catalyzes inactivation of factors Xa (FXa) and thrombin. The volume of PVT was measured by CECT before and after treatment, and cases in which the volume was reduced by 75% or more compared with that before treatment were designated effective. PVT volume was significantly decreased from 6.1 ± 8.9 mL to 2.5 ± 7.4 mL; effective cases comprised 53.8%. In patients with low blood antithrombin (AT) activity, the effective rate was increased by increasing AT activity to 70% or more by combining AT preparations with danaparoid sodium. In all 52 cases, better prognosis was seen in Child-Pugh class A cases than in Child-Pugh class B and C cases. When limited to 39 cases of Child-Pugh class B and C, prognosis in the effective cases was 2262 days, which was significantly better than the 818 days in the non-effective cases (Fig. 6.8).
Prognosis of cirrhosis patients with PVT. (a) Comparison by hepatic reserve capacity: patients with compensated cirrhosis (Child-Pugh class A, n = 13) show significantly better prognosis than those with decompensated cirrhosis (Child-Pugh class B and C, n = 39; P = 0.0127). (b) Comparison by treatment effect: no significant difference is seen between the effective group (n = 28) and ineffective group (n = 24; P = 0.7128). (c) Comparison by presence of hepatocellular carcinoma (HCC): no significant difference is seen between presence of HCC (n = 21) and absence of HCC (n = 31; P = 0.0618). (d) Comparison by treatment effect in Child-Pugh class B and C decompensated cirrhosis: effective group (n = 20) shows significantly better prognosis than ineffective group (n = 19; P = 0.0179). (Reproduced from [15])
The therapeutic effect was an independent prognostic factor that was not affected by HCC, and no hemorrhagic complications were observed. Similarly, in 90 patients who used danaparoid sodium (Child-Pugh score 5–12 points, median 7 points), the 1-year survival rate was 83% and the 3-year survival rate was 60% [16]. Prognosis was significantly better in patients who achieved complete disappearance of PVT due to treatment compared to partial responders or non-responders. Prognosis was good for patients without HCC. Recently, the results on the use of direct oral anticoagulants (DOACs) have begun to be reported. Edoxaban, which is a DOAC, has the effect of inhibiting FXa similarly to danaparoid sodium, but it does not require AT. When either warfarin or edoxaban was given as maintenance therapy for 6 months after induction therapy with danaparoid sodium, edoxaban was superior to warfarin in reducing thrombosis [17]. On the other hand, hemorrhagic complications were observed in 15% of edoxaban users and 7% of warfarin users.
Although retrospective in design, the results showed that the better the thrombosis reducing effect, the better the prognosis of patients with PVT. On the other hand, it must be recognized that anticoagulant therapy given with the aim of improving prognosis can pose a risk of worsening prognosis. Large-scale randomized controlled trials are needed to determine whether treatment of PVT can really improve prognosis and which cases are good candidates for treatment.
6.5 Conclusions
In this chapter, we have discussed the effects of PVT on the clinical course and prognosis of liver cirrhosis. In some cases of PVT, the thrombus is partial and asymptomatic, spontaneously disappears, and has no significant effect on short-term prognosis. Conversely, in many cases after the appearance of PVT, ascites increases, variceal bleeding ensues, and liver failure progresses, resulting in poor prognosis. Differences in the course of PVT are thought to result from differences in the balance and severity of the three factors (i.e., portal vein blood flow stagnation, abnormal coagulation factors, and vascular endothelial damage). Shrinkage of PVT after anticoagulant therapies well correlates with favorable prognosis. Although prevention of PVT by anticoagulants may benefit cirrhosis patients (by improving intestinal microcirculation and inhibiting bacterial translocation), it is unrealistic to target all cases of cirrhosis. Future large-scale, long-term prospective studies should clarify the impact of PVT on cirrhosis and when and what therapeutic intervention is safe and effective.
References
Nery F, Chevret S, Condat B, et al. Causes and consequences of portal vein thrombosis in 1,243 patients with cirrhosis: results of a longitudinal study. Hepatology. 2015;61(2):660–7.
Maruyama H, Okugawa H, Takahashi M, et al. De novo portal vein thrombosis in virus-related cirrhosis: predictive factors and long-term outcomes. Am J Gastroenterol. 2013;108(4):568–74.
Luca A, Caruso S, Milazzo M, et al. Natural course of extrahepatic nonmalignant partial portal vein thrombosis in patients with cirrhosis. Radiology. 2012;265(1):124–32.
Stine JG, Shah PM, Cornella SL, et al. Portal vein thrombosis, mortality and hepatic decompensation in patients with cirrhosis: a meta-analysis. World J Hepatol. 2015;7(27):2774–80.
Villa E, Cammà C, Marietta M, et al. Enoxaparin prevents portal vein thrombosis and liver decompensation in patients with advanced cirrhosis. Gastroenterology. 2012;143(5):1253–60.
Tsochatzis EA, Senzolo M, Germani G, et al. Systematic review: portal vein thrombosis in cirrhosis. Aliment Pharmacol Ther. 2010;31(3):366–74.
Loffredo L, Pastori D, Farcomeni A, et al. Effects of anticoagulants in patients with cirrhosis and portal vein thrombosis: a systematic review and meta-analysis. Gastroenterology. 2017;153(2):480–7.
Dell’Era A, Iannuzzi F, Fabris FM, et al. Impact of portal vein thrombosis on the efficacy of endoscopic variceal band ligation. Dig Liver Dis. 2014;46(2):152–6.
DeLeve LD, Valla DC, Garcia-Tsao G. Vascular disorders of the liver. Hepatology. 2009;49(5):1729–64.
Rajani R, Björnsson E, Bergquist A. The epidemiology and clinical features of portal vein thrombosis: a multicentre study. Aliment Pharmacol Ther. 2010;32(9):1154–62.
European Association for the Study of the Liver. EASL clinical practice guidelines: vascular diseases of the liver. J Hepatol. 2016;64(1):179–202.
Ghabril M, Agarwal S, Lacerda M, et al. Portal vein thrombosis is a risk factor for poor early outcomes after liver transplantation: Analysis of risk factors and outcomes for portal vein thrombosis in waitlisted patients. Transplantation. 2016;100(1):126–33.
Zanetto A, Rodriguez-Kastro KI, Germani G, et al. Mortality in liver transplant recipients with portal vein thrombosis—an updated meta-analysis. Transpl Int. 2018;31(12):1318–29.
Qi X, Han G, Fan D. Management of portal vein thrombosis in liver cirrhosis. Nat Rev Gastroenterol Hepatol. 2014;11(7):435–46.
Hayashi T, Takatori H, Horii R, et al. Danaparoid sodium-based anticoagulation therapy for portal vein thrombosis in cirrhosis patients. BMC Gastroenterol. 2019;19(1):217. https://doi.org/10.1186/s12876-019-1140-8.
Fujiyama S, Saitoh S, Kawamura Y, et al. Portal vein thrombosis in liver cirrhosis: incidence, management, and outcome. BMC Gastroenterol. 2017;17(1):112. https://doi.org/10.1186/s12876-017-0668-8.
Nagaoki Y, Aikata H, Daijyo K, et al. Efficacy and safety of edoxaban for treatment of portal vein thrombosis following danaparoid sodium in patients with liver cirrhosis. Hepatol Res. 2018;48(1):51–8. https://doi.org/10.1111/hepr.12895.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Takatori, H., Hayashi, T., Nakagawa, H., Kaneko, S. (2021). Impact of Non-malignant Portal Vein Thrombosis on Outcomes of Liver Cirrhosis. In: Qi, X., Xie, W. (eds) Portal Vein Thrombosis. Springer, Singapore. https://doi.org/10.1007/978-981-33-6538-4_6
Download citation
DOI: https://doi.org/10.1007/978-981-33-6538-4_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-33-6537-7
Online ISBN: 978-981-33-6538-4
eBook Packages: MedicineMedicine (R0)