Abstract
Follow-up study can provide clinicians with prognosis information about the patients with fulminant myocarditis. The prognosis of patient with fulminant myocarditis seems different according to published literature, which is related to histological classification, clinical manifestation and treatments received. With the gaining knowledge about the endocardial biopsy of myocarditis, the infiltrated immune cells can provide guidance to the clinical treatment decision and prognosis. In this chapter, we reviewed the prognostic value of histological examinations, and the relationship of the prognosis with patient’s clinical manifestation and treatment received. Of note, the effect of control of chronic inflammation with immunosuppressive treatment to the long-term prognosis seems negative, but related clinical trials are still carrying on. Importantly, our patients with fulminant myocarditis who received “life support based comprehensive treatment regimen” treatments not only have much lower in-hospital mortality, but also have much better long term prognosis in one year follow-up.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
The clinical manifestations of myocarditis vary from mild dyspnea or chest pain, which can gradually disappear without special treatment for cardiogenic shock leading to death [1]. The prognosis can also vary. A prospective study including 174 cases of myocarditis confirmed by endocardial biopsy with a median follow-up of 23.5 months found that the positive polymerase chain reaction (PCR) detection of viral genes on endocardial biopsy was a factor contributing to poor prognosis [2]. Another study of 222 patients diagnosed with viral myocarditis by endocardial biopsy with a median follow-up of 4.7 years reported a mortality rate of 19.2% [3]. The prognosis of myocarditis is related to its etiology, clinical manifestations, and disease stages [2, 4, 5]. This chapter describes the prognosis of patients with viral myocarditis with different pathological tissue types, clinical symptoms, auxiliary examination findings, and clinical treatments.
19.1 Histopathological Type and Prognosis
The histological examination of endocardial biopsy in myocarditis shows cellular infiltration (Fig. 19.1), usually of tissue cells and mononuclear cells, and possibly with myocardial cell damage [6]. The specific histopathological types of myocarditis are mainly divided into lymphocytic myocarditis, eosinophilic myocarditis, and giant cell myocarditis.
Endocardial biopsies of four types of myocarditis. (a) Lymphocytic myocarditis (CD45RO immunoperoxidase stain, 200×); (b) eosinophilic myocarditis (250× with HE staining); (c) giant cell myocarditis (HE staining, 200×); (d) granulomatous myocarditis (HE staining, 200×) [6]. HE hematoxylin and eosin
Viral infection, the most common cause of lymphocytic myocarditis [1, 7, 8], usually begins within 3–5 days or up to 2 weeks of infection. Its course varies. The disease can be subclinical, indolent, or fulminant. When the disease is indolent, it gradually progresses to dilated cardiomyopathy (DCM); when it is fulminant, it may lead to death, or the patient may fully recover after reasonable treatment such as short-term hemodynamic support [9]. A study of 27 patients with lymphocytic myocarditis or critical myocarditis confirmed by endocardial biopsy reported a 5-year survival rate of 56%, which did not differ significantly from that of specific DCM [10]. In an earlier retrospective study of 112 patients with histopathologically diagnosed myocarditis, 66 (59%) had lymphocytic myocarditis. The 1- and 5-year follow-up rates of the total population without heart transplantation were 79% and 56%, respectively. A multivariate regression analysis suggested that histopathological findings (lymphocytic, granulomatous, or giant cell myocarditis) are among the factors predicting patient death or need for heart transplantation [11].
Giant cell myocarditis is a rare and severe autoimmune myocarditis that is virus-negative and usually fatal for which immunosuppressive therapy may be effective [12]. Compared with other pathological myocarditis, giant cell myocarditis has a higher mortality and heart transplantation rate [13]. Eosinophilic myocarditis, characterized by eosinophilic infiltration of the myocardium, is seen in malignancies, parasitic infections, allergic myocarditis, endocardial fibrosis, and idiopathic hypereosinophilic syndromes. In a meta-analysis of 264 patients with eosinophilic myocarditis, the average left ventricular ejection fraction (LVEF) at admission was 35%, 16.8% of patients required transient cardiopulmonary bypass, and the in-hospital mortality rate was 22.3% [14]. In recent years, immune checkpoint inhibitors have been widely used in tumor treatment and achieved significant effects. However, it cannot be ignored that 20–30% of patients have myocardial injury, 1–2% of patients develop fulminant myocarditis, and the risk of death is as high as 46–75% [15].
However, the above results were all reported by Western scholars and published before our report entitled “Life-support based comprehensive treatment regimen.” Therefore, it does not represent the latest research conclusions.
19.2 Clinical Manifestations and Prognosis
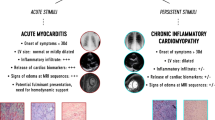
Fulminant myocarditis, the most serious and special type of myocarditis, is characterized by sudden-onset and extremely rapid disease progression. The patient rapidly develops abnormal hemodynamics and severe arrhythmia that may be accompanied by respiratory failure and liver and kidney failure, and the early mortality rate is extremely high. It is worth noting that some studies have found that, although the hospital mortality rate of this disease is high, once the acute crisis is passed, the long-term prognosis is good. An 11-year follow-up study in 2000 (Fig. 19.2) showed that, among patients diagnosed with myocarditis by endocardial biopsy, 14 patients with fulminant myocarditis had a significantly higher survival rate than 132 patients with normal acute myocarditis (93% and 45%, respectively), and the long-term survival rate was similar to that of the general population [16]. However, two articles published in 2017 and 2019 reported the opposite conclusion. In a previous study (Fig. 19.3), myocarditis was diagnosed by endocardial biopsy or cardiac magnetic resonance, and 34 patients with fulminant myocarditis and 96 patients with acute viral myocarditis were followed for 9 years [17]. In the latter study, myocarditis was diagnosed by histological examination, and 165 patients with fulminant myocarditis and 55 patients with acute viral myocarditis were followed for 60 days and 7 years, respectively [12]. Both studies showed that the survival rate without heart transplantation in patients with fulminant myocarditis was significantly lower than that in patients with acute viral myocarditis [12, 17]. The difference in results was related to the fact that all patients in the study published in 2000 were diagnosed with lymphocytic myocarditis by endocardial biopsy, whereas few patients were assisted with extracorporeal mechanical circulation for fulminant myocarditis at that time.
Survival of fulminant myocarditis versus acute lymphocytic myocarditis groups [ 16]
Survival of fulminant myocarditis versus non-fulminant myocarditis groups [17]
In a study published in 2017, only some patients underwent endocardial biopsy, and the pathological types of myocarditis were not classified and compared [18]. In a study published in 2019, although all of the patients underwent endocardial biopsies and lymphocytic myocarditis pathological types were compared, the changes in treatment methods for myocarditis (such as the use of external mechanical circulation assistance in patients with fulminant myocarditis) and differences in etiology led to somewhat different conclusions [19] (Fig. 19.4).
Short-term (left) and long-term (right) follow-up results of lymphocytic fulminant versus non-fulminant myocarditis [12]
Ammirati’s two studies also suggested that a QRS duration greater than 120 ms on admission is also a predictor of poor prognosis in patients with myocarditis [12, 17]. A prospective study of 174 patients with biopsy-diagnosed myocarditis found that biventricular dysfunction severity was a major predictor of death or heart transplantation in patients with myocarditis [18]. A follow-up study in Germany of 77 patients diagnosed with viral myocarditis by endocardial biopsy (EMB) reported that the presence of late gadolinium enhancement (LGE) of cardiac magnetic resonance was the most independent predictor of all-cause and cardiogenic death in patients [19]. A follow-up study in 2018 enrolled 443 patients with acute myocarditis diagnosed by endocardial biopsy or biomarker elevation plus meeting two cardiac magnetic resonance criteria for myocarditis (edema and late gadolinium enhancement in non-ischemic mode) reported that patients with complex acute myocarditis (defined as an initial LVEF of less than 50%, persistent ventricular arrhythmia, or low cardiac output syndrome requiring positive inotropic drugs or mechanical circulation support) had significantly higher mortality and heart transplant rates than patients with uncomplicated acute myocarditis [20]. Secondary pulmonary hypertension is a predictor of a poor outcome. In another study, 93 patients with myocarditis who underwent right heart catheterization and EMB were followed up for 4.4 years, and mean arterial pressure was the most important predictor of death [21].
The persistence of viral genomes in the myocardium may be an important factor in predicting the outcome of viral myocarditis. In one study, 172 patients with viral myocarditis diagnosed by biopsy were included, of whom 151 were infected with only one virus. After a median follow-up of 6.8 months, endocardial biopsy was repeated. Of them, 55 had spontaneous clearance of the virus genome and a significant increase in LVEF but a significant decline in LVEF was found in patients with persistent viral genome [22]. However, another follow-up study showed that the viral genome detected by endocardial biopsy at admission was not associated with poor prognosis, suggesting that the viral genome may sometimes reflect latent rather than active viral infection [23].
Serological markers, particularly soluble Fas ligands and interleukin-10 (IL-10), may be helpful for predicting the outcomes of acute severe myocarditis. A case series showed that the serum concentrations of soluble Fas and Fas ligand in patients with acute myocarditis were significantly higher than those in normal subjects or patients with old myocardial infarction, and the concentrations of Fas and Fas ligands in patients with myocarditis who died during hospitalization was significantly higher than that in patients with myocarditis who were discharged after recovery [24]. Another study included 20 patients with recently onset idiopathic DCM and the results further support the role of Fas [25]. A higher serum IL-10 concentrations at admission in patients with fulminant myocarditis may indicate cardiogenic shock and death [26].
Cardiac-specific autoantibodies can be identified in patients with acute or chronic myocarditis. The presence of autoantibodies is associated with an increased risk of chronic myocarditis progressing to DCM [27]. A case series of 33 patients with chronic myocarditis showed that patients with anti-α-myosin autoantibodies were less likely to have improved left ventricular systolic and diastolic function. Patients with this antibody showed no improvement in LVEF at 6 months, while those without this antibody showed a 9% absolute increase in LVEF. Similarly, in relatives of patients with idiopathic DCM, the presence of anti-cardiac antibodies predicts future left ventricular dilatation [27].
In summary, we found that the difference between myocardial enzyme spectrum changes in patients with myocarditis and myocardial infarction is that there is no obvious enzyme peak, indicating that the lesion is a gradual change, while the continuous increase indicates that the myocardium is continuously damaged and aggravated, indicating a poor prognosis. The levels of brain natriuretic peptide (BNP) or N-terminal proBNP are usually significantly increased, indicating that cardiac function is seriously impaired, and it is an important indicator for diagnosing cardiac insufficiency, evaluating its severity, and judging its progression and outcome. Neutropenia is a sign of poor prognosis, as is persistent thrombocytopenia.
19.3 Clinical Treatment and Prognosis
The treatment of patients with viral myocarditis can be divided into three parts: symptomatic and supportive treatment, immunosuppressive therapy, and immunomodulatory therapy [28]. Some clinical studies have discussed the effects of these treatments on the prognosis of patients with viral myocarditis. When hemodynamically stable heart failure with a reduced LVEF is present in patients with myocarditis, diuretic therapy on demand, diuretic therapy should be used as needed, angiotensin converting enzyme inhibitors (ACEI) or angiotensin II receptor blockers should be started as soon as possible, and beta-blockers should be used on an evidence-based basis. ACEI and beta-blockers have been shown to reduce complications and death in patients with systolic heart failure [29]. Animal experiments have shown that ACEI can improve myocardial necrosis and adverse outcomes caused by myocarditis [30, 31].
Cardiopulmonary bypass devices are necessary for patients with myocarditis and severe hemodynamic disorders. Although studies have found that intra-aortic balloon pumping (IABP) cannot improve the prognosis of patients with myocardial infarction [32], and large randomized controlled studies on the efficacy of IABP in patients with myocarditis are currently lacking, some retrospective studies or case reports have found that IABP can help patients with fulminant myocarditis in the acute phase [33,34,35]. The therapeutic effect of extracorporeal membrane oxygenation (ECMO) on fulminant myocarditis has been supported by a large number of clinical data [36,37,38,39,40]. In a study of 57 patients with fulminant myocarditis who received adjuvant ECMO therapy, 41 (71.9%) survived and were discharged from the hospital with a 5-year follow-up survival rate of 65.2% [36]. A statistical analysis of 3846 patients with cardiogenic shock treated with veno-arterial ECMO from January 2003 to December 2013 showed that 1601 patients (42%) survived and were discharged from the hospital, while chronic renal failure, hypotension, and low bicarbonate ion levels were associated with high mortality rates [38].
The widespread use of glucocorticoids in clinical practice is largely based on the doctors’ clinical experience [8]. Preliminary studies reported that immunosuppressive agents may be beneficial for certain patients with chronic myocarditis, but the effectiveness of immunosuppressive therapy for acute lymphocytic myocarditis of unknown etiology has not been proven. In 1989, two controlled studies showed that glucocorticoids did not improve the survival rate of patients with myocarditis (Fig. 19.5) [41]. One study included 102 patients with myocarditis and reported that the LVEF in the glucocorticoid treatment group was significantly improved compared with that of the control group within 3 months [42]. Long-term follow-up found no significant intergroup difference in LVEF [43]. Glucocorticoids combined with azathioprine therapy benefits the prognosis of patients with chronic myocarditis [43, 44]. In a study in 2001, 84 patients diagnosed with DCM more than 6 months prior with chronic inflammation confirmed by biopsy were randomly divided into hormone treatment and control groups, and the former was treated with steroids and azathioprine. After a 2-year follow-up, the hormone treatment group had significant improvements in LVEF and cardiac function classification compared with the control group (Fig. 19.6) [43].
Effect of prednisone on survival of patients with acute myocarditis [41]
Comparison of cardiac function between the immunosuppressive therapy and control groups of patients with chronic inflammatory dilated cardiomyopathy [43]
In 2009, the TIMIC study included 85 patients with chronic stable DCM who were confirmed to have myocarditis by endocardial biopsy, the viral genome was not found on endocardial biopsy, and the patients were randomly divided into hormone treatment and control groups. The hormone treatment group was treated with prednisone and azathioprine. After 6 months of follow-up, the hormone treatment group had significantly improved cardiac function and cardiac outcome than the control group (Fig. 19.7) [44]. The Myocarditis Treatment Trial is a randomized controlled trial that included 111 patients with an LVEF of less than 45% and histopathologically diagnosed myocarditis of unknown cause, and there was no significant difference in cardiac function improvement or survival between the hormone treatment group treated with glucocorticoids and cyclosporine or azathioprine and the control group (Fig. 19.8) [45]. A meta-analysis published in 2013 with a total of 719 patients summarized 8 clinical trials on the use of glucocorticoids in the treatment of viral myocarditis and found that, although there was no intergroup difference in mortality, the LVEF of the treatment group was significantly better than that of the control group during 1–3 months of follow-up [46].
TIMIC study [44] of patients with chronic stable dilated cardiomyopathy who had myocarditis confirmed by endocardial biopsy and had no viral genome detected by endocardial biopsy. Group 1 was the hormone therapy group (n = 43; prednisone and azathioprine), and group 2 was control group (n = 42). *Indicates a statistically significant difference. The black shadow indicates the data after 6 months of follow-up, while the white shadow indicates the baseline data. The follow-up for 6 months showed that the ejection fraction of the hormone treatment group increased significantly after 6 months and the cardiac structure recovered. The results were worse in the control group
The Myocarditis Treatment Trial [45]. The left figure compares the cumulative mortality, with the dashed line as the control group and the solid line as the immunosuppressive treatment group. The figure on the right compares the left ventricular ejection fractions of the two groups. The figure above compares the total population, while the follow-up data of some groups are missing. The figure below compares the follow-up data of the three groups. The results showed no significant difference in cardiac function and survival in patients with a left ventricular ejection fraction of less than 45% and histopathological diagnosed myocarditis of unknown cause received hormone and cyclosporine or azathioprine treatment compared with the control group
Immunoglobulin has antiviral and immunomodulatory effects, suggesting that it may be helpful in the treatment of viral myocarditis. A multicenter clinical study in Japan on 41 patients with acute myocarditis showed that high-dose intravenous immunoglobulin (IVIG; 1–2 g/kg body weight for 2 days) significantly improved patient survival, the 1-month mortality rate showed a downward trend, and the inflammatory factors in the peripheral blood were significantly reduced [47]. A retrospective study of 58 patients with explosive myocarditis in Guangdong, China, showed that IVIG 400 mg/kg for 5 days can significantly improve the patient’s LVEF and left ventricular end diastolic diameter after 4 weeks, and significantly reduce malignant arrhythmias and mortality [48]. However, a systematic review concluded that the current rigorous data are insufficient to enable recommendation of routine immunoglobulin therapy in patients with acute myocarditis [49]. Preliminary data suggest that antiviral therapy with interferon β may be beneficial in patients with chronic dilation who have viral genomes in endocardial biopsy confirmed by PCR. A study included 143 patients with biopsy-diagnosed viral myocarditis complicated with symptoms of heart failure who were randomly divided into an antiviral therapy group (interferon β1b) and a control group. The results showed that antiviral therapy could improve viral clearance and cardiac function in chronic viral myocarditis [50].
In 2017, the Chinese Society of Cardiology Expert Consensus Statement on the Diagnosis and Treatment of Adult Fulminant Myocarditis put forward a “life-support based comprehensive treatment regimen” which emphasized the early treatment of mechanical life support therapy, immunoregulatory therapy, and neuraminidase inhibitor therapy (Fig. 19.9) [15]. A multicenter controlled trial showed that treatment in accordance with the “comprehensive treatment plan” can significantly reduce the in-hospital mortality of fulminant myocarditis from 50% to 3.7%, and the use of cardiopulmonary bypass devices, antiviral therapy, and immunoglobulin were associated with improved outcomes for fulminant myocarditis [51]. In another multicenter study further confirmed its effacacy [52]. In our study center (Tongji Hospital), myocarditis patients were followed up for an average of 12 months, and we only found 24.2% fuliminant myocarditis patients had sustained LVEF <55% after discharge but no cases of cardiac death or heart transplantation in 66 discharged patients with fulminant myocarditis were reported [53]. Related research is ongoing, and the current follow-up situation is shown in Fig. 19.9.
Flowchart of clinical decision-making process in the treatment of fulminant myocarditis [50]
19.4 Follow-Up of Fulminant Myocarditis Patients
Although the association between troponin, BNP, CRP, and the prognosis of fulminant myocarditis is controversial, monitoring these indicators can help clinicians assess the patient’s condition, so as to timely adopt aggressive treatment and adjust the medication regimen. Therefore, for patients with fulminant myocarditis, we recommend that cardiac troponin (CTN), BNP, and CRP be tested, and cardiac ultrasound and magnetic resonance examination be performed regularly. It is recommended that patients be tested 1, 6, and 12 months after discharge. If all indicators and cardiac ultrasound were normal, the follow-up reexamination time could be extended.
References
Cooper LJ Jr. Myocarditis. N Engl J Med. 2009;360(15):1526–38. https://doi.org/10.1056/NEJMra0800028.
Caforio AL, Calabrese F, Angelini A, Tona F, Vinci A, Bottaro S, Ramondo A, Carturan E, Iliceto S, Thiene G, et al. A prospective study of biopsy-proven myocarditis: prognostic relevance of clinical and aetiopathogenetic features at diagnosis. Eur Heart J. 2007;28:1326–33. https://doi.org/10.1093/eurheartj/ehm076.
Grün S, Schumm J, Greulich S, Wagner A, Schneider S, Bruder O, Kispert EM, Hill S, Ong P, Klingel K, et al. Long-term follow-up of biopsy-proven viral myocarditis: predictors of mortality and incomplete recovery. J Am Coll Cardiol. 2012;59:1604–15. https://doi.org/10.1016/j.jacc.2012.01.007.
Kindermann I, Barth C, Mahfoud F, Ukena C, Lenski M, Yilmaz A, Klingel K, Kandolf R, Sechtem U, Cooper LT, et al. Update on myocarditis. J Am Coll Cardiol. 2012;59(9):779–92. https://doi.org/10.1016/j.jacc.2011.09.074.
Sagar S, Liu PP, Cooper LT Jr. Myocarditis. Lancet. 2012;379:738–47. https://doi.org/10.1016/S0140-6736(11)60648-X.
Aretz HT, Billingham ME, Edwards WD, Factor SM, Fallon JT, Fenoglio JJ, Olsen EG, Schoen FJ. Myocarditis. A histopathologic definition and classification. Am J Cardiovasc Pathol. 1987;1(1):3–14.
Rose NR, Neumann DA, Herskowitz A. Coxsackievirus myocarditis. Adv Intern Med. 1992;37:411–29.
Trachtenberg BH, Hare JM. Inflammatory cardiomyopathic syndromes. Circ Res. 2017;121:803–18. https://doi.org/10.1161/CIRCRESAHA.117.310221.
Kociol RD, Cooper LT, Fang JC, Moslehi JJ, Pang PS, Sabe MA, Shah RV, Sims DB, Thiene G, Vardeny O, American Heart Association Heart Failure and Transplantation Committee of the Council on Clinical Cardiology. Recognition and initial management of fulminant myocarditis: a scientific statement from the American Heart Association. Circulation. 2020;141:e69–92. https://doi.org/10.1161/CIR.0000000000000745.
Grogan M, Redfield MM, Bailey KR, Reeder GS, Gersh BJ, Edwards WD, Rodeheffer RJ. Long-term outcome of patients with biopsy-proved myocarditis: comparison with idiopathic dilated cardiomyopathy. J Am Coll Cardiol. 1995;26:80–4. https://doi.org/10.1016/0735-1097(95)00148-s.
Magnani JW, Danik HJ, Dec GW Jr, DiSalvo TG. Survival in biopsy-proven myocarditis: a long-term retrospective analysis of the histopathologic, clinical, and hemodynamic predictors. Am Heart J. 2006;151(2):463–70. https://doi.org/10.1016/j.ahj.2005.03.037.
Cooper LT Jr, Hare JM, Tazelaar HD, Edwards WD, Starling RC, Deng MC, Menon S, Mullen GM, Jaski B, Bailey KR, et al. Usefulness of immunosuppression for giant cell myocarditis. Am J Cardiol. 2008;102:1535–9. https://doi.org/10.1016/j.amjcard.2008.07.041.
Brambatti M, Matassini MV, Adler ED, Klingel K, Camici PG, Ammirati E. Eosinophilic myocarditis: characteristics, treatment, and outcomes. J Am Coll Cardiol. 2017;70:2363–75. https://doi.org/10.1016/j.jacc.2017.09.023.
Moslehi JJ, Salem JE, Sosman JA, Lebrun-Vignes B, Johnson DB. Increased reporting of fatal immune checkpoint inhibitor-associated myocarditis. Lancet. 2018;391:933. https://doi.org/10.1016/S0140-6736(18)30533-6.
Precision Medicine Group, Chinese Society of Cardiovascular Diseases, Working Group of Adult Fulminant Myocarditis, Chinese Journal of Cardiovascular Disease Editorial Board. Diagnosis of fulminant myocarditis in adults. Chin J Cardiovasc Dis. 2017;45(9):742–52.
McCarthy RE III, Boehmer JP, Hruban RH, Hutchins GM, Kasper EK, Hare JM, Baughman KL. Long-term outcome of fulminant myocarditis as compared with acute (nonfulminant) myocarditis. N Engl J Med. 2000;342:690–5. https://doi.org/10.1056/NEJM200003093421003.
Ammirati E, Cipriani M, Lilliu M, Sormani P, Varrenti M, Raineri C, Petrella D, Garascia A, Pedrotti P, Roghi A, et al. Survival and left ventricular function changes in fulminant versus nonfulminant acute myocarditis. Circulation. 2017;136:529–45. https://doi.org/10.1161/CIRCULATIONAHA.117.026386.
Cooper LT Jr. When lightning strikes: fulminant myocarditis in the realm of inflammatory cardiomyopathies. Circulation. 2017;136:546–8. https://doi.org/10.1161/CIRCULATIONAHA.117.029340.
Moslehi JJ, Brinkley DM, Meijers WC. Fulminant myocarditis: evolving diagnosis, evolving biology, evolving prognosis. J Am Coll Cardiol. 2019;74:312–4. https://doi.org/10.1016/j.jacc.2019.05.026.
Ammirati E, Cipriani M, Moro C, Raineri C, Pini D, Sormani P, Mantovani R, Varrenti M, Pedrotti P, Conca C, et al. Clinical presentation and outcome in a contemporary cohort of patients with acute myocarditis. Circulation. 2018;138:1088–99. https://doi.org/10.1161/CIRCULATIONAHA.118.035319.
Cappola TP, Felker GM, Kao WH, Hare JM, Baughman KL, Kasper EK. Pulmonary hypertension and risk of death in cardiomyopathy: patients with myocarditis are at higher risk. Circulation. 2002;105:1663–8. https://doi.org/10.1161/01.cir.0000013771.30198.82.
Kühl U, Pauschinger M, Seeberg B, Lassner D, Noutsias M, Poller W, Schultheiss HP. Viral persistence in the myocardium is associated with progressive cardiac dysfunction. Circulation. 2005;112:1965–70. https://doi.org/10.1161/CIRCULATIONAHA.105.548156.
Kindermann I, Kindermann M, Kandolf R, Klingel K, Bültmann B, Müller T, Lindinger A, Böhm M. Predictors of outcome in patients with suspected myocarditis. Circulation. 2008;118:639–48. https://doi.org/10.1161/CIRCULATIONAHA.108.769489.
Fuse K, Kodama M, Okura Y, Ito M, Hirono S, Kato K, Hanawa H, Aizawa Y. Predictors of disease course in patients with acute myocarditis. Circulation. 2000;102:2829–35. https://doi.org/10.1161/01.cir.102.23.2829.
Sheppard R, Bedi M, Kubota T, Semigran MJ, Dec W, Holubkov R, Feldman AM, Rosenblum WD, McTiernan CF, McNamara DM, Investigators IMAC. Myocardial expression of fas and recovery of left ventricular function in patients with recent-onset cardiomyopathy. J Am Coll Cardiol. 2005;46:1036–42. https://doi.org/10.1016/j.jacc.2005.05.067.
Nishii M, Inomata T, Takehana H, Takeuchi I, Nakano H, Koitabashi T, Nakahata J, Aoyama N, Izumi T. Serum levels of interleukin-10 on admission as a prognostic predictor of human fulminant myocarditis. J Am Coll Cardiol. 2004;44:1292–7. https://doi.org/10.1016/j.jacc.2004.01.055.
Lauer B, Schannwell M, Kühl U, Strauer BE, Schultheiss HP. Antimyosin autoantibodies are associated with deterioration of systolic and diastolic left ventricular function in patients with chronic myocarditis. J Am Coll Cardiol. 2000;35:11–8. https://doi.org/10.1016/s0735-1097(99)00485-4.
Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno-Blanes J, Felix SB, Fu M, Heliö T, Heymans S, Jahns R, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34:2636–2648d. https://doi.org/10.1093/eurheartj/eht210.
Guo WQ, Li L. Angiotensin converting enzyme inhibitors for heart failure with reduced ejection fraction or left ventricular dysfunction: a complementary network meta-analyses. Int J Cardiol. 2016;214:10–2. https://doi.org/10.1016/j.ijcard.2016.03.173.
Rezkalla S, Kloner RA, Khatib G, Khatib R. Effect of delayed captopril therapy on left ventricular mass and myonecrosis during acute Coxsackievirus murine myocarditis. Am Heart J. 1990;120:1377–81. https://doi.org/10.1016/0002-8703(90)90251-r.
Tominaga M, Matsumori A, Okada I, Yamada T, Kawai C. Beta-blocker treatment of dilated cardiomyopathy. Beneficial effect of carteolol in mice. Circulation. 1991;83:2021–8. https://doi.org/10.1161/01.cir.83.6.2021.
Thiele H, Zeymer U, Neumann FJ, Ferenc M, Olbrich HG, Hausleiter J, de Waha A, Richardt G, Hennersdorf M, Empen K, et al. Intra-aortic balloon counterpulsation in acute myocardial infarction complicated by cardiogenic shock (IABP-SHOCK II): final 12 month results of a randomised, open-label trial. Lancet. 2013;382:1638–45. https://doi.org/10.1016/S0140-6736(13)61783-3.
Ihdayhid AR, Chopra S, Rankin J. Intra-aortic balloon pump: indications, efficacy, guidelines and future directions. Curr Opin Cardiol. 2014;29:285–92. https://doi.org/10.1097/HCO.0000000000000075.
Okai I, Inoue K, Maruyama M, Maruyama S, Komatsu K, Nishizawa H, Okazaki S, Fujiwara Y, Sumiyoshi M, Daida H. Transbrachial intra-aortic balloon pumping for a patient with fulminant myocarditis. Heart Vessel. 2012;27:639–42. https://doi.org/10.1007/s00380-012-0231-z.
Wang Q, Pan W, Shen L, Wang X, Xu S, Chen R, Ge J. Clinical features and prognosis in Chinese patients with acute fulminant myocarditis. Acta Cardiol. 2012;67:571–6. https://doi.org/10.1080/ac.67.5.2174132.
Lorusso R, Centofanti P, Gelsomino S, Barili F, Di Mauro M, Orlando P, Botta L, Milazzo F, Actis Dato G, Casabona R, et al. Venoarterial extracorporeal membrane oxygenation for acute fulminant myocarditis in adult patients: a 5-year multi-institutional experience. Ann Thorac Surg. 2016;101:919–26. https://doi.org/10.1016/j.athoracsur.2015.08.014.
Diddle JW, Almodovar MC, Rajagopal SK, Rycus PT, Thiagarajan RR. Extracorporeal membrane oxygenation for the support of adults with acute myocarditis. Crit Care Med. 2015;43:1016–25. https://doi.org/10.1097/CCM.0000000000000920.
Schmidt M, Burrell A, Roberts L, Bailey M, Sheldrake J, Rycus PT, Hodgson C, Scheinkestel C, Cooper DJ, Thiagarajan RR, et al. Predicting survival after ECMO for refractory cardiogenic shock: the survival after venoarterial-ECMO (SAVE)-score. Eur Heart J. 2015;36:2246–56. https://doi.org/10.1093/eurheartj/ehv194.
Nakamura T, Ishida K, Taniguchi Y, Nakagawa T, Seguchi M, Wada H, Sugawara Y, Funayama H, Mitsuhashi T, Momomura S. Prognosis of patients with fulminant myocarditis managed by peripheral venoarterial extracorporeal membranous oxygenation support: a retrospective single-center study. J Intensive Care. 2015;3:5. https://doi.org/10.1186/s40560-014-0069-9.
Pozzi M, Banfi C, Grinberg D, Koffel C, Bendjelid K, Robin J, Giraud R, Obadia JF. Veno-arterial extracorporeal membrane oxygenation for cardiogenic shock due to myocarditis in adult patients. J Thorac Dis. 2016;8:E495–502. https://doi.org/10.21037/jtd.2016.06.26.
Latham RD, Mulrow JP, Virmani R, Robinowitz M, Moody JM. Recently diagnosed idiopathic dilated cardiomyopathy: incidence of myocarditis and efficacy of prednisone therapy. Am Heart J. 1989;117:876–82. https://doi.org/10.1016/0002-8703(89)90626-1.
Parrillo JE, Cunnion RE, Epstein SE, Parker MM, Suffredini AF, Brenner M, Schaer GL, Palmeri ST, Cannon RO III, Alling D. A prospective, randomized, controlled trial of prednisone for dilated cardiomyopathy. N Engl J Med. 1989;321:1061–8. https://doi.org/10.1056/NEJM198910193211601.
Wojnicz R, Nowalany-Kozielska E, Wojciechowska C, Glanowska G, Wilczewski P, Niklewski T, Zembala M, Polonski L, Rozek MM, Wodniecki J. Randomized, placebo-controlled study for immunosuppressive treatment of inflammatory dilated cardiomyopathy: two-year follow-up results. Circulation. 2001;104:39–45. https://doi.org/10.1161/01.cir.104.1.39.
Frustaci A, Russo MA, Chimenti C. Randomized study on the efficacy of immunosuppressive therapy in patients with virus-negative inflammatory cardiomyopathy: the TIMIC study. Eur Heart J. 2009;30:1995–2002. https://doi.org/10.1093/eurheartj/ehp249.
Mason JW, O’Connell JB, Herskowitz A, Rose NR, McManus BM, Billingham ME, Moon TE. A clinical trial of immunosuppressive therapy for myocarditis. The myocarditis treatment trial investigators. N Engl J Med. 1995;333:269–75. https://doi.org/10.1056/NEJM199508033330501.
Chen HS, Wang W, Wu SN, Liu JP. Corticosteroids for viral myocarditis. Cochrane Database Syst Rev. 2013;2013:CD004471. https://doi.org/10.1002/14651858.CD004471.pub3.
Kishimoto C, Shioji K, Hashimoto T, Nonogi H, Lee JD, Kato S, Hiramitsu S, Morimoto SI. Therapy with immunoglobulin in patients with acute myocarditis and cardiomyopathy: analysis of leukocyte balance. Heart Vessel. 2014;29:336–42. https://doi.org/10.1007/s00380-013-0368-4.
Yu DQ, Wang Y, Ma GZ, Xu RH, Cai ZX, Ni CM, Chen P, Zhu ZD. Intravenous immunoglobulin in the therapy of adult acute fulminant myocarditis: a retrospective study. Exp Ther Med. 2014;7:97–102. https://doi.org/10.3892/etm.2013.1372.
Robinson JL, Hartling L, Crumley E, Vandermeer B, Klassen TP. A systematic review of intravenous gamma globulin for therapy of acute myocarditis. BMC Cardiovasc Disord. 2005;5(1):12. https://doi.org/10.1186/1471-2261-5-12.
Schultheiss HP, Piper C, Sowade O, Waagstein F, Kapp JF, Wegscheider K, Groetzbach G, Pauschinger M, Escher F, Arbustini E. Betaferon in chronic viral cardiomyopathy (BICC) trial: effects of interferon-beta treatment in patients with chronic viral cardiomyopathy. Clin Res Cardiol. 2016;105:763–73. https://doi.org/10.1007/s00392-016-0986-9.
Li S, Xu S, Li C, Ran X, Cui G, He M, Miao K, Zhao C, Yan J, Hui R. A life support-based comprehensive treatment regimen dramatically lowers the in-hospital mortality of patients with fulminant myocarditis: a multiple center study. Sci China Life Sci. 2019;62:369–80. https://doi.org/10.1007/s11427-018-9501-9.
Zhou N, Zhao Y, Jiang J, Shen L, Li J, Wan J, Ma X, Zhang J, Ammirati E, Wang DW. Impact of mechanical circulatory support and immunomodulation therapy on outcome of patients with fulminant myocarditis: Chinese registry of fulminant myocarditis. Signal Transduct Target Ther. 2021;6(1):350. https://doi.org/10.1038/s41392-021-00700-6.
Jiang JG, Liu C, Cui GL, Chen C, Zuo HJ, Li R, Wang DW.Long term prognosis of fulminant myocarditis and predictors related to impaired cardiac function post discharge. Chin J Cardiol. 2022;50:263–9.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Jiang, J., Wang, D.W. (2022). Follow-Up and Long-Term Prognosis of Myocarditis and Fulminant Myocarditis. In: Wang, D.W. (eds) Fulminant Myocarditis. Springer, Singapore. https://doi.org/10.1007/978-981-19-5759-8_19
Download citation
DOI: https://doi.org/10.1007/978-981-19-5759-8_19
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-5758-1
Online ISBN: 978-981-19-5759-8
eBook Packages: MedicineMedicine (R0)